Research Article - (2025) Volume 9, Issue 1
Efficacy of Combined Use of Follicular Stem Cell Suspension with Platelet Rich Plasma Vs Platelet Rich Plasma Alone in Native and Transplanted Hair during Hair Transplant: A Randomized Study
2Consultant Dermatologist, AB Centre for Skin and Hair, New Delhi, India
33Assistant professor, Department of Dermatology, Amrita institute of medical science and research ce, India
4Consultant Dermatologist, Aura Skin Institute, Chandigarh, India
5Assistant Professor, Department of Transfusion Medicine, Punjab institute of Liver and Biliary Scien, India
Received Date: Nov 18, 2025 / Accepted Date: Dec 18, 2025 / Published Date: Dec 31, 2025
Copyright: ©2025 Suruchi Garg, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Garg, S., Bansal, A., Gupta, S., Sharma, G., Grag, A. (2025). Efficacy of Combined Use of Follicular Stem Cell Suspension with Platelet Rich Plasma Vs Platelet Rich Plasma Alone in Native and Transplanted Hair during Hair Transplant: A Randomized Study. Stem Cell Res Int, 9(1), 01-10.
Abstract
Background: Androgenetic alopecia (AGA) involves progressive hair follicle miniaturization. Platelet-rich plasma (PRP) has shown promising results in AGA, and combining it with hair follicle stem cell suspension (HFSCS) can enhance the outcome.
Methodology: This randomized, prospective, single-blind study of forty male AGA patients (Stage III–VII), aged 20–55 years, compared intraoperative PRP+HFSCS with PRP alone over one year. Statistical analysis included Kruskal- Wallis, t-tests, Mann-Whitney U, Chi-square and Fisher's exact test.
Results: The PRP+HFSCS group showed significantly higher density of transplanted follicles at 8 weeks (p=0.0177) and greater hair length at 4 months (p=0.02) than PRP alone group. The PRP+HFSCS group also had significantly higher number of multirooted follicles in native (nontransplanted) hairs at 4 weeks, 8 weeks, 4 months (p<=0.001, 0.001, 0.035 respectively). Temporal and vertex hair growth beyond transplanted area was significantly better in PRP+HFSCS at 8 weeks and 4 months. At one year, PRP+HFSCS group demonstrated statistically significant hair growth beyond transplanted areas in a greater number of subjects in temporal (20%) and vertex (35%) areas (p=0.025, 0.04).
Conclusion: The PRP+HFSCS demonstrated statistically significant early improvements in hair density and length with a similar trend at one year though statistically insignificant; native hair growth in nontransplanted sites was statistically significant at one year.
Keywords
Androgenetic Alopecia, Male Pattern Hair Loss, Stem Cell Therapy, Hair Follicle Stem Cell Suspension, Platelet-Rich Plasma, Follicular Unit Extraction, Hair Regeneration, Multirooted Hair Growth, Dermatological Therapy, Hair Transplant, Regenerative Medicine, Interventional Dermatology, Progenitor Stem Cells
Introduction
Androgenetic alopecia (AGA) is associated with a progressive miniaturization of the hair follicles (HFs) and a continuous reduction in anagen phase[1]. Platelet rich plasma (PRP) significantly improves hair density in AGA and can be used as monotherapy or as an adjunct to hair transplant [2]. Growth factors released from PRP act on stem cells (SCs) in the bulge area of the HFs, promoting proliferation, inhibiting apoptosis of dermal papillary cells and stimulating new follicular development and neovascularization [3]. Current data indicate that HFs contain multipotent SCs located in the bulge area, and progenitor stem cells in the infrabulbar area, which can result in the regeneration of a complete HF, a process known as neogenesis. This knowledge is utilized in the management of AGA where hair follicle stem cell suspension (HFSCS) alone or in combination with PRP has produced favourable results [4-7]. This study aimed to compare the effect of PRP+HFSCS versus PRP alone in follicular unit extraction (FUE) hair transplant.
Materials and Methods
This randomized, prospective, single-blind comparative study was conducted in a hair transplant clinic in North India. Forty male AGA patients aged 20–55 years with Hamilton–Norwood stages III–VII, with no medical or surgical AGA treatment in the past 3 months were recruited for the study. AGA was diagnosed through history, clinical examination, and trichoscopy by a dermatologist. All patients had been using topical minoxidil 5% twice daily and stopped only 3 weeks prior to hair transplant. Patients on anticoagulant therapy, having non-AGA hair loss, bleeding disorders, autoimmune disorders, infection at the local site and immunocompromised individuals were excluded. Informed consent was taken and Helsinki guidelines were followed. Institutional Ethics Committee approval was granted for the study. Patients were randomly assigned to PRP+HFSCS and PRP groups using a computerized random number generator. PRP was prepared using two Renew cell kits and a Remi cold centrifuge at 3200 RPM and 1350 RCF for 4 minutes single spin to process 25 ml of venous blood, harvesting 6 ml of PRP with 3 times platelet concentration from baseline [8]. HFSCS was prepared by incubating 100 grafts from the occipital area in 5 ml trypsin-EDTA solution at 40°C for 2 hours. The trypsin was washed off with platelet-poor plasma, followed by mechanical agitation and sterile sieve filtration in platelet-poor plasma. Finally, it was centrifuged at 3200 rpm for 10 minutes. The supernatant was discarded and remaining pellet with high concentration of HFSC, was thoroughly mixed with 6 ml of PRP.
PRP and PRP+HFSCS were administered intraoperatively during FUE hair transplantation, with 0.05 ml injected at intradermal and subcutaneous level at a gap of 1 cm each into the whole androgenetic sensitive area after slitting. Pre-operative evaluations included hematology, biochemistry, coagulation profile, viral markers, iron studies with ferritin, vitamin D and B12 levels. The subjects found deficient in micronutrients, were started on therapeutic doses as per recommended guidelines. Scalp hair was harvested with minimal transection, and grafts with complete roots were transplanted at a density of 40–45 grafts/ cm² in both groups. Post-transplant, patients received topical 5% minoxidil lotion once daily, peptide-based serum, oral multivitamin containing biotin, minerals and amino acid-based supplement, and a plant-based protein supplement for next 1 year. No oral or topical finasteride was prescribed in either group. Each patient was photographed with clinical and trichoscopic images. An area of 1×1 cm was marked in the frontal, temporal, and vertex region in every patient. Trichoscopy and clinical photographs were taken at 4 weeks, 8 weeks, 4 months, 6 months and 1 year to assess hair density, shaft length, type of native (nontransplanted) or original hair follicles (single or multirooted) and hair growth beyond the transplanted area. Fixed anatomical landmarks (glabella to frontal hairline, highest point of eyebrow to temporal hairline, occipital eminence to highest point in vertex) were used for consistent measurements in each patient. Outcome assessments (hair counts, trichoscopy, photographic analysis) were performed by blinded assessors not involved in treatment allocation or procedures. Images were coded to maintain blinding Clinical photographs were taken with fixed camera distance, uniform lighting, and consistent head positioning.
Flow cytometry was performed to assess the composition of HFSCS, focusing on presence of CD34 positive stem cells (progenitor cells) and CD45 stem cells. The sample was labelled with the name, specimen type and combination of fluorochrome- conjugated Monoclonal antibodies (McAbs) prior to staining. Stain-Lyse-Wash method was used. We pipetted 50μl sample (hair follicle suspension) into a round-bottom tube and added 10μl of fluorochrome-conjugated McAbs (Fluorescein isothiocyanate (FITC), CD34 McAb and Phycoerythrin(PE) CD45 McAb. The sample was incubated in the dark for 15 minutes at room temperature. BD FACS lysing solution was used to lyse red cells. After incubation, we centrifuged the sample at 100 rpm, washed it twice in phosphate buffer saline (PBS) and resuspended the sample in PBS. Data was acquired on flow cytometer (BD FAClyric (Bectson, Dickinson and Company). Data were acquired according to the 2014 British Committee for standards in Hematology guidelines.
Statistical Analysis
Sample size was determined to draw useful inferences and calculate statistically significant differences and was based on previous experience by the authors. The quantitative variables were expressed as mean±SD and compared between groups using Kruskal–Wallis test, unpaired t-test and Mann–Whitney U-test wherever applicable. Qualitative variables were expressed as frequencies/percentages and compared between groups using Chi- square test and Fisher's exact test. Data were analysed using SPSS, and a p-value <0.05 was considered significant and <0.01 as highly significant.
Results
Baseline Characteristics
Out of a total of 55 screened patients, 40 patients were included and randomised into 2 groups. Fifteen patients were excluded due to thrombocytopenia, altered coagulation, insufficient donor area, or inability to follow up. The mean age was 35 years. Baseline characteristics were similar in both groups. All the patients completed follow-up at 4 weeks, 8 weeks, 4 months, 6 months, and 1 year ( Supplementary Figure 1, Supplementary Table 1).
Hair Density of Transplanted Follicles
To measure the density of transplanted follicles, we used 2-week post-transplant photographs as baseline (100% density), since retained follicles are actively growing at this stage. Over the next 4-8 weeks, the follicles enter a dystrophic phase resulting in reduced density. However, over subsequent months, hair density recovers gradually. We categorised the densities into 4 categories: <25%, 25–50%, 50–75% and >75% of the baseline hair density. In the PRP+HFSCS group, 35% had retained >75% density at 8 weeks, significantly outperforming PRP group (5% with P=0.0177). The density was later again comparable in both groups at 6 months and 1 year (Supplementary Figure 2).
Hair Shaft Length
In the PRP+HFSCS group, 15% of patients displayed average hair shafts length >6 cm at 4 months, significantly more than 7.5% in PRP group (p-value = 0.020). This pattern of longer follicle continued at 6 months though the difference was not statistically significant (Supplementary Figure 2).
Native Hair Follicle Type
Native or original hair follicles were assessed for single or multirooted types, with multirooted defined as those in which >50% of visible follicular units contained multiple hair shafts. More multirooted hair follicles were observed in PRP+HFSCS group (55% subjects) as early as 4 weeks, a trend which persisted in 80% at 8 weeks and in 100% subjects at 4 months. In comparison, the PRP group had lesser growth of multirooted follicles with 5% at 4 weeks, 15% at 8 weeks and 80% subjects at 4 months (p values=0.01, 0.001, 0.035 respectively) (Supplementary Figure 2). Trichoscopy at 8 weeks also revealed more terminal and intermediate hairs in the PRP+HFSCS group (Figure 1).
Hair Growth Beyond Transplanted Area
Native hair growth beyond the transplanted area was defined as the clinical and trichoscopic growth of dormant hair follicles in areas with previously reduced hair density. At 8 weeks, PRP+HFSCS group showed significantly more growth than PRP group in the temporal (80% vs 10%, p=0.001) and vertex regions (60% vs 5%, p=0.0001). Similar trends were seen at 4 months follow up. At one year, PRP+HFSCS group demonstrated statistically significant (p < 0.05) hair growth beyond the transplanted areas in more number of subjects in the temporal (20%), and vertex (35%) areas compared to the PRP group (Figure 2). Quantitative and qualitative evaluation of stem cells was done using flowcytometry FITC. Total CD45 positive stem cells population in the sample was 11% and we found that CD34 positive stem cell comprised of 0.3% population in follicular suspension (Supplementary Figure 4).
Discussion
AGA is characterised by progressive miniaturization of the HFs, producing microscopic hairs.1 Hair follicle stem cells (HFSCs), located in the bulge region of the hair follicle, play a critical role during the anagen phase, supporting hair follicle formation. The regulation of HFSC involves the arrector pili, stem cell bulge region and epidermal basement membrane, collectively termed ‘golden anchorage.’9 The latter operates through the nephronectin –integrin pathway, working like a lock and key mechanism for signal activation. In addition, other factors like extracellular matrix, hypoxia, shear stress, cellular metabolism, environmental factors are closely associated with stem cell niche [9,10]. HFSCs are typically dormant in the bulge region, but can migrate, proliferate, and differentiate to maintain tissue balance [11]. They give rise to "primed stem cells" (progenitor cells), which further differentiate into structures like the inner root sheath and hair shaft [12]. These progenitor cells are present below the bulge region and are CD34 positive [13].
Studies have shown that the number of HFSCs remains normal, but it is noteworthy that there's a reduced pool of actively multiplying CD34+ progenitor cells. This indicates that the issue in AGA is not the quantity of HFSCs but rather the depletion of progenitor cells and subsequent signalling and regulation of other stem cells [14]. Occipital hairs, which are resistant to hormonal influences in AGA are considered the best source for extraction of stem cells [15]. Stem cells also exert positive effects via paracrine action by secreting factors like platelet derived growth factor, hepatocyte growth factor, vascular endothelial growth factor, Insulin like growth factor binding protein precursors, and fibronectin which regulates hair regrowth primarily through ERK activation and Wnt signalling [16]. Several studies have shown the effectiveness of stem cell therapy for alopecia. Gentile et al. reported a 29 ± 5% increase in hair density in 11 AGA patients treated with autologous HFSCS, compared to < 1% in the placebo group [4]. Similarly, Kadry et al. on 60 AGA patients showed significant improvements in hair density with both PRP and stem cell therapies. The mean change in terminal hair density was 10.73 ± 3.66 for the PRP group and 19.30 ± 13.65 for the stem cell group.5 In another investigation of 38 AGA patients, Tak et al. observed that topical application of adipose-derived stem cell extract led to a 28.1% increase in hair count and 14.2% improvement in hair diameter at 16 weeks, significantly surpassing the control group's 7.1% increase in hair count and 6.3% improvement in hair diameter.6 Elmaadawi et al. found that intradermal injections of autologous bone marrow-derived mononuclear cells and autologous follicular suspension cells showed a mean improvement of 52% and 42%, with no statistical difference [7].
Our study substantiates the results of previous studies and we observed beneficial effect of using HFSCS, enriched with CD34 + stem cells into the scalp tissue during FUE hair transplantation. Our results showed significant improvements in the group receiving PRP+HFSCS. At 8 weeks, this group exhibited significantly higher hair density compared to the PRP group (35% retained >75% density with PRP+HFSCS vs. 5% in PRP). This suggests that the catagen shedding and breakage of hair follicle was better controlled with addition of HFSCS, with direct continuation into anagen growth phase. Improved hair density and length can be attributed to the stronger hair shafts, reduced breakage and fragility due to an early transition to the anagen phase facilitated by stem cells.
However, at 1 year, the differences between the groups were not statistically significant, suggesting the need for further long-term evaluations. The presence of stem cells in the scalp tissue during recovery period could potentially lead to modified cross talk and improved environment via gene expression, signalling pathway activation, oxidative balance and alignment of cytoskeletal filaments [17]. Interestingly, our study revealed a remarkable effect on activation of dormant hair follicles and improvement in native hair growth. In PRP+HFSCS group, we observed hair growth beyond the transplant sites in the temporal, and vertex regions (Figure 2). This phenomenon was significantly evident at 8 weeks. More importantly, a larger number of patients in this group exhibited significant effect throughout the 1-year follow-up. Additionally, we also observed multirooted follicles in PRP+HFSCS group suggesting addition of stem cell may have led to activation of secondary hair roots around stem cell bulge region in otherwise dormant hair follicle unit. This was visually supported by serial trichoscopy, showing progressive vellus-to- terminal hair transformation over one year (Figure 3). All patients continued daily minoxidil use throughout the study period. Therefore, the observed improvements in native hair regrowth should be interpreted as effects additive to standard medical therapy rather than standalone treatment effects. The improvement in native hair growth is a dynamic variable, largely dependent on paracrine signalling, which may be active in some areas and less so in others. For example, treatments like minoxidil and finasteride often show better hair growth in the central region compared to frontotemporal region due to differences in hormonal sensitivity and follicular density.
This outcome highlights the potential of stem cell-induced paracrine signalling to stimulate androgen-sensitive native hair follicles, resulting in the regeneration of dormant secondary hair that were retained at 1 year. One plausible explanation for this could be due to role of stem cell-secreted bioactive molecules, including nucleic acids, extracellular vesicles (exosomes), proteins, collectively known as the secretome. There is a continuous cross talk amongst these molecules and native cells which plays vital role maintaining the hair growth, well-coordinated by HFSCs, vasculature, lymphatic capillaries, stromal cells, adipocytes, and immune regulators [18]. To illustrate these findings, authors propose a mechanism illustrating the role of stem cell in promoting hair growth (Figure 4).
Previous studies report that the effect of PRP weans off at around 9 months [19]. In our study, however, the significant benefit on native hair persisted at one year without addition of oral or topical finasteride. A limitation of our study design is the absence of a true placebo control group receiving FUE without any adjuvant therapy (PRP or HFSCS). This design choice was made on ethical grounds, as PRP therapy during hair transplantation is now considered standard practice with established benefits. Additionally, the control group received only a single PRP session during transplantation, whereas the recommended protocol typically involves monthly PRP sessions for 3 months. The drawbacks include lack of performing scalp biopsy and subsequent evaluation of stem cell markers. Furthermore, we evaluated only CD34 and CD45 stem cell markers, and other markers such as CK15, CD200 and CD44 with previously documented credibility in HFSCs population could not be studied [7].
Conclusion
The PRP+HFSCS demonstrated statistically significant (p<0.05) improvement in catagen fall, early improvement in hair density and length at 8 weeks. However, these improvements were statistically insignificant in transplanted hair at 1-year. On the contrary, there was statistically significant improvement (p<0.05) on native hair growth by dormant hair follicle activation in nontransplanted sites that persisted at one-year interval. It seems prudent to utilize PRP along with HFSCS as a cocktail of live cells to augment the sustainable regenerative results, though well researched multicentre trials are warranted.
• Figure Legend
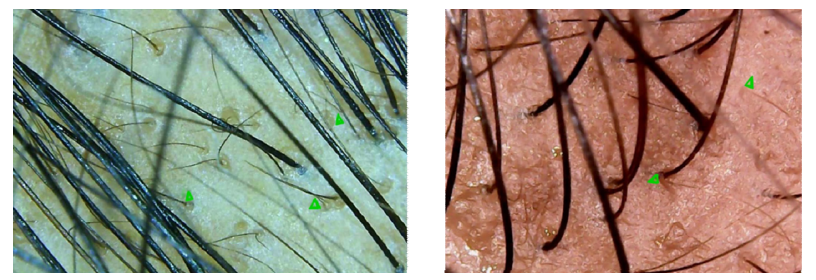
Figure 1: Trichoscopy at 8 weeks a) PRP + HFSCS group shows vellus hairs transitioning into intermediate hair {(Terminal + Intermediate + Vellus ) n = 47; T:I:V::21:8:18} b) PRP group demonstrating activity in vellus hair, visibly much less in number (n = 27; T: I: V ::12:8:7)
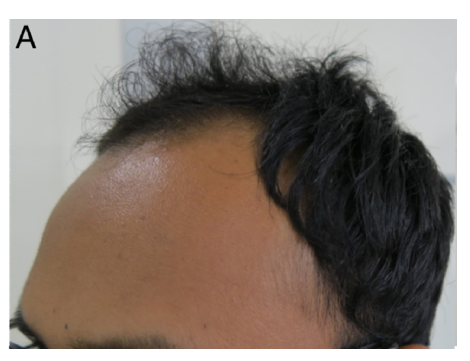
Figure 2A: Baseline image illustrates the initial state of hair density in a patient of grade IV Androgenetic Alopecia (AGA) before any treatment
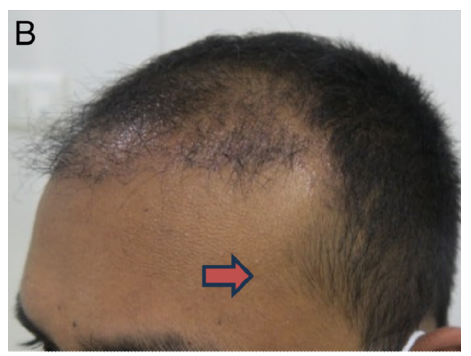
Figure 2B: 4 Months Post-Treatment image shows visible improvement in hair growth over the fronto-parietal area beyond transplanted sites, following hair transplant coupled with PRP+HFSCS at 4 months
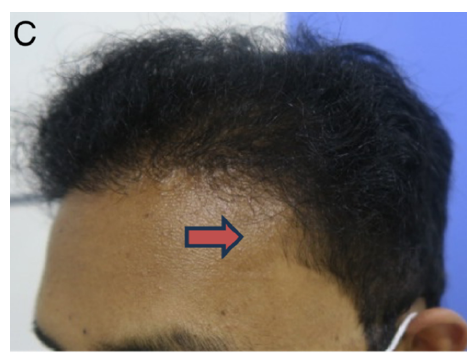
Figure 2C: One Year Post-Treatment image displays further improvement in hair growth over the fronto-parietal area beyond transplanted sites, demonstrating the effectiveness of the combined treatment over a one-year interval
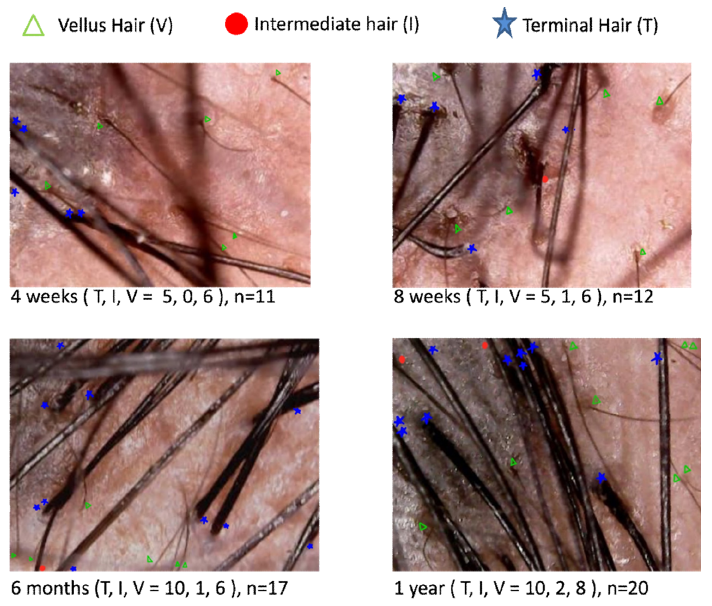
Figure 3: Trichoscopy demonstrating a visible increase in the density and number of terminal, intermediate, and vellus hairs in the PRP+HFSCS group at (a) 4 weeks, (b) 8 weeks, (c) 6 months, and (d) 1 year. Trichoscopy was performed at the same scalp site each time, using the melanocytic nevus in the background as a fixed reference point
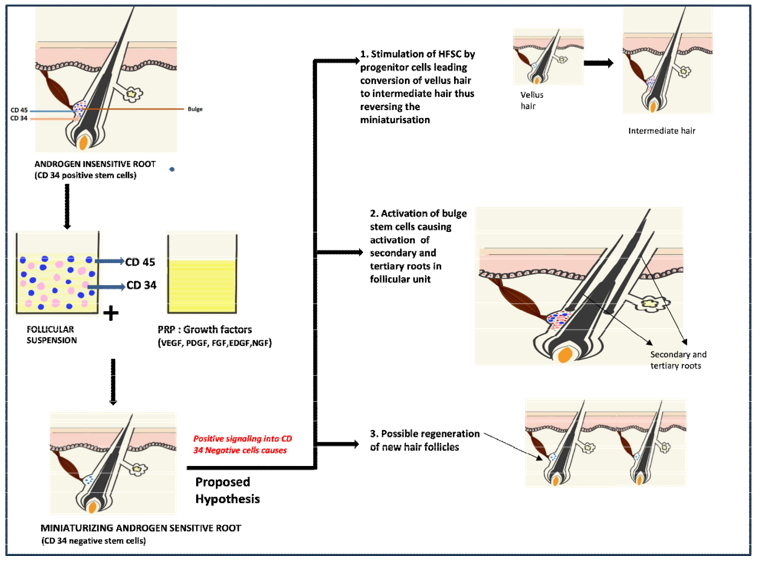
Figure 4: The diagram is demonstrating proposed hypothesis of PRP enriched CD34+ progenitor stem cell suspension in FUE hair transplant. The loss of progenitor stem cells contributes to progressive miniaturization and male pattern baldness. Follicular suspension containing CD34+ stem cells has potential to stimulate androgen-sensitive dormant native hair follicles, resulting in conversion of vellus hair into intermediate hair, activation of secondary and tertiary hair alongside the primary follicle and possible new hair formation in FUE hair transplant
Supplementary Table: Patient distribution at baseline by Hamilton-Norwood stages III-VII in PRP+HFSCS and PRP-only groups.
|
Baseline Hamilton-Norwood stage |
PRP+HFSCS group (No. of patients) |
PRP-only group No. of patients) |
|
Grade III |
5 |
5 |
|
Grade IV |
4 |
6 |
|
Grade V |
7 |
4 |
|
Grade VI |
3 |
4 |
Supplementary Table 1: Patient distribution at baseline by Hamilton-Norwood stages III-VII in PRP+HFSCS and PRP-only groups
Supplementary Figure 1: Consort flowchart diagram depicting phases of randomized trial of two groups (enrolment, intervention, allocation, follow-up and data analysis)
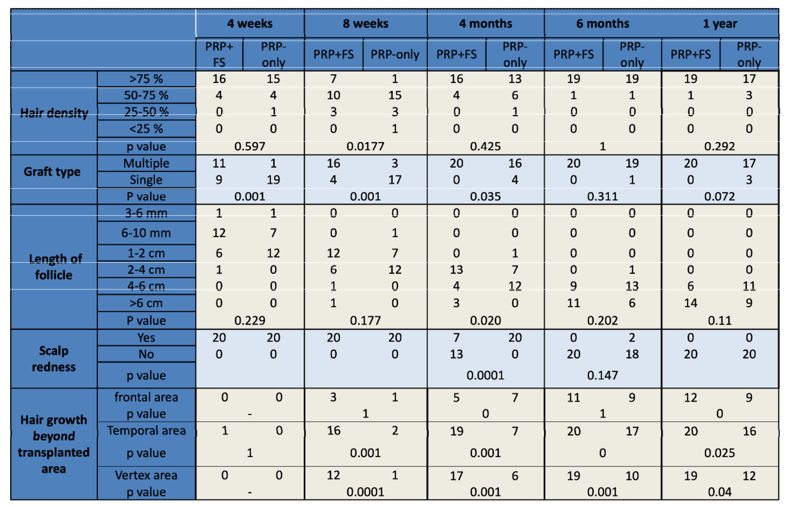
Supplementary Figure 2: Comparison of various parameters observed in two groups over a period of 4 weeks, 8 weeks, 4 months, 6 months and 1 year time period following FUE hair
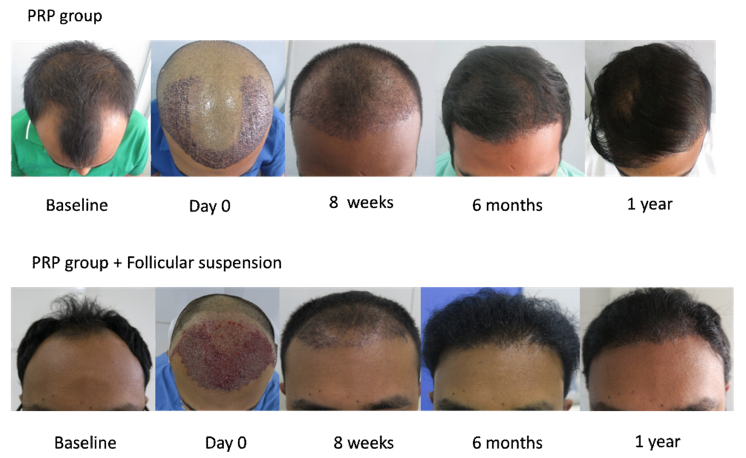
Supplementary Figure 3: Images showing sequential comparison of visible hair growth in PRP (AGA Grade IV) group versus PRP+HFSCS (AGA Grade IV) group at at baseline (pre-transplant), Day 0 (immediately post-transplant), 8 weeks, 6 months, and 1 year
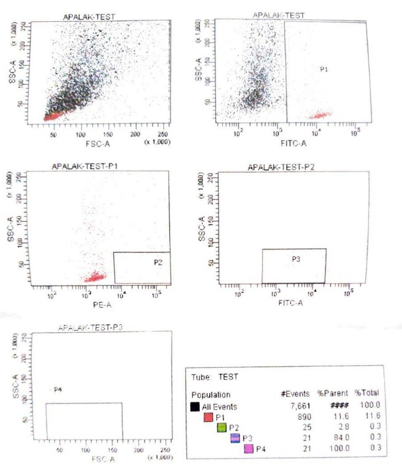
Supplementary Figure 4: Cells detected by flow cytometry FITC (fluorescein isothiocyanate) in hair follicle stem cells in vitro. The percentage of CD34 positive progenitor cells detected was 0.3%. The last figure with tube test depicts, P1-total CD45 cells, P2-CD34 positive cells, P3-dim CD45 positive cells, P4-stem cell count
References
- Paus R, Cotsarelis G. The Biology of Hair Follicles. N Engl J Med. 1999;341:491-497.
- Gentile P, Garcovich S. Systematic Review of Platelet- Rich Plasma Use in Androgenetic Alopecia Compared with Minoxidil®, Finasteride®, and Adult Stem Cell-Based Therapy. Int J Mol Sci. 2020;21(8):2702.
- Jiang S, Zhao L, Purandare B, et al. Differential expression of stem cell markers in human follicular bulge and interfollicular epidermal compartments. Histochem Cell Biol. 2010;133:455–465.
- Gentile P, Scioli MG, Bielli A, Orlandi A, Cervelli V. Stem cells from human hair follicles: first mechanical isolation for immediate autologous clinical use in androgenetic alopecia and hair loss. Stem Cell Investig. 2017;4:58.
- Kadry MH, El-Kheir WA, El-Sayed SM, El Shahid AR, Metwally HG. Autologous Adipose Derived Stem Cell versus Platelet Rich Plasma Injection in the Treatment of Androgenetic Alopecia: Efficacy, Side Effects and Safety. J Clin Exp Dermatol Res. 2018;9:447.
- Tak YJ, Lee SY, Cho AR, Kim YS. Arandomized, double-blind, vehicle-controlled clinical study of hair regeneration using adipose-derived stem cell constituent extract in androgenetic alopecia. Stem Cells Transl Med. 2020;9(8):839-849.
- Elmaadawi IH, Mohamed BM, Ibrahim ZAS, et al. Stem cell therapy as a novel therapeutic intervention for resistant cases of alopecia areata and androgenetic alopecia. J Dermatolog Treat. 2018;29(5):431-440.
- Garg S. Outcome of Intra-operative Injected Platelet-rich Plasma Therapy During Follicular Unit Extraction Hair Transplant: A Prospective Randomised Study in Forty Patients. J Cutan Aesthet Surg. 2016;9(3):157-164.
- Garg S, Manchanda S. Platelet-rich plasma-an ‘Elixir’ for treatment of alopecia: personal experience on 117 patients with review of literature. Stem Cell Investig. 2017;4:64.
- Li, K. N., Jain, P., He, C. H., Eun, F. C., Kang, S., & Tumbar, T. (2019). Skin vasculature and hair follicle cross- talking associated with stem cell activation and tissue homeostasis. Elife, 8, e45977.
- Yu H, Fang D, Kumar SM, et al. Isolation of a Novel Population of Multipotent Adult Stem Cells from Human Hair Follicles. Am J Pathol. 2006;168:1879–1888.
- Kang S, Chovatiya G, Tumbar T. Epigenetic control in skin development, homeostasis and injury repair. Exp Dermatol. 2019;28:453–463.
- Jiang S, Zhao L, Purandare B, et al. Differential expression of stem cell markers in human follicular bulge and interfollicular epidermal compartments. Histochem Cell Biol. 2010;133:455–465.
- Garza LA, Yang C-C, Zhao T, et al. Bald scalp in men with androgenetic alopecia retains hair follicle stem cells but lacks CD200-rich and CD34-positive hair follicle progenitor cells. J Clin Investig. 2011;121:613–622.
- Teumer J, Cooley J. Follicular Cell Implantation: An Emerging Cell Therapy for Hair Loss. Semin Plast Surg. 2005;19:193- 200.
- Cervelli, V., Garcovich, S., Bielli, A., Cervelli, G., Curcio, B. C., Scioli, M. G., ... & Gentile, P. (2014). The effect of autologous activated platelet rich plasma (AAâ?PRP) injection on pattern hair loss: clinical and histomorphometric evaluation. BioMed research international, 2014(1), 760709.
- Tian et al. Mechanoresponse of stem cells for vascular repair. World J Stem Cells. 2019;11(12):1104-1114.
- Egger, A., Tomic-Canic, M., & Tosti, A. (2020). Advances in stem cell-based therapy for hair loss. CellR4--repair, replacement, regeneration, & reprogramming, 8, e2894.
- Gkini MA, Kouskoukis AE, Tripsianis G, et al. Study of platelet-rich plasma injections in the treatment of androgenetic alopecia through an one-year period. J Cutan Aesthet Surg. 2014;7(4):213-219.


