Case Study - (2025) Volume 10, Issue 10
Effects of Multiple Sessions of Direct Current Brain Stimulation (tDCS) on Dysarthria Rehabilitation: A Scientific Review and Case Description
2Health and Disability Services, ARESS Fabiola, Termini Imerese, Palermo, Italy
3UOC Spinal Unit, Villa delle Ginestre Hospital, Provincial Health Authority Palermo, Italy
Received Date: Jun 06, 2025 / Accepted Date: Jul 15, 2025 / Published Date: Oct 06, 2025
Copyright: ©Copyright: Ã?©2025 Maria Luisa Capuana. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Capuana, M.L., Mandala, S., Saraniti, A., Tomasello, S., & Mandal
Abstract
Objective: To evaluate the effects of multiple sessions of transcranial direct current stimulation (tDCS) combined with speech ther-apy on dysarthria rehabilitation in a patient with acquired brain injury.
Introduction: Transcranial direct current stimulation (tDCS) is a non-invasive brain stimulation technique capable of modulating cortical excitability and influencing cognitive functioning. One major limitation of this method is the transient nature of its neuro-modulatory effects, which aim to promote neuroplasticity mechanisms. Emerging evidence suggests that repeated sessions are necessary to achieve sustained therapeutic outcomes.
Materials and Methods: We present the case of a 57-year-old male patient with tetraparesis and dysarthria resulting from organo-phosphate poisoning, who underwent a neuromodulation protocol consisting of multiple cycles of tDCS combined with speech therapy. Severity of dysarthria was assessed using the Italian version of the Bogenhausen Dysarthria Scales (BoDyS) at baseline and following each treatment cycle (T0–T4) [1].
Results: The patient tolerated the tDCS sessions well, with no reported adverse events. At baseline, speech intelligibility was severely compromised. Over a four-month treatment period, progressive improvements in phonation and articulation were documented. Quantitative evaluation showed BoDyS scores above 27 in all domains except prosody, which remained moderately impaired.
Conclusion: This case suggests that when integrated with multidisciplinary rehabilitation, tDCS may support functional recovery in dysarthria. These findings encourage further validation through randomized controlled trials to establish efficacy and guide treat-ment protocols.
Keywords
Transcranial direct current stimulation, Neuromodulation, Dysarthria, Rehabilitation, Speech therapy
Introduction
Transcranial direct current stimulation (tDCS) is a non-invasive brain stimulation technique capable of modulating cortical excitability and influencing cognitive functioning. One major limitation of this method is the transient nature of its neuromodulatory effects, which aim to promote neuroplasticity mechanisms. Emerging evidence suggests that repeated sessions are necessary to achieve sustained therapeutic outcomes [2]. tDCS is a portable, wearable brain stimulation technique that delivers low-intensity electrical currents to the scalp. Standard protocols typically consist of daily sessions lasting 20–30 minutes, five days a week, for two weeks, using currents ranging from 1 to 2 mA. Electrode-anodal (positive) and cathodal (negative) electrodes are positioned at targeted scalp locations, to influence specific cortical regions. While the current is insufficient to trigger action potentials, it can modulates neural activity by altering membrane potential, thereby affecting synaptic plasticity [3] and cortical excitability [4], both crucial for learning. When paired with behavioral training, tDCS may augment neuroplasticity and amplify the effects of rehabilitation on neural circuits. It has been studied in various conditions, including depression, schizophrenia, aphasia, epilepsy, addiction, chronic pain, and both cognitive and motor rehabilitation. Whitin the European Union, tDCS is approved for the treatment of pain and depression. Recent literature suggests that higher session frequency, prolonged session duration (up to three hours), and shorter inter-session intervals may enhance recovery through long-term potentiation (LTP) mechanisms [5–7]. In disorders of consciousness (DOC), multi-target and multi-session tDCS has been found to improve connectivity between affected and preserved cortical networks [8]. tDCS has also been investigated for its role in promoting neuroplasticity and rehabilitation in motor speech disorders such as dysarthria [4,9]. The presence of dysarthria has a marked effect on both communication ability and the life situation of patients with neurologically induced pathologies. Traditional speech therapy is mainstay of treatment; however, results are often unsatisfactory, especially in chronic cases. tDCS is often administered concurrently with individualized cognitive and speech therapy to potentially enhance outcomes [10,11]. Most consistent effects on language outcomes regard high frequency repetitive Trancranial Magnetic Stimulation (rTMS) applied to the lesioned hemisphere. Similarly, therapeutic benefits have also been reported following tDCS, although the findings are less consistent than is the case with rTMS. Improved articulatory function and speech intelligibility has been observed with excitatory rTMS in Parkinson's disease. The combined use of brain stimulation techniques and conventional therapies may represent an innovative future direction for treating acquired communication disorders [12]. The effectiveness of tDCS in enhancing cerebral blood flow, neuroplasticity, motor rehabilitation and treatment of dysarthria and other speech and language disorders has been been corroborated by neuroimaging [11]. As regard stimulation parameters and protocols and about electrode placement, Most studies targeting speech, use the left primary motor cortex or Broca’s area with the cathode over the contralateral supraorbital region. Common protocols apply 1–2 mA for 15–30 minutes per session over 5–10 sessions. There is also some more (but not only anecdotal) evidence for the combination of tDCS and speech therapy in post-stroke dysarthria, from randomized controlled trials (RCTs). For instance, a double-blind trial with twelve patients following acute middle cerebral artery infarction demonstrated that anodal tDCS (2 mA, 30 min, 5 times/week for 2 weeks), when combined with speech therapy, significantly improved phonation time and speech motor rates compared to sham stimulation [13]. Additionally, a randomised pilot study showed that anodal tDCS targeting the left inferior primary motor cortex (2 mA, 15 minutes, 10 sessions) improved speech intelligibility and kinematic parameters of speech production in chronic post-stroke dysarthric patients, compared to sham stimulation, suggesting enhanced speech motor control [11]. Improvements have been noted across speech subsystems, including phonation, articulation, and velar movement, with some studies reporting better speech intelligibility and functional communication [13,14]. Despite promising results, few studies have specifically examined efficacy in dysarthria rehabilitation, and results remain inconsistent. Here, we describe a case of dysarthria treated with multiple tDCS sessions combined with speech therapy, aiming to report preliminary outcomes and contribute to the existing evidence base.
Materials and Methods
A 57-year-old male with tetraparesis and dysarthria secondary to organophosphate poisoning was admitted for intensive neuromotor rehabilitation on May 21, 2025, fifteen days after the acute event. He had previously been hospitalized in the intensive care and internal medicine units, where he gradually regained consciousness, basic motor responses, and limited vocalizations. At admission, the patient was alert and cooperative, with persistent severe dysarthria. A speech-language pathology assessment revealed marked deficits in articulation, prosody, and speech intelligibility, despite preserved respiratory and facial motor control. Previous spontaneous recovery had plateaued in the preceding week, with no significant improvements in speech production prior to transfer. After informed consent, the patient underwent a neuromodulation protocol combining anodal tDCS and speech therapy. The stimulation was delivered at 2 mA over the left primary motor cortex (M1, site C3) for 30 minutes per session, five times a week. Electrodes (25 cm²) were placed with the cathode over the contralateral supraorbital area. Two consecutive two-week cycles of tDCS were administered during inpatient rehabilitation, interspersed with two-week breaks during which speech therapy continued. Two additional outpatient cycles (each consisting of 10 sessions) were performed over the following months, following the same parameters. Dysarthria severity was assessed using the Italian version of the BoDyS scale at baseline (T0) and after each treatment cycle (T1-T4). A follow-up assessment was conducted one month post-treatment.
Results
The patient completed all tDCS sessions without adverse effects. At baseline (T0), speech was barely intelligible, with marked pneumophonoarticulatory incoordination, low vocal pitch, hypoarticulation, slow vocal rhythm, and poor prosodic modulation. Severely impaired domains (score <16) included prosody, rhythm, and articulation. Subsequent evaluations showed progressive improvements in tone, rhythm, intensity, and vocal quality, with BoDyS scores increasing to 24–30. By the end of the treatment, a notable reduction also in symptom frequency was observed, with BoDyS scores exceeding 27 across all domains except prosody, which, while improved, remained moderately impaired (score 25). Assessment with the BoDyS scale at one month following the final tDCS cycle demonstrated sustained therapeutic gains. See Table 1 [Table 1. Progression of Vocal Parameter Scores] for quantitative evaluations of vocal parameters at four different timepoints (T1–T4) and at one-month follow-up.
|
Parameter |
T1 |
T2 |
T3 |
T4 |
Follow-Up (1 Month) |
|
Breathing |
20 |
24 |
27 |
30 |
30 |
|
Pitch and Vocal Intensity |
24 |
24 |
27 |
30 |
31 |
|
Vocal Quality |
20 |
26 |
27 |
32 |
32 |
|
Vocal Stability |
20 |
27 |
27 |
30 |
30 |
|
Articulation |
10 |
29 |
30 |
30 |
30 |
|
Nasality |
26 |
30 |
30 |
31 |
30 |
|
Articulatory rate |
10 |
27 |
27 |
30 |
30 |
|
Fluency |
20 |
35 |
37 |
37 |
35 |
|
Prosodic Modulation |
10 |
15 |
25 |
25 |
25 |
Table 1: Progression of vocal parameter scores across four evaluation timepoints (T1–T4) and at one-month follow-up. Each line represents the performance trend in specific domains such as breathing, pitch and intensity, vocal quality, and prosodic modulation
Scores are expressed on the BoDys clinical scale. See Figure 1 (Figure 1). Trends in vocal function parameters across timepoints] for a graphic representation of trends in vocal function parameters across five timepoints (T1 to 1-month follow-up).
The graph displays scores for nine vocal domains: breathing, pitch and vocal intensity, voice quality, vocal stability, articulation, nasality, articulatory rate, fluency, and prosodic modulation. Each colored line represents a different evaluation timepoint: T1 (after the first tDCS cycle), T2, T3, T4, and 1-month follow-up. Higher scores indicate better performance.
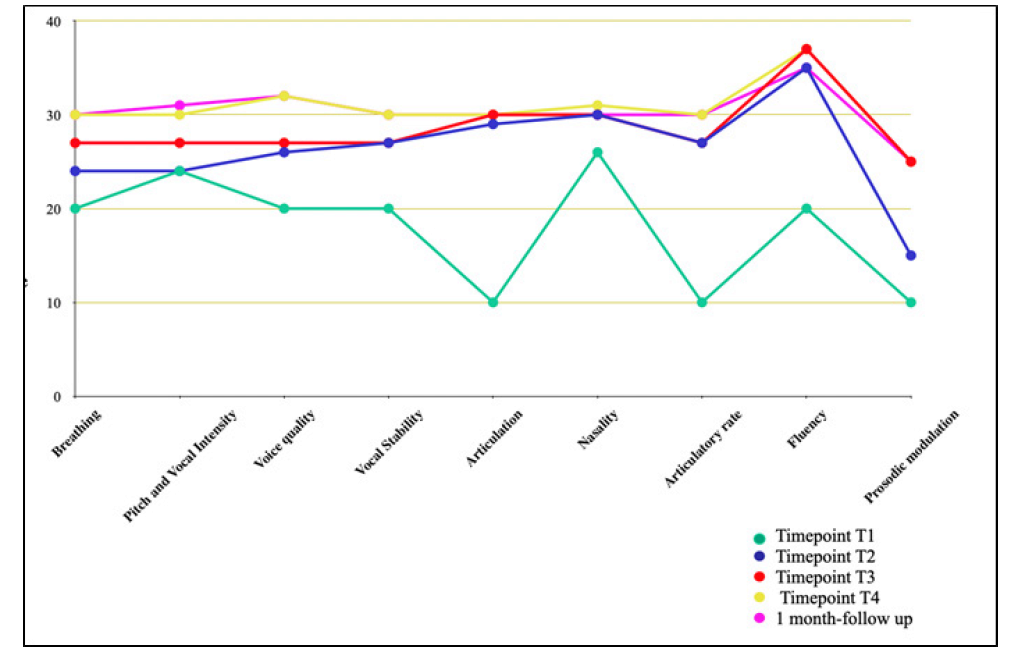
Figure 1: Trends in vocal function parameters across five timepoints (T1 to 1-month follow-up). The graph displays scores for nine vocal domains: breathing, pitch and vocal intensity, voice quality, vocal stability, articulation, nasality, articulatory rate, fluency, and prosodic modulation. Each colored line represents a different evaluation timepoint and 1-month follow-up. Higher scores indicate better performance.
Discussion
This case demonstrates that anodal tDCS over M1 may augment the effects of conventional speech therapy in dysarthria. As a motor speech disorder commonly resulting from stroke or traumatic brain injury, dysarthria affects articulation, phonation, and prosody, severely impacting communication and quality of life. Although traditional speech therapy remains the standard of care, outcomes are often limited, particularly in chronic cases. tDCS is gaining attention for its potential to enhance neuroplasticity and motor recovery, including speech function [9]. Several RCTs have shown positive outcomes when combining tDCS with speech therapy in post-stroke dysarthria. For example, a trial involving 12 patients with acute middle cerebral artery infarction found that anodal tDCS (2 mA, 30 min, 5 sessions/week for 2 weeks), significantly improved phonation time, alternative and sequential motion rates compared to sham [13]. A randomized pilot study involving nine chronic post-stroke dysarthric patients also demonstrated improved speech intelligibility and motor control after ten sessions of anodal tDCS targeting the left primary motor cortex. These findings suggest tDCS can improve dysarthria outcomes also in a chronic phase of disease [11]. In traumatic brain injury (TBI), case reports indicate that tDCS paired with targeted speech therapy programs, like “Be Clear,” can improve speech intelligibility and reduce symptoms like palilalia, with effects persisting for months [15]. Despite these encouraging results, existing studies vary widely in protocol, participant characteristics, and outcome measures. Many suffer from small sample sizes and methodological limitations, including potential bias and confounding factors. Some observed benefits may reflect spontaneous recovery rather than treatment effects. Meta-analyses have found no significant differences between active and sham stimulation in some dysarthria measures, due to these limitations [9]. We present this case to highlight a favorable clinical outcome in a domain where high-quality evidence is lacking. Although causality cannot be established from a single case, the observed speech improvements followed a temporally coherent pattern after the application of tDCS, in a patient who had already reached a clinical plateau. The patient’s spontaneous recovery was limited in the week preceding rehabilitation admission, with persistent severe dysarthria despite intact consciousness and cooperation. The improvements recorded across phonation, articulation, and intelligibility coincided with the neuromodulation phases, and were sustained at follow-up. These factors strengthen the plausibility of a treatment effect, particularly considering the subacute-to-early chronic stage of intervention, and the use of a standardized, targeted protocol. Additionally, we aim to stress the potential of using prolonged stimulation protocols to maintain therapeutic gains. To our knowledge, there are no studies exploring positive effects of prolonged and multi-cycle tDCS stimulation protocols in dysarthria treatment. Although rapid improvement were observed, prosody remained partially impaired. We hypothesize that this could be due to the right hemisphere’s role in prosodic control, which was not targeted in this protocol. The lack of improvement in prosodic features suggests domain-specific limitations, possibly reflecting the need for more comprehensive stimulation strategies, including targeting of right-hemisphere regions. Literature supports the link between right hemisphere damage and aprosodia, affecting both expressive and receptive prosody [16, 17]. A multi-target stimulation approach including right hemisphere regions would likely have provided additional benefits.
Conclusion
When integrated into multidisciplinary rehabilitation, neuromodulation techniques like tDCS may support and enhance speech recovery by promoting synaptic plasticity. Although preliminary findings are encouraging, further high-quality studies—particularly randomized controlled trials with standardized protocols and larger cohorts—are necessary to validate and optimize tDCS in dysarthria rehabilitation.
Authors Contribution
Conceptualization, MLC; resources, AS and ST; data curation, MLC; writing—original draft preparation, MLC and SM; validation, writing—review and editing, MLC and GM. All authors have read and agreed to the published version of the manuscript.
Conflict of Interest
The authors declare no conflict of interest.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
References
- Ziegler, W., Staiger, A., Schölderle, T., & Vogel, M. (2017). Gauging the auditory dimensions of dysarthric impairment: Reliability and construct validity of the Bogenhausen Dysarthria Scales (BoDyS). Journal of Speech, Language, and Hearing Research, 60(6), 1516-1534.
- Goldsworthy, M. R., & Hordacre, B. (2017). Dose dependency of transcranial direct current stimulation: implications for neuroplasticity induction in health and disease. The Journal of physiology, 595(11), 3265.
- Kronberg, G., Rahman, A., Sharma, M., Bikson, M., & Parra, L. C. (2020). Direct current stimulation boosts hebbian plasticity in vitro. Brain Stimulation, 13(2), 287-301.
- Tarantino, V., Fontana, M. L., Buttà, A., Ficile, S., Oliveri, M., Mandalà, G., & Smirni, D. (2024). Increase in EEG alpha-to-theta ratio after transcranial direct current stimulation (tDCS) in patients with disorders of consciousness: a pilot study. NeuroRehabilitation, 55(4), 440-447.
- Monte-Silva, K., Kuo, M. F., Hessenthaler, S., Fresnoza, S., Liebetanz, D., Paulus, W., & Nitsche, M. A. (2013). Induction of late LTP-like plasticity in the human motor cortex by repeated non-invasive brain stimulation. Brain stimulation, 6(3), 424-432.
- Agboada, D., Mosayebi-Samani, M., Kuo, M. F., & Nitsche, M. A. (2020). Induction of long-term potentiation-like plasticity in the primary motor cortex with repeated anodal transcranial direct current stimulation–better effects with intensified protocols?. Brain stimulation, 13(4), 987-997.
- Moirand, R., Imbert, L., Haesebaert, F., Chesnoy, G., Bediou, B., Poulet, E., & Brunelin, J. (2022). Ten sessions of 30 min tDCS over 5 days to achieve remission in depression: a randomized pilot study. Journal of Clinical Medicine, 11(3), 782.
- Zhang, X., Liu, B., Li, Y., Duan, G., Hou, J., & Wu, D. (2021). Multi-target and multi-session transcranial direct current stimulation in patients with prolonged disorders of consciousness: a controlled study. Frontiers in Neuroscience, 15, 641951.
- Balzan, P., Tattersall, C., & Palmer, R. (2022). Non-invasive brain stimulation for treating neurogenic dysarthria: A systematic review. Annals of physical and rehabilitation medicine, 65(5), 101580.
- Mandala, G. (2023). Rehabilitation of motor and cognitive functions in patients with right cerebral hemisphere stroke. American Journal of Medical and Clinical Research & Reviews, 2(8), 1-4.
- Wong, M. N., Baig, F. N., Chan, Y. K., Ng, M. L., Zhu, F. F., & Kwan, J. S. K. (2022). Transcranial direct current stimulation over the primary motor cortex improves speech production in post-stroke dysarthric speakers: A randomized pilot study. Plos one, 17(10), e0275779.
- Murdoch, B. E., & Barwood, C. H. (2013). Non-invasive brain stimulation: a new frontier in the treatment of neurogenic speech-language disorders. International Journal of Speech-Language Pathology, 15(3), 234-244.
- You, D. S., Chun, M. H., Kim, D. Y., Han, E. Y., & Jung, S. E. (2010). The Effects of Transcranial Direct Current Stimulation on Dysarthria in Stroke Patients. Journal of the Korean Academy of Rehabilitation Medicine, 34(1), 10-14.
- Rossini Peters, R., Cielo, C. A., & Pagliarin, K. C. (2025). Transcranial direct current stimulation on speech intelligibility and acoustic analysis in Parkinson’s disease: a pilot study. Journal of Clinical Practice in Speech-Language Pathology, 27(1), 53-64.
- Bayat, M., Sabeti, M., Rao, K. S., & Nami, M. (2020). The clinical outcome of concurrent speech therapy and transcranial direct current stimulation in dysarthria and palilalia following traumatic brain injury: A case study. NeuroRegulation, 7(3), 118-118.
- Sheppard, S. M., Stockbridge, M. D., Keator, L. M., Murray, L. L., Blake, M. L., Right Hemisphere Damage working group, & Evidence-Based Clinical Research Committee. (2022). The company prosodic deficits keep following right hemisphere stroke: a systematic review. Journal of the International Neuropsychological Society, 28(10), 1075-1090.
- Mandalà S, Capuana ML, Mandalà G. Transcranial Direct Current Stimulation in the Treatment of Autism: Literature Review. Int Clin Med Case Rep Jour (ICMCRJ) 2025, 4, 5.



