Research Article - (2024) Volume 7, Issue 1
Evaluation of Aqueous Methanol Stem Bark Extract of Stereospermum kunthianum (pink jacaranda) for Wound Healing Effects in Alloxan-induced Diabetic Rats
2Department of Pharmacology and Toxicology, Faculty of Veterinary Medicine, University of Abuja, Nigeria
3Department of Pharmacology Toxicology, Faculty of Veterinary Medicine, University of Abuja, Nigeria
44Department of Pharmacology and Toxicology, Faculty of Veterinary Medicine, University of Abuja, Nigeria
5Veterinary Surgery, University of Abuja, Nigeria
Received Date: Oct 02, 2023 / Accepted Date: Jan 03, 2024 / Published Date: Feb 20, 2024
Copyright: ©Ã?©2024 Ifeadi Chekwube Nkemjika1, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Ifeadi, C. N., Nwnyi, C. F., Ode, O. J., Ramon-Yusuf, Babatunde, S. B., Onah, J. A. (2024). Evaluation of Aqueous Methanol Stem Bark Extract of Stereospermum kunthianum (pink jacaranda) for Wound Healing Effects in Alloxan-induced Diabetic Rats. Toxi App Phar Insig, 7(1), 09-23.
Abstract
Background and Purpose: The stem bark of Stereospermum kunthianum is used extensively in traditional medicine to treat wounds, inflammatory and other pain related health conditions in humans. The aim of this study was to evaluate the wound healing properties of the stem bark extract of S. kunthianum in alloxan-induced diabetic rats.
Experimental Approach: The aqueous methanol extract of S. kunthianum was analysed for phytochemical constituents, acute toxicity tests in rats. The stem bark extract of S. kunthianum was evaluated for wound healing using excision and incision wound healing models in diabetic rats.
Results: The aqueous methanol extract of S. kunthianum revealed that saponins, terpenes, tannins and steroids were present. The acute toxicity study in rats up to 72 hrs. Showed no observed toxicity sign up to the dose of 5000 mg/kg. The S. kunthianum stem bark extract (100, 200, 400 mg/kg, p.o) caused significant reduction of wound area. The excision wound contraction (%) in rats treated with S. kunthianum stem bark extract was most effective and had 100% wound contraction. In the incision wound studies, the S. kunthianum stem bark extract showed a progressive rate of wound repair with increasing doses of the extract.
Conclusion: The results corroborated the ethno-medicinal claims and have given the scientific justification for the use of S. kunthianum stem bark extract for wound healing.
Implication: The S. kunthianum stem bark extract therefore has the potential to be developed as a wound healing agent.
Keywords
Wound Healing, Diabetes Mellitus, Stereospermum Kunthianum Stem Bark Extract, Surgical Wounds (Excision and Incision), Tensile Strength, Phytoconstituents, Wound Contraction, Mean Wound Closure Time, Wound Epithelialization.
Introduction
There is urgent need for discovery of targeted drugs that will enhance wound healing in diabetic patients. The skin is the body’s largest organ which covers the entire external surface of the body [1]. The skin as the external envelope of the body serves as a physical barrier, mainly by the structure of the stratum corneum which constitutes the primary defence against environmental physical aggressions and external pathogen invasion [2]. Wounds are injuries that break or cause disruption of the cellular and anatomical continuity of the skin or other body tissues [3]. Causes of injury may be mechanical, chemical, microbial, immunological, electrical, thermal, or nuclear sources [4]. Following skin injury, the damaged tissue is repaired through the coordinated biological actions that constitute cutaneous healing [5]. In mammals, repaired skin is not identical to intact uninjured skin; however, this disparity may be caused by differences in the mechanisms that regulate post-natal cutaneous wound repair compared to embryonic skin development [5]. Wounds represent a major global health challenge, which puts much economic, financial, and social stress on health institutions, caregivers, patients, and their families causing disability and loss productivity [6]. A chronic non-healing wound (CNHW) is typically correlated with comorbidities such as diabetes [7,8]. Diabetes is related to impaired wound restoration, making patients at risk of continual non-restoration wounds [9]. Diabetes mellitus is a chronic, non-communicable metabolic disease characterized by elevated levels of blood glucose (or blood sugar), which leads to serious damage of the heart, blood vessels, eyes, kidneys and nerves [10]. This disease is also associated with symptoms such as polyuria, fatigue, weight loss, delayed wound healing, blurred vision, increases in urine glucose levels, etc [11]. Impaired healing in diabetes is the result of a complex pathophysiology involving vascular, neuropathic, immune, and biochemical components [12]. One of the main consequences of diabetes is the impairment of self-repairing abilities [13]. Diabetes mellitus is one of the leading and major contributors to chronic wound healing problems. Diabetic patients with ulcers are at high risk for major complications which include infection and amputation [14,15]. The risk for lower extremity amputation is 15 to 40 times higher in people with diabetes than people without diabetes [16]. The main reason for skin wound healing is the restoration of the barrier function in order to prevent further damage or infection. This process of wound healing requires the distinct interplay and crosstalk of a multitude of cells and mediators from the onset [17]. Wound healing is a dynamic, interactive, complex process that involves soluble mediators, blood cells, extracellular matrix, parenchymal cells, and tissue layers of replacing devitalized and missing cellular structures, and tissue layers [18,19]. The intricate skin repair process has been organized in four sequential and overlapping steps: the hemostasis phase, the inflammatory phase, the proliferative phase, and the remodeling phase [20]. Due to simplicity in the measurement of wound healing responses, the excision and incision skin-wound healing models in rodents are by far the most convenient and reliable methods of study for potential new therapeutic agents [21]. Medicinal plants are the richest biosource of drugs of traditional systems of medicine, modern medicines, nutraceuticals, food supplements, folk medicines, pharmaceutical intermediates, and chemical entities for synthetic drugs [22]. In current years, numerous researches had been completed on natural pills to explicate their capability in wound management and these natural remedies proved their effectiveness as an alternative treatment to available synthetic drugs for the treatment of wounds [23]. Stereospermum kunthianum belonging to the family Bignoniaceae, also referred to as pink jacaranda in English, is an African deciduous shrub found widespread across Africa to the Red Sea. There are over thirty species within Central Africa and Asian distribution [24,25]. The Stereospermum kunthianum, Cham, Sandrine Petit (Family: Bignoniaceae) is used in traditional medicine to treat bronchitis, pneumonia and cough, gastritis, wounds, rheumatic arthritis, ulcers, dysentery, leprosy and venereal diseases in humans [26]. The plant is mainly used by the local people of Mayo–Danay Cameron as a wound healing agent [21]. The present study aimed at evaluating the stem bark extract of S. kunthianum to authenticate its ethnomedicinal usage for wound healing in alloxan-diabetic rats and for future drug development.
Materials and Methods
Study Area
The study was carried out at the Department of Pharmacology and Toxicology, Faculty of Veterinary Medicine, University of Abuja, Nigeria.
Plant Collection and Identification
The fresh stem bark of Stereospermum kunthianum was collected by the author from Apia village in Suleja district of Niger State, situated at 10000’N 6000’E, Nigeria. This was done during the rainy season specifically between May and June and in accordance with the Good Field collection practices by NMPB. The research was conducted at the Department of Pharmacology and Toxicology, Faculty of Veterinary Medicine, University of Abuja, Nigeria. The plant sample was identified by a plant Taxonomist, Mr. Muazzu, employed at the Herbarium Unit, Department of Medicinal Plants Research and Traditional Medicine, National Institute for Pharmaceutical Research and Development (NIPRD), Abuja. The plant’s authenticity was confirmed by comparing it to relevant voucher specimens. Afterwards, the samples were then deposited in the Pharmacology laboratory at the University of Abuja, located in Nigeria.
Preparation of Plant Extract
Stem bark of Stereospermum kunthianum was washed with clean water after collection and then cut into small pieces with a knife. Then, the stem bark of S. kunthianum was air-dried and pulverized using a mortar and pestle. One thousand six hundred grams (1.60 kg) of the pulverized sample was macerated successively in 5 litres of 80% v/v methanol under a temperature of 40oC on a shaker (GFLD 3006 mgH, Germany) with agitation to ensure maximum extraction. Double maceration was done over a period of 24 h each and the extract was then filtered with Whatman size 1 filter paper. The filtrate was concentrated using rotary evaporator (KNF RC 900, Neuberger, USA). The concentrate was then placed over a water bath to ensure proper dryness of the extract. The percentage yield of the extract was calculated as follows:
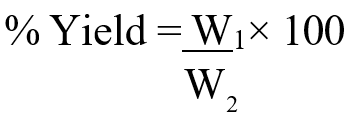
Where W1 = weight of dry extract after extraction; W2 = weight of the stem bark before extraction. The extract was stored in a closed container, kept in a refrigerator at 4oC for subsequent studies.
Phytochemical Analyses
The phytochemical screening of the crude extract was done using the standard method of Trease and Evans [27]. The extract was screened for the presence or absence of various chemical constituents like saponins using the froth test, terpenes using Libermann-Buchard test, tannins using ferric chloride test, steroids using Salkowski test, flavonoids using ferric chloride test, anthraquinones using Borntrager’s test, carbohydrates using Molisch test and alkaloids using Dragendorff’s test.
Phytochemical Screening Froth for Saponins Using the Test
A 2 ml of filtered sample was added to 4 ml of distilled water in a test tube. It was mixed well and shaken vigorously. Foam was produced after ten minutes which persisted on constant shaking; this confirms the presence of saponins.
Phytochemical Screening for Terpenes Using the Libermann-Buchard Test
A 1ml of the extract was added to chloroform, acetic anhydride and few drops of H2SO4 in a test tube and vigorously shaken. The formation of red-violet color indicates the presence of terpenoids.
Phytochemical Screening for Tannins Using the Ferric Chloride Test
A 1 ml of the extract was diluted in distilled water and added 2 drops of ferric chloride in a test tube. A greenish-black precipitate indicates the presence of tannins.
Phytochemical Screening for Steroids Using the Salkowski Test
This test was performed by adding 2 ml of chloroform and 1 ml of concentrated sulfuric acid in a test tube. Then, 10 drops of the extract was dissolved in isopropyl alcohol, which was slowly added into the test tube until double phase formation and it was shaken. A reddish-brown color in the middle layer of the test tube is indicative of the presence of steroids.
Phytochemical Screening for Flavonoids Using the Aluminum Chloride Test
A 4 ml of the extract was added to 1 ml of 1% aluminum chloride solution in a test tube; it was shaken and observed for light yellow coloration. A yellow precipitate indicates the presence of flavonoids.
Phytochemical Screening for Anthraquinones Using the Borntrager’s Test
A 4 ml of the extract was added to 4 drops of diluted sulphuric acid, and then it was boiled for 5 min and filtered and cooled. Then 4 drops of benzene was added to the cooled filterate and was shaken, and then aqueous ammonia was added and shaken very well. The formation of pink-red precipitate indicates the presence of anthraquinones.
Phytochemical Screening for Carbohydrates Using the Molisch Test
A 2 ml of the extract was added to 1 ml of concentrated sulphuric acid through the sides of test tube to form a layer without shaking. A purplish-reddish colour at the interface of the two liquids revealed the presence of carbohydrates.
Phytochemical Screening for Alkaloids Using the Dragendorff’s Test
This screening was performed by adding 1 ml of Dragendorff’s reagent to 2 ml of the extract; an orange red precipitate was formed, indicating the presence of alkaloids.
Chemicals and Drugs
Methanol (Fluka Chemie, Switzerland), Alloxan Monohydrate (Sigma Aldrich, USA), Accu-Chek active test strips (mg/dl: Indiana USA), Xylazine (20 mg/ml: Bioverta Czech Republic), Ketamine Hydrochloride injection (Jawa Ketamine, India), Vitamin C (Ascorbic acid; Emzor Pharmaceutical Industries Ltd., Lagos Nigeria), were used for the studies.
Animals
In this experimental study, fifty healthy adult Wistar rats of both sexes weighing (150-200 g) of approximately the same age (4-6 weeks of age) were used for the studies. The rats were procured from the Animal Facility Centre, Department of Pharmacology and Toxicology, National Institute for Pharmaceutical Research and Development (NIPRID), Abuja. The adult Wistar rats were grouped into 5 groups of 5 rats each (n=5). The experimental rats were housed in stainless steel cages for at least two weeks in the experimental room of the Laboratory Animal Unit of the Faculty of Veterinary Medicine, University of Abuja, Nigeria; at room temperature of (26°C - 27°C) and under a normal 12 h light/dark illumination cycle (lights on 07:00 am-19:00 pm) daily. The rats were housed for a period of 2 weeks prior to the commencement of the experiments for acclimatization. They were allowed access to standard feed and water ad libitum throughout the experimental period except when starvation was needed in the course of the experiment. To prevent the influence of circadian rhythm, all tests were conducted between 8:00 am and 11:00 am. The reason rats were chosen is because they share genetic, biological, and behavioral traits with humans, making them a suitable model for studying human conditions as many symptoms can be reproduced in rats.
Acute Toxicity Study
The modified method of Lorke was adopted for the study [29]. The approach was utilized to determine the quantity of the S. kunthianum extract required to cause the death of half of the rats (50%) that were treated (known as the median lethal dose or LD50) after 72 hours. The research was performed on healthy adult Wistar rats, and it involved administering the stem bark extract of S. kunthianum orally. The research was divided into two distinct phases (first and second phase). Nine rats were divided into three groups (groups A, B, and C: n=3) with each group containing three rats. The initial weights of the rats were also recorded. The remedy made from the stock sample was carefully divided and mixed evenly for each group. The rats received the correct dosage based on their weight and were then monitored for 72 hours to count the number of deaths. In the first phase of the research, groups A, B, and C were given the oral stem bark extract of S. kunthianum at different doses of (10, 100, and 1000 mg/kg) and their reactions were monitored. No deaths were reported after 72 hours. During the second phase of the research, a new group of nine adult Wistar rats were divided into three groups, with each group consisting of one rat (a total of three rats). Groups A, B, and C were administered the extract at doses of 2000, 3000, and 5000 mg/kg, respectively, using the oral route following the same procedure outlined for the first phase. After the rats received treatment, the rats were monitored for a period of 72 hours to check for any possible changes in their behavior or signs of toxicity. These changes included signs of nervousness, lack of coordination, increased activity, heightened awareness, reduced alertness, and mortality. The lethal dose 50% (LD50) of the extract was calculated using the formula [30].
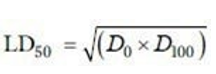
Where D0 = Highest dose that gave no mortality, and D100 = Lowest dose that produced mortality. All rats that survived were further monitored for two weeks for toxic effects.
Induction of Diabetes Mellitus Using Alloxan Monohydrate and Blood Glucose Monitoring Alloxan monohydrate from (Sigma Aldrich Inc. USA) was used to induce diabetes mellitus in the rats. The drug was prepared for injection by dissolving it in distilled water at a concentration of (20 mg/ml). Fifty (50) adult Wistar rats weighing (150 - 200 g) were fasted for 24 h (depriving them access to feed and water), after which each rat was injected with alloxan monohydrate. The fasting blood sugar of the rats was measured before the alloxan injection. Alloxan monohydrate at (115 mg/ kg) was administered using the intraperitoneal route to each rat. The rats resumed normal feeding after the induction. The blood glucose level in mmol/L of each rat was measured after 48 h of induction using a glucometer by using the tail tipping blood sample technique. Blood was drawn from the tail of each rat, spotted on glucose test strips and the baseline blood glucose levels of the rats were obtained using Accu-chek glucometer and glucose test strips. Rats with blood glucose of ≥ 11 mmol/L were considered diabetic. Diabetes in rats is frequently defined as fasting blood glucose above 7 mmol/L (126 mg/dL). The rats with diabetes were later utilized for additional investigations. The model was employed to provoke and sustain the development of type II diabetes. Before the study began, a pilot study for the suitable dose range of alloxan was experimented. When rats were given a high dose of 130 mg/kg of alloxan, a toxic glucose substitute, it resulted in the death of many of the rats. However, a lower dose of 110 mg/kg did not lead to the development of diabetes in the rats. During the experiment, rats were given alloxan monohydrate at a dosage of 115 mg/kg. This dose caused the rats to develop diabetes, but did not result in any deaths. Based on these findings, it can be concluded that the 115 mg/kg dosage was considered the safest and most effective for this particular experiment. Alloxan monohydrate can be administered through the intraperitoneal, intravenous, and subcutaneous methods. The intraperitoneal method leads to a slight increase in blood sugar levels and no deaths were observed. This is in contrast to the intravenous route, as it leads to a sudden increase in blood glucose levels in the rats, resulting in a high number of recorded deaths. In the case of subcutaneous administration of alloxan monohydrate, the majority of the rats did not develop diabetes [31,32]
Wound Healing Studies in Diabetic Rats
Excision and incision wound models were used to evaluate the wound healing activity of the stem bark extract of Stereospermum kunthianum in diabetic-induced rats.
Excision Wound Study in Diabetic Rats
Twenty-five diabetes-induced adult Wistar rats (150 g -200 g) of both sexes were used for the study. The rats were inflicted with excision wounds using the modified methods of Rupesh, et al. and Nwinyi, et al. [33,34]. The predetermined areas of wound infliction at the back of the rats were prepared for surgery by shaving their dorsal hair with shaving sticks and razor blades followed by cleaning with 70% alcohol. Each of the rats was anaesthetized using xylazine (10 mg/kg, i.p.) followed by ketamine (80 mg/kg, i.p.) prior to the creation of the wounds. The excision wound site was outlined on the back of each of the rats using a marker and ruler. The excision was inflicted on the dorsal region 1.5 cm away from the vertebral column and 5 cm away from the ear. A measuring ruler was used to measure the distance marked for wound creation. A full thickness of excision wound area of 4 cm2 (2 cm by 2 cm) was created along the outlined area of the shaved back using a surgical blade, toothed forceps, and pointed scissors. The entire wound was left open according to Diwan, et al. [35]. Haemostasis was achieved by dabbing the wound with a cotton wool swabs soaked in normal saline [34,36,37]. Following recovery from anaesthesia after surgery (creation of wounds), the rats were housed individually and allotted into five groups (A-E), of five rats each. Distilled water (10 ml/kg, p.o.) was administered to the rats in group A and served as a negative control. Three doses (100, 200, 400 mg/kg) of the stem bark extract of Stereospermum kunthianum were administered orally to the second, third, and fourth groups of rats (groups B, C and D), respectively. Vitamin C (100 mg/ kg p.o.) was given to the fifth group (group E) and serves as the positive control group. Treatment commenced on the day of wound creation and continued until the wound healed (i.e., until complete epithelialization). Changes in the size of the excision wound in each experimental rat on days 5, 10, 15, 20 and 25 were recorded accordingly [38]. Gross observation of the wounds was made; wound contraction and period of epithelialization were also determined. The diabetic level of each rat was monitored throughout the period of the research.
Wound Contraction
The size of the wound on each of the diabetic-induced rats was traced on transparent paper and placed on millimetre scale graph paper to estimate the wound area every five days until the wound was completely healed. Wound contraction contributes to wound closure and was expressed as a reduction in the percentage of the original wound size from day of wound creation until the day of complete epithelialization. It was used to calculate the degree of wound healing and represented as a percentage of healing wound area [39]. Percentage wound contraction was calculated using the formula:
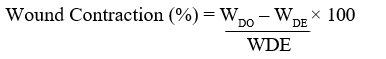
WDE Where: WD0= wound area on Day 0; WDE = wound area post excision day on or before complete epithelialization.
Period of Epithelialization
This was expressed as the number of days required for falling of the eschar (dead tissue remnants) without any residual raw wound. Wounds were considered closed (completely healed) when moist granulation tissue was no longer apparent and the wound was covered with new epithelium [40,41]. Epithelialization is useful for assessing the progress of wound healing in patients. In each wound, the percentage of wound epithelialization was calculated by the following formula [42].
Percentage epithelialization = (open wound area/total wound area) × 100
Mean Wound Closure Time (WC50)
This is the time taken for 50% of wound closure and was read off on a plot of wound closure (%) against time (days).
Incision Wound Study in Diabetic Rats
Twenty-five diabetic-induced adult Wistar rats (150 g-200 g) of both sexes were used for the study. The dorsal hair of each rat was clipped and disinfected with 70% alcohol. Each of the rats was sedated with xylazine (10 mg/kg, i.p.) followed by ketamine (80 mg/kg, i.p.) to achieve anaesthesia prior to the creation of the wounds. Two (2) paravertebral incisions were made through the skin and cutaneous muscles at a distance of 1.5 cm from the midline on either shaved side of the vertebral column with a sterile sharp blade. Each incision was 4 cm in length. A measuring ruler was used to measure the distance marked for wound creation and was used on all rats to ensure the accuracy of dimensions for the incision wounds [43]. After complete haemostasis, which was achieved by dabbing the wound with cotton wool soaked in normal saline, the parted skin was stitched with interrupted sutures at intervals of 0.5 –0.6 cm using a black braided silk surgical thread (size no.0) and a curved needle (3/8 semicircle-curved cutting: 35 mm). The wounds were left undressed and mopped with cotton wool [44,45]. The alloxan-induced diabetic rats were grouped into five groups (A-E), of five rats each. Distilled water (10 ml/kg, p.o.) was administered to the first group of rats and served as a negative control. Three doses (100, 200, 400 mg/kg) of the stem bark extract of S. kunthianum were administered orally to the second, third, and fourth groups of rats, respectively. Vitamin C (100 mg/kg, p.o.) was given to the fifth group (group E) and to serve as the positive control group. On the 7th day of the wound creation and of treatment, the sutures were removed while the treatment continued. The skin-breaking strength of the healed wound was measured on the 10th day according to the method described by Garg, et al. [46]. Krishnaveni, et al. and Nwinyi, et al. [34,47]. The anaesthetized rats were secured on the table and a line was drawn on either side of the wound 3 mm away from the suture line. These lines were gripped using two forceps applied firmly on to the line facing each other. One of the forceps was supported firmly; whereas the other was connected to a freely suspended light-weight bag. Weight was added gradually to the bag. A gradual increase in weight got transmitted to the wound site pulling apart the wound edges. As soon as wound gaping appeared, the addition of weight was stopped and the weight added to the bag was determined by weighing it on a measuring scale. The procedure was repeated on the contra lateral wound of each of the rats. The two values obtained from each rat were summed together and the mean weight for each rat was obtained to represent the tensile strength of the wound of each rat. The mean reading for the group was then given as the tensile strength for a given group which was measured in grammes. Gross observations of the incision wounds were made and determination of tensile strength of the incision wounds in respect of the varying group treatments at different time intervals was performed accordingly. The diabetic level of each rat was monitored throughout the period of the research.
Data Analysis
IBM SPSS Statistics version 23 was used for the statistical analyses. The results of the study were expressed as mean + SEM. The data generated from the studies were analysed using One- way analysis of Variance (ANOVA) where appropriate. Tukey Post Hoc Test was used to determine the differences between treatment groups. P values<0.05 were taken to be statistically significant. Results were presented as tables, figures, and plates as appropriate.
Results
Plant Extract
The weight of the pulverized S. kunthianum stem bark was 1600.0 g, while the weight of S. kunthianum extract was 279.0 g. The percentage yield was 17.44%. The extract was dark brown in colour, oily, and slurry in consistency.
Phytochemical Analysis
The phytochemical analyses carried out on the crude stem bark extract showed the presence of saponins, terpenes, tannins, and steroids (Table I).
|
Chemical Constituents |
Inference |
|
Anthraquinones |
- |
|
Carbohydrates |
- |
|
Flavonoids |
- |
|
Saponinins |
+ |
|
Steroids |
+ |
|
Tannins |
+ |
|
Terpenes |
+ |
|
Key: + = Present; - = Absent |
|
Table I: Phytochemical constituents of aqueous methanol extract of S. kunthianum stem bark extract
Acute Toxicity Studies
The results of the acute toxicity tests with S. kunthianum stem bark extract are shown in Table II. No toxic signs or death was observed in the rats 72 h after oral treatment with S. kunthianum stem bark extract at doses between 10 and 5,000 mg/kg. The estimated oral median lethal dose (LD50) of the extract in rats was therefore ≥ 5,000 mg/kg. The experimental rats did not exhibit acute signs of toxicity or death within 72 h even at the maximal test dose of 5000 mg/kg following oral of S. kunthianum stem bark extract. The oral median lethal dose (LD50) of the extract in rats was estimated therefore to be equal or greater than 5,000 mg/kg.
|
Treatment |
Number of dead rats |
Number of rats alive |
|
S.kunthianum (p.o.) |
||
|
Phase I |
||
|
10 mg/kg |
0/3 |
3/3 |
|
100 mg/kg |
0/3 |
3/3 |
|
1000 mg/kg |
0/3 |
3/3 |
|
Phase II |
||
|
2000 mg/kg |
0/3 |
3/3 |
|
3000 mg/kg |
0/3 |
3/3 |
|
5000 mg/kg |
0/3 |
3/3 |
|
Key: 0/3 =Number of animals which died/number of animals used; 3/3 = Number of animals alive/number of animals used. |
||
Excision Wound Studies in Diabetic Rats
Gross Observation of the Excision wounds
Gross observation of the excision wounds on the diabetic rats showed that rats in the negative control group (distilled water: 10 ml/kg, p.o.) healed more slowly with scar tissues which were very visible when compared with rats treated with S. kunthianum at 100, 200, 400 mg/kg; p.o. from day 5 to day 25 after excision. The wounds in the positive control group (Vitamin C; 100 m/ kg, p.o.) were fresh and healed at a much slower pace, while all wounds in rats treated with Stereospermum kunthianum stem bark extract at 100, 200, 400 mg/kg; p.o. completely healed with no scar formation by the 25th day post treatment (Plates I, II, III, IV, V).
On Day 0 of creating excision wounds, the mean wound size of all the groups was 4.00 cm2.
Wound Contraction
The effect of S. kunthianum stem bark extract on the wound closure of diabetic rats can be seen in Table 3. The percentage wound contraction post treatment in each experimental group was determined on days 5, 10, 15, 20 and 25. At day 5, administration of distilled water (10 ml/kg, p.o.) caused the wound to decrease in size from 4.00 ± 0.00 cm2 to 3.58 ± 0.4 cm2. Thus, a 10.5% contraction was produced. This contraction was significantly (p<0.05) different from the contraction in the treatment groups. Vitamin C produced 40.5% contraction while S. kunthianum extract had 41.0%, 42.5%, and 46.0% contraction at 100, 200, and 400 mg/kg, respectively (Table III, Table IV, Figure 1, Plate I).
At day 10, The mean excision wound size of non-treated control group (Group A; distilled water; 10 ml/kg, p.o.) was 1.57 ± 0.3 cm relative to 3.58 ± 0.4 cm at Day 5; the percentage wound contraction was 60.8%. The wound size in 100 mg/kg extract treated rats was 1.11 ± 0.1 cm compared to 2.36 ± 0.1 cm2 on day 5; the percentage wound contraction produced was 72.3%. Similarly, the extract at 200 and 400 mg/kg significantly (p<0.05) reduced the excision wound sizes of 2.30 ± 0.0 cm and 2.16 ± 0.1 on day 5 to 0.93 ± 0.1 cm and 0.64 ± 0.1 cm which are equivalent to 76.8% and 84.0% wound contraction, respectively. The contraction produced by the extract was significantly (p<0.05) different from the negative control at all tested doses (100, 200, 400 mg/kg, p.o.). Vitamin C, however, produced the lowest wound contraction of 57.0% compared to the other groups; the mean wound size was reduced from 2.38 ± 0.2 to 1.72 ± 0.2 cm (Table III, Table IV, Figure 1, and Plate II).
At day 15, administration of distilled water (10 ml/kg, p.o.) was able to reduce the excision wound size from 3.58 ± 0.4 cm to 0.90 ± 0.1 cm, (77.5% wound contraction) within the period. S. kunthianum stem bark extract at 100 and 200 mg/kg, p.o. achieved a reduction in wound sizes between 2.36 ± 0.1 cm to 0.58 ± 0.1 cm (85.5%) and 2.30 ± 0.0 to 0.62 ± 0.0 cm (84.5%), respectively. The extract at 400 mg/kg, p.o. was however, able to significantly (p<0.05) produced a reduction in the size of excision wound from 2.16 ± 0.1 cm to 0.42 ± 0.1 cm (89.5% wound contraction). In the positive control treated with Vitamin C (100 mg/kg, p.o.), the wound size was reduced from 2.38 ± 0.2 cm to 1.33 ± 0.2 cm, (66.8% wound contraction). Vitamin C was observed to have exerted the lowest percentage wound contraction in all test groups (Table III, Table IV, Figure 1, and Plate III).
At day 20, administration of distilled water (10 ml/kg, p.o) was able to reduce the excision wound size in rats from 3.58 ± 0.4 cm to 0.64 ± 0.1 cm, (84.0% wound contraction) within the period. S. kunthianum stem bark extract at 100, 200, 400 mg/kg, p.o. achieved significant (p<0.05) reductions in the wound sizes in rats from 2.36 ± 0.1 cm to 0.25 ± 0.0 cm (93.8%) 2.30 ± 0.0 to 0.27 ± 0.0 cm (93.3%) and 2.16 ± 0.1 cm to 0.18 ± 0.0 cm respectively. In the positive control treated with Vitamin C (100 mg/kg, p.o.), the wound size got reduced from 2.38 ± 0.2 cm to 0.74 ± 0.1 cm, (81.5% wound contraction (Table III, Table IV, Figure 1, Plate IV).
At day 25; administration of distilled water (10 mg/kg, p.o) reduced the excision wound size of 3.58 ± 0.4 cm to 0.31 ± 0.1 cm, (92.3% wound contraction). S. kunthianum stem bark extract at 100, 200, 400 mg/kg, p.o. significantly (p<0.05) achieved a complete reduction in the wound size of 100% in all treated groups. In the positive control (Vitamin C; 100 mg/kg, p.o.) group, the wound size reduced from 2.38 ± 0.2 cm to 0.48 ± 0.1 cm (88.0% wound contraction) (Table III, Table IV, Figure 1, Plate V).
The wound area contraction effect caused by the S. kunthianum stem bark extract (100, 200, and 400 mg/kg, p.o) was significant compared to that caused by Vitamin C (100 mg/kg, p.o.) on the days post treatment (Table IV, Figure 1). The S. kunthianum stem bark extract (100, 200, 400 mg/kg p.o) caused significant (p< 0.05) contraction of wound area on days 5, 10, 15, 20 and 25 when compared to the negative control (distilled water; 10 ml/kg p.o.; Table III). However, S. kunthianum stem bark extract (400 mg/kg, p.o.) was the most effective dose (Plate V).
|
Treatment |
Wound Contraction (cm2) |
||||
|
Day 5 |
Day 10 |
Day 15 |
Day 20 |
Day 25 |
|
|
Distilled water (10 ml/kg) |
3.58 + 0.4 |
1.57 + 0.3 |
0.90 + 0.1 |
0.64+ 0.1 |
0.31 + 0.1 |
|
S. kunthianum (100 mg/kg p.o) |
2.36 + 0.1* |
1.11 + 0.1* |
0.58 + 0.1* |
0.25+ 0.0* |
0.00 + 0.0* |
|
S. kunthianum (200 mg/kg p.o) |
2.30 + 0.0* |
0.93 + 0.1* |
0.62 + 0.0* |
0.27+ 0.0* |
0.00 + 0.0* |
|
S. kunthianum (400 mg/kg p.o) |
2.16 + 0.1* |
0.64 + 0.1* |
0.42+ 0.1* |
0.18+0.0* |
0.00 + 0.0* |
|
Vitamin C (100 mg/kg p.o) |
2.38 + 0.2* |
1.72 + 0.2 |
1.33 + 0.2 |
0.74 +0.1 |
0.48 + 0.1 |
|
S.k = Stereospermum kunthianum; n=5 Values are expressed as mean ±SEM (n=5);*P<0.05, significantly different from the control; One-way ANOVA; Tukey post hoc; *The mean difference is significant at the level of 0.05. |
|||||
Table III: Effect of aqueous methanol stem bark extract of Stereospermum kunthianum administered orally on excision wound contraction in diabetic rats
Wound contraction (%) Days
Treatment (mg/kg) Day 5, Day10, Day15, Day 20, Day 25
D.w (10 ml/kg) 10.5% 60.8% 72.5% 84.0% 92.3%
S.k (100) 41.0% 72.3% 85.5% 93.8% 100%
S.k (200) 42.5% 76.8% 84.5% 93.3% 100%
S.k (400) 46.0% 84.0% 89.5% 95.5% 100% Vitamin C (100) 40.5% 57.0% 66.8% 81.5% 88.0%
S.k – Stereospermum kunthianum.
D.w – distilled water Values are expressed as percent wound contraction (n=5)
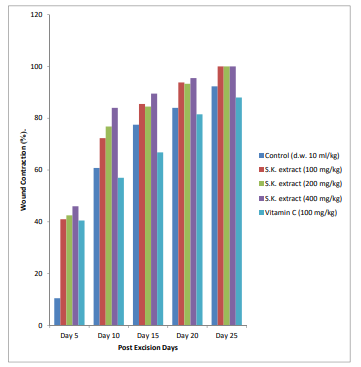
Figure 1: Effect of aqueous- methanol extract of S. kunthianum stem bark on excision wound contraction percentage (%) in diabetic rats
Significantly (p<0.05) different compared to control; d.w. = distilled water; S.K. = Stereospermum kunthianum stem bark extract.
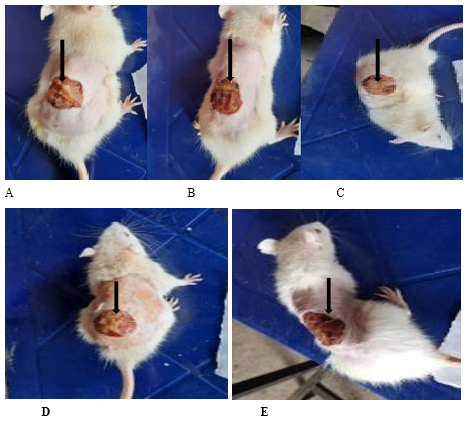
Plate I: The wound healing effect of Stereospermum kunthianum stem bark extract on excision wound in diabetic rats on day 5 post excision
A: Negative control (distilled water: 10 ml/kg p.o); B: S.kunthianum (100 mg/kg p.o.); C: S.kunthianum (200 mg/kg p.o.); D: S.kunthianum (400 mg/kg p.o.); E: Vitamin C (100 mg/kg p.o.)
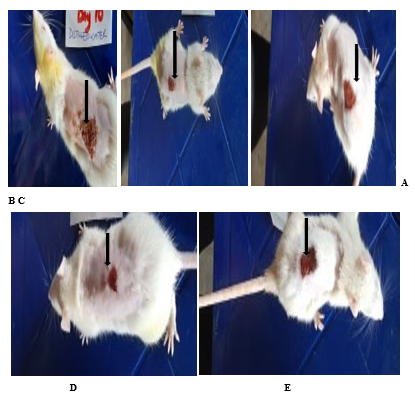
Plate II: The wound healing effect of Stereospermum kunthianum stem bark extract on excision wound in diabetic rats on day 10 post excision
A: Negative control (distilled water: 10 ml/kg p.o.); B: S.kunthianum (100 mg/kg p.o.); C: S. kunthianum (200 mg/kg p.o.); D: S.kunthianum (400 mg/kg p.o.); E: Vitamin C (100 mg/kg p.o.)
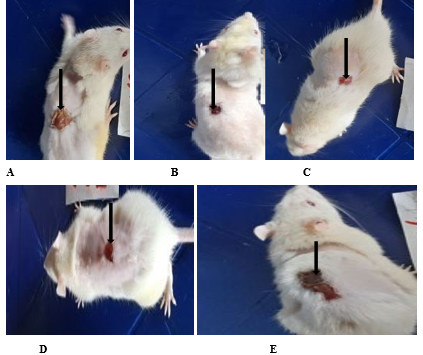
Plate III: The wound healing effect of Stereospermum kunthianum stem bark extract on excision wound in diabetic rats on day 15 post excision.
A: Negative control (distilled water: 10 ml/kg p.o.); B: S. kunthianum (100 mg/kg p.o); C: S. kunthianum (200 mg/kg p.o.); D: S. kunthianum (400 mg/kg p.o.); E= Vitamin C (100 mg/kg p.o.)
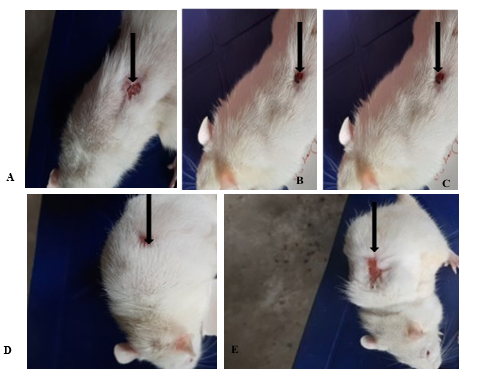
Plate IV: The wound healing effect of Stereospermum kunthianum stem bark extract on excision wound in diabetic rats on day 20 post excision.
A: Negative control (distilled water: 10 ml/kg p.o.); B: S. kunthianum (100 mg/kg p.o.); C: S. kunthianum (200 mg/kg p.o.); D: S. kunthianum (400 mg/kg p.o.); E: Vitamin C (100 mg/kg p.o.)

Plate V: The wound healing effect of Stereospermum kunthianum stem bark extract on excision wound in diabetic rats on day 25 post excision.
A: Negative control (distilled water: 10 ml/kg p.o.); B: S.kunthianum (100 mg/kg p.o.); C: S. kunthianum (200 mg/kg p.o.); D: S. kunthianum (400 mg/kg p.o.); E: Vitamin C (100 mg/kg p.o.)
Period of Epithelialization
In all test rats, there was no epithelialization of the excision wounds at days 1 and 5 (Table V). Epithelialization commenced, however, on day 10 in the negative control (distilled water, 10 mg/kg, p.o.) rats had 65% compared to 62.3%, 63%, 64% with the respective oral doses of 100, 200, 400 mg/kg of S. kunithanum extract. Vitamin C (100 mg/kg p.o.) however, induced 62% epithelialization. Moreover at day 15, distilled water produced 69% epithelialization compared to 71%, 72%, 80%, and 74% and for Vitamin C (100 mg/kg), 100, 200 and 400 mg/kg of the extract respectively. In the same manner, distilled water induced 72%, Vitamin C 77%, but 100, 200, and 400 mg/kg of the extract had 81%, 90%, and 88% accordingly. All doses (100, 200 and 400 mg/kg) of the stem bark extract of S. kunthianum produced complete (100%) epithelialization at day 25, while the negative control (distilled water; 10 ml/kg p.o.) had 85% and Vitamin C (100 mg/kg p.o.) had 86% epithelialization at the end of the study period. Stem bark extract of S. kunthianum at 100 mg/kg p.o, 200 mg/kg p.o and 400 mg/kg p.o. produced the shortest epithelialization period of 25 days each while, animals in the negative control (distilled water; 10 ml/kg, p.o) and positive control (Vitamin C; 100 mg/kg, p.o.) did not achieve full epithelialization period at 25 days post excision (Table 5). All doses (100, 200, 400 mg/kg) of the extract enhanced excision wound healing with greater percentage wound contraction and complete epithelialization with no scar tissue formation compared to either distilled water or Vitamin C, the reference drug from day 10 to the end of the study. Again, at day 25, the various doses of the extract displayed maximal wound healing potency with 100% wound contraction relative to Vitamin C (100 mg/kg, p.o.) which produced 88.0% within the same period. The extract doses (100, 200, 400 mg/kg, p.o.) demonstrated greater wound healing efficacy compared to Vitamin C (100 mg/kg, p.o).
|
Treatment (mg/kg) |
Degree (%) and Day of Epithelialization |
||
|
Day10 Day15 |
Day 20 |
Day 25 |
|
|
Distilled water (10 ml/kg p.o.) |
65 69 |
72 |
85 |
|
S.kunthianum (100 mg/kg p.o.) |
62 72 |
81 |
100 |
|
S.kunthianum (200 mg/kg p.o.) |
63 80 |
90 |
100 |
|
S.kunthianum (400 mg/kg p.o.) |
64 74 |
88 |
100 |
|
Vitamin C (100 mg/kg p.o.) |
62 71 |
77 |
86 |
|
S.K.= Stereospermum kunthianum; n=5 |
|||
Table V: Effect of aqueous-methanol stem bark extract of S. kunthianum given orally to diabetic rats on epithelialization period of excision wounds
Values are expressed as percent of number of the rats with complete epithelialization of the excision wound within a period.
Mean Wound Closure Time (WC50) in Diabetic Rats
Diabetic rats treated with the stem bark extract of S.kunthianum at 100, 200, and 400 mg/kg p.o. had a mean wound closure times of 6, 7, and 5 days, respectively when compared to the control groups (distilled water 10 ml/kg p.o. and Vitamin C 100 mg/kg p.o.) with median wound closure time of 8 days (Table VI).
|
Treatment (mg/kg) |
Median wound closure Time (Days) |
|
Distilled water 10 ml/kg |
8 |
|
S.kunthianum 100 mg/ kg |
6 |
|
S.kunthianum 200 mg/ kg |
7 |
|
S. kunthianum 400 mg/ kg |
5 |
|
Vitamin C 100 mg/kg |
8 |
|
S.K. = Stereospermum kunthianum; n=5 |
|
Table VI: Effect of aqueous methanol stem bark extract of S. kunthianum given orally to diabetic rats on the Mean WoundClosure Time (WC50)
Incision Wound Study in Diabetic Rats
Gross Observation of the Incision wounds
Rats treated with the stem bark extract of S. kunthianum at 100, 200, and 400 mg/kg had complete epithelialization at day 10. Healing was observed to occur in the negative control group with scar tissue formation, but a thin line depicted the area of wound healing in each of the treated rats. It was observed that wounds in rats treated with S. kunthianum extract at 100, 200, 400 mg/kg, p.o. healed completely by the 10th day with completeepithelialization. The wounds in the control group healed with scar tissue formation. Incision wounds on the diabetic rats in the treatment groups (100, 200, 400 mg/kg, and Vitamin C) healed completely leaving a thin lines of scars.
Tensile strength of incision wounds on diabetic rats treated with S. kunthianum stem bark extract
Diabetic rats treated with aqueous methanol stem bark extract of S. kunthianum at 100, 200, 400 mg/kg, p.o. showed a progressively significant (p<0.05) increase in the tensile strength of the healed wound muscle fibres in the diabetic rats compared to the control groups (Plate XIII). The S. kunthianum extract at 100, 200, and 400 mg/kg, p.o. produced a dose-dependent increase in the tensile strength of incision wounds in diabetic rats with values of 983 g, 984.4 g and 1066.0 g respectivelycompared to 446.3 g (distilled water) and 542.0 g (Vitamin C) of the control groups. The test doses of the extract (100, 200, 400 mg/kg) produced a significantly (p<0.05) higher tensile strength compared to the control groups (distilled water and Vitamin C) (Figure 2).
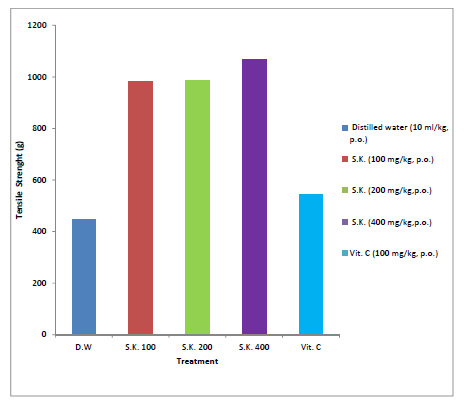
Figure 2: Effect of aqueous-methanol stem bark extract of Stereospermum kunthianum on the tensile strength of healed incision wounds in diabetic rats.
Significant difference at p<0.05; S.K. = Stereospermum kunthianum stem bark; Vit. C= Vitamin C; n=5.
Discussion
The phytochemical analyses of the stem bark extract of S. kunthianum revealed the presence of saponins, terpenes, tannins, and steroids. These plant constituents are responsible for the biological activity of the plant including antidiabetic and wound healing properties. This finding agrees with Bhattacharjee, et al. [48]. which show that these phytochemicals have bioactive properties. The aqueous methanol stem bark extract of Stereospermum kunthianum was evaluated for its acute toxicity (72 h) in rats using the oral and intraperitoneal routes. The extract did not produce any toxic effect up to a dose of 5000 mg/kg in the adult Wistar rats. The extract induced no acute toxic manifestation or death of the Wistar rats at 5000 mg/kg. According to the guidelines set by OECD, this level is considered practically non-toxic [49]. Similarly, Lorke showed that LD50 value greater than 1g (1000 mg/kg) for a test substance or chemical is considered as only slightly toxic (relatively safe) [29]. In diabetes mellitus, glucose cannot be converted into energy due to lack of insulin or resistance of body cells to insulin or both [50]. The acute complications of diabetes mellitus are hyperglycaemia, ketoacidosis, and hyperosmolar non-ketonic coma, while the delayed systemic complications include diabetic nephropathy, which may lead to amputation of joints, microangiopathy, diabetic neuropathy retinopathy, artherosclerosis, and infections [51]. Wound healing is generally delayed in diabetic patients due to factors considered localized to the wound and these include desiccation, infection or abnormal bacterial presence, maceration, necrosis, pressure, trauma and oedema. Alloxan monohydrate is a toxic glucose analogue used to induce diabetes in laboratory animals; it selectively destroys insulin-producing cells in the pancreas [52,53]. Excision and incision wound models were then used to evaluate the wound healing effect of Stereospermum kunthianum since the plant is used by traditional herbalists in northern regions of Cameroon for treating wounds. In this study that effect was evaluated to further justify the use of the plant in wound treatment [21]. The extract at all doses tested had a significant effect on wound healing. The stem bark extract doses demonstrated greater wound healing ability when compared with the standard control drug (Vitamin C). The underlying reason behind the reduced efficacy of Vitamin C observed in this study is not very clear. However, increased doses of Vitamin C may be useful in further studies. Vitamin C reportedly plays critical roles in tissue regeneration [54-56]. Tissue repair and regeneration are influenced by vitamin C through the synthesis of connective tissue, particularly collagen; it provides tensile strength to newly formed collagen to stretch without tearing, acts as an antioxidant which removes and neutralizes oxidants in the body and increases the proliferation of dermal fibroblast [57]. The extract probably facilitated excision wound healing by primary intention, hence scar tissue formation was not observed, while the treated and untreated control groups healed by secondary intention with visible scars [58]. The extract of S. kunthianum stem bark may also have caused wound healing effects due to its antimicrobial properties whereby bacterial activities due to wound contamination were greatly suppressed, allowing the wound to heal, devoid of scar formation. This corroborates the findings of [59-61]. The extract of S. kunthianum did not only induce the healing of the incision wounds in the diabetic rats but also increased the tensile strength of the healed wounds. This effect could be attributed to increased collagen concentration and stabilization of the fibres [62,63]. The stem bark extract of S. kunthianum produced a dose-dependent increased in tensile strength of the wound. This finding corroborated the report by Nwinyi, et al. that S. kunthianum stem bark extract enhanced wound healing in a dose-dependent manner in normal rats [34]. Thus, S. kunthianum extract aided wound healing in diabetic rats by inducing greater percentage wound contraction and increase in the tensile strength of the muscle fibres involved in the healing process. The wound healing potential of S. kunthianum may be attributable to the presence of a mixture of phytochemical constituents (saponins, terpenes, tannins, and steroids) present in the plant. The role of phytochemical constituents in wound healing is supported by different studies. Saponins extracted from ginseng, known as ginsenoside, have been shown to accelerate neovascularization in burn wounds of the skin in mice and increase vascular endothelial growth actor and interleukin (IL)-1β which is one of the inflammatory cytokines known to induce the accumulation of macrophages at skin wound sites and accelerate wound healing [64]. Tannins also inhibit bacterial growth and are shown to be active detoxifying agents, which has antioxidant and anti-inflammatory properties which helps to stabilize collagen needed for wound healing [65,66]. Terpenoids promote the wound healing process mainly due to their astringent and antimicrobial properties [67]. Therefore, this study has revealed the possible scientific justification for the use of S. kunthianum for the treatment and management of wounds in traditional medicine.
Conclusion
In conclusion, thestem bark of S. kunthianum extract was relatively safe in adult Wistar rats; the oral and intraperitoneal LD50 of the extract was found to be ≥ 5000 mg/kg. Phytoconstituents detected in the extract were saponins, terpenes, tannins, and steroids. At all tested doses of the extract tested (100, 200, 400 mg/kg, p.o.), the extract enhanced wound healing with greater percentage contraction, epithelialization, and tensile strength of the muscle fibers which may be due to the phytochemical constituents of the S. kunthianum stem bark extract suggesting a remarkable wound healing effect of the extract in diabetic rats. The results validated the ethno-medicinal use of S. kunthianum stem bark extracts in the treatment of wounds. The stem bark extract of S. kunthianum therefore has the potential to be developed as a wound healing agent in diabetic patients.
Recommendations
I recommend an isolation and identification of the active compound (s) of the stem bark of S. kunthianum associated with wound healing. The stem bark extract of S. kunthianum should be evaluated for its effects on mediators of inflammation such as prostaglandins, leukotrienes, histamine, bradykinin, platelet activating factor and interleukin-1. Sub-acute and chronic toxicity studies are required for determining the effects of long terms, low dose exposure to the stem bark extract of S. kunthianum. The mechanism of action of the extract on the measured parameters should be evaluated.
Ethical Approval
The study was approved by the Ethical Committee on Animal Use (UAECAU), Faculty of Veterinary Medicine, University of Abuja, Nigeria, with reference number: UAECAU/2020/0002. The animal handling was in accordance with the Guide for the Care and Use of Laboratory Animals of the National Research Council [28].
Conflict of Interests
No conflict of interest was declared by the authors.
Funding
The study was self-sponsored
Availability of Data and Materials
All relevant data and materials have been presented in the manuscript
Acknowledgement
Adamu Mohammed, a Laboratory Scientist with the Department of Pharmacology and Toxicology, Faculty of Veterinary Medicine, University of Abuja, Nigeria.
References
- Yousef, H., Alhajj, M., & Sharma, S. (2022). Anatomy, skin (integument), epidermis. StatPearls. Treasure Island.
- Pistone, D., Meroni, G., Panelli, S., D’Auria, E., & Acunzo, M., et al. (2021). A journey on the skin microbiome: pitfalls and opportunities. International journal of molecular sciences, 22(18), 9846.
- Sharma, Y., Jeyabalan, G., Singh, R., & Semwal, A. (2013). Current aspects of wound healing agents from medicinal plants: a review. Journal of Medicinal plants studies, 1(3), 2320-3862.
- Wedro, B. (2017). https://www.medicinenet.com/heart_ disease_coronary_artery_disease/article.html.
- Bielefeld, K. A., Amini-Nik, S., & Alman, B. A. (2013). Cutaneous wound healing: recruiting developmental pathways for regeneration. Cellular and Molecular Life Sciences, 70, 2059-2081.
- Benbow, M. (2011). Wound care: ensuring a holistic and collaborative assessment. British Journal of Community Nursing, 16(Sup9), S6-S16.
- Sen, C. K. (2021). Human wound and its burden: updated 2020 compendium of estimates. Advances in wound care, 10(5), 281-292.
- Sen, C. K. (2019). Human wounds and its burden: an updated compendium of estimates. Advances in wound care, 8(2), 39-48.
- Eming, S. A., Martin, P., & Tomic-Canic, M. (2014). Wound repair and regeneration: mechanisms, signaling, and translation. Science translational medicine, 6(265), 265sr6- 265sr6.
- Ziamajidi, N., Nasiri, A., Abbasalipourkabir, R., & Sadeghi Moheb, S. (2017). Effects of garlic extract on TNF-α expression and oxidative stress status in the kidneys of rats with STZ+ nicotinamide-induced diabetes. Pharmaceutical biology, 55(1), 526-531.
- Antu, K. A., Riya, M. P., Mishra, A., Sharma, S., & Srivastava,A. K., et al. (2014). Symplocos cochinchinensis attenuates streptozotocin-diabetes induced pathophysiological alterations of liver, kidney, pancreas and eye lens in rats. Experimental and Toxicologic Pathology, 66(7), 281-291.
- Dinh, T., Elder, S., & Veves, A. (2011). Delayed wound healing in diabetes: Considering future treatments. Diabetes Management, 1(5), 509-5019.
- Maffi, P., & Secchi, A. (2017). The burden of diabetes: emerging data. Management of Diabetic Retinopathy, 60, 1-5.
- Cunha, W. R., Arantes, G. M., Ferreira, D. S., Lucarini, R., & Silva, M. L. A., et al. (2008). Hypoglicemic effect of Leandra lacunosa in normal and alloxan-induced diabetic rats. Fitoterapia, 79(5), 356-360.
- Greenhalgh, D. G. (2003). Wound healing and diabetes mellitus. Clinics in plastic surgery, 30(1), 37-45.
- Center for Disease Control and Prevention. (2008). New Cases of Diagnosed Diabetes on the Rise. https://www.cdc. gov/media/releases/2008/index.html.
- Landén, N. X., Li, D., & Ståhle, M. (2016). Transition from inflammation to proliferation: a critical step during wound healing. Cellular and Molecular Life Sciences, 73(20), 3861-3885.
- Sorg, H., Tilkorn, D. J., Hager, S., Hauser, J., & Mirastschijski, U. (2017). Skin wound healing: an update on the current knowledge and concepts. European Surgical Research, 58(1-2), 81-94.
- Mercandetti, M., Cohen, A. (2021). Wound Healing and Repair.
- Cañedo-Dorantes, L., & Cañedo-Ayala, M. (2019). Skin acute wound healing: a comprehensive review. International journal of inflammation, 2019. 3706315.
- Tsala, D. E., Joseph, M. N., Heroyine, L. T., & Solomon,H. (2016). In vivo wound healing effect of water extract of stereospernum kunthianum stem-bark. Journal of medicinal herbs and ethnomedicine, 2, 1-6.
- Nafiu, M. O., Hamid, A. A., Muritala, H. F., & Adeyemi,S. B. (2017). Preparation, standardization, and quality control of medicinal plants in Africa. Medicinal spices and vegetables from Africa, 7, 171-204.
- Kumar, B., Vijayakumar, M., Govindarajan, R., & Pushpangadan, P. (2007). Ethnopharmacological approaches to wound healing—exploring medicinal plants of India. Journal of ethnopharmacology, 114(2), 103-113.
- Hutchison, J. (1963). Dalziel JM. Flora of West Tropical Africa. Crown Agents for Overseas Government and Administration, London. p.435-436.
- Von Maydell, H. J. (1986). Trees and shrubs of Sahel-their characteristics and uses. GTZ. 6. MBH. Eschborn. P111.
- Ching, F. P., Omogbai, E., & Otokiti, I. (2009). Aqueous stem bark extract of Stereospermum kunthianum (Cham, Sandrine Petit) protects against generalized seizures in pentylenetetrazole and electro-convulsive models in rodents. African Journal of Traditional, Complementary and Alternative Medicines, 6(4), 544-548.
- Trease, G. E., Evans, W. C. (1989). Phenols and phenolic glycosides. Textbook of Pharmacognosy. Bailliere Tindall and Co publishers, London, UK. vol. 13, p343 - 383.
- National Research Council: Guide for the care and use of laboratory animals. (2011). 8th edition, National Academic Press, Washington DC.
- Lorke, D. (1983). A new approach to practical acute toxicitytesting. Archives of toxicology, 54(4), 275-287.
- Chinedu, E., Arome, D., & Ameh, F. S. (2013). A new method for determining acute toxicity in animal models. Toxicology international, 20(3), 224-226.
- Alberti, K. G. M. M., & Zimmet, P. Z. (1998). Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus. Provisional report of a WHO consultation. Diabetic medicine, 15(7), 539-553.
- Skovsø, S. (2014). Modeling type 2 diabetes in rats using high fat diet and streptozotocin. Journal of diabetes investigation, 5(4), 349-358.
- Thakur, R., Jain, N., Pathak, R., & Sandhu, S. S. (2011). Practices in wound healing studies of plants. Evidence- based complementary and alternative medicine, 2011, 17, 438056.
- Nwinyi, F.C., Abdu, P., Saidu, N. A. S., Adamu, M. (2021). Pharmacological Justification for the ethnomedicinal use of stem bark extract of Sterospermum kunthianum Cham. (Family: Bignoniaceae) for wound healing effects. J of Medi Plants Stu, 9(2), 19-27.
- Diwan, A. R., Coker-Vann, M., Brown, P., Subianto, D. B., & Yolken, R., et al. (1982). Enzyme-linked immunosorbent assay (ELISA) for the detection of antibody to cysticerci of Taenia solium. The American Journal of Tropical Medicine and Hygiene, 31(2), 364-369.
- Nayak, B. S., & Pinto Pereira, L. M. (2006). Catharanthus roseus flower extract has wound-healing activity in Sprague Dawley rats. BMC Complementary and Alternative medicine, 6(1), 1-6.
- Nagar, H. K., Srivastava, A. K., Srivastava, R., Kurmi, M. L., Chandel, H. S., & Ranawat, M. S. (2016). Pharmacological investigation of the wound healing activity of Cestrum nocturnum (L.) ointment in Wistar albino rats. Journal of Pharmaceutics, 2016.
- Masson-Meyers, D. S., Andrade, T. A., Caetano, G. F., Guimaraes, F. R., & Leite, M. N., et al. (2020). Experimental models and methods for cutaneous wound healing assessment. International journal of experimental pathology, 101(1-2), 21-37.
- Murthy, S., Gautam, M. K., Goel, S., Purohit, V., & Sharma, H., et al. (2013). Evaluation of in vivo wound healing activity of Bacopa monniera on different wound model in rats. BioMed Research International, 972028.
- Singh, S. D. J., Krishna, V., Mankani, K. L., Manjunatha,B. K., & Vidya, S. M., et al. (2005). Wound healing activity of the leaf extracts and deoxyelephantopin isolated from Elephantopus scaber Linn. Indian journal of pharmacology, 37(4), 238-242.
- Shenoy, C., Patil, M. B., Kumar, R., Patil, S. (2009). Preliminary phytochemical investigation and wound healing activity of Allium cepa Linn (Liliaceae). Int. J Pharma Pharmaceuti Sci, 2(2), 167-175.
- Bloemen, M. C., Boekema, B. K., Vlig, M., van Zuijlen, P. P., & Middelkoop, E. (2012). Digital image analysis versus clinical assessment of wound epithelialization: a validation study. Burns, 38(4), 501-505.
- Grada, A., Mervis, J., & Falanga, V. (2018). Research techniques made simple: animal models of wound healing. Journal of Investigative Dermatology, 138(10), 2095-2105.
- Perez G, R. M., Vargas S, R., & Ortiz H, Y. D. (2005). Wound healing properties of Hylocereus undatus on diabetic rats. Phytotherapy Research: An International Journal Devoted to Pharmacological and Toxicological Evaluation of Natural Product Derivatives, 19(8), 665-668.
- Bhat, R. S., Shankrappa, J., & Shivakumar, H. G. (2007). Formulation and evaluation of polyherbal wound treatments. Asian Journal of Pharmaceutical Sciences, 2(1), 11-17.
- Garg, V. K., Khosa, R. L., & Paliwal, S. K. (2009). Wound healing activity of aqueous extract of Cynodon dactylon. Pharmacologyonline, 1, 1246-1255.
- Krishnaveni, B., Neeharika, V., Venkatesh, S., Padmavathy, R., & Reddy, B. M. (2009). Wound healing activity of Carallia brachiata bark. Indian journal of pharmaceutical sciences, 71(5), 576-578.
- Bhattacharjee, S., Waqar, A., Barua, K., Das, A., & Bhowmik, S., et al. (2018) Phytochemical and Pharmacological evaluation of methanolic extract of Lathyrus sativus L. seeds. Clinical Phytosci. 4(20).
- OECD/OCDE 420. OECD Guidelines for Testing of Chemicals, Acute Oral Toxicity–Fixed Dose Procedure. (2001).
- Wilcox, G. (2005). Insulin and insulin resistance. Clinical biochemist reviews, 26(2), 19-39.
- Ullah, F., Afridi, A. K., Rahim, F., Ashfaq, M., & Khan, S., et al. (2015). Knowledge of diabetic complications in patients with diabetes mellitus. Journal of Ayub Medical College Abbottabad, 27(2), 360-363.
- Lenzen, S. (2008). https://doi.org/10.1007/s00125-007-0886-7.
- Bingham, J. T., Etz, B. D., DuClos, J. M., & Vyas, S. (2021). Structure and Reactivity of Alloxan Monohydrate in the Liquid Phase. The Journal of Organic Chemistry, 86(21), 14553-14562.
- Mohammed, B. M., Fisher, B. J., Kraskauskas, D., Ward, S., & Wayne, J. S., et al. (2016). Vitamin C promotes wound healing through novel pleiotropic mechanisms. International wound journal, 13(4), 572-584.
- Mohit, V., Harshal, G., Neha, D., Vilasrao, K., & Rajashree,H. (2011). A comparative study of complexation methods for cefdinir-hydroxypropyl-β-cyclodextrin system. Journal of Inclusion Phenomena and Macrocyclic Chemistry, 71(12),57-66.
- Gunton, J. E., Girgis, C. M., Lau, T., Vicaretti, M., & Begg, L., et al. (2021). Vitamin C improves healing of foot ulcers: A randomised, double-blind, placebo-controlled trial. British Journal of Nutrition, 126(10), 1451-1458.
- Bechara, N., Flood, V. M., & Gunton, J. E. (2022). A systematic review on the role of vitamin C in tissue healing. Antioxidants, 11(8), 1605.
- Chetter, I., Arundel, C., Bell, K., Buckley, H., & Claxton, K., et al. (2020). The epidemiology, management and impact of surgical wounds healing by secondary intention: a research programme including the SWHSI feasibility RCT. Programme Grant for Applied Research. Southampton (UK), NIHR J Lib, 8(7).
- Aliyu, A. B., Ibrahim, M. A., Ibrahim, H., Dambatta, M. B., & Oyewale, A. O. (2017). GC-MS analysis of Pavetta corymbosa lipophilic extract and its antimicrobial activity. Ife Journal of Science, 19(2), 363-368.
- Adegun, A. A., Adesegun, S. A., & Usman, A. R. (2023). Thin Layer Chromatography Bio-autography guided identification of Antibacterial constituents of leaf extract of Stereospermum kunthianum Cham. (Bignoniaceae): https:// doi. org/10.51412/psnnjp. 2023.18. The Nigerian Journal of Pharmacy, 57(1), 582-591.
- Wangso, H., Laya, A., Leutcha, P. B., Koubala, B. B., & Laurent, S., et al. (2022). Antibacterial and antioxidant activities and phytochemical composition of Stereospermum kunthianum root bark. Natural Product Research, 36(22), 5665-5675.
- Udupa, A. L., Kulkarni, D. R., & Udupa, S. L. (1995). Effect of Tridax procumbens extracts on wound healing. International Journal of Pharmacognosy, 33(1), 37-40.
- Sorushanova, A., Delgado, L. M., Wu, Z., Shologu, N., & Kshirsagar, A., et al. (2019). The collagen suprafamily: from biosynthesis to advanced biomaterial development. Advanced materials, 31(1), 1801651.
- Navarro, P., Giner, R. M., Recio, M. C., Máñez, S., & Cerdá- Nicolás, et al. (2001). In vivo anti-inflammatory activity of saponins from Bupleurum rotundifolium. Life Sciences, 68(10), 1199-1206.
- Kimura, Y., Sumiyoshi, M., Kawahira, K., & Sakanaka,M. (2006). Effects of ginseng saponins isolated from Red Ginseng roots on burn wound healing in mice. British journal of Pharmacology, 148(6), 860-870.
- Mongalo, N. I., McGaw, L. J., Segapelo, T. V., Finnie, J. F., & Van Staden, J. (2016). Ethnobotany, phytochemistry, toxicology and pharmacological properties of Terminalia sericea Burch. ex DC.(Combretaceae)–A review. Journal of ethnopharmacology, 194, 789-802.
- Chirchir, D. K., Mshimba, S. M., Cheruiyot, G., & Adongo,J. O. (2014). Antimicrobial activity and phytochemical investigation of crude extracts of the fruits of Solanum incanum (Solananceae) and Dovyalis abbysinica (Flacourtiaceae). Sci J Microbiology, 193:4.


