Research Article - (2022) Volume 3, Issue 2
Evaluation of Biochemical, Hematological and Oxidative Parameters in Mice Exposed To the Binapacryl
2Department of Microbiology, University of Lagos (UNILAG), Akoka, Lagos, Nigeria
3Department of Health and Health Care Administration, Swami Rama Himalayan University (SRHU), Dehradu, India
Received Date: Apr 26, 2022 / Accepted Date: May 02, 2022 / Published Date: May 17, 2022
Copyright: ©Copyright: ©2022 Kehinde Sowunmi, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Kehinde S. Adebayo S. M., Kade E. A., Adesiyan A. L. and Gurpreet K. (2022). Evaluation of Biochemical, Hematological and Oxidative Parameters in Mice Exposed To the Binapacryl. J Vet Heal Sci, 3(2), 150-156.
Abstract
We evaluated the toxicity of hepatic, hematological, and oxidative effects of binapacryl on female albino mice. The animals were treated orally with 50, 100, 150 and 200 mg/kg body weight of the binapacryl, on a daily basis for a period of 15 days. Distilled water was used as control treatment. Samples of blood and hepatic tissue were collected at the end of the treatment. Hepatotoxicity was monitored by quantitative analysis of the serum enzymes ALT, AST, γ-GT, urea and creatinine. We also investigated liver tissues histopathologically. Alterations of hematological parameters were monitored by RBC, WBC, hemoglobin, hematocrit, MCV, MCH, and MCHC. Significant increases in the levels of hepatic enzymes (ALT, AST, and γ-GT) were observed for both binapacryl treatments, but no considerable differences were found by histological analysis. The hematological parameters showed significant alterations (200mg/kg body weight) with reductions of RBC, hematocrit, and hemoglobin, together with a significant increase of MCV. There was an important increase in lipid peroxidation at both dosage levels, together with an NPSH decrease in the hepatic tissue, and significant changes in these parameters were observed only at the higher dose rate. The results of this study indicate that binapacryl can promote hematological and hepatic alterations, even at subacute exposure, which could be related to the induction of reactive oxygen species.
Keywords
Binapacryl, Hepatotoxicity, Hematological Damage, Oxidative Stress
Introduction
Binapacryl (Organochlorines) are the compounds, which contain a minimum of one covalently bonded chlorine atom. It is exhibit a large variety of structures with much diverse chemical properties [1]. Due to high atomic weight of chlorine, these compounds are found to be denser than water [2]. These compounds Binapacryl could enter an organisms’ body across the skin, from the lungs and could also be absorbed from the gut wall. Cyclodienes, hexachlor-ocyclohexane, endosulfan and lindane can easily pass through the skin, while the absorption is less in case of dicofol, toxaphene, DDT, mirex and methoxychlor [1]. It has been observed that absorption of binapacryl through skin and gut wall is greatly in¬creased by fat and fat solvents. These compounds are volatile and their significant part is stored in fat tissue and is excreted through biliary and urinary pathways, while storable lipophilic compounds could be egxcreted from maternal milk. They affect central nerv¬ous system causing hyper-excitable state in brain, convulsions, tremor, hyper-reflexia and ataxia [3, 4].
The binapacryl compounds have largely been attributed to the dec-lination of many species [5]. Effect of binapacryl compounds on the chromosomal abberancy & nuclear DNA content variation was also studied. A significant level of 1, 1 Dichloro 2, 2 bis (p- bichlo- rophenyl) ethylene (DDE) contamination was observed in both the populations. Females were found to have lower levels of bina¬pacryl compounds than males. A negative relationship between DDE concentration, carcass tissue and brain of bats was found [6, 7]. Boyd and de Castro researched on the relation of protein-de-ficient diet and DDT toxicity [8]. Higher concentrations of bina¬pacryl stimulate the tissues to produce more of hepatic microsomal drug metabolizing enzymes.
Regulatory agencies and scientific institutions worldwide have concluded that binapacryl does not present a risk to human health [9]. However, recent studies have suggested that long-term expo¬sure to the chemical can cause toxicity in pregnant rats, with bone development deficiency in the fetus (Dallegrave, 2003), changes in cellular metabolism cutaneous lesions and increased rates of non-Hodgkin’s lymphoma (De Ross et al., 2003). Furthermore, studies using low doses of binapacryl have shown that the product can cause significant hepatic changes, as well as nasal bleeding without interfering in platelet aggregation [10-12].
Hematological parameters, such as hematocrit, hemoglobin, and numbers of erythrocytes and white blood cells, can be used as in¬dicators of toxicity and have a broad potential application in envi- ronmental and occupational monitoring [13, 14].
Biochemical markers of hepatic and renal function, as well as of oxidative stress, are important for biomonitoring the exposure to environmental pollutants [15]. Many pollutants can induce damage in biological systems, including the mammalian liver, which is the main site in the body for detoxification and biotransformation pro¬cesses. These involve formation of reactive oxygen species (ROS) such as hydrogen peroxide (H2O2), the superoxide anion (O2–), and the hydroxyl radical (•OH) [15-17]. Due to their high reactivity, these species can damage lipids, proteins, carbohydrates, and nu- cleic acids leading to serious damage to health [18].
In order to neutralize ROS, animals possess an antioxidant defense mechanism composed of enzymes including superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), and glu¬tathione reductase (GR), as well as non-enzymatic antioxidants including non-protein thiols, especially glutathione (GSH). When the defenses of the organism are insufficient for neutral izing the ROS, oxidative damage can occur, one of the most serious types of which is membrane lipid peroxidation [19]. This has been reported in several species of fish [20-22]. Meanwhile, the activities of an¬tioxidant enzymes, as well as the occurrence of oxidative damage, have been proposed as indicators’ of oxidative stress caused by pollutants [23, 24].
Given the increasing use of binapacryl, along with the lack of in-formation on its toxicity in mammals, the objective of this work was to evaluate the effects of the product on hematological, bi-ochemical, and oxidative stress parameters, using female Albino mice.
Materials and Methods Chemicals
The animals were treated using the commercial binapacryl formu¬lation Original® (Monsanto, St, Louis, MO, USA), which contains DDT-type compounds the active ingredient, and the chlorinat-ed alicyclic. The compounds 5,5’-dithiobis(2-nitrobenzoic) acid, reduced glutathione (GSH), malondialdehyde, and thiobarbituric acid (TBA) were obtained from Sigma (St. Louis, MO, USA). All other chemicals used were of the highest grade available commer¬cially [25-27].
Animals
Adult female albino mice, aged 90 days and weighing around 25 g, were housed in plastic cages containing a layer of sawdust that was changed every 3 days to maintain hygienic conditions. Throughout the experimental period, the animals were kept in colonies, with free access to water and food. The temperature was controlled at 22±2 °C, and an illumination cycle of alternating 12-hour periods of light and dark was used.
Treatment
The animals were organized into three groups of 10 individuals each (both sexes). The control group received distilled water, while the test groups received either 50 or 500 mg/kg body weight of binapacryl diluted in distilled water. The pesticide was admin-istered orally, by gavage, on a daily basis for a period of 15 days. Collections of blood and hepatic tissue were made at the end of the period. All animal experiments were conducted in accordance with the guidelines published by the Society of Toxicology in July 1989 (Guiding Principles in the Use of Animals in Toxicology), and all experiments were approved by the Committee for the Ethical Use of Animals, University of Lagos.
Biochemical Evaluation
The blood was first centrifuged at 1,500 × g for 10 min at ambi¬ent temperature. The serum was then separated and used for liver function assessment employing measurements of the enzymes as-partate aminotransferase (AST), alanine aminotransferase (ALT), and gamma-glutamyl transferase (γ-GT). Renal function was eval¬uated using serum concentrations of urea and creatinine. These tests were performed using disposable kits obtained from Labtest Diagnostica South Africa.
Histopathological Analysis
Samples of hepatic tissue were obtained from the animals by sur¬gical excision following euthanasia. In all cases, a standardized 0.5 cm section of sample was removed from the same hepatic lobe. The samples were fixed using 0.1 M phosphate buffer solution (pH 7.4) containing 10% formaldehyde, then washed, dehydrated in alcohol, clarified using xylene, and mounted in paraffin blocks.
The tissues were sectioned into 5 μm slices, stained with hema-toxylin-eosin, and evaluated by electron microscopy. Indices of oxidative stress Lipoperoxidation in the hepatic tissue was eval¬uated using the thiobarbituric acid reactive substances (TBARS) technique described by Bird and Draper (1984) in which malon¬dialdehyde and the final products of lipid peroxidation react with barbituric acid, forming a colored complex. The tissue samples were homogenized in 10 mM phosphate buffer (pH 7.0, 1:10 w/v), containing 150 mM NaCl and 0.1% Triton X-100, using a Potter Elvehjem glass homogenizer. The mixtures were cooled, and then centrifuged at 10,000 × g for 10 min at 4 °C. The supernatant was removed and incubated at 100 ºC for 1 h with equal volumes of buffer (60 mM Tris-HCl at pH 7.4, containing 0.1 mM DPTA), 12% trichloroacetic acid and 0.73% thiobarbituric acid. The mix¬ture was cooled and then centrifuged at 10,000 × g for 5 min. The absorbance of the supernatant was measured at 535 nm. The concentration of TBARS in the sample was calculated from the malondialdehyde analytical curve and the results were expressed as nM/g of tissue.The concentration of non-protein thiols (NPSH) was determined as described by Ellman (1959). This method is based on the reaction of NPSH with 5,5’- dithiobis(2- nitrobenzoic acid) (DTNB), generating the thiolate anion (TNB), which can be measured spectrophotometrically at 412 nm. Samples of hepatic tissue were homogenized in 12% trichloroacetic acid (1:10, w/v), using the Potter Elvehjem homogenizer. The samples were then centrifuged at 10,000 × g, 4 °C for 10 min and the supernatant was added to the reaction medium (20 μM of DTNB, and 200 mM of sodium phosphate buffer, at pH 8.0). After 10 min at ambient tem¬perature, the absorbance was measured at 412 nm. The concentra¬tion of NPSH was calculated using the GSH analytical curve and the results were expressed as mM/g of tissue. Hematological eval¬uation Hematological parameters, such as red blood cell (RBC), white blood cell (WBC), lymphocyte and neutrophil counts were determined according to the serum content of hemoglobin, hema-tocrit, mean corpuscular hemoglobin (MCH), mean corpuscular volume (MCV) and mean corpuscular hemoglobin concentration (MCHC).
Statistical Analysis
The results were expressed as mean ± standard error of the mean. Differences between the groups were determined using one-way ANOVA, followed by Duncan’s test where appropriate. Signifi¬cant differences were indicated by p-values ≤ 0.05.
Results
The results showed that binapacryl can affect hepatic metabolism, causing important hematological alterations and oxidative damage to the hepatic tissue. Assessment of hepatic biochemical parame¬ters showed that at both concentration levels employed, the bina¬pacryl formulation induced significant liver damage, as indicated by increased levels of the enzymes ALT, AST, and γ-GT, in female mice (Table 1).
Table 1: Biochemical Parameters of Female Mice Submitted To Binapacryl for 15days
|
Parameters |
Control |
50mg/kg |
100mg/kg |
150mg/kg |
200mg/kg |
|
ALT (IU/L) |
1.01±0.00 |
2.44±0.01 |
*3.02±0.00 |
*3.98±0.01 |
*4.96±0.01 |
|
AST (IU/L) |
12.5±0.36 |
14.0±0.51 |
14.0±0.38* |
15.56±0.38 |
*14.0±0.26 |
|
γ-GT (IU/L) |
4.00±0.19 |
3.57±0.02 |
*3.8±0.02 |
*3.67±0.02 |
*3.56±0.03 |
|
Urea (mg/dL) |
24.14±0.11 |
25.31±0.08 |
25.2±0.12 |
23.06±0.16 |
*64±0.12 |
|
Creatinine (mg/dL) |
43.55±0.15 |
45.56±0.20 |
*44.4±0.20 |
*45.95±0.13 |
*44.4±0.20 |
|
TBARS(nM/gtissue) |
15±2 |
12±0.7* |
11±0.88* |
12.7±0.5 |
10.3±0.84* |
|
NPSH(mM/g tissue) |
94±18 |
180±15* |
303±57* |
230±19* |
404±29* |
* Significant Difference Relative to the Control (p≤0.05)
Nonetheless, histological analysis of the hepatic tissue did not reveal any significant differences compared to the control samples (Figure 1). The liver damage could be related to the capacity of binapacryl to cause oxidative stress since it induced lipoperoxidation and reduced the levels of non-protein thiols in the hepatic tissue (Table 1). The animals treated with binapacryl at a dose of 50-250 mg/kg body weight showed lower weight gain over the 15-day experimental period compared to the controls. Over the same period, the animals that received 200 mg/kg body weight showed significant weight reduction of ~10% (Figure 1).
(A) (B) (C)
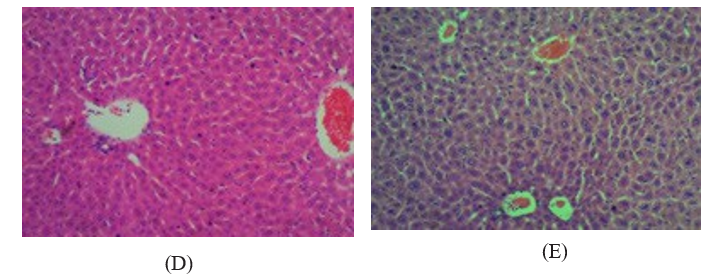
Figure 1: Histogical analysis of liver lobes of female submitted to oral treatment with BINAPACRYL for 15days dose rate 50-200mg/ kg body weight. A=control, B=50mg/kg, C= 100 mg/kg, D=150 mg/kg, E=200 mg/kg.
Table 2 summarizes the blood parameters of all groups. Our result data show that the median values of blood parameters decreased in animals treated with binapacryl at a dose of 50-200 mg/kg body weight, indicative of anemic syndrome. There was a significant reduction in the number of erythrocytes and of hemoglobin concentration, with reduced hematocrit and increased MCV, characteristic of macrocytic anemia.
Table 2: Hematological Parameters of Female Mice Submitted to BINAPACRYL for 15 days
|
PARAMETERS |
Control |
50mg/kg |
100mg/kg |
150mg/kg |
200mg/kg |
|
Red Blood cell |
17.34±0.40 |
*6.57±0.37 |
*0.95±0.08 |
*6.57±0.08 |
*3.55±0.55 |
|
Haemoglobin |
15.25±0.15 |
12.05±0.05 |
*2.0±0.30 |
*12.05±0.10 |
5.65±0.15 |
|
PVC |
35.50±0.0 |
24.5±0.50 |
*23.5±0.50 |
34.5±2.0 |
*22.00±0.50 |
|
MCV |
48.00±3.0 |
49.5±1.50 |
*50.5±1.50 |
*49.5±2.0 |
*47±2.00 |
|
MCH |
18.50±0.50 |
17.00±0.0 |
*17.5±0.50 |
*17.00±1.0 |
*17.5±0.50 |
|
MCHC |
58.00±1.00 |
*33.00±1.0 |
*38.00±1.0 |
*33.00±1.0 |
*39.5±1.50 |
|
RDW |
12.9±0.70 |
11.85±0.75 |
*11.75±0.25 |
*11.85±0.10 |
*11.3±0.20 |
|
White Cell count |
4.35±1.15 |
3.4±0.20 |
*3.85±0.15 |
*3.4±0.10 |
*4.15±0.15 |
|
Neutrophils |
18.76±0.29 |
14.5±0.04 |
*12.11±0.01 |
*13±0.02 |
*9±0.01 |
|
Lymphocytes |
4.02±0.58 |
2.72±0.28 |
*3.83±0.06 |
*2.72±0.11 |
*3.02±0.01 |
|
Monocytes |
0.05±0.03 |
*0.025±0.01 |
*0.015±0.05 |
*0.025±0.00 |
*0.015±0.01 |
|
Eosinophile |
0.18±0.06 |
0.15±0.03 |
*0.02±0.05 |
*0.15±0.00 |
*0.02±0.01 |
|
Basophils |
0.37±0.23 |
0.29±0.16 |
*0.11±0.07 |
*0.29±0.01 |
*0.18±0.01 |
|
Platelets count |
920±247 |
905±262 |
*223±7.0 |
*605±11 |
*399±2.50 |
* Significant difference relative to the control (p≤0.05). RBC: red blood cell number; WBC: leukocytes count; MCV: mean corpuscular volume; MCH: mean corpuscular hemoglobin; MCHC: mean corpuscular hemoglobin concentration.
Discussion
Previous works have shown that the toxic effects of different formulations based on BINAPACRYL may be associated with liver and oxidative damage. However, few studies were performed using mammals and most of the earlier works used aquatic organisms sensitive to the chemical [17, 28, 29]. It is therefore important to investigate the effects on mammals to establish relevant toxicity parameters, as well as to identify possible treatments in cases of occupational or accidental poisoning.
The haematological studies showed severe anaemia, which may imply inhibition of globin synthesis, depression of erythropoiesis, or a decreased level of folic acid [30-32]. Extract administration might have caused destruction of erythrocytes directly or the decreased RBC count may be due to the effect of extract on erythropoietic tissue [30]. The manifestation of hypochromic anaemia is due to reduction in the number of red blood cells orhaemoglobin or impaired production of erythrocytes ([1, 32]. DDT might be responsible for the decreased RBCs and haemoglobin levels due to increased level of pro-inflammatory cytokines that induced iron retention by reticulo-endothelial system, gastrointestinal tract and liver, thereby exerting inhibitory effect on erythroid precursors [40]. The significant decrease in WBC observed in this study may be alluded to suppression of the haematopoetic system, which consequently reduces the production of WBCs and bioconcentration of the toxicant in the kidney and liver [41]. Also, decreased level of white blood cell counts were observed mainly in mice exposed to chlorine in the cases of severe liver dysfunction and as a result of decreased defence mechanism against probable attack of toxic molecules duringextract toxicosis [42, 43]. Decreased in haematocrit observed in this study can be attributed to the reduction in RBC count caused by either destruction or reduction in size [44].
Variation in MCV, MCH, and MCHC values observed in this study may imply that the macrocytic anaemia which can lead to very slow production of erythroblasts in bone marrow which make them grow over in size with shape and have fragile membranes called megaloblast which is characteristic of pernicious anaemia which can lead to megaloblastanaemia [45, 46]. The reduction in Hb, RBC, WBC, MCV, MCH, and MCHC indicated that there is slow development of blood in the haemopoitic cells due to the presence of which has been reported to as reported to suppress haematopoiesis of all blood cells [47-50].
In conclusion, the data in this study presents the effect of binapacryl pesticides to be vast and devastating. Binapacryl, being non-biodegradable remain ubiquitous in environment and are the major pollutants. Binapacryl have been studied for their toxic effects on animals. They show multiple effects on the major physiological systems of the body including nervous, circulatory, respiratory and reproductive system. The present study reveals that binapacryl, at some critical growth periods, may generate severe health disturbances. Conclusively, the exposure to binapacryl should be reduced so as to minimize the associated environmental and human health hazard.
Acknowledgments
This work was supported by students and provided by Department of cell biology and Genetics. The authors are also grateful to Lagos State University Teaching Hospital for the histological analyses.
Conflict of Interest Declaration
All authors declare: No conflict of interest in this work
References
- Echobichon D J, Klaasen C D, ed Cassarett. (1996). Doull’s toxicology: Toxic effects of pesticides. The Basic Science of Poisons 5th edition, New York, 649.
- Russell, P. F. (1972). DDT Toxicology. Science, 177(4047),387-388.
- Shaffi, S. A. (1982). DDT toxicity: gluconeogenic enzymes and non-specific phosphomonoesterases in three teleosts. Toxicology Letters, 13(1-2), 11-15.
- Shaffi, S. A., & Dubey, R. P. (1989). DDT toxicity: variations in tissue non-specific phosphomonoesterases and gluconeogenic enzymes in three teleosts. Acta Physiologica Hungarica, 74(1), 57-62.
- Jaga, K., & Duvvi, H. (2001). Risk reduction for DDT toxicity and carcinogenesis through dietary modification. The journal of the Royal Society for the Promotion of Health, 121(2), 107-113.
- Lotufo, G. R., Farrar, J. D., Duke, B. M., & Bridges, T. S. (2001). DDT toxicity and critical body residue in the amphipod Leptocheirus plumulosus in exposures to spiked sediment. Archives of environmental contamination and toxicology, 41(2), 142-150.
- De Perre, C., Trimble, A. J., Maul, J. D., & Lydy, M. J. (2014). Ecological bioavailability of permethrin and p, p′-DDT: Toxicity depends on type of organic matter resource. Chemosphere, 96, 67-73.
- Boyd, E. M., & De Castro, E. S. (1968). Protein-deficient diet and DDT toxicity. Bulletin of the World Health Organization, 38(1), 141.
- EPA, Environmental protection Agency 1975 http://www2. epa.gov/aboutepa/ddt-ban-takes- effect. .
- Khayat, R., Dupuy, A., Pansé, I., Bagot, M., & Cordoliani,F. (2013, May). Sclerodermatous changes in porphyria cutanea tarda: six cases. In Annales de Dermatologie et de Venereologie (Vol. 140, No. 10, pp. 589-597).
- Wallaeys, E., Thierling, U., Lang, E., Neumann, N. J., & Frank,J. (2014). Porphyria cutanea tarda mit sklerodermiformen Veränderungen und Hämochromatose. Der Hautarzt, 65(4),272- 274.
- Andersen, J., Gjengedal, E., Sandberg, S., & Råheim, M. (2015). A skin disease, a blood disease or something in between? An exploratory focus group study of patients’ experiences with porphyria cutanea tarda. British Journal of Dermatology, 172(1), 223-229.
- Booth, N. H., & McDowell, J. R. (1975). Toxicity of hexachlorobenzene and associated residues in edible animal tissues. Journal of the American Veterinary Medical Association, 166(6), 591- 595.
- Song, L., Zhao, J., Jin, X., Li, Z., Newton, I. P., Liu, W., & Zhao, M. (2014). The organochlorine p, p′-dichlorodiphenyltrichloroethane induces colorectal cancer growth through Wnt/β-catenin signaling. Toxicology Letters, 229(1), 284-291.
- Arrebola, J. P., Belhassen, H., Artacho-Cordón, F., Ghali, R., Ghorbel, H., Boussen, H., & Olea,N. (2015). Risk of female breast cancer and serum concentrations of organochlorine pesticides and polychlorinated biphenyls: A case–control study in Tunisia. Science of the Total Environment, 520, 106-113.
- Kim, S. A., Kim, K. S., Lee, Y. M., Jacobs, D. R., & Lee,D. H. (2015). Associations of organochlorine pesticides and polychlorinated biphenyls with total, cardiovascular, and cancer mortality in elders with differing fat mass. Environmental research, 138, 1-7.
- Koutros, S., Langseth, H., Grimsrud, T. K., Barr, D. B., Vermeulen, R., Portengen, L., & Engel,L. S. (2015). Prediagnostic serum organochlorine concentrations and metastatic prostate cancer: a nested case–control study in the Norwegian Janus Serum Bank cohort. Environmental health perspectives, 123(9), 867-872.
- Lewis-Mikhael, A. M., Olmedo-Requena, R., Martínez-Ruiz, V., Bueno-Cavanillas, A., & Jiménez-Moleón, J. J. (2015). Organochlorine pesticides and prostate cancer, Is there an association? A meta-analysis of epidemiological evidence. Cancer causes & control, 26(10), 1375-1392.
- Parada Jr, H., Wolff, M. S., Engel, L. S., White, A. J., Eng, S.M., Cleveland, R. J., & Gammon,
- M. D. (2016). Organochlorine insecticides DDT and chlordane in relation to survival following breast cancer. International journal of cancer, 138(3), 565-575.
- Rivero, J., Luzardo, O. P., Henríquez-Hernández, L. A., Machín, R. P., Pestano, J., Zumbado, M., & Valerón, P. F. (2015). In vitro evaluation of oestrogenic/androgenic activity of the serum organochlorine pesticide mixtures previously described in a breast cancer case–control study. Science of the total environment, 537, 197-202.
- Dierauf, L., & Gulland, F. M. (Eds.). (2001). CRC handbook of marine mammal medicine: health, disease, and rehabilitation. CRC press.Colabuono, F. I., Taniguchi, S., & Montone, R. C. (2012).Organochlorine contaminants in albatrosses and petrels during migration in South Atlantic Ocean. Chemosphere, 86(7), 701-708.
- Salem, D. M. A., El Sikaily, A., & El Nemr, A. (2014). Organochlorines and their risk in marine shellfish collected from the Mediterranean coast, Egypt. The Egyptian Journal of Aquatic Research, 40(2), 93-101.
- Zhou, H. Y., Cheung, R. Y. H., & Wong, M. H. (1999).Residues of organochlorines in sediments and tilapia collected from inland water systems of Hong Kong. Archives of Environmental Contamination and Toxicology, 36(4), 424-431.
- Wang, Y., Qiu, Y. L., Fei, Y., Li, L., Zhu, Z. L., Zhao, J. F.,& Yao, Y. X. (2011). Measurement and preliminary human health risk assessment of representative organochlorines in farmed Mandarin fish. Huan jing ke xue= Huanjing kexue, 32(8), 2385-2390.
- Tao, Y., Pan, L., Zhang, H., & Tian, S. (2013). Assessment of the toxicity of organochlorine pesticide endosulfan in clams Ruditapes philippinarum. Ecotoxicology and environmental safety, 93, 22-30.
- Phillips, P. J., Nowell, L. H., Gilliom, R. J., Nakagaki, N., Murray, K. R., & VanAlstyne, C. (2010). Composition, distribution, and potential toxicity of organochlorine mixtures in bed sediments of streams. Science of the total environment, 408(3), 594-606.
- Veses, O., Mosteo, R., Ormad, M. P., & Ovelleiro, J. L. (2012). Potential toxicity of polycyclic aromatic hydrocarbons and organochlorine pesticides in sediments from the Ebro River basin in Spain. Bulletin of environmental contamination and toxicology, 88(4), 644-650.
- Deribe, E., Rosseland, B. O., Borgstrøm, R., Salbu, B.,Gebremariam, Z., Dadebo, E., ... & Eklo,M. (2014). Organochlorine pesticides and polychlorinated biphenyls in fish from Lake Awassa in the Ethiopian Rift Valley: human health risks. Bulletin of environmental contamination and toxicology, 93(2), 238-244.
- Yohannes, Y. B., Ikenaka, Y., Saengtienchai, A., Watanabe, K. P., Nakayama, S. M., & Ishizuka M. (2014). Concentrations and human health risk assessment of organochlorine pesticides in edible fish species from a Rift Valley lake—Lake Ziway, Ethiopia. Ecotoxicology and environmental safety, 106, 95-101.
- Yohannes, Y. B., Ikenaka, Y., Nakayama, S. M., & Ishizuka,M. (2014). Organochlorine pesticides in bird species and their prey (fish) from the Ethiopian Rift Valley region, Ethiopia. Environmental pollution, 192, 121-128.
- Zhang, Y., Lin, N., Su, S., Shen, G., Chen, Y., Yang, C., & Tao, S. (2014). Freeze drying reduces the extractability of organochlorine pesticides in fish muscle tissue by microwave-assisted method. Environmental pollution, 191, 250-252.
- Byrne, S., Miller, P., Waghiyi, V., Buck, C. L., von Hippel,F. A., & Carpenter, D. O. (2015). Persistent organochlorinepesticide exposure related to a formerly used defense siteon St. Lawrence Island, Alaska: data from sentinel fish and human sera. Journal of Toxicology and Environmental Health, Part A, 78(15), 976-992.
- Cui, L., Ge, J., Zhu, Y., Yang, Y., & Wang, J. (2015).Concentrations, bioaccumulation, and human health risk assessment of organochlorine pesticides and heavy metals in edible fish from Wuhan, China. Environmental Science and Pollution Research, 22(20), 15866-15879.
- Wang, J., Liang, W., Henkelmann, B., Pfister, G., & Schramm,K. W. (2015). Organochlorine pesticides accumulated by SPMD-based virtual organisms and feral fish in Three Gorges Reservoir, China. Environmental pollution, 202, 160-167.
- Rahmawati, S., Margana, G., Yoneda, M., & Oginawati, K. (2013). Organochlorine pesticide residue in Catfish (Clarias sp.) collected from local fish cultivation at Citarum watershed, West Java Province, Indonesia. Procedia Environmental Sciences, 17, 3-10.
- Capkin, E. R. O. L., Altinok, I., & Karahan, S. (2006). Water quality and fish size affect toxicity of endosulfan, an organochlorine pesticide, to rainbow trout. Chemosphere, 64(10), 1793-1800.
- Mittal, P. K., Adak, T., & Sharma, V. P. (1991). Acute toxicity of certain organochlorine, organophosphorus, synthetic pyrethroid and microbial insecticides to the mosquito fish Gambusia affinis (Baird and Girard). Indian journal of malariology, 28(3), 167-170.
- Salvo, L. M., Bainy, A. C., Ventura, E. C., Marques, M. R., Silva, J. R. M., Klemz, C., & Silva de Assis, H. C. (2012). Assessment of the sublethal toxicity of organochlorine pesticide endosulfan in juvenile common carp (Cyprinus carpio). Journal of Environmental Science and Health, Part A, 47(11), 1652-1658.
- Da Cuña, R. H., Vazquez, G. R., Piol, M. N., Guerrero, N. V.,Maggese, M. C., & Nostro, F. L.L. (2011). Assessment of the acute toxicity of the organochlorine pesticide endosulfan in Cichlasoma dimerus (Teleostei, Perciformes). Ecotoxicology and Environmental Safety, 74(4), 1065-1073.
- Thies, M. L., Thies, K., & McBee, K. (1996). Organochlorine pesticide accumulation and genotoxicity in Mexican free-tailed bats from Oklahoma and New Mexico. Archives of environmental contamination and toxicology, 30(2), 178-187.
- Wang, D., Shelver, W. L., Atkinson, S., Mellish, J. A., & Li, Q. X. (2010). Tissue distribution of polychlorinated biphenyls and organochlorine pesticides and potential toxicity to Alaskan northern fur seals assessed using PCBs congener specific mode of action schemes. Archives of environmental contamination and toxicology, 58(2), 478-488.
- Sharma, H., Zhang, P., Barber, D. S., & Liu, B. (2010). Organochlorine pesticides dieldrin and lindane induce cooperative toxicity in dopaminergic neurons: role of oxidative stress. Neurotoxicology, 31(2), 215-222.
- Pelletier, G., Masson, S., Wade, M. J., Nakai, J., Alwis, R.,Mohottalage, S., ... & Vincent, R. (2009). Contribution of methylmercury, polychlorinated biphenyls and organochlorine pesticides to the toxicity of a contaminant mixture based on Canadian Arctic population blood profiles. Toxicology letters, 184(3), 176-185.
- Yu, H., & Yang, K. (2007). Mechanisms of mitogen-activated protein kinase pathway in male reproductive toxicity induced organochlorine pesticides. Wei Sheng yan jiu= Journal of Hygiene Research, 36(6), 756-8.
- Pandey, N., Gundevia, F., Prem, A. S., & Ray, P. K. (1990). Studies on the genotoxicity of endosulfan, an organochlorine insecticide, in mammalian germ cells. Mutation Research/ Genetic Toxicology, 242(1), 1-7.
- Tilson, H. A., Hong, J. S., & Mactutus, C. F. (1985). Effects of 5, 5-diphenylhydantoin (phenytoin) on neurobehavioral toxicity of organochlorine insecticides and permethrin. Journal of Pharmacology and Experimental Therapeutics, 233(2), 285-289.
- Maslansky, C. J., & Williams, G. M. (1981). Evidence for an epigenetic mode of action in organochlorine pesticide hepatocarcinogenicity: a lack of genotoxicity in rat, mouse, and hamster hepatocytes. Journal of Toxicology and Environmental Health, Part A Current Issues, 8(1-2), 121-130.
- Mikhail, T. H., Aggour, N., Awadallah, R., Boulos, M. N., El-Dessoukey, E. A., & Karima, A. I. (1979). Acute toxicity of organophosphorus and organochlorine insecticides in laboratory animals. Zeitschrift für Ernährungswissenschaft, 18(4), 258-268.
- Cárdenas-González, M., Gaspar-Ramírez, O., Pérez-Vázquez,F. J., Alegría-Torres, J. A., González-Amaro, R., & Pérez-Maldonado, I. N. (2013). p, p′-DDE, a DDT metabolite, induces proinflammatory molecules in human peripheral blood mononuclear cells â??in vitroâ??. Experimental and Toxicologic Pathology, 65(5), 661-665


