Research Article - (2025) Volume 8, Issue 1
Evaluation of Residual Total Petroleum Hydrocarbon (Tph) in Rhizosphere of Cowpea (Vigna Unguiculata) and Non-Rhizosphere Soil Contaminated with Crude Oil Under Different Organic Manure Treatments
2Department of Geography and Natural Resource management, University of Uyo, Nigeria
Received Date: Feb 07, 2025 / Accepted Date: Mar 18, 2025 / Published Date: Mar 25, 2025
Copyright: ©2025 Tombari Bodo, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Ordu, J. O., Bodo, T. (2025). Evaluation of Residual Total Petroleum Hydrocarbon (Tph) in Rhizosphere of Cowpea (Vigna Unguiculata) and Non-Rhizosphere Soil Contaminated with Crude Oil Under Different Organic Manure Treatments. Earth Envi Scie Res & Rev, 8(1), 01-08.
Abstract
This study was undertaken to evaluate residual Total petroleum hydrocarbon (TPH) in rhizosphere of Vigna unguiculata (cowpea) and non-rhizosphere soils contaminated with crude oil under different organic manure treatments, with the rhizosphere microbial community acting as plant-microbe interaction at subsurface environment. Enumeration of total heterotrophic bacteria and total saprophytic fungi were carried out using plant rhizosphere soil samples. The rhizosphere soil samples were collected from crude oil contaminated soil amended with organic nutrients: poultry droppings, goat faeces, cow dung and fish pond waste water i.e. water from pond containing Clariesgariepenus mixed with its faeces and feeds. The plant rhizosphere sample was cowpea (Vigna unguiculata). A non-rhizosphere soil was used as control. The representative strains or genera of both bacteria and fungi were identified. Total heterotrophic bacterial count range from 3.5x104 to 8.21x106 colony forming units per log10 CFU/g log 10of sample while total fungal counts ranged from 4.30x105 to 8.67x106 log 10 CFU/g; Non-rhizosphere soil (control) samples, had colony counts within the range of 4.30x103 to 4.71x105 for both bacteria and fungi; the rhizosphere were also screened quantitatively for the presence ofhydrocarbon utilizing fungi (HUF) andhydrocarbon utilizing bacteria (HUB) and to evaluate the rate of degradation of crude oil by pure culturesover a period of 28 days in mineral salt broth with crude oil as sole source of carbon. While an experimental set-up mineral salt broth with crude oil without any inoculums served as control. The analysis of pH and total viable counts were carried out at weekly intervals while total petroleum hydrocarbon content was determined at bi weekly interval. Results showed percentages of crude oil degradation in the control and crude oil polluted bioremediated rhizosphere soils, which percentages of crude oil degraded range in polluted soils amended with organic nutrients were as follows: poultry droppings (19.53%), cow dung (13.8%), Goat faeces (12.30%), fish pond waste water (10.69%) and polluted soil without addition of nutrients (9.40%). The result also showed that there was a constant decrease in PH from range 6.08 to 5.04. Total Petroleum Hydrocarbon (TPH) also followed the same pattern of steady decrease. Within days 7, the total TPH was degraded from 28600mg/kg to 1502.877mg/kg, and at day 14 it was reduced to 943.871mg/kg. Thus, this study has succeeded in evaluating the residual TPH levels in the rhizosphere of (cowpea) vigna unguiculata and also in the non- rhizosphere soils contaminated with crude oil under different organic manure treatments.
Keywords
Crude Oil, Vigna Unguiculata (Cowpea), Rhizosphere Soil, Total Petroleum Hydrocarbon
Introduction
Petroleum hydrocarbon play important role in the socio-economic life of Nigeria [1]. The activities associated with oil exploration and production, transportation and pipelines, refining, depots and terminal, service stations and underground storage sometimes lead to spillage in the environment [2]. This anthropogenic situation has led to land contamination by petroleum pollutants such as (TPHS, BTEX, PAHS and other inorganic additives that are toxic). This contamination by petroleum contaminants on soils results to loss of agricultural land, ground water and other health implication [3,4]. These creates unsustainable development situation that sometimes lead to restiveness in host communities [2]. There areso many hydrocarbon polluted sites in Nigeria, most of which are within the Niger Delta region. There are several technologies for the remediation or cleanup of hydrocarbon polluted soil. However, remediation industries especially the International Oil Companies are looking for technology that are: (1) effective and cost- effective (2) less invasive or in-situ technologies and that which will (3) take advantage of nature capacity to degrade hydrocarbon pollutants. One of such technology is phytoremediation which is also refer to as “GREEN” technology, in general term, phytoremediation is the use of plants and their associated microbial community for environmental clean-up of hydrocarbon polluted. This technology is becoming an important approach owing to its cost- effective and environmentally friendly features. The efficiency of phytoremediation depends mostly on the establishment of robust plant-microbe”s interactions. Plants through their “rhizosphere effects” support hydrocarbon–degrading microbes that assist in phytoremediation in the root zone, for example root activities in perennial ryegrass and alfalfa increases the number of rhizobacteria capable of petroleum degradation in the soil. In turn, healthy microbial communities enhances soil nutrients availability to the plants. However, petroleum hydrocarbon are known to be harmful not only to plants growth and development, but also to microbial process. This is because; petroleum hydrocarbons negatively affect photosynthesis and therefore reduce nutrient assimilation and biomass accumulation. In addition, petroleum pollution often results in altered microbial community structure and negatively influence rate of soil resources. A better understanding of the impact of petroleum pollution in plant-microbe interaction is required to improve the sustainability and feasibility of rhizoremediation as little is known about how plant-microbes interactions can be influenced by petroleum pollution itself. In this study, we evaluated the residual Total petroleum hydrocarbon (TPH) in rhizosphere of cowpea (Vigna unguiculata) and non rhizosphere soils contaminated with crude oil under different organic manure treatments.
The rhizosphere by definition is the soil region in close contact with the plant root. The term “rhizosphere soil” generally refers to thin layers of soil adhering to a root system after the loose soil has been removed by shaking [5]. The rhizosphere is basically divided into two general areas; the inner rhizosphere at the very root surface and the outer rhizosphere embracing the immediate adjacent soil. The microbial populations are more in the inner zones where the biochemical interaction between organisms are most pronounced. In the rhizosphere we have the rhizoplane which is the surface directly covering the root. Within the rhizosplane and rhizosphere, the inner organisms contribute excretory products and sloughed off tissues. Some of the interactions (such as mutualism) are beneficial to the plant, while some (parasitism) are detrimental[5]. Evaluation of residual Total Petroleum Hydrocarbon (TPH) in rhizosphere of plant have been reported by [6].
Microroganism-plant interactions may be classified as neutralistic, commensalism, synergism, mutualism, amensalism, competition and parasitism. In the rhizosphere, the plant root has a direct influence on the soil bacteria known as rhizosphere effect. The microbial populations in the rhizosphere also have an influence on the growth of plants. The interaction of plant root and rhizosphere microorganisms are based largely on interactive modifications of the chemical soil environment by the process such as water uptake by the plant system, release of organic chemicals to the soil by the plant roots, microbial production of plant growth factors and microbial mediated availability of micro mineral nutrients [2]. Microbial populations are generally abundant in the rhizosphere than in the soil that is free of plants roots. Bacteria population in rhizosphere are frequently in excess of 109/g. The rhizosphere effect may be expressed in terms of R/S ratio and most R/S ratios, range from 5 to 20. The R/S ratio is the ratio of the number of corresponding microorganism in the rhizosphere soil to the number in soil free roots. The population of algae, protozoa and fungi are often higher in the rhizosphere than the root-free soil and this is not as great as that of bacteria.
Materials & Methods
Study Area
The (Vigna unguiculata (cowpea) species, rhizosphere and non-rhizosphere soil samples for the quantitative evaluation of hydrocarbon utilizing microorganisms employed in this study were obtained from Umuechem in Etche Local Government Area, Rivers State. The soil used for plant propagation came from a large area of hydrocarbon-polluted crude oil spills, whereas there was no known history of crude oil pollution in the control soil samples. Screening of the rhizosphere of the plant species and collection of soil samples for plant propagation took place between April, 2019 to March, 2020.
Collection of Crude Oil Contaminated Soils
The soil samples were randomly collected at two different locations designated as Umuechem 1 and Umuechem 2. The crude oil contaminated samples were taken from Umuechem 1 from non- crude oil contaminated area (control) at a depth of 0-15 cm with a clean soil auger into sterile polythene bags. The samples (about 300g each) were labelled and transported to the laboratory in a cooler packed with ice block for analysis. Non-rhizosphere soils (i.e. soils from unpolluted rhizosphere) were taken at different points of Umuechem I and Umuechem 2, were also collected at depth of 0-15cm, about 5-10g of the soil was collected into a sterile polythene bag using a sterile hand trowel and allowed to air dry at ambient temperature. Five replicate samples of rhizosphere from the plant species and non-rhizosphere soils collected randomly within each of the sampling areas were pooled together and taken straight for microbiological and physicochemical analysis after thorough mixing. Analysis of collected samples were performed within 24 hours of sampling from field to laboratory.
Physico-chemical and Microbiological Analysis
Total phosphorus, total nitrogen and total petroleum hydrocarbon contents in the soil samples were determined.
Analysis of Total Petroleum Hydrocarbon (TPH)
United State Environmental Protection Agency (USEPA) method 8015C, (2007) was employed in the analysis of soil TPH. Crude oil contaminated soil samples were first dried using anhydrous sodium sulphate. Ten (10g) of the dried soil sample was thereafter put into a clean extracting amber glass bottle of which 10ml of extractant (n-Hexane) was added. Extraction of oil from soil sample was carried out by shaking the mixture of extracting and soil samples together in a mechanical shaker for 30 minutes. The extract was collected and stored in a dry organic free vial, observing the volume of the extract (V1). From the Vial, 1.0ul of the extract was collected and injected into already calibrated Gas Chromatograph (Dani Master GC model) which was equipped with capillary column, identification and data processing software (Data Apex clarity).

Processing of Samples
The soil samples were processed using the method of Almansoory [7]. Ten grams of the soil samples were weighed and added to 90ml of sterile distilled water to get an aliquot. One millilitre (1ml) of the aliquots, were then serially diluted using the ten-fold serial dilution method as described by Barba [8].
Microbial Analysis
Isolation and Enumeration of Total Heterotrophic Bacteria, (THB), Total Fungi (TF) Hydrocarbon Utilizing Bacteria (HUB) and Hydrocarbon Utilizing Fungi (HUF)
Plate count technique was used for the enumeration of non- rhizosphere populations of total Heterotrophic bacteria and fungi, Hydrocarbon utilizing bacteria and fungi. The Rhizosphere and non-rhizosphere soil samples were obtained and 1.0g of the respective soil samples was weighed and introduced into sterile normal physiological Saline (8.5g of NaCl in 1000ml of de- ionized water), under aseptic conditions, in a test tube. From here, ten-fold dilutions were carried out up to 10-6dilution. Afterwards a 0.1ml aliquot of each diluted soil sample was then (CFU/g).
Percentage Frequency of Occurrence of HUB and HUF in the Soil Samples
The percentage non-rhizosphere and rhizosphere microbial Colony counts of Hydrocarbon utilizing Bacteria (HUB) and fungi (HUF) of the plants were estimated by expressing overall total counts of HUB and HUF as percentage generic distribution in number of the entire Total Heterotrophic Bacteria and Saprophytic fungi (TSF) counts respectively.
Processing of Samples
The soil samples were processed using the method of Almansoory [7]. Ten grams of the soil samples were weighed and added to 90ml of sterile distilled water to get an aliquot. One millilitre (1ml) plated out aseptically onto agars that have properly dried using spread plate method. The respective dilution was spread plated, in triplicate plates onto potato dextrose Agar (PDA), Nutrient Agar (NA) and Oil Mineral Salt Agar (OMSA) (with tetracycline antibiotics added for hydrocarbon utilizing fungi to prevent the growth of bacteria). This was incubated at room temperature 37oC for 24 – 48 hours.
Counts of Total Heterotrophic Bacteria (THB) and Total fungi (TF) were carried out after incubation for 24 hours at 37oC and 48 hours for 5 days. On the other hand, incubated plates of Hydrocarbon Utilizing Bacteria and fungi were incubated at 37oC for 5 – 7days on the Oil Mineral Salt Agar (OMSA). All microbial counts were performed and expressed as Colony forming unit per gram of soil
Evaluation of the Effects of Organic Manure Treatment on the growth of (Vigna unguiculata (cowpea).
Treatments of Soil with Organic Manure Samples
Four treatments; each of poultry droppings, cow dung, goat faeces and fish pond waste water (water gotten from the raring of Clariasgariepeneus with deposites of feeds, and faeces), were applied to the Crude Oil Contaminated Soil (COCS) to serve as enrichment media for the microbial growth on the COCS. The poultry droppings and cow dung were collected from the poultry farm and abattoir belonging to the Faculty of Agriculture of Rivers State University. The Goat faces (GF) were collected from the Mile 3 Goat Market behind the park whilst Fish pond waste water was supplied to me by Mr. Barine Rogers of Animal and Environmental Biology Department of the Rivers State University, who operates a fish farming business within the University. All these treatments samples were sent to laboratory and were homogenized, composited separately before use [9].
Results and Discussions
Results of Physicochemical Analysis of Petroleum Hydrocarbon Polluted Soils
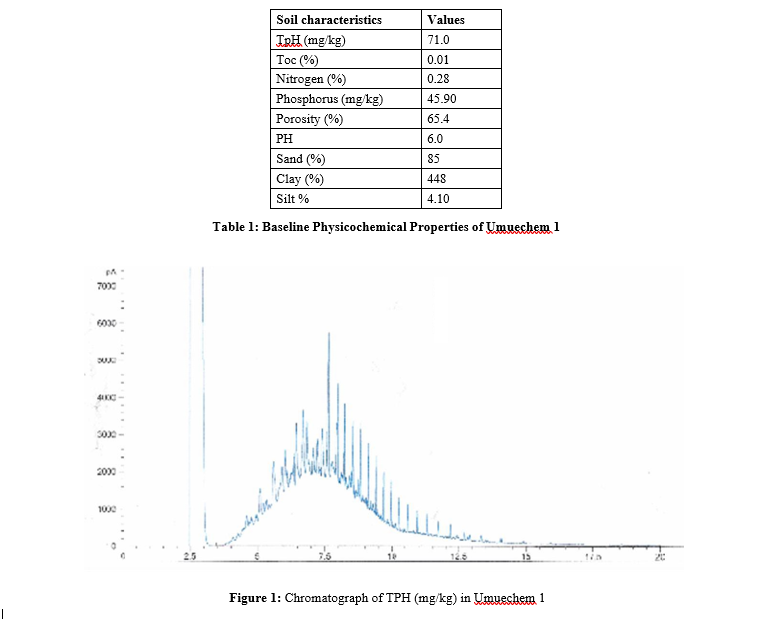
The results of the baseline physicochemical properties of the two locations Umuechem 1 and Umuechem 2 from where rhizosphere and non-rhizosphere soil samples were collected for studies are presented in (tables 1, 2 and figures 1, 2, 3, 4, 5 & 6) shows the chromatogram of TPH removal with (in weeks) from Vigna unguiculata rhizosphere grown at 6% w/w organic manure under crude oil concentrations PSO. The result of total culturable heterotrophic rhizosphere and bulk soil bacteria and fungi counts (CFU/g) in the Umuechem 1 and Umuechem 2 revealed that rhizosphere soils harboured total culturable heterotrophic bacterial population of 106 CFU/g irrespective of plant. Total culturable rhizosphere fungal population was found ranging from 105 to 106 CFU/g among the plant species in both environments. The bulk soil from both environments also harboured heterotrphic bacteria and fungi which range from 105 to 106 CFU/g of soil. The rhizosphere heterotrophic bacterial population and hydrocarbon utilizers differed significantly among plant species at (P<0.05) in locations. The results of the baseline physic-chemical properties of sample locations Umuechem1 and Umuechem 2, from whererhizosphere and bulk soil samples were collected for studies were presented in tables 1 and 2
|
Soil characteristic |
Values |
|
TPH (mg/kg) |
189.10 |
|
TOC (%) |
0.02 |
|
Nitrogen (%) |
0.15 |
|
Phosphorus (mg/kg) |
50.50 |
|
Porosity |
76.0 |
|
pH |
5.7 |
|
Sand (%) |
81.0 |
|
Clay (%) |
6.9 |
|
Silt (%) |
3.4 |
Table 2: Baseline Physicochemical Properties of Umuechem 2, Uncontaminated Soil
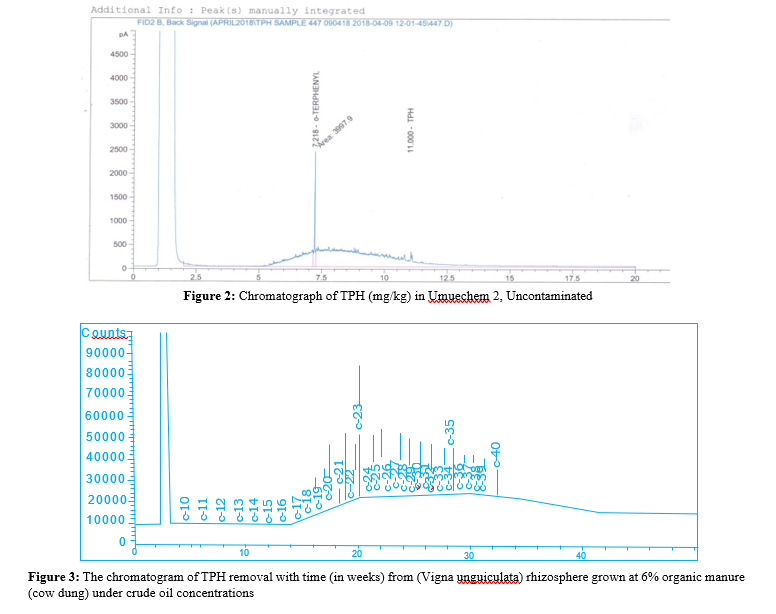
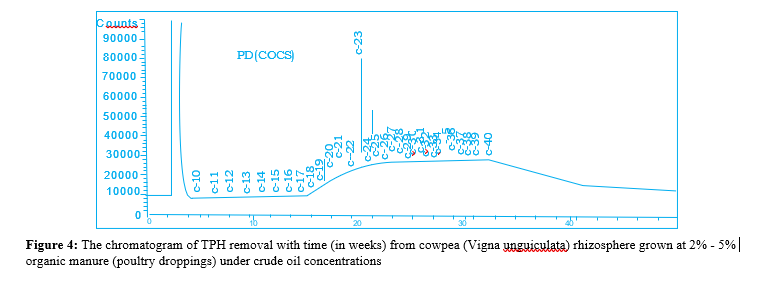
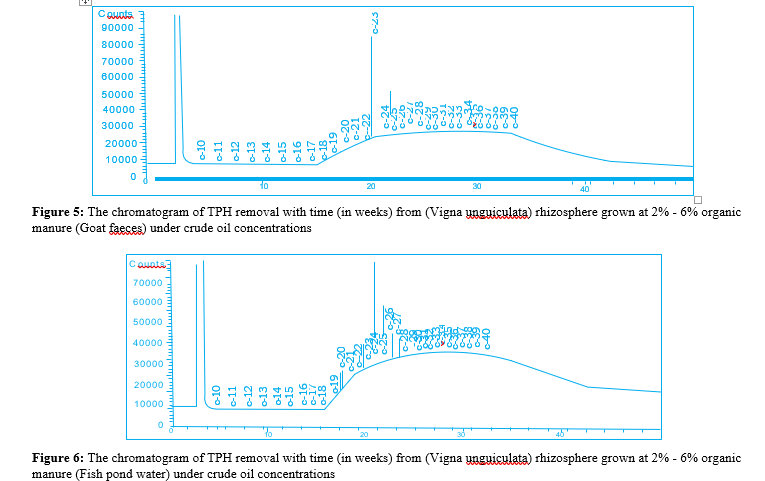
There were significant changes that occurred in Total Petroleum hydrocarbon (TPH) in Crude Oil Contaminated Soil (COCS) amended/treatment with organic manures; Poultry Droppings (PD) Cow Dung (CD), Goat Faeces (GF) and Fish Pond Waste Water (FPWW). The TPH in COCS amended with PD decreased the TPH from initial TPH level from 28,500 mg kg to 13,305 mg/ kg and 744.32 mg/kg by the 4th week. In the cow dung amended treatment, the TPH recorded initial of 5576 mg/kg and reduced weeks after planting to 2135.06 mg/kg and 1314 mg/kg at the 4th week. Amended COCS with goat faeces had initial TPH value of 125.48 mg/kg which was brought down to 35.36 mg/kg and 415.66 mg/kg at the 4th week. The TPH value of COCS amended with organic manure in fish pond at the beginning was 138.63mg/ kg week after planting. The TPH in the rhizosphere was reduced to 327.98 mg/kg finally less than 20mg/kg at the 4th week. In the control (un-amended COCS) a decrease in TPH level from 417 mg/ kg for the beginning of 2 weeks and TPH value for crude oil with plant and organic nutrient amended had a remarkable reduction for TPH level from 2858.91 mg/kg to 70.04 mg/kg respectively. The Carbon 1-Carbon 17 (C1-CI7) fraction were not seen on COCS within the environment while the high molecular carbon chain (C17-C40) fraction were predominant, however in different treatment, it significantly reduced at different rates. The lowest reduction was observed in the control (unamended) as shown in the chromatograph peak height after 4th week. The highest reduction in TPH value was recorded in PD, and CD in the FPWW reduction was heavier in the higher molecular weight fraction in the crude oil after the expiration of the 4th week of the experiment
Discussion of Physicochemical Analysis of Petroleum Hydrocarbon Polluted Soil
Residual Total Petroleum Hydrocarbon (TPH), in the cowpea Rhizosphere and Non-Rhizosphere Soils Contaminated with Crude Oil under different Organic Manure Treatments
In the uncontaminated rhizosphere of the plant used for the study, residual TPH also showed a progressive decrease with time throughout the study period in all the treatments both in high concentration and low concentration of crude oil at 3500.0mg/kg and 7190.0mg/kg. The residual TPH reduced from 35600mg/kg at week 2 to 1820.0, 490.0, 400.0 and 470.0mg/kg in treatment, PSO + organic manure (PSOOM) at week 12, the TPH lose recorded in the rhizosphere was higher in treatment with PSOOM for all the tested plants while treatments with SOOM recorded the highest TPH lost for the non-rhizosphere soil. The highest TPH lose recorded at week 12 for Vigna unguculata were 82.1% and 76.8%, while that of the non-rhizosphere soil was 65.8 and 5.3% respectively from the initial 3500.0mg/kg and 71900mg/ kg contamination levels at the first week, revealing a decrease in crude oil biodegradation by bacterial consortium from 71% to 49% as oil concentration increased from 1% to 10%.Also, TPH at high concentration have been reported to be phytotoxic, and responsible for the reduction of the growth of the plants (Tang et al., 2010). The toxicity effects or inhibitory action of petroleum hydrocarbon does not necessarily show up in the soils where biodegradation conditions were favourable [5].On the overall interest, the concentration of heavy metals exceeding above 2500ppm or crude oil exceeding above 25000ppm are regarded as inhibitor or toxicant to aerobic microorganisms in soils (US EPA, 1994). The Vigna unguiculata recorded the higher rate of TPH removal with time when supplemented with cow dung and poultry droppings from contaminated soils.The TPH degradation potentials of plants vary widely with plant species. Tang et al., (2010) tested the following plants alfalfa, tall fescue, ryegrass and cotton for their capability to bioremediate TPH and reported TPH biodegradation in the range of 33.2-47:8% with tall fescue > ryegrass > alfalfa > cotton in order of effectiveness.From this study, Vigna unguculata recorded greater percentage population of hydrocarbon utilizing microflora in the rhizospherewhich also showed more tolerance as the plants recorded higher growth in the tested concentration of crude oil amended with cow dung and poultry droppings than in control (uncontaminated soil).The earlier works of Medalla, reported that there was increase in height growth valued of 10.0, 20.0, 30.0ml of crude oil contaminated soil with 10 – 18 litres of water which Vigna unguculata was propagated than in the control (i.e. water without crude oil) in14 weeks interval [10].By this present study, it does appear that Vigna unguculata utilized the part of the oil for its growth and this may be as a result of higher TPH removal observed in the rhizosphere of the plant than the corresponding non-rhizosphere soils. Vigna unguiculata has been known to have remarkable capability to absorb and metabolize phenols and trace organic elements [11]. Vigna unguculata rhizosphere bacteria have been reported to decrease naphthalene within ten days by up to 46% in waste water [12].It also has the ability of enhancing oil- refinery effluent quality up to 20% reduction level in the total petroleum hydrocarbon content [13].
There was a progressive reduction in the percentage phosphorus (P) content of the soil with time throughout the study period both at the high and low concentration of crude oil contamination. The phosphorus content decreased progressively from 41.15mg/kg at week 2 to 5.19mg/kg at week 14 treatment with PSO from 43.80mg/ kg in all level of organic manure treatments. It is imperative to note that nitrogen and phosphorus serve as limiting factors in soil biodegration of petroleum hydrocarbon [14]. Other workers also reported the same decrease in nitrogen and phosphorus reduction with time during petroleum hydrocarbon degradation [15,16]. The Observable progressive reduction with time in this study recorded in both rhizosphere and non- rhizosphere soils was as a result of increased demand for nitrogen and phosphorus utilization for metabolic activities as the plants and microorganisms also increase in growth level [17]. In this study, effort was made to evaluate how plant Eco- physiological traits, soil nutrients, rhizosphere and microbial activities are influenced by petroleum hydrocarbon pollution in Vigna unguiculata (cowpea) [18-20].
References
- Bodo, T. (2019). Deep issues behind the crisis in the Niger Delta Region: The case of oil exploration in Ogoniland, Rivers State, Nigeria. Asian Journal of Geographical Research, 2(1), 1-12.
- Bodo, T., Gimah, B. G., & Seomoni, K. J. (2020). Illegal oil bunkering in the Niger Delta region of Nigeria: a challenge to Nigeria’s development. European Scientific Journal, 16(29), 134-154.
- David, L. K., & Bodo, T. (2019). Environmental pollution and health challenges of the Ogoni People, Rivers State, Nigeria. International Journal of Advanced Research and Publication, 2(2), 28-32.
- Gimah, B. G., & Bodo, T. (2019). Creation of awareness through environmental adult education as a solution to the problem of habitat loss in Ogoni, Rivers State, Nigeria. International Journal of Advanced Research and Publications, 3(1), 22-28..
- Atlas, R. M. (1981). Microbial degradation of petroleum hydrocarbons: an environmental perspective. Microbiological reviews, 45(1), 180-209.
- Obire, O., & Anyanwu, E. C. (2009). Impact of various concentrations of crude oil on fungal populations of soil. International journal of environmental science & technology, 6, 211-218.
- Almansoory, A. F., Hasan, H. A., Idris, M., Abdullah, S. R. S., & Anuar, N. (2015). Potential application of a biosurfactant in phytoremediation technology for treatment of gasoline- contaminated soil. Ecological Engineering, 84, 113-120.
- Barba, A. A., Acierno, D., & d’Amore, M. (2012). Use of microwaves for in-situ removal of pollutant compounds from solid matrices. Journal of hazardous materials, 207, 128-135.
- Obire, O., Anyanwu, E. C., & Okigbo, R. N. (2008). Saprophytic and crude oil degrading fungi from cow dung and poultry droppings as bioremediating agents. Journal of Agricultural Technology, 4(2), 81-89.
- Medalla, F., Hoekstra, R. M., Whichard, J. M., Barzilay,E. J., Chiller, T. M., Joyce, K., ... & Griffin, P. M. (2013). Increase in resistance to ceftriaxone and nonsusceptibility to ciprofloxacin and decrease in multidrug resistance among Salmonella strains, United States, 1996–2009. Foodborne pathogens and disease, 10(4), 302-309.
- Wolverton, B., & McDonald, R. C. (1976). Don't wastewaterweeds.
- Tang, J. C., Wang, R. G., Niu, X. W., Wang, M., Chu, H. R., &Zhou, Q. X. (2010). Characterisation of the rhizoremediation of petroleum-contaminated soil: effect of different influencing factors. Biogeosciences, 7(12), 3961-3969.
- Tang, S. Y., & Lu, X. W. (1993). The use of Eichhornia crassipes to cleanse oil-refinery wastewater in China. Ecological Engineering, 2(3), 243-251.
- Obire, O. M. O. K. A. R. O., & Isoboye, M. A. (2014).Microorganisms associated with the rhizosphere ofMoringa oleifera. Current studies in Comparative Education. Science and Technology. ISCEST Journal, 1, 131-138.
- Muratova, A., Hübner, T., Narula, N., Wand, H., Turkovskaya,O., Kuschk, P., ... & Merbach, W. (2003). Rhizosphere microflora of plants used for the phytoremediation of bitumen-contaminated soil. Microbiological research, 158(2), 151-161.
- Lynch, J. M., Benedetti, A., Insam, H., Nuti, M. P., Smalla,K., Torsvik, V., & Nannipieri, P. (2004). Microbial diversityin soil: ecological theories, the contribution of molecular techniques and the impact of transgenic plants and transgenic microorganisms. Biology and fertility of soils, 40, 363-385.
- Bodo, T., & Gimah, B. G. (2019). Oil crisis in the Niger Delta region of Nigeria: genesis and extent. European Scientific Journal, 15(36), 141-158


