Research Article - (2025) Volume 9, Issue 2
Fabrication and Characterization of Ascorbyl Propyl Hyaluronate for Skin Anti-Aging
2Biomaterials Research center, CELLINBIO Co., Ltd., Suwon-si, Republic of Korea
3CAPABIOSCIENCE Co., Ltd., Guri-si, Republic of Korea
Received Date: Apr 10, 2025 / Accepted Date: May 05, 2025 / Published Date: May 14, 2025
Copyright: ©Ã?©2025 Jin-Su Kim, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Yu, J. W., Choi, H., Park, W., Kim, J., Kim, G. H. (2025). Fabrication and Characterization of Ascorbyl Propyl Hyaluronate for Skin Anti-Aging. J App Mat Sci & Engg Res, 9(2), 01-12.
Abstract
Background: Ascorbic acid (AA) is a powerful antioxidant that can help slow premature skin aging, improve the appearance of acne, wrinkles and dark spots, and against sun damage. Despite these advantages, the low oxidative stability and skin penetration rate of AA are major obstacles to commercial applications and require improvement.
Methods: In this study, we developed ascorbyl propyl hyaluronate (APH), a hyaluronic acid conjugated with ascorbic acid, to overcome the disadvantages of AA. To confirm the characteristics of the developed APH, the thermal stability, skin penetration rate, and antioxidant capacity were evaluated, and the cell viability, cell proliferation, and skin-related cell regeneration efficacy were compared with the conventional AA.
Results: We demonstrated that APH is improved thermal stability than AA while maintaining antioxidant activity. In addition, APH was not cytotoxic and increased cell proliferation in a dose-dependent manner. Furthermore, APH not only reduces inflammatory cytokine expression, melanin content, and cellular tyrosinase activity, but also increases type 1 collagen production, Enhanced cell barrier function and actin filament development.
Conclusion: This strategy using APH with improved oxidative stability and tissue penetration efficiency is expected to provide a useful source for maximizing the effectiveness of anti-aging and skin tissue regeneration in the future when combined with various modern skin aging treatments.
Keywords
Hyaluronic Acid, Ascorbic Acid, Drug Stability, Penetration, Skin Aging
Introduction
Skin is an organ that exists in the outermost layer of the body and is an important organ that has a barrier function to prevent evaporation of moisture in the body and to protect the body from invasion of foreign substances such as chemicals or microorganisms from the outside [1-3]. However, the skin barrier is damaged by external stimuli factors caused by climate change and environmental pollution, and the proportion of patients with skin sensitivity, such as skin inflammation and dryness due to moisture loss, is increasing [4-6]. Continuous exposure to external stimulants increases the generation of reactive oxygen species (ROS), which induces damage and death of skin related cells and increases skin aging [7,8]. In addition, it has been reported that inflammatory factors activated by external stimuli damage the skin barrier and reduce skin elasticity by promoting the production of MMP-1 (matrix metalloproteinase-1), an enzyme that degrades collagen protein due to active ROS [9,10].
L-ascorbic acid (AA or vitamin C, Figure. 1A), which is abundant in plants, fruits, vegetables, and algae, is used in human tissues as a non-enzymatic antioxidant and reduces oxidative damage to cellular organelles by reducing the reactivity of free radicals [11,12]. Recently, the biochemical properties of AA have attracted considerable attention due to their skin protective effects, such as photo-protection effects against UV and inhibition effects of melanin production [13,14]. However, AA, a hydrophilic substance, has difficulty penetrating the hydrophobic stratum corneum and is easily oxidized when exposed to various environmental factors such as heat, light, moisture, and oxygen due to its low stability [15,16]. Therefore, despite many advantages, a poor oxidative stability of AA is one of the major obstacles to its application in various fields such as cosmetics, dermatology, and pharmaceuticals [17,20]. Some AA derivatives such as AA2G (Ascorbic acid 2-glucoside), AAE (3-o-ethyl-l-ascorbic acid), MAP (magnesium ascorbyl-2- phosphate) and AA6P (Ascorbyl 6-palmitate) have been proposed in order to overcome the production process of AA and instability within dosage form[21,24]. Unfortunately, the enhanced stability of ascorbic acid also accompanied the impaired permeability or lowered activity during the topical application on human skin (25).
Figure 1A
Meanwhile, Hyaluronic acid (HA, Figure. 1B) is a biopolymer formed by combining N-acetylglucosamine (UDP-GlcNAc) and glucuronic acid (UDP-GlcUA) together in the form of a chain and is known as an important constituent of connective tissue such as skin or cartilage tissues [26,28]. In addition, several previous studies have reported that hyaluronic acid can be used as an important biomedical material for drug delivery and tissue engineering, and that transdermal delivery efficiency can be improved by delivering conjugated with HA and drugs or hormones [29-32].
Figure 1B
In the present study, on the basis of positive effect of HA, we hypothesized that it is possible to conjugate of HA-AA (Ascorbyl Propyl Hyaluronate, APH, Figure. 1C) for enhancing the stability of AA and creating synergistic effects of conjugates. The APH was synthesized by introducing an alkyl group into the hydroxyl group at C3 of ascorbic acid using the Mitsunobu synthesis method and then etherifying the modified oligo-HA (O-HA)[33]. The purpose of this study was to determine whether the newly synthesized APH could improve the instability of AA while maintaining antioxidant and anti-inflammatory effects and prevents skin aging through the moisturizing and cell penetration effects of HA. In order to evaluate the anti-aging effect of the newly synthesized APH, we investigated the APH activity test in three stages: in vitro thermal stability and release test, in vitro cell behavior and antioxidant test, and in vitro anti-aging test.
Materials & Methods
Materials
Dulbecco’s modified Eagle’s medium (DMEM) and fetal bovine serum (FBS) were purchased from Invitrogen-Gibco (Grand Island, NY. USA). Neonatal human dermal fibroblast (HDF, ATCC Catalog No. PCS-201-010), murine macrophage J774A.1 cells (ATCC Catalog No. TIB-67), and murine melanoma B16F10 cells (ATCC Catalog No. CRL-6475) were purchased from ATCC (Manassas, VA, USA). Procollagen type I C-peptide (PIP) ELISA kit was purchased from Takara (Shiga, Japan) and PEG2, and TNF-α ELISA kit were purchased from R&D Systems (Minneapolis, MN, USA). The α-melanocyte stimulating hormone (α-MSH), arbutin, ascorbic acid, and L-DOPA were obtained from Sigma-Aldrich (St. Louis, MO. USA). Oligo hyaluronic acid, with an average molecular weight is 5,000 Da was obtained from SK bioland (Dongnam, Korea). Strat-M Synthetic membrane was obtained from Merck Millipore (Merck KGaA, Darmstadt, Germany).
For the preparation of APH various chemicals were used such as: Triphenylphosphine, DIAD, 1-bromopropanol, L-Ascorbic-Acid- Acetonide, and THF (ACS grade) were purchased from Sigma Aldrich. NMR spectra were recorded with 400 MHz for 1H NMR spectrometer. The chemical shifts are reported in parts per million related to tetramethyl silane as an internal standard. 1H NMR spectra are reported relative to residual D2O (d = 4.79 ppm).
Synthesis of HA-ascorbic acid (APH) Conjugate
Briefly, an alkyl group is introduced into the hydroxyl group at C3 of ascorbic acid protected in the form of an acetal using the Mitsunobu synthesis method, and then the protective group is removed. This compound was reacted with hyaluronic acid tetrabutylammonium salt in DMSO for about 24 hours to obtain the final compound, ascorbyl propyl hyaluronate (APH). In more detail, the synthesis of APH consists of the following four steps: 4-(3-bromopropoxy)-5-(2,2-dimethyl-1,3-dioxolan-4-yl)-3- hydroxyfuran-2(5H)-one (1): To a stirred solution of Ph3P (1.2 eq) in dry THF at 0ºC was added DIAD (1.2 eq). After 15 minutes at 0ºC, white solid (Mitsunobu betaine) was formed. Stirring was continued at 0ºC for additional 10 minutes. Then a solution of L-Ascorbic-Acid-Acetonide (1 eq) in THF was added. After 5 minutes, the mixture was treated with 1-bromopropanol (1.2 eq) and stirred for 3 hours at RT. The solvents were removed under reduced pressure. The resulting oil was purified by column chromatography on silica gel (1:1 hexane/EtOAc) to give a viscous.
4-(3-bromopropoxy)-5-(1,2-dihydroxyethyl)-3-hydroxyfuran- 2(5H)-one (2): To a stirred solution of compound 1 in MeOH (10 mL) was treated with conc.HCl (3 mL) and stirred for 3 hours at RT. The solvents were removed under reduced pressure to give a colorless viscous oil, which was used for the subsequent reaction without purification.
HA-TBA salt (3): The sodium salt of HA is converted to a tetrabutylammonium (TBA; Sigma) salt by acidic ion exchange (room temperature, 8 hours) with Dowex 50 W × 8–100 resin, neutralized in aqueous TBA hydroxide. The solution was then freeze-dried and resulting TBA-HA was kept in a freezer until further use.
Ascorbyl Propyl Hyaluronate (4): HA-TBA (1 eq) are solubilized in 10 mL DMSO, and then compound 2 (1.2 eq) and tetrabutylammonium iodide (0.1 eq) were added. The resulting solution was stirred for 24 hours at RT and slowly poured into acetone under agitation. A precipitate is formed which is filtered and washed three times acetone and finally vacuum dried for 8 hours.
NMR Analysis
Three kinds of samples (AA, O-HA and APH) were prepared for solution NMR analysis. 25 mg of each sample was dissolved in 600 μL 100% D2O and final concentration of samples were 142 μM, 65 μM and 3.1 μM respectively. All NMR spectra were recorded using a Bruker Avance III spectrometer operating 400 MHz (Bruker Biospin, Germany) equipped with a z-gradient unit. The complex point of 1D 1H NMR spectra was 65 K and spectral widths were 8 KHz. Acquisition time and relaxation delay were 0.5 and 10 seconds respectively. All experiments were carried out at 50ºC.
In Vitro Thermal Stability Test
APH and AA were dissolved in water at concentrations of 1000 μg/mL and 150 μg/mL, respectively, and incubated at 50°C. After incubation, samples taken at 0, 24, and 48 hours were analyzed by HPLC (Agilent technology 1200 series, Santa Clara, CA. USA). The area value of 0-hour sample was set as control and calculated as 100. Area values were calculated after 24 and 48 hours and compared with the control group.
Ascorbic Acid Assay
Ascorbic acid was measured using ascorbic acid colorimetric assay kit (BioVision, Korea). First, prepare test samples to a final volume of 120 µL/well with ascorbic acid assay buffer in a 96-well microplate. Second, 30 μL of catalyst was added to each well, and 50 μL of the reaction mixture (46 µL Ascorbic acid assay buffer, 2 µL Ascorbic acid probe, 2 µL Ascorbic acid enzyme) was added with blocking of light. Absorbance measurements were performed at 570 nm using a microplate reader.
DPPH Scavenging Activity Assay
The antioxidant potential of AA, O-HA and APH was assessed using the DPPH scavenging photometric assay. For DPPH assay, the color intensity reduction of each sample was performed at 517 nm using a UV spectrometer in a 96-well plate. A 100 µL of each concentration sample (ranging from 1.25 µg/mL to 250 µg/mL) was mixed with 0.2 mM of methanolic DPPH and the color change was measured using methanol as a blank. DPPH inhibition percent was calculated according to the following the equation 1.
DPPH scavenging percent (%) = [(Ac. – As.) / Ac.] x 100
Here, Ac. = absorbance value of the control group, and As. = absorbance value of the test sample.
In Vitro Skin Permeation Studies
In this study, a skin permeation experiment of APH was performed using Strat-M Transdermal Diffusion Membrane. The Strat-M membrane without pre-soaking treatment was mounted on the static diffusion device (Franz diffusion cell) of a transdermal absorption system (Phoenix Robotic Diffusion Station, Hanson Research, USA) Then, 150 μL of AA and APH dissolved in water at a concentration of 20 mg/mL were added into the donor chamber of the static diffusion device. The reaction was performed for 24 hours under the experimental conditions of 32.5°C and 600 rpm. To evaluate the skin permeation of the test substance, 450 μL of samples from the receptor chamber of the static diffusion device were collected eight times (0, 2, 4, 6, 8, 10, 12, and 24 hours) and quantitatively analyzed by HPLC (Agilent technology 1200 series, Santa Clara, CA, USA). The cumulative AA permeation per unit of Strat-M skin surface area was obtained by equation 2.
Tn = Cn / A
Where, Tn (μg) is the cumulative amount of AA per unit area, Cn is the measured AA concentration in the nth sample, and A is the area of Strat-M membrane.
Steady-state flux (Jss, μg/h/cm2) represents the rate of penetration, which can be calculated from the amount of permeant per unit time and area for the initial concentration applied to the upper skin area. Permeability coefficient (Kp, cm/h) can be calculated from flux value divided by the initial concentration of the test substance.
Cell Culture and Proliferation Assay
The cells were maintained in DMEM supplemented with 10% (v/v) FBS (and 1% penicillin/streptomycin) at 37°C and 5% CO2. The cell proliferation effect of APH on HDF was evaluated using WST-1 assay. 3 x 103 cells were suspended in 200 μL of assay medium, seeded on 96-well cell culture plate, and incubated for 24 hours. Cells were treated with various concentrations of APH and the equivalent amount of AA contained in the APH conjugate as control and incubated for 72 hours. Then, WST-1 reagent was added to each well and the plates were incubated at 37°C in the tissue culture incubator for 1 hour. The absorbance was measured at 450 nm using a microplate reader.
Enzyme-Linked Immunosorbent Assay (ELISA) and Treatment
Collagen biosynthesis and anti-inflammation effects were measured by ELISA. All ELISAs were performed according to the protocols provided from the manufactures. For procollagen type 1 assay, HDF cells were seeded at 5 x 103 cells/well into 48- well tissue culture plate, starved in serum-free media for 24 h, and cultured in the presence or absence of AA or APH for 72 hours. Collagen assay was performed using a commercial PIP ELISA Kit according to the protocols provided from the manufactures. For anti-inflammation assay, 5 x 104 J774A.1 macrophage cells were seeded into 24-well plate. Cells were pre-treated with various doses of AA and APH for 2 hours and then with LPS (150 ng/ mL). After 24 hours, cell culture supernatant was harvested and the concentration of PEG2 and TNF-α was measured by ELISA according to the protocols provided from the manufactures.
Scratch Wound Healing Assay
Cell migration and motility were determined by a scratch wound healing assay. HDF was seeded at a density of 1 x 106 cells/well in 6-well cell culture plates and cultured for 24 hours. To determine the wound healing potential of APH, each well was artificially wounded by scratching the cell monolayer with a 10 uL plastic pipette tip, and then the cells were irradiated with UVB (100 mJ/ cm2). Cells were washed with fresh medium and treated with AA or APH for additional 12 hours. Images of scratch wounds were taken after post-wounding, and the wound width measurements 12 hours were subtracted from the wound width at 0-hour to obtain the net wound closure rate. Wound closure area was quantitatively analyzed by using the ImageJ software (NIH, version 1.25p).
Fluorescent Immunohistochemistry
Epidermal keratinocytes were seeded and grown on the NUNC Lab-Tek chamber slide system (Rochester, NY, USA) to 40% - 50% confluence, serum-deprived overnight and then treated with APH for 24 hours. At the end of experiment, cells were fixed with 3.7% formaldehyde for 10 minutes at room temperature and then pre-incubated with blocking solution (2.5% bovine serum albumin in phosphate buffered saline) for 40 minutes. Zonula occludens-1 (ZO-1) in cultured epidermal keratinocytes was stained with rabbit polyclonal antibodies (Molecular Probes, Waltham, MA. USA). After brief washing with PBS, cells were incubated with Alexa 488 anti-rabbit secondary antibodies (Molecular Probes, Waltham, MA. USA). Cell nuclei were stained with DAPI (Molecular Probes, Waltham, MA. USA) for 10 minutes. Images were observed using a confocal microscope (FluoView 1000; Olympus Corporation, Tokyo, Japan).
Melanin Contents Assay and Cellular Tyrosinase Activity Assay
For melanin measurement, murine B16F10 melanoma cells were treated with arbutin, AA, or APH for 72 hours with or without α-MSH. After 72 hours, the cells were washed with PBS and dissolved in 100 mL of 2 N NaOH at 60°C and absorbance was measured at 450 nm. Tyrosinase activity was examined in melanoma B16F10 cells. After treatment with arbutin, AA or various concentrations of APH, the cells were stimulated with or without 0.1 μg/mL of α-MSH. After 72 hours, the cells were lysed and the L-Dopa oxidation activity of tyrosinase was measured at 475 nm. The melanin/tyrosinase activity inhibition ratio was obtained by the equation 3.
Inhibition rate (%) = [(A - C) / (A – B)] x 100
Where, A. Melanin/tyrosinase activity contents of α-MSH-treated control
B. Melanin/tyrosinase activity contents of not-treated control
C. Melanin/tyrosinase activity contents of test sample-treated
Statistical Analysis
All of the experiments were performed in triplicate and repeated three times. The data were represented as the mean ± SD. The two- way analysis of variance (ANOVA) test was performed to analysis of quantitative values, and the Tukey’s post hoc test was performed for all pair-wise comparisons among groups. Statistically significant differences among the means were considered P-value of less than 0.05.
Results
Synthesis of APH
The synthesis of 4-(3-bromopropoxy)-5-(1,2-dihydroxyethyl)- 3-hydroxyfuran-2(5H)-one (2), key intermediate for coupling with HA-TBA, are outlined in Figure. 1D. We have introduced Mitsunobu conditions for the preparation of 3-O-Alkyation derivative of L-ascorbic acid (1) in good yield. Deprotection of the 5,6-O,O-protected derivative of L-ascorbic acid (1) with Methanolic HCl gave 4-(3-bromopropoxy)-5-(1,2- dihydroxyethyl)-3-hydroxyfuran-2(5H)-one (2). This compound was reacted with HA-TBA salt in DMSO for about 24 hours to obtain the HA-AA conjugate (APH).
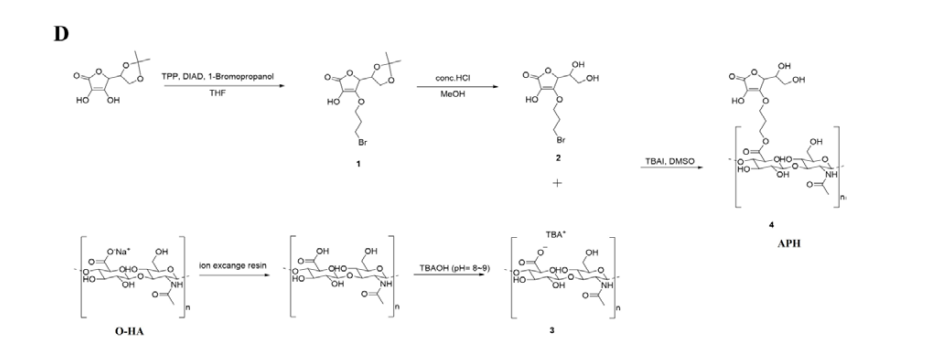
Figure 1D
NMR Analysis
To investigate the structure and conjugate ratio of APH, we comparatively analyzed O-HA, AA, and APH using 1H NMR (Proton NMR). Figure. 2A is a representative 1H NMR spectrum of HA-TBA salt before conjugating with AA, in which the Tetra butyl group of TBA shows four peaks (red arrows) and the signal corresponding to the proton of the methyl group of the HA N-acetyl group (CH3) is found in the 2.0 ppm region (purple arrow). The synthesis of HA–Vita C (APH) conjugates was further confirmed peak was identified at 2.0 ppm along with ascorbic acid H at 4.9 ppm. Singlet peaks at 2.2 ppm were attributed to -CH2- group (red circle) present in ascorbic acid moiety in the conjugate. Additionally, the conjugation yield is defined as the amount of modified material relative to the initial material. The conjugation yield was calculated using the following calculations:
Conjugation yield (%) = [(Ha. / 2) / (Hb. / 3)] x 100
Where Ha. corresponds to the integration area of protons located at 2.2 ppm (propyl linker, red circle) and Hb. are the integration area of protons located at 2.0 ppm (HA N-acetyl group, purple circle). All proton peaks used to determine the conjugate ratio were selected without any overlap, and yields of 70–80% were obtained for all samples. As a result of solution NMR analysis, the synthesis, chemical structure, and conjugation yield of APH were confirmed, and it was confirmed that our new synthesis process could effectively synthesize AA derivatives.
Figure 2A and 2B
Characterization of AA in APH
Antioxidant effect of APH was measured using DPPH assay. Unlike HA, AA and APH showed free radical scavenging activities (Figure. 3A). The AA has a rapid free radical scavenging activity from the low concentration and shows almost 96% at 7.81 μg/ mL, whereas APH gradually increases and observes free radical scavenging activity similar to AA in 125 μg/mL. In addition, we compared the thermal stability of AA bound to APH with the conventional AA. The AA bound to APH remained 66.16% after 24 hours incubation and 45.52% after 48 hours incubation, whereas AA remained only 12.95 and 1.08%, respectively (Figure. 3B). In vitro ascorbic acid colorimetric assay was performed to observe the conjugation stability of APH in the absence of esterase. In the experiment, the conventional AA shows dose-dependent detection results, while APH was confirmed that the AA bound to the APH was not detected even at high concentrations (Figure. 3C). Additionally, we performed skin permeability tests using an artificial membrane (Strat-M Transdermal Diffusion Membrane), a synthetic non-animal model useful for predicting human skin diffusion, which is known to correlate more closely with human skin than animal skin models[34,36]. Unfortunately, AA did not permeate the artificial membrane (data not shown). According to a report by Vayachuta et al., the Strat-M membrane can only permeate chemicals with log P between -0.131 and 6.9 due to its high hydrophobicity, whereas AA has difficulty permeating the artificial membrane because its log P is -2.15[37]. However, unlike AA, APH showed sustained skin permeation for 24 hours. Figure. 3D depicts the amount of AA permeated into the receiving chamber through a unit area of skin for 24 hours. The permeated AA content started at 548.18 μg/cm2 in the initial 30 minutes and showed a cumulative permeation amount of 6995.12 μg/cm2 over 24 hours, with a steady-state flux (Jss, μg/h/cm2) of 734.44 and a permeability coefficient (Kp, cm/h) of 621.74. Collectively, these results demonstrate that the advantages of AA were maintained while the disadvantages of low heat stability and skin penetration efficiency were dramatically improved by conjugation with HA, and the binding stability of AA-HA was confirmed, which did not release free AA under the absence of esterase.
Figures 3A to 3D
Effect of APH on Proliferation and Collagen Synthesis of HDFs
Increased cell death was observed as the treated AA concentration increased (Figure. 4A), and more than 50% of cell death was observed at 50 μg/mL (data not shown). Schmidt et al reported a study on the cytotoxicity of AA at high concentrations [11]. This result showed the same tendency as the previously reported results. On the other hand, APH significantly increased the proliferation of HDF in dose-dependent manner (Figure. 4B). When AA was treated at 15 and 30 μg/mL, the cell death was increased to 7.50 and 23.75%, respectively, whereas the APH containing equivalent amount of AA promoted 117.25 and 119.50% cell growth. The conjugation of AA and HA did not only reduce the cytotoxicity of AA at high concentrations but also promoted cell proliferation. To investigate the effect of APH on collagen biosynthesis, HDF was treated with APH at different concentrations for 72 hours. Collagen synthesis ability was analyzed using type I procollagen ELISA, and ascorbic acid was treated as a positive control. The amount of type I procollagen released from the HDF was measured. In the AA treatment group, the amount of type I procollagen was increased at a low concentration depending on AA concentration but started to decrease from 7.5 μg/mL. In particular, the amount of procollagen at 30 μg/mL AA was reduced to 96.15% compared to the untreated group (Figure. 4C). The group treated with 100 and 200 μg/mL APH conjugate containing equivalent amount of 15 and 30 μg/mL AA showed a statistically significant increase in the synthesis of procollagen by 115.55 and 122.80% compared to the untreated group (Figure. 4D). These results are thought to be the effect of reducing cytotoxicity and enhancing cell activity while continuously releasing AA from APH.
Anti-Inflammatory Effect of APH on LPS-Induced J774A.1
Murine macrophages J774A.1 were treated with LPS in the absence or the presence of AA or APH. PGE2 and TNF-α were measured in the supernatants of culture media at 24 hours after LPS treatment. The PGE2 level increased over four-fold from baseline levels with LPS treatment, suggesting that macrophages were stimulated. When treated with 7.5 μg/mL of AA, LPS induced PGE2 decreased by 22.65%, whereas when treated with APH at 100 and 200 μg/mL, the PGE2 decreased to 38.80 and 47.71%, respectively (Figure. 4E). In addition, LPS-stimulated TNF-α production was reduced by 8.70% in the 7.5 μg/mL of AA treated group. However, APH decreased the TNF-α production by 16.43 and 23.19% at concentrations of 100 and 200 μg/mL, respectively (Figure. 4F). These results showed that APH enhances the anti- inflammatory effect more than AA treatment group.
Figures 4A to 4F
Efficacy of Improving Skin Anti-Aging by APH
To determine whether APH has an effect on improving skin from environmental stress factors, it induced skin cell damage and confirmed wound healing, skin barrier function, and whitening effects. As shown in Figure. 5A, dermal fibroblasts exposed to APH showed statistically higher wound healing efficacy compared to the untreated control and UVB treated groups. In detail, AA showed a low wound recovery of 49.59% compared to 0-hour under UVB treatment conditions, but APH showed a rapid wound recovery of 89.23% from low concentration (10 ug/mL), and it was confirmed that it increased in a APH dose-dependent manner (Figure. 5B). Secondly, we observed the expression of tight junction protein ZO-1, which plays an important role in the skin barrier function [38] in UVB-induced human skin keratinocytes. As with the previous wound healing, even in low-concentration APH conditions, ZO-1 expression was shown at 26.82%, which was higher than in the untreated condition without UVB treatment, and it was observed that the cell barrier was strengthened in a dose-dependent manner of APH (Figure. 5C, D). These results confirmed that APH is effective in suppressing skin damage caused by UVB through strengthening the skin barrier and improving cell migration ability. Finally, we measured melanin pigmentation (Figure. 5E) and tyrosinase activity (Figure. 5F) in APH-treated B16F10 melanocytes to investigate the inhibitory effect of APH on melanogenesis. Arbutin, a melanin pigmentation inhibitor, was used as a positive control and AA was used as a control. The production of melanin pigment was markedly increased after α-MSH addition, APH decreased melanin pigmentation in dose-dependent manner. The inhibition of pigmentation was increased to 19.30, 63.16, 80.70 and 116.14%, respectively, as the concentrations of APH were increased to 10, 50, 100 and 200 μg/ mL. This was superior to the 81.75% inhibition rate of 100 μg/ mL arbutin, well known pigmentation inhibitor. AA showed the inhibition rate of 66.67% when treated with 7.5 μg/mL, but when treated with higher concentration, cell death would occur due to its cytotoxicity (Figure. 4A). Interestingly, 100 μg/mL of APH containing 15 μg/mL of AA showed no cytotoxicity (Figure. 4B) and inhibited melanin pigmentation by 80.70%. Moreover, the inhibition of tyrosinase activity was increased to 16.07, 53.57, 83.93 and 183.93%, respectively, as the concentrations of APH were increased to 10, 50, 100 and 200 μg/mL. Our results of inhibition of melanin pigmentation through B16F10 cells were interpreted as a result that APH inhibited tyrosinase activity and thereby suppressed melanin pigmentation, because pigmentation was suppressed in a pattern similar to that of APH-induced reduction of tyrosinase activity.
Figure 5A to 5F
Discussion
Human skin aging that forms the wrinkles and pigmentation of skin is the result of two biologically independent processes, which forms the wrinkles of skin[39,40]. The first is age-associated intrinsic or innate aging, an inevitable, genetically determined process. The second is extrinsic skin aging, which is the result of environmental factors (i.e. air pollution, solar ultraviolet radiation, nutrition, etc.) and it is commonly referred to as photoaging or environmental and lifestyle aging[41]. Especially, it is known that extrinsic aging not only causes cosmetic problems such as deep wrinkles and pigmentation, but also causes various skin diseases including skin cancer[42,44].
Several previous studies have reported that AA has protective potential against both extrinsic and intrinsic skin aging by effectively preventing cellular aging in vitro and in vivo[45,48]. AA plays an indispensable and important role in the human body, but most AA is easily oxidized to dehydroascorbic acid (DHA) by various environmental factors [49,50]. Recently, researchers demonstrated that chemically modified AA derivatives have the more stable oxidative stability than AA and may be easily used replacing the C-2 hydroxyl group of vitamin C with glucose and is considered one of the most important derivatives in the field of cosmetics. However, it is known that the activity of AA2G does not appear significantly because of the low concentration of glucosidase in the skin.
Therefore, in the present study, we explored the potential of researching novel AA derivatives that it is more suitable for the skin environment while effectively improving the low oxidation stability of AA for anti-aging skin treatment. In addition, we confirmed the possibility that a novel AA derivative chemically combined with HA, a natural polymer that can be used as a skin moisturizer and drug delivery agent, can be used as a skin anti- aging raw material. To this end, we developed a process to produce a new AA derivative (APH) by chemically combining HA and AA through a propyl linker decomposed by esterase (Figure. 1D).
First, we confirmed whether the newly synthesized APH was chemically bound to HA and AA via a propyl linker and the binding ratio by NMR. In the 1H NMR results, unlike HA-TBA salt (Figure. 2A), the tetra butyl peak of TBA disappeared in APH, and the peaks of the propyl linker (red circle) and ascorbic acid (blue circle) were identified, indicating that HA was substituted with a thiolated side chain (Figure. 2B). Furthermore, 1H NMR analysis showed that yields of 70-80% were obtained for all samples.
An interesting result was obtained while investigating the mechanical properties of AA in APH. In this study, APH significantly improved the thermal stability (Figure. 3B) and skin permeability (Figure. 3D) while maintaining the antioxidant capacity (Figure. 3A), which is one of the characteristics of AA. It is well known that chemical modification of AA derivatives plays a central role in the regulation of oxidative stability[51,53]. Furthermore, several researchers have confirmed that conjugation with other substances has advantages such as chemical stability of vitamin C, maintenance of ROS consumption, efficient penetration into the skin lipid barrier, and control of vitamin C degradation [55,57]. As mentioned above, in our study, it was also confirmed that AA release can be suppressed even at high concentrations when degradation selectivity by esterase is imparted through the propyl linker in the free ascorbic acid detection test (Figure. 3C). Taken together, these finding suggests that chemical bonding with HA via a propyl linker can play an important role in improving the oxidative stability, skin permeability and release control of AA while maintaining its antioxidant capacity.
As an additional in-vitro study, we investigated the effects of newly synthesized APH on both cell behaviors and anti-inflammatory ability in dermal fibroblasts and macrophage cell. In earlier reports AA-based cell therapy, it was verified that AA not only stimulates the reduced proliferative capacity of elderly dermal fibroblasts, but also increasing collagen synthesis in elderly cells by similar degrees as in newborn cells[58,59]. However, Schmidt et al demonstrated that high concentrations of ascorbic acid induced high cytotoxic levels to fibroblast cells in culture through the glutathione status of the cells unfavorably[60]. As in previous studies, in our findings, cell proliferation and type I collagen synthesis were significantly decreased when AA was present in the higher levels (Figure. 4A, C). On the other hand, newly synthesized APH significantly increased cell proliferation and type I collagen synthesis in dose-dependent manner (Figure. 4B, D). Moreover, high-dose APH also effectively down-regulated the expression level of pro-inflammatory factors unlike AA (Figure. 4E and 4F). In that respect, our new AA derivative is superior in that it allows the use of high doses of AA an intact chemical form. The important aspect of this present study results is that even when APH is used at high doses, cytotoxicity is minimal, while anti inflammatory effects and skin compatibility can be improved, making it very valuable as a raw material for skin regeneration.
Finally, we investigated the effect of APH on the skin regeneration in of aged dermal fibroblasts, epidermal keratinocyte and melanocyte. Recently, clinical studies have directly demonstrated that consistent use of topical AA or AA derivatives significantly improves the skin barrier, wrinkles, skin tone and dark spots, and gives it a more youthful appearance[46,61,63]. Our results demonstrated that high-dose APH treatment in skin cells not only improved cell migration and skin barrier function, but also reduced melanin production through tyrosinase inhibition more effectively than AA (Figure. 5). These in vitro results provide evidence that APH reverses cellular senescence of skin cells more than AA, enhancing cell activity and increasing the recovery rate of aged skin tissue. However, in order to apply APH to a wider range of skin diseases and regeneration, further investigation is needed to elucidate the clear intracellular mechanism of action and long- term effects of APH.
In conclusion, the most important point of this study is that APH is a material that improves oxidation stability and skin penetration while maintaining the advantages of AA. In addition, APH has the advantage of selectively releasing AA by esterase, which is abundantly present in the skin, after penetrating the epidermal layer. This is a new strategy that can maximize the benefits of each substance: the antioxidant, anti-inflammatory, and anti- aging effects of AA, and the moisturizing, elasticity maintenance, wrinkle prevention, and skin aging relief effects of HA. Therefore, the newly developed APH can be utilized as an ideal cosmetic and therapeutic ingredient because it can suppress or delay skin damage caused by environmental stress factors of the skin.
Acknowledgements
This work was supported by the Korea Institute of Marine Scinece & Technology Promotion (KIMST) funded by the Ministry of Oceans and Fisheries (RS-2025-02303933) and by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (RS-2023-KH135936).
Data Availability Statement
The data that support the findings of this study are accessible from the corresponding author upon reasonable request.
Conflict of Interest Statement
Ji Won Yu, Hayoung Choi, and Jin-Su Kim are employees of CELLINBIO. Wonchoul Park is the Chief Scientific Officer of CAPABIOSCIENCE. This does not alter the authors' adherence to the journal's policy on data and material sharing. Gwang Hoon Kim has no conflicts of interest to declare.
Ethical Statement
No animal experiments was carried out in this article.
Transparency Statements
The lead author, Jin-Su Kim, Gwang Hoon Kim that this manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned (and, if relevant, registered) have been explained.
References
- Sparr, E., Millecamps, D., Isoir, M., Burnier, V., Larsson, Å., & Cabane, B. (2013). Controlling the hydration of the skinthough the application of occluding barrier creams. Journal ofThe Royal Society Interface, 10(80), 20120788.
- Nguyen, A. V., & Soulika, A. M. (2019). The dynamics of the skin’s immune system. International journal of molecular sciences, 20(8), 1811.
- Martic, I., Jansen-Dürr, P., & Cavinato, M. (2022). Effects of air pollution on cellular senescence and skin aging. Cells, 11(14), 2220.
- Kim, B. E., Kim, J., Goleva, E., Berdyshev, E., Lee, J., Vang,K. A., ... & Ahn, K. (2021). Particulate matter causes skinbarrier dysfunction. JCI insight, 6(5), e145185.
- Parrado, C., Mercado-Saenz, S., Perez-Davo, A., Gilaberte, Y., Gonzalez, S., & Juarranz, A. (2019). Environmental stressors on skin aging. Mechanistic insights. Frontiers in pharmacology, 10, 759.
- Green, M., Kashetsky, N., Feschuk, A., & Maibach, H. I. (2022). Transepidermal water loss (TEWL): Environment and pollution—A systematic review. Skin health and disease, 2(2), ski2-104.
- K. (2025). Antioxidants in Skin Disorders. Antioxidants: Nature's Defense Against Disease, 551-572 Kumar, S., Subba,D. P., Seema, Firdous, S. M., Odeku, O. A., Kumar, S., &Sindhu, R..
- Pillai, S., Oresajo, C., & Hayward, J. (2005). Ultraviolet radiation and skin aging: roles of reactive oxygen species, inflammation and protease activation, and strategies for prevention of inflammationâ?ÂÂinduced matrix degradation–a review. International journal of cosmetic science, 27(1), 17- 34.
- Ansary, T. M., Hossain, M. R., Kamiya, K., Komine, M., & Ohtsuki, M. (2021). Inflammatory molecules associated with ultraviolet radiation-mediated skin aging. International Journal of Molecular Sciences, 22(8), 3974.
- Pittayapruek, P., Meephansan, J., Prapapan, O., Komine, M., & Ohtsuki, M. (2016). Role of matrix metalloproteinases in photoaging and photocarcinogenesis. International journal of molecular sciences, 17(6), 868
- Nihro, Y., Miyataka, H., Sudo, T., Matsumoto, H., & Satoh,T. (1991). 3-O-alkylascorbic acids as free-radical quenchers: synthesis and inhibitory effect on lipid peroxidation. Journal of medicinal chemistry, 34(7), 2152-2157..
- Colven, R. M., & Pinnell, S. R. (1996). Topical vitamin C inaging. Clinics in dermatology, 14(2), 227-234.
- 13. Darr, D. O. U. G. L. A. S., Dunston, S. T. A. N. L. E.Y., Faust, H. O. L. L. Y., & Pinnell, S. H. E. L. D. O. N.(1996). Effectiveness of antioxidants (vitamin C and E) with and without sunscreens as topical photoprotectants. Acta Dermato-Venereologica, 76(4), 264-268
- Al-Niaimi, F., & Chiang, N. Y. Z. (2017). Topical vitamin C and the skin: mechanisms of action and clinical applications. The Journal of clinical and aesthetic dermatology, 10(7), 14..
- Tikekar, R. V., Anantheswaran, R. C., & LaBorde, L. F. (2011). Ascorbic acid degradation in a model apple juice system and in apple juice during ultraviolet processing and storage. Journal of food science, 76(2), H62-H71..
- Yin, X., Chen, K., Cheng, H., Chen, X., Feng, S., Song,Y., & Liang, L. (2022). Chemical stability of ascorbic acid integrated into commercial products: A review on bioactivity and delivery technology. Antioxidants, 11(1), 153.
- Yamamoto, I., Tai, A., Fujinami, Y., Sasaki, K., & Okazaki,S. (2002). Synthesis and characterization of a series of novel monoacylated ascorbic acid derivatives, 6-O-acyl-2-O-α- D-glucopyranosyl-L-ascorbic acids, as skin antioxidants. Journal of medicinal chemistry, 45(2), 462-468.
- Gallarate, M., Carlotti, M. E., Trotta, M., & Bovo, S. (1999). On the stability of ascorbic acid in emulsified systems for topical and cosmetic use. International Journal of Pharmaceutics, 188(2), 233-241.
- Austria, R., Semenzato, A., & Bettero, A. (1997). Stability of vitamin C derivatives in solution and topical formulations. Journal of Pharmaceutical and Biomedical Analysis, 15(6), 795-801.
- Špiclin, P., Gašperlin, M., & Kmetec, V. (2001). Stability of ascorbyl palmitate in topical microemulsions. International journal of pharmaceutics, 222(2), 271-279.
- Stamford, N. P. (2012). Stability, transdermal penetration, and cutaneous effects of ascorbic acid and its derivatives. Journal of cosmetic dermatology, 11(4), 310-317.
- Iliopoulos, F., Sil, B. C., Moore, D. J., Lucas, R. A., & Lane,M. E. (2019). 3-O-ethyl-l-ascorbic acid: Characterisation and investigation of single solvent systems for delivery to the skin. International Journal of Pharmaceutics: X, 1, 100025.
- Kameyama, K., Sakai, C., Kondoh, S., Yonemoto, K., Nishiyama, S., Tagawa, M., ... & Blanock, K. (1996). Inhibitory effect of magnesium L-ascorbyl-2-phosphate (VC- PMG) on melanogenesis in vitro and in vivo. Journal of the American Academy of Dermatology, 34(1), 29-33.
- Liu, Y., Liu, C., & Li, J. (2020). Comparison of vitamin C and its derivative antioxidant activity: evaluated by using density functional theory. ACS omega, 5(39), 25467-25475.
- Ravetti, S., Clemente, C., Brignone, S., Hergert, L., Allemandi, D., & Palma, S. (2019). Ascorbic acid in skin health. Cosmetics, 6(4), 58.
- Volpi, N., Schiller, J., Stern, R., & Soltes, L. (2009). Role, metabolism, chemical modifications and applications of hyaluronan. Current medicinal chemistry, 16(14), 1718-1745.
- Purcell, B. P., Kim, I. L., Chuo, V., Guenin, T., Dorsey, S. M., & Burdick, J. A. (2014). Incorporation of sulfated hyaluronic acid macromers into degradable hydrogel scaffolds for sustained molecule delivery. Biomaterials science, 2(5), 693- 702.
- Fraser, J. R. E., Laurent, T. C., & Laurent, U. B. G. (1997). Hyaluronan: its nature, distribution, functions and turnover. Journal of internal medicine, 242(1), 27-33.
- 29. Yang, J. A., Kim, E. S., Kwon, J. H., Kim, H., Shin, J.H., Yun, S. H., ... & Hahn, S. K. (2012). Transdermal delivery of hyaluronic acid–human growth hormone conjugate. Biomaterials, 33(25), 5947-5954.
- Guan, S., Zhang, K., Cui, L., Liang, J., Li, J., & Guan,F. (2022). Injectable gelatin/oxidized dextran hydrogel loaded with apocynin for skin tissue regeneration. Biomaterials Advances, 133, 112604..
- Chen, Y. C., Su, W. Y., Yang, S. H., Gefen, A., & Lin, F. H.(2013). In situ forming hydrogels composed of oxidized high molecular weight hyaluronic acid and gelatin for nucleus pulposus regeneration. Acta biomaterialia, 9(2), 5181-5193.
- Huang, G., & Huang, H. (2018). Application of hyaluronic acid as carriers in drug delivery. Drug delivery, 25(1), 766- 772.
- But, T. Y. S., & Toy, P. H. (2007). The Mitsunobu reaction: origin, mechanism, improvements, and applications. Chemistry–An Asian Journal, 2(11), 1340-1355.
- Pulsoni, I., Lubda, M., Aiello, M., Fedi, A., Marzagalli, M., von Hagen, J., & Scaglione, S. (2022). Comparison between Franz diffusion cell and a novel micro-physiological system for in vitro penetration assay using different skin models. SLAS technology, 27(3), 161-171.
- Neupane, R., Boddu, S. H., Renukuntla, J., Babu, R. J., & Tiwari, A. K. (2020). Alternatives to biological skin in permeation studies: Current trends and possibilities. Pharmaceutics, 12(2), 152.
- Arce, F. J., Asano, N., See, G. L., Itakura, S., Todo, H., & Sugibayashi, K. (2020). Usefulness of artificial membrane, Strat-M®, in the assessment of drug permeation from complex vehicles in finite dose conditions. Pharmaceutics, 12(2), 173.
- Vayachuta, L., Leang, M., Ruamcharoen, J., Thiramanas, R., Prateepchinda, S., Prompinit, P., ... & Waranuch, N. (2023). Printable-Microencapsulated Ascorbic Acid for Personalized Topical Delivery. ACS Applied Bio Materials, 6(12), 5385- 5398.
- Peltonen, S., Riehokainen, J., Pummi, K., & Peltonen, J. (2007). Tight junction components occludin, ZO-1, and claudin-1,-4 and-5 in active and healing psoriasis. British Journal of Dermatology, 156(3), 466-472.
- Baumann, L. (2007). Skin ageing and its treatment. The Journal of Pathology: A Journal of the Pathological Society of Great Britain and Ireland, 211(2), 241-251.
- Langton, A. K., Sherratt, M. J., Griffiths, C. E. M., & Watson,R. E. B. (2010). A new wrinkle on old skin: the role of elastic fibres in skin ageing. International journal of cosmetic science, 32(5), 330-339.
- Ralf Paus, L., Berneburg, M., Trelles, M., Friguet, B., Ogden, S., Esrefoglu, M., ... & Thappa, D. M. (2008). How best to halt and/or revert UVâ?ÂÂinduced skin ageing: strategies, facts and fiction. Experimental dermatology, 17(3), 228-229.
- Yaar, M., Eller, M. S., & Gilchrest, B. A. (2002, December). Fifty years of skin aging. In Journal of Investigative DermatologySymposium Proceedings (Vol. 7, No. 1, pp. 51-58). Elsevier.
- Gallagher, R. P. (2005). Sunscreens in melanoma and skin cancer prevention. Cmaj, 173(3), 244-245.
- Mona, A., & Amal K, K. (2004). Ultraviolet carcinogenesis in nonmelanoma skin cancer. Part I: incidence rates in relation to geographic locations and in migrant populations. SKINmed: Dermatology for the Clinician, 3(1), 29-35.
- Crisan, D., Roman, I., Crisan, M., Scharffetter-Kochanek, K., & Badea, R. (2015). The role of vitamin C in pushing back the boundaries of skin aging: an ultrasonographic approach.Clinical, cosmetic and investigational dermatology, 463-470.
- Pullar, J. M., Carr, A. C., & Vissers, M. C. (2017). The roles of vitamin C in skin health. Nutrients, 9(8), 866.
- Wang, K., Jiang, H., Li, W., Qiang, M., Dong, T., & Li, H. (2018). Role of vitamin C in skin diseases. Frontiers in physiology, 9, 378515.
- Carr, A. C., & Maggini, S. (2017). Vitamin C and immune function. Nutrients, 9(11), 1211.
- Cisternas, P., Silvaâ?ÂÂAlvarez, C., Martínez, F., Fernandez, E., Ferrada, L., Oyarce, K., ... & Nualart, F. (2014). The oxidized form of vitamin C, dehydroascorbic acid, regulates neuronal energy metabolism. Journal of neurochemistry, 129(4), 663- 671.
- Ferrada, L., Magdalena, R., Barahona, M. J., Ramírez, E., Sanzana, C., Gutiérrez, J., & Nualart, F. (2021). Two distinct faces of vitamin C: AA vs. DHA. Antioxidants, 10(2), 215.
- Segall, A. I., & Moyano, M. A. (2008). Stability of vitamin C derivatives in topical formulations containing lipoic acid, vitamins A and E. International journal of cosmetic science, 30(6), 453-458.
- Telang, P. S. (2013). Vitamin C in dermatology. Indian dermatology online journal, 4(2), 143-146.
- Enescu, C. D., Bedford, L. M., Potts, G., & Fahs, F. (2022). A review of topical vitamin C derivatives and their efficacy. Journal of Cosmetic Dermatology, 21(6), 2349-2359.
- Bae, H. K., Lee, S. B., Park, C. S., Shim, J. H., Lee, H. Y., Kim, M. J., ... & Park, K. H. (2002). Modification of ascorbic acid using transglycosylation activity of Bacillus stearothermophilus maltogenic amylase to enhance its oxidative stability. Journal of agricultural and food chemistry, 50(11), 3309-3316.
- Chakraborty, A., & Jana, N. R. (2017). Vitamin C-conjugated nanoparticle protects cells from oxidative stress at low doses but induces oxidative stress and cell death at high doses. ACS applied materials & interfaces, 9(48), 41807-41817.
- Dang, X., Yang, M., Shan, Z., Mansouri, S., May, B. K., Chen, X., ... & Woo, M. W. (2017). On spray drying of oxidized corn starch cross-linked gelatin microcapsules for drug release. Materials Science and Engineering: C, 74, 493-500.
- Uddin, M. S., Hawlader, M. N. A., & Zhu, H. J. (2001). Microencapsulation of ascorbic acid: effect of process variables on product characteristics. Journal of Microencapsulation, 18(2), 199-209.
- Phillips, C. L., Combs, S. B., & Pinnell, S. R. (1994). Effects of ascorbic acid on proliferation and collagen synthesis in relation to the donor age of human dermal fibroblasts. Journal of Investigative Dermatology, 103(2), 228-232.
- Hata, R. I., & Senoo, H. (1989). L-ascorbic acid 2-phosphate stimulates collagen accumulation, cell proliferation, and formation of a three-dimensional tissuelike substance by skin fibroblasts. Journal of cellular physiology, 138(1), 8-16.
- Schmidt, R. J., Chung, L. Y., Andrews, A. M., & Turner, T.D. (1993). Toxicity of L-ascorbic acid to L929 fibroblast cultures: Relevance to biocompatibility testing of materials for use in wound management. Journal of biomedical materials research, 27(4), 521-530.
- Neves, J. R., Grether-Beck, S., Krutmann, J., Correia, P., Gonçalves Jr, J. E., Sant’Anna, B., & Kerob, D. (2022). Efficacy of a topical serum containing L-ascorbic acid, neohesperidin, pycnogenol, tocopherol, and hyaluronic acid in relation to skin aging signs. Journal of Cosmetic Dermatology, 21(10), 4462-4469.
- Fitzpatrick, R. E., & Rostan, E. F. (2002). Double-blind,half-face study comparing topical vitamin C and vehicle for rejuvenation of photodamage. Dermatologic surgery, 28(3), 231-236.
- Woolery-Lloyd, H., Baumann, L., & Ikeno, H. (2010). Sodium L-ascorbyl-2-phosphate 5% lotion for the treatment of acne vulgaris: a randomized, double-blind, controlled trial. Journal of cosmetic dermatology, 9(1), 22-27.


