Research Article - (2018) Volume 1, Issue 1
Expression of E-Cadherin in Epidermis from Wounds Treated with Costus afer Stem Juice Extract
2Community Health Officers Training School, University of Uyo Teaching Hospital, Uyo, Nigeria
Received Date: Nov 15, 2018 / Accepted Date: Nov 21, 2018 / Published Date: Dec 05, 2018
Copyright: ©Copyright: ©2018 Kudighe Patrick Udoh. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
This study investigated the expression of e-cadherin in the epidermis of re-epithelialized wound treated with Costus afer stem juice extract. Twenty-five adult female albino rats were used for the study. They were divided equally into 5 treatment groups of: A – no treatment; B – iodine treatment only on the first day; C – iodine treatment for 7 days; D – Costus afer stem juice extract only on the first day; and E – Costus afer stem juice extract for 7 days. The rats were aseptically wounded on their dorsum under ketamine hydrochloride anaesthesia. They were treated according to their groups and closely monitored till the wounds healed. On healing, the rats were euthanized and samples of the healed skin were collected and processed for immunohistochemical staining using antibodies for e-cadherin. It was found that group D showed highest intensity of e-cadherin expression among the treatment groups, suggesting the influence of Costus afer stem juice extract on the process of mesenchymal-epithelial transition.
Keywords
Wound Healing, Costus Afer, E-Cadherin
Introduction
E-cadherin or cadherin-1 is a classical member of the cadherin superfamily expressed in epithelial tissues. It is a calcium-dependent cell-cell adhesion glycoprotein composed of five extracellular cadherin repeats, a transmembrane region and a cytoplasmic tail [1]. It is a major component of adherens junctions which link neighbouring cells through their actin cytoskeleton. The extracellular domain of e-cadherin binds to Ca2+ and establishes interactions with cadherin from the same cell in the case of cis-interaction or neighbouring cell in the case of trans-interaction [2-4]. Its cytoplasmic domain binds to p120-catenin and β-catenin [5-7].
During the wound healing process, e-cadherin expression is downregulated at the basal layer of the epithelium bordering the edge of the wound. This is to allow cell proliferation and cell migration, characteristics of epithelial-mesenchymal transition (EMT). Initially, it was thought that the loss of e-cadherin expression initiated EMT, but recent studies have shown that it is a downstream mechanism of EMT initiation cascade as the processes had already been initiated in the cytoplasm of the cells before the loss of e-cadherin at the cell membrane is observed. Growth factors such as epithelial growth factor (EGF), Snai1, transforming growth factor-beta 1 (TGF-β1) have been implicated in e-cadherin down-regulation during wound healing [8,9].
However, e-cadherin expression is not completely turned off during the wound healing process. It is required for collective cell migration, which is essential for proper re-epithelialisation [9-12]. E-cadherin has been reported to mechanically coordinate and direct the migration of large epithelial sheets. In the absence of e-cadherin, the migrating cells are scattered over the surface of the wound without proper alignment. When the cells at the migratory tongue of the keratinocytes which lack e-cadherin get in contact with other migrating cells, the cells undergo mesenchymal-epithelial transition (MET), regain their e-cadherin expression and begin to proliferate to re-establish the stratified nature of the epidermis [13-15].
The epidermis of scars and keloids have been reported to have low expression of e-cadherin compared to normal epidermis and the scar tissues retain many of the characteristics of mesenchymal tissue such as the expression of n-cadherin. In addition, e-cadherin expression in healing tissue has been found to be inversely related to fibrosis [16-20].
Costus afer has been used traditionally for the treatment of several ailments, including malaria, cough and tachycardia. Its stem has been found to be antimicrobial, antinociceptive, and antioxidative [21-24]. Studies by Udoh and colleagues showed that the stem juice extract of Costus afer promote wound healing and drive the healing process towards regeneration of lost tissue [25]. It has also been reported to significantly increase the rate of re-epithelialization and enhance the outcome of the healing process [26]. This study was therefore carried out to investigate the expression of e-cadherin in the healed epidermis of wounds treated with Costus afer stem juice extract.
Methodology
Experimental Animals
The albino rats for this study were obtained from the Animal House of the Department of Pharmacology and Toxicology, Faculty of Pharmacy, University of Uyo. The ethical approval for the study was duly obtained from the Ethical Committee of the Faculty of Basic Medical Sciences, University of Uyo, Uyo, Nigeria. A total of twenty-five (25) adult female albino rats weighing between 243- 320g, were used for the study [27]. Before commencement of the study, all apparatus was obtained, assessed for functionality and put in place within 2 weeks to the study. Three days before the commencement of the experiment, the room and the apparatus were soaked in concentrated antiseptic solution and left overnight for asepsis. The next morning, they were washed and cleaned with soapy water and the rats were brought in 3 days after, when the concentration of antiseptic in the atmosphere of the room had dissipated. The rats were housed individually in rubber cages between 22 – 26°C temperature, humidity range of 50-60%, and 12- hour light/dark lighting. They were fed with standard rat pelletized diet, (Vital Feed Growers, Green Cereals Nigeria Ltd.) and water ad libitum.
Plant Collection and Preparation of Extract
Identification and registration of Costus afer were done with the Herbarium of the Department of Pharmacognosy, Faculty of Pharmacy, University of Uyo with the herbarium number UUPH26(a) under the family Costacae as identified by the taxonomist. After the identification and confirmation, fresh stem of Costus afer were collected from bushes within Uyo local government area in February, 2017. The foliage leaves were removed and the stems were washed and cut into pieces and crushed in an electric blender (NAKAI-Japan; HR-2818). The juice obtained from the blender was filtered with a chess cloth. Aqueous crude extract of the filtrate was obtained by evaporation in beakers placed in water bath at 40°C [28]. The extract was made into a semiliquid paste of 4g/ml concentration with water [25].
Experimental Protocol
The albino rats were divided equally into 5 groups of A, B, C, D and E for each treatment procedure (Table 3.1). After one (1) week of acclimatization, all the rats were wounded on their dorsum and treated according to group specifications.
Table 3.1: Experimental Design
|
Group |
Number |
Treatment |
|
A |
5 |
Control |
|
B |
5 |
Iodine ointment for 1 day |
|
C |
5 |
Iodine ointment for 7 days |
|
D |
5 |
Costus afer paste for 1 day |
|
E |
5 |
Costus afer paste for 7 days |
The health status of the rats were monitored closely by regular assessment of their weight and stress indicators according to National Centre for the Replacement, Refinement and Reduction of Animals in Research, as well as internationally accepted standard guide for care and use of laboratory animals promulgated and adopted by the National Institute of Health (NIH) publication number 85, revised 1996 and related ethics and regulation [23,29]. All animals were handled humanely with care. Throughout the experiment, all procedures were carried out with high degree of asepsis and no infection was observed in the wounds.
Wound Creation
All surgical interventions were carried out under general anaesthesia with ketamine hydrochloride (120mg/kg b.w. i.p.). A predetermined area of fur, 5cm from the ear centrally, was shaved off the rat dorsum using clipper (model number GM-3005). The shaved area was cleaned using an antiseptic soap solution and methylated spirit. Within the shaved area, another 1.5x1.5cm2 was marked off as the wounding site using methylene blue. The target area was excised with surgical blade, scissors and forceps to create a full thickness square wound. Immediately after wounding, sterile gauze was used to apply pressure on the wound until haemostasis was attained. Thereafter, the wounded rat was treated according to group.
Application of Substances
The extract was made into a semiliquid paste of 4g/ml concentration with distilled water. Povidone iodine ointment (Wosan Iodine, Jawa International Nigeria Limited) with 5%w/w concentration was used for the study. The ointment was obtained commercially from pharmaceutical stores. Wounds of group A rats were left untreated, group B were treated with 0.2ml of iodine ointment only on the first day, group C were treated with 0.2ml of iodine ointment daily for 7days, group D were treated with 0.2ml of Costus afer paste only on the first day, and group E were treated with 0.2ml of Costus afer paste daily for 7days.
Immunohistochemical Study
The tissues obtained from the wound site were processed for immunohistochemical assessment of e-cadherin expression. The method used was the Avidin Biotin Complex (ABC) method also referred to as the Avidin Biotin Imuunoperoxidase method with formalin fixed and paraffin embedded tissues. The e-cadherin antibodies used for the work were manufactured by Ventana Medical Systems, Inc., USA with ref number 790-4497 and lot number G01366. The processed tissues were sectioned at 2µ on the rotary microtome and placed on the hot plate at 70°C for at least 1hour. Sections were brought down to water by passing them on 2 changes of xylene, then 3 changes of descending grades of alcohol and finally to water. Antigen retrieval was performed on the sections by heating them on a citric acid solution of pH 6.0 using the microwave at power 100 for 15minutes. The sections were equilibrated gradually with cool water to displace the hot citric acid for at least 5min for the sections to cool. Peroxidase blocking were done on the sections by simply covering sections with 3% hydrogen peroxide (H2O2) for 15min. Sections were washed with PBS and protein blocking were performed using avidin for 15min. Sections were again washed with phosphate buffered saline (PBS) and endogenous biotin in tissue were blocked using biotin for 15min. After washing with PBS sections were incubated with diluted e-cadherin antibody of 1:100 dilution factor for 60 min. Excess antibodirs were washed off with PBS and a secondary antibody (LINK) was applied on sections for 15min. Sections were washed and horsradish peroxidase (HRP) label was applied on the sections for 15min. A working 3,3’-diaminobenzidine (DAB) solution was made up by mixing 1 drop (20microns) of the DAB chromogen to 1ml of the DAB substrate. This working solution was applied on sections after washing off the HRP with PBS for 5min. The brown reactions began to appear at this moment especially for a positive target. Excess DAB solution and precipitate were washed off with water. Sections were counterstained with Haematoxylin solution for 2min and blued briefly. Sections were dehydrated in alcohol, cleared in xylene and mounted in DPX.
Result
E-cadherin expression was found to be higher in the Costus afer treated groups D and E than in the control and iodine treated groups B and C (Figure 1). Group D (single dose treatment with Costus afer) had the highest expression of e-cadherin.
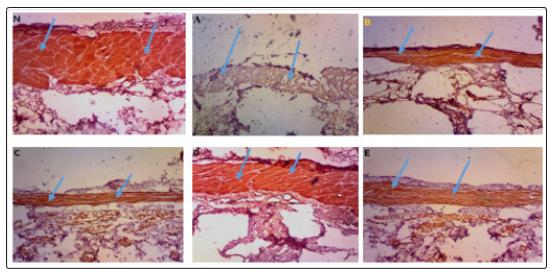
Figure 1: E-cadherin expression in normal epidermis and healed epidermis (arrows) (x40).
N: E-cadherin expression in normal uninjured epidermis.
A: E-cadherin expression in epidermal tissue of control, group A of which wounds were left untreated.
B: E-cadherin expression in epidermal tissue of group B treated only on the first day with iodine ointment.
C: E-cadherin expression in epidermal tissue of group C treated for 7 days with iodine ointment.
D: E-cadherin expression in epidermal tissue of group D treated only on the first day with aqueous extract of Costus afer stem.
E: E-cadherin expression in epidermal tissue (arrows) of group E treated for 7 days with aqueous extract of Costus afer stem.
Discussion
The stem of Costus afer is used traditionally for the treatment of wounds. Its sap has been reported to be rubefacient, to have burning sensation on open wounds with anodyne and healing properties [24,30]. It is also used traditionally to treat a range of skin ailments, veneral diseases and colic [22,31]. Scientific studies have reported that the stem extract of Costus afer has significant antibacterial, antifungal and amoebicidal activity in vitro. Its antinociceptive and antioxidant properties have also been reported [21-24,32,33]. Phytochemical analysis has shown that it contains significant wound healing biomolecules including alkaloids, flavonoids, quinones, saponins, glycosides and triterpenes [25,26,33,38]. Each of these molecules have been scientifically proven to promote wound healing through their effect on different phases of wound healing and wound healing cells [25,26]. Several minerals and ions that facilitate the healing process have also been identified in the stem of Costus afer. These minerals include copper, potassium, calcium, sodium, magnesium, manganese and phosphorus [25,26,34,38].
The presence of these biomolecules and minerals makes Costus afer stem a potential wound healing agent and supports its traditional use in wound treatment. Furthermore, studies by Udoh and colleagues showed that treatment of wounds with Costus afer stem juice extract maintained a moist healing environment and showed a considerable improvement in the healing process and outcome [25]. High rate of re-epithelialization and random organisation of dermal fibers with high tensile strength has also been reported in full thickness excisional wound model treated with Costus afer stem juice extract. Regeneration of hair follicles has also been reported, suggesting the regenerative potential of Costus afer stem [25,26].
This study showed that Costus afer stem juice treatment enhances the process of mesenchymal-epithelial transition as shown in the high intensity of e-cadherin expression in the epidermis of the healed skin. This is based on the reports that migratory keratinocytes which lose e-cadherin to become mesenchymal cells undergo contact inhibition and regain their e-cadherin when they come in contact with opposite migratory cells [13,15]. Furthermore, the present findings support the report of Ekanem and colleagues that Costus afer promotes wound healing through its effect on different activities of the healing process [26]. These findings can be related to its effect on the re-epithelialization process as it has been reported that Costus afer stem juice extract increases the rate of re-epithelialization, and E cadherin is known to contribute significantly to collective cell migration and the stability of epithelial sheet by holding the cells together [3,4,13]. This may indicate the potential of Costus afer stem juice in the effective treatment of wounds in keloid prone skin as well as the treatment of keloids, since proper mesenchymal-epithelial transition has been a major drawback in this condition [16-18].
Conclusion
The process of mesenchymal-epithelial transition is a major event that re-establishes the epidermis of an injured skin. Impairment of this process has been implicated in keloid formation. The results from this study have shown that Costus afer stem juice extract could be effectively used to promote this process through its enhancement of e-cadherin expression in the injured skin. However, more studies are recommended to ascertain its effect on this process as other factors may be implicated.
References
- Li L, Wang B, Steffany AL, Wang L (2012) Role of E-cadherin and other cell adhesion molecules in survival and differentiation of human pluripotent stem cells. Cell Adhesion and Migration 6: 59-70.
- Baumgartner W, Hinterdorfer P, Ness W, Raab A, Vestweber D, et al. (2000) Cadherin interaction probed by atomic force microscopy. Proceedings of the National Academy of Sciences of the United States of America 97: 4005-4010.
- Harrison OJ, Jin X, Hong S, Bahna F, Ahlsen G, et al. (2011) The extracellular architecture of adherens junctions revealed by crystal structures of type I cadherins. Structure (London, England : 1993) 19: 244-256.
- Marie PJ, Haÿ E, Modrowski D, Revollo L, Mbalaviele G, et al. (2014) Cadherin-Mediated Cell–Cell Adhesion and Signaling in the Skeleton. Calcified Tissue International 94: 46-54.
- Meng W, Takeichi M (2009) Adherens junction: molecular architecture and regulation. Cold Spring Harbor perspectives in biology 1: a002899.
- Harris TJC, Tepass U (2010) Adherens junctions: from molecules to morphogenesis. Nature reviews Molecular cell biology 11: 502-514.
- Wu Y, Kanchanawong P, Zaidel-Bar R (2015) Actin-delimited adhesion-independent clustering of E-cadherin forms the nanoscale building blocks of adherens junctions. Developmental cell 32: 139-154.
- Chavez MG, Buhr CA, Petrie WK, Wandinger-Ness A, Kusewitt DF, et al. (2012) Differential downregulation of E-Cadherin and desmoglein by epidermal growth factor. Dermatology Research and Practice 2012: 309587.
- Maeda M, Johnson KR, Wheelock MJ (2005) Cadherin switching: essential for behavioral but not morphological changes during an epithelium-to-mesenchyme transition. Journal of cell science 118: 873-887.
- Etienne-Manneville S (2011) Control of polarized cell morphology and motility by adherens junctions. Seminars in Cell and Developmental Biology 22: 850-857
- Hunter MV, Lee DM, Harris TJC, Fernandez-Gonzalez R (2015) Polarized E-cadherin endocytosis directs actomyosin remodeling during embryonic wound repair. Journal of Cell Biology 210: 801-816.
- Kardash E, Reichman-Fried M, Maître JL, Boldajipour B, Papusheva E, et al. (2010) A role for Rho GTPases and cell-cell adhesion in single-cell motility in vivo. Nature Cell Biology 12: 47-53.
- Hwang S, Zimmerman NP, Agle KA, Turner JR, Kumar SN, et al. (2012) E-Cadherin is Critical for Collective Sheet Migration and is Regulated by the Chemokine CXCL12 Protein During Restitution. Journal of Biological Chemistry 287: 22227-22240.
- Jiang J, Li L, He Y, Zhao M (2013) Collective cell migration: Implications for wound healing and cancer invasion. Burns and Trauma 1: 21.
- Kuwahara M, Hatoko M, Tada H, Tanaka a (2001) E-cadherin expression in wound healing of mouse skin. Journal of Cutaneous Pathology 28: 191-199.
- Agarwal SK (2014) Integrins and cadherins as therapeutic targets in fibrosis. Frontiers in Pharmacology 5: 1-7.
- Hahn JM, McFarland KL, Combs KA, Supp DM (2016) Partial Epithelial-Mesenchymal Transition in Keloid Scars: Regulation of Keloid Keratinocyte Gene Expression by Transforming Growth Factor-β1. Burns and Trauma 4: 30.
- Liu Y, Li Y, Li N, Teng W, Wang M, et al. (2016) TGF-β1 promotes scar fibroblasts proliferation and transdifferentiation via up-regulating MicroRNA-21. Scientific Reports 6: 32231.
- Nural-Guvener HF, Zakharova L, Nimlos J, Popovic S, Mastroeni D, et al. (2014) HDAC class I inhibitor, Mocetinostat, reverses cardiac fibrosis in heart failure and diminishes CD90+ cardiac myofibroblast activation. Fibrogenesis and Tissue Repair 7: 10.
- Yan C, Grimm WA, Garner WL, Qin L, Travis T, et al. (2010) Epithelial to mesenchymal transition in human skin wound healing is induced by tumor necrosis factor-alpha through bone morphogenic protein-2. The American Journal of Pathology 176: 2247-2258
- Uchegbu RI, Akalazu JN, Ibe CO, Ahuchaogu AA, Amadikwa CU (2016) Chemical Composition of the Stem Extract of Costus afer ( Bush Cane ) and Its Antimicrobial Activity. British Journal of Pharmaceutical Research 10: 1-9.
- Aweke G (2007) Costus afer Ker Gawl [Internet]. Schmelzer GH, GuribFakim A, editors. Wageningen, Netherlands: PROTA (Plant Resources of Tropical Africa / Ressources végétales de l’Afrique tropicale) 4-7.
- Ejiogu MC, Ezeibekwe IO, Egbucha KC (2016) Antifungal activity of the plant Costus afer extract on yam (dioscorea species) rot pathogen in owerri. south-east nigeria. Journal of Botany and Research 1: 1-6.
- Fern K (2014) Costus afer - useful tropical plant [Internet].Usefule Tropical Plant Database 6-8.
- Udoh KP, Ekanem AU, Peter AI (2018) Investigating the Wound Healing Potential of Costus afer Ker Gawl Stem Juice on Cutaneous Wounds of Albino Rats. Cell, Stemm cells and Regenerative Medicine 3: 1-8.
- Ekanem AU, Udoh KP, Peter AI (2018) Morphometric and Histological Studies of Effects of Aqueous Extract of Costus afer Stem Juice on Cutaneous Wound of Albino Rats. International Journal of Advanced Research 6: 1011-1022.
- Dorsett-Martin WA (2004) Rat models of skin wound healing :A review. 2004; Wound Repair and Regeneration 12: 591-599.
- Akpantah AO, Akpan U, Eluwa MA, Isamoh TE, Udonkang M, et al. (2016) Blood Parameters and Spleen Histology Following Chronic Consumption of Ethanolic Extract of Costus afer Stem and Juice on Albino Wistar Rats. British Journal of Medicine and Medical Research 13: 1-10.
- Sotocina SG, Sorge RE, Zaloum A, Tuttle AH, Martin LJ, et al. (2011) The Rat Grimace Scale: A Partially Automated Method for Quantifying Pain in the Laboratory Rat via Facial Expressions. Molecular Pain 7: 1744-8069.
- Burkill HM (1985) Costus afer Ker–Gawl. [family Costacae] [Internet]. JSTOR Global plants. JSTOR; 1985.
- Omokhua GE (2011) Medicinal and Socio-Cultural Importance of Costus Afer (Ker Grawl) in Nigeria. African Research Review 5: 282-287.
- Ijioma SN, Nwosu CO, Emelike CU, Okafor AI, Nwankwo AA (2014) Antinociceptive Property of Costus afer Ker Stem Juice and Ethanol Leaf Extract in Albino Rats. Comprehensive Journal of Medical Sciences 2: 14-19.
- Tchamgoue AD, Tchokouaha LRY, Tarkang PA, Kuiate J, Agbor GA (2015) Costus afer Possesses Carbohydrate Hydrolyzing Enzymes Inhibitory Activity and Antioxidant Capacity In Vitro. Evidence-based complementary and alternative medicine : eCAM 2015: 987984.
- Anyasor GN, Onajobi FD, Osilesi O, Adebawo O (2014) Proximate composition, mineral content and in vitro antioxidant activity of leaf and stem of Costus afer (Ginger lily). Journal of Intercultural Ethnopharmacology 3: 128-134.
- Anyasor GN, Ogunwenmo KO, Oyelana OA, Akpofunure BE (2010) Phytochemical constituents and antioxidant activities of aqueous and methanol stem extracts of Costus afer Ker Gawl. (Costaceae). African Journal of Biotechnology 9: 4880-4884.
- Ukpabi CF, Agbafor KN, Ndukwe OK, Akuagwu A, Nwachukwu SN (2012) Phytochemical Composition of Costus Afer Extract and Its Alleviation of Carbon Tetrachloride – induced Hepatic Oxidative Stress and Toxicity. International Journal of Modern Botany 2: 120-126.
- Akaninwor JO, Essien EB, Tonkiri A, Uvoh SM (2014) Phytoconstituents of Costus afer Methanolic Stem Extract and Its in vitro Radical Scavenging Activities. Journal of Agriculture and Biodiversity Research 3: 99-110.
- Akpabio UD, Udo UE, Akpakpan AE (2012) Evaluation of phytochemical, proximate and mineral element composition of stem of Costus afer (Bush cane). Asian Journal of Plant Science and Research 2: 607-612.


