Research Article - (2025) Volume 3, Issue 1
Effect of Trachyspermum Ammi (Ajwain) Fruit's Aqueous Extract on Atropine Induced Dry Eye in Male Mice
2Faculty of pharmacy, Department of pharma- ceutics, Azad university of Medical Sciences, Tehran, Iran
3Herbal Pharmacology Research Center, School of Medicine, Dept. of Pharmacology, Tehran Medical Scien, Iran
Received Date: Jan 17, 2025 / Accepted Date: Feb 03, 2025 / Published Date: Feb 07, 2025
Copyright: ©Â©2025 Ayda Chavoshi Aghdam, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Aghdam. A. C., Zandkarimi. M., Jahromy. H. M. (2025). Effect of Trachyspermum ammi (Ajwain) fruit's aqueous extract on atropine induced dry eye in male mice, Trans Med OA, 3(1), 01-08.
Abstract
Dry eye syndrome (DES) is a common eye disorder that is described as a tear film disorder caused by reduced tear production, poor tear quality, or excessive tear evaporation. This disorder is associated with symptoms of eye discomfort such as irritation, foreign body sensation, redness and itching. This disease affects the lives of millions of people and its prevalence reaches 14.5%, which is still increasing. Many evidences indicate that the increase in tear film osmolality and ocular surface inflammation caused by T cells play a role in the pathogenesis of the disease. However, the exact pathogenesis of DES is not fully understood. Oxidative stress has recently been found to play a prominent role in the development of dry eye.
Purpose of study: To investigate the therapeutic effects of topical administration of the aqueous extract of the fruit of the trachyspermum ammi (TA) plant with antioxidant effect in the experimental model of dry eye in adult male mice.
Method: In the first step the aqueous extract was prepared from the fruits of the TA. After checking the pH, osmolality and total phenol content (TPC) of the extract by Folin Ciocalteu (FC) method, three different percentages of the extract were prepared. After development of dry eyes using atropine, sterile eye drops containing solvent and extract with percentages of 1%, 2.5% and 5% were used in different groups. Eye surface moisture level was assessed using Schirmer's test (ST). After the study, the mice were euthanized and a number of corneas from each group were collected for pathological examination and comparison with the control group.
Results: According to the pathological examinations and statistical data from ST, there was no significant decrease in the moisture content of the eye surface of the control group. But over time, the moisture level of the eye surface in the atropine and atropine with solvent group, decreased significantly and the destruction of the epithelium was observed much more significant. In the atropine plus 1% extract group and atropine plus 2.5% extract group, the decrease in eye surface moisture was less and slighter inflammation and destruction were observed than groups without treatment. In the atropine with 5% extract group, according to the results of the ST (p-value<0.001), the moisture of the eye surface did not decrease significantly over time, and the results were much closer to the control group and the destruction of the epithelium and the infiltration of inflammatory cells caused by dry eye were insignificant. Conclusion Topical use of eye drops containing 5% extract of TA as a medicinal plant improved clinical symptoms, reduced inflammation, and DES induced damages in the eye of mice based on statistical evaluations using ST results (p-value<0.001) and pathological examinations.
Keywords
Dry Eye, Aqueous Extract of Trachyspermum Ammi, Corneal Epithelium; Schirmer's Test, Total Phenol Content
Abbreviations DES
Dry eye syndrome, FC: Folin Ciocalteu, TA: trachyspermum ammi, ST: Schirmer's test, TPC: total phenol content, H&E: Hematoxylin and Eosin, GA: Gallic acid, UV: ultra violet, GAE: Gallic acid equivalent, DW: distilled water, ANOVA: analysis of variance
Introduction
DES is a multifactorial disease of the eye surface that occurs as a result of a decrease in tear production or an increase in tear evaporation from the eye surface and is associated with discomfort, burning sensation, blurred vision, visual impairment, tear film instability and possible damage to the eye surface. Therefore, it reduces the quality of life of people [1-2]. It is estimated that 100 million people worldwide may be affected by dry eye. It is also reported that the global prevalence of dry eye ranges from 5 to 34%, so it is one of the most common eye diseases. Therefore, effective treatment for dry eyes has become an issue that needs more attention [2].
Various environmental factors such as dry air, smoke, allergens, systemic diseases and age lead to dry eyes. This disease has increased in recent years with long-term use of computers and is one of the most important reasons for visiting eye clinics, which is characterized by symptoms such as burning, redness, scratching, and the feeling of a foreign body in the eye [2,3,4]. Dry eye has a multifactorial etiology with challenging treatment. The two main types of dry eye are: aqueous-deficient dry eye and evaporative dry eye [5]. Regardless of the types, the pathologic events involved in ocular surface injury are similar.
It has been proven that the underlying pathogenesis of DES involves an inflammatory process mediated by T lymphocytes [1,6]. The two main mechanisms of DES are tear hyperosmolarity and tear film instability [7]. Increased evaporation or decreased tear flow leads to tear hyperosmolarity, which stimulates the production of pro-inflammatory cytokines. Inflammatory mediators cause the destruction of goblet cells, decrease in mucin production and subsequent tear film instability. Since DES is associated with ocular surface inflammation and tear hyperosmolarity is known to be an effective factor in ocular surface inflammation [8]. drops containing anti-inflammatory agents may effectively improve the signs and symptoms of dry eye [7,8]. Therefore, we decided to select a plant and evaluate its effectiveness in improving dry eye in order to take a step forward in treating this common problem.
Dry eye treatment is based on tear replacement. Newer treatments focus on specific drugs such as lubricants, antibiotics, immuno- suppressants, and new anti-inflammatory drugs. Meanwhile, herb- al treatments have been used in the treatment of various diseases due to their low cost and less side effects [4,9]. There are several formulations of lubricating eye drops that can adequately relieve the symptoms of dryness and serve therapeutic purposes Consid- ering that the mechanism of dry eye is an inflammatory process, artificial tear formulations containing anti-inflammatory agents may effectively improve the signs and symptoms of dry eye [9- 11]. However, few clinical trials have been conducted to study the effect of herbal medicines on dry eye [2]. Herbal remedies for the prevention and treatment of dry eye are currently under examina- tion [12]. In addition, the beneficial effects of herbal medicines such as antioxidant, anti-inflammatory and antimicrobial activities may enhance the healing process [13]. Compared to artificial tear drops, herbal remedies can be useful supplements to relieve dry eye symptoms. Of course, it should be noted that excessive use of some herbal medicines may have adverse effects. Therefore, the side effects of these drugs should also be carefully considered [2].
Trachyspermum Ammi (TA)
TA, also known as Ajwain, is an annual plant belonging to the medicinally valuable family, Apiaceae. Ajwain is widely cultivated in different regions such as Iran, Pakistan, Afghanistan and India as well as Europe, while it is native to Egypt. Usually, the grayish- brown fruits of Ajwain are used for medical and nutritional purposes. Considering that the mechanism of dry eye is an inflammatory process, the formulation of artificial tears containing anti-inflammatory agents may effectively improve the signs and symptoms of dry eye. Ajwain has been used in traditional medicine systems for various medicinal and medical aspects. Fenugreek has also been evaluated for its anti-inflammatory effect. Based on this, both total alcoholic extract and total aqueous extract have a significant anti-inflammatory effect in vivo [14].
Generally, medicinal plants increase tear film stability by reducing osmolality and increasing tear production. Several mechanisms including reduction of reactive oxygen species or ROS (antioxidant activity), prevention of cell apoptosis and modulation of inflammatory factors can affect tear glands and membrane cells and thus help to treat dry eye [4,11].
In this article we evaluated the effect of TA aqueous extract on experimental atropine induced dry eye in male mice. ST have been used to determine eye surface moisture and tear production. TPC was measured by FC method using its specific reagent and uv- visible spectroscopy to estimate antioxidant and anti-inflammatory effects of extract. Histopathological changes of cornea have been also evaluated by microscopical tissue assessment using Hematoxylin and eosin (H&E) staining procedure. Various effects of Ajwain have been investigated in recent studies [14]. However, this research discusses a new issue in order to investigate the effects of Ajwain in dry eye.
Experimental and Method
Development of Experimental Dry Eye Model in Mice To check the effectiveness of the dry eye model, 14 adult male mice weighing 20-25 grams were prepared. After a few days to adapt to the laboratory environment, the mice were divided into two groups. A group as control group did not receive any substance. The second group received daily atropine eye drops to cause dry eye. The moisture of the eye surface in the control group and the atropine group was measured before receiving the drops on days 1, 3 and 5 with ST paper.
TA's (Ajwain) fruit from West Azerbaijan province was prepared from herbal medicine store in Tehran. After confirming the authenticity and quality of the fruits, the extraction process started. The fruits of the plant were ground first. The obtained powder was mixed with distilled water (DW) (100 grams of ajwain with 1000 cc equivalent to 1 liter of DW) at a weight-volume ratio of one tenth. Then it was soaked in a hot water bath below the boiling point for 15 to 20 minutes, and after filtering the obtained solution, the solution was again placed in a hot water bath (below the boiling point) to evaporate the excess water and finally the brown colored extract was obtained. We poured the extract on the glass and let the water evaporate completely. The obtained powder was collected and 5 grams of it was dissolved in 25 cc of DW.
Measurement of TPC
Total phenolic content was determined using the FC method. To perform this method, FC reagent, 7.5% sodium carbonate solution and Gallic acid (GA) are required as standards. --Preparation of sodium carbonate solution To prepare a 7.5% solution, 7.5 grams of sodium carbonate were weighed and dissolved with DW up to 100 ml in a volumetric flask. -- Preparation of stock and standard solutions of GA To prepare the stock solution, 0.250 grams of GA powder produced by Merck co. was weighed and then dissolving in DW up to 100 ml in a flask. GA standard solutions with concentrations of 0.25, 0.5, 0.75, 1 and 1.25 mg/ml were prepared from the stock solution. (respectively by diluting 1, 2, 3, 4 and 5 ml of the stock solution with distilled water up to 10 ml)
FC Method and Ultra Violet (Uv) Spectroscopy
A small amount of extract (0.5 ml) was poured into the test tube. Then, 0.5 ml of FC reagent from SIGMA-ALDRICH was added. After a few minutes, 1.5 ml of 7.5% sodium carbonate solution was added to the mixture and the volume was made up to 10 ml with DW, and it was placed in a dark environment for two hours to be checked in a UV Spectrophotometer. This process was also done for standard solutions and blank solution.
Preparation of Blank Solution
The blank solution was prepared using 0.5 ml of FC reagent, 1.5 ml of 7.5% sodium carbonate and DW to a volume of 10 ml. The final solution was kept in a dark environment for two hours.
Uv-Visible Spectroscopy
After preparing the solutions and the UV-spectrophotometry device, a small amount of the standard GA solution was placed in the device to check the maximum absorption, which was measured as the wavelength of 722.5 nm. According to similar studies, this number varies between 720 and 760 nm.
Then, by adjusting the device, the absorbance of the standard solutions and the sample was measured, and the concentration was calculated using the calibration curve equation of the standards, and the results are reported as milligrams GA equivalent (GAE) per gram of dried extract powder. This process was repeated 3 times with samples and standard solutions to ensure the accuracy of the method used. It should be noted that due to the high concentration of samples and standards, all solutions were diluted 10 times before being placed in the cell of the device.
Preparation of Eye Drops
After extraction, three different percentages were diluted using normal saline, and the pH of the extract and the diluted percentages were measured. which varied between 6 and 7, so they were suitable for use in the eyes.
Osmolality Measurement
The osmolality of Diluted percentages and bulk extract were adjusted using normal saline and measured by the Wescor Vapor Pressure Osmometer VAPRO (Model 5520) device in the brain and cognition research laboratory in tarbiat modarres university and results were reported as millimole/kilogram (table1). The method of osmolality Determination was vapor pressure. At the first step the device was calibrated by 290 mmol/kg standard solution. Then a 10 microliter specimen is aspirated into a micropipette tip. The specimen is then inoculated into a solute- free paper disc in the sample holder, whereupon the sample holder is pushed into the instrument and the sample chamber is locked. Locking initiates, the automatic measurement sequence. At the end of each measurement, the micropipette tip and paper disc were replaced and the chamber was washed out.
Animal Study
42 adult male mice with a weight of 20 to 25 grams were prepared to investigate the effectiveness of the TA's fruit extract in atropine- induced dry eye. The mice were divided into 6 groups of 7 and after two days of keeping them in the laboratory to adapt to the environmental conditions, the study began.
Therefore, the first group or healthy control did not receive any drops. The second group received only atropine, the third group received atropine and solvent, the fourth group received atropine and 1% extract, the fifth group received atropine and 2.5% extract, and finally the sixth group received atropine and 5% extract.
The study was continued for 18 days until the desired results were reached, after which the mice were euthanized and a number of eyes were isolated from each group and collected for histopathological examination and preserved in formalin.
Histopathological Study
After separating the cornea, a tissue cross section was prepared from each sample. Tissue sections were stained with hematoxylin-eosin method and slides were prepared for microscopic examination.
Statistical Analysis
The results were expressed as average amount of each group from ST results. The data were subjected to one-way analysis of variance (ANOVA). P values <0.05 were considered significant.
Results
Dry Eye Model
The results of a pilot study of experimental dry eye in male mice with atropine drops are shown in table 1. It is obvious that after 5 days of daily use of atropine drops, dry eye has been developed
|
|
Day 1 |
Day 3 |
Day 5 |
|
Control group |
2.4 |
1.83 |
1.65 |
|
Atropine group |
2 |
1.05 |
0.16 |
Table 1: ST Average Results in Animal Model of Dry Eye Development Measured in Atropine and Control Group. Dry Eye has been Developed by using Atropine Eye Drop After 5 days
Fc Method and Uv Spectroscopy
GA maximum absorption wavelength: The maximum wavelength of GA absorption in FC method was measured 722.5nm, which is in the reported range by previous studies.
Tpc Assessment
As directed the absorbance of the standard solutions and the sample was measured, this process was repeated 3 times to ensure the accuracy of the method and the concentration was calculated using the calibration curve. and the results were reported as mgGAE/g. Based on the samples` concentrations average TPC is calculated 24.78 mgGAE/g extract dry powder.
Osmolality measurement results are presented in table 2. The osmolality was adjusted by using normal saline and was suitable for administration of solutions as eye drops.
|
No. |
Sample |
Osmolality (mmol/kg) |
|
1 |
Bulk extract |
383 |
|
2 |
%1 |
272 |
|
3 |
%2.5 |
283 |
|
4 |
%5 |
297 |
Table 2: Osmolality of Bulk Aqueous Extract is Measured 383 mmol/kg, While Osmolality of%1, %2.5 and %5 extract is 272, 283 and 297 mmol/kg of Solution Respectively
ST Results
Total of 6 groups were included in the study, which were evaluated 1, 4, 7, 10, 13, 16, and 18 days after the intervention by ST. According to statistical data from ST (p-value<0.001), there was no significant decrease in the moisture content of the eye surface of the control group. But over time, the moisture level of the eye surface in the atropine and atropine with solvent group, decreased significantly. In the atropine plus 1% extract group and atropine plus 2.5% extract group, the decrease in eye surface moisture was much less and slighter inflammation and destruction were observed than groups without treatment. In the group receiving atropine plus 5% extract, according to the results of the ST, the moisture of the eye surface did not decrease significantly over time, and the results were much closer to the control group (table 3).
|
Groups / Days |
1 |
4 |
7 |
10 |
13 |
16 |
18 |
|
control |
2.13 |
2.05 |
2.13 |
1.93 |
2.12 |
2.08 |
2.05 |
|
atropine |
1.93 |
1.07 |
0.23 |
0 |
0 |
0 |
0 |
|
atropine and solvent |
1.93 |
1.33 |
0.43 |
0.17 |
0 |
0 |
0 |
|
Atropine and 1% extract |
2.13 |
2.05 |
1.27 |
1.32 |
1.43 |
1.45 |
1.57 |
|
Atropine and 2.5% extract |
2.05 |
1.88 |
1.65 |
1.53 |
1.68 |
1.82 |
1.82 |
|
Atropine and 5% extract |
1.98 |
1.97 |
2 |
1.95 |
2.05 |
2.02 |
2.08 |
Table 3: Schirmer's test (ST) Average Results Based on Daily Evaluation on Days 1, 4, 7, 10,13, 16, 18 of Study Demonstrates %5 Extract Group Has the Most Optimal Impact on Eye Surface Moisture
Accordingly figure 1 compares the test results, which confirms the atropine plus %5 extract has shown the best improvement over time. Although the groups receiving %2.5 and %1 extract has shown few improvements in eye moisture level, they have been less effective
Figure 1: Eye Surface Moisture Level Based on Schirmer's Test (ST) Comparing Different Groups of Study.(*: p-value<0.01,
**:p-value<0.05 and ***:p-value<0.001)
Histopathology Evaluations
Photomicrograph of a section of cornea from the control group, shows the stratified squamous nonkeratinized corneal Epithelium formed of normal cell layers. The basal epithelial cells are columnar with oval nuclei resting on Bowman’s membrane. The Stroma is formed of parallel arranged collagen bundles with spindle-shaped keratocytes in between (Figure 2A).
In the histopathological examination of the atropine group, inflammation of the epithelium and infiltration of inflammatory cells, degeneration and necrosis are significant and extensive. In the stroma area, the collagen fibers are very distant and the fibers are disintegrated. The nuclei of epithelial cells are hypertrophied in areas without necrosis. Keratinocytes are destroyed and necrotic (Figure 2B).
In the atropine and solvent group, the epithelium is inflamed and has necrosis and local destruction, and the nuclei of the basal cells are hypertrophic. In the stroma, the distance between the collagen fibers is large and they are moderate to severely edematous. Keratinocytes are hypertrophic and out of spindle state. Few inflammatory cells are seen. Descemet’s membrane is slightly damaged and thinner than the healthy control group (Figure 2C).
The destruction of the epithelium in atropine and %1 extract is mild, but destruction is still observed. The nuclei of the basal cells are hypertrophic. The distance between the collagen fibers is less than the previous two groups and they have mild edema. Few inflammatory cells are seen. Keratinocytes are observed as spindle shaped and slightly hypertrophic (Figure 2D).
In the of atropine and 2.5% extract group, inflammation is much less and hypertrophy is mild. The distance between collagen fibers is reduced and spindle-shaped keratinocytes are observed. Infiltration of inflammatory cells is not observed (Figure 2E). Destruction of the epithelium is very mild in the group receiving atropine plus %5 extract. Collagen fibers are observed parallel in most areas. Keratinocytes are spindle-shaped and infiltration of inflammatory cells was not observed (Figure 2F).
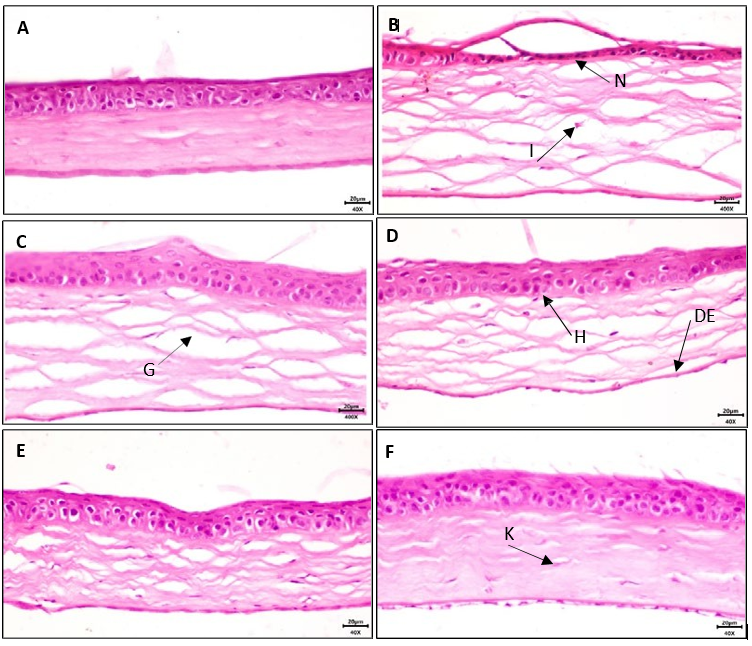
Figure 2: Corneal Tissues Stained with h&e Method (40x). Histopathology of Corneal Tissuesfrom Control Group (a), Atropine Group (b), Atropine and Solvent Group (c), Atropine and%1 Extract (d), Atropine and %2.5 Extract (e) and Atropine and %5 Extract (f): Inflammatory Cells Infiltration (i) and Extensive Epithelium Degeneration and Necrosis (n) are Presented in Atropine Group. Collagen Fibers (g) and keratinocytes (k), Hypertrophic Nuclei in Epithelial Basal Cells (h), Descemet's Membrane (de). Bar is Equal to 20 μm
Discussion
DES is a common eye problem that can reduce vision function and quality of life. The two main mechanisms involved in dry eye disease are increase in tear osmolarity and ocular surface inflammation. Activation of inflammatory mediators leads to cellular apoptosis, glycocalix mucin loss, and epithelial damage that result in tear film instability [11]. Oxidative stress also can be an important factor in the development of dry eye disease such as inflammation [15]. Lubricants, which are usually prescribed as the first line of treatment for dry eye, are recommended at all levels of disease severity. There are several formulations of lubricating eye drops that can adequately relieve the symptoms of dryness and serve therapeutic purposes [9-11].
For many years, the rabbit was generally the preferred animal model for eye research. But in recent years, mice have been widely used in cornea research and studies related to other eye areas such as retina and lens. The advantages of using mice are that it is easy to breed, grows quickly, and is affordable [16]. An obvious disadvantage of the mouse as a dry eye model is that the diameter of its cornea is much smaller than that of humans. However, the mouse cornea is histologically very similar to the structure of the human cornea [16-18].
The cornea is a clear, avascular tissue that protects the structures inside the eye, acts as a protective barrier against fluid loss and pathogen penetration, and provides a transparent environment for vision [17]. The corneal epithelium is the outermost tissue of the eye, which is often attacked by physical, chemical and pathogenic environmental pollution and undergoes repeated damage and repair. Continuous replacement of epithelial cells is essential to ensure its function as a protective barrier [19].
The chemical composition of Ajwain fruit essential oil is influenced by various factors. Different parameters such as geographical region, soil type and extraction method (pressure, temperature and extraction time) have a significant effect on the percentage and composition of essential oil [20].
Usually, thymol and carvacrol are the main components of ajwain essential oil and may be extracted between 35 and 60%. The non- thymol (thymen) part of the extract contains paracymene, gamma- terpinene, alpha-pinene, betapinene, alpha-terpinene, styrene, delta-3-carene, betaphilendrene, terpinen-4-l and carvacrol. On the other hand, in a research, carvone, limonene and dilapiol were introduced as other essential oil compounds [14,21].
Plants produce an extraordinary variety of phenolic metabolites that contain one or more acidic hydroxyl moieties attached to an aromatic arene (phenyl) ring. Phenolic compounds are secondary metabolites that are synthesized in plants. They have biological properties such as: antioxidant, anti-apoptotic, anti-aging, anti- cancer, anti-inflammatory, anti-arteriosclerosis [22]. Phenolic compounds are excellent absorbers of oxygen radicals because the reduction potential of phenolic radicals is lower than the reduction potential of oxygen radicals and also phenoxyl radicals are generally less reactive than oxygen radicals. Therefore, phenolic compounds can eliminate reactive oxygen intermediates without causing more oxidative reactions [23].
Eye drops containing herbal extracts for the prevention and treatment of dry eye are currently under examination and as well as artificial tears, can be effective in relieving the symptoms of dry eye. Although hundreds of plants are used to prevent or treat diseases, there is no clinical evidence in most cases.
Potential mechanisms by which TA aqueous extract may improve ocular surface disease in dry eye were not evaluated in this study, but anti-inflammatory effects of TA have been demonstrated in several studies [14,24,25,26]. Accordingly, both total alcoholic extract and total aqueous extract possess in vivo significant anti– inflammatory effect [27].
Conclusion
In this study we found that eye drops containing Trachyspermum ammi L. fruit's aqueous extract can be beneficial in improving atropine induced dry eye. Pathological investigation demonstrated significantly reduced inflammation, and improved dry eye induced damages in the cornea of mice specially with daily use of eye drops containing Trachyspermum ammi L. fruit's aqueous extract 5% extract.
Conflict of interest
The authors declare no competing financial interest.
References
- Lemp, M. A., & Foulks, G. N. (2007). The definition and classification of dry eye disease. Ocul Surf, 5(2), 75-92.
- Reyhani, M., Aghamollaei, H., Jadidi, K., & Barzegar, A. (2021). Herbal-Based Drugs for Dry Eye; Treatment and Adverse Reactions. Indian Journal of Traditional Knowledge (IJTK), 20(1), 33-40.
- Martin, E., Oliver, K. M., Pearce, E. I., Tomlinson, A., Simmons, P., & Hagan, S. (2018). Effect of tear supplements on signs, symptoms and inflammatory markers in dry eye. Cytokine, 105, 37-44.
- Memarzadeh, E., Luther, T., & Heidari-Soureshjani, S. (2018). Effect and Mechanisms of Medicinal Plants on DryEye Disease: A Systematic Review. Journal of Clinical & Diagnostic Research, 12(9).
- Tsubota, K., Yokoi, N., Watanabe, H., Dogru, M., Kojima, T., Yamada, M., ... & Shimazaki, J. (2020). A new perspective on dry eye classification: proposal by the Asia dry eye society. Eye & contact lens, 46, S2-S13.
- Ouyang, W., Wu, Y., Lin, X., Wang, S., Yang, Y., Tang, L., ... & Liu, Z. (2021). Role of CD4+ T helper cells in the development of BAC-induced dry eye syndrome in mice. Investigative Ophthalmology & Visual Science, 62(1), 25-25..
- Su, Y. C., Hung, J. H., Chang, K. C., Sun, C. C., Huang, Y. H.,Lee, C. N., ... & Lai, E. C. C. (2022). Comparison of sodium- glucose cotransporter 2 inhibitors vs glucagonlike peptide-1 receptor agonists and incidence of dry eye disease in patients with type 2 diabetes in Taiwan. JAMA network open, 5(9), e2232584-e2232584.
- Chen, H. C., Chen, Z. Y., Wang, T. J., Drew, V. J., Tseng, C. L., Fang, H. W., & Lin, F. H. (2017). Herbal supplement in a buffer for dry eye syndrome treatment. International journal of molecular sciences, 18(8), 1697.
- Mohamed, H. B., Abd El-Hamid, B. N., Fathalla, D., & Fouad,E. A. (2022). Current trends in pharmaceutical treatment of dry eye disease: A review. European Journal of Pharmaceutical Sciences, 175, 106206.
- Barabino, S., Benitez-Del-Castillo, J. M., Fuchsluger, T., Labetoulle, M., Malachkova, N., Meloni, M., ... & Rolando, M. (2020). Dry eye disease treatment: the role of tear substitutes, their future, and an updated classification. European Review for Medical & Pharmacological Sciences, 24(17).
- Haji-Ali-Nili N, Khoshzaban F, Karimi M, Rahimi R, Ashrafi E, Ghaffari R, Ghobadi A, Behrouz MJ. Effect of a natural eye drop, made of Plantago ovata mucilage on improvement of dry eye symptoms: A randomized, double-blind clinical trial. Iranian Journal of Pharmaceutical Research: IJPR. 2019;18(3):1602.
- Shukla, A. K., Yadav, V. K., Yadav, V. P., Tiwari, A., Tiwari, P., Verma, G., ... & Sharma, M. (2023). An overview of the herbs used to treat conjunctivitis. Current Advances in Biosciences, 207.
- Biswas, N. R., Gupta, S. K., Das, G. K., Kumar, N., Mongre, P. K., Haldar, D., & Beri, S. (2001). Evaluation of Ophthacare® eye drops—a herbal formulation in the management of various ophthalmic disorders. Phytotherapy Research, 15(7), 618-620.
- Zarshenas, M. M., Moein, M., Samani, S. M., & Petramfar,P. (2014). An overview on ajwain (Trachyspermum ammi) pharmacological effects; modern and traditional. Journal of natural Remedies, 98-105.
- Uchino, Y., Kawakita, T., Ishii, T., Ishii, N., & Tsubota, K. (2012). A new mouse model of dry eye disease: oxidative stress affects functional decline in the lacrimal gland. Cornea, 31, S63-S67.
- Henriksson, J. T., McDermott, A. M., & Bergmanson, J.P. (2009). Dimensions and morphology of the cornea in three strains of mice. Investigative ophthalmology & visual science, 50(8), 3648-3654.
- Zhang, Y., Liu, X., Liang, W., Dean, D. C., Zhang, L., & Liu, Y. (2021). Expression and function of ZEB1 in the cornea. Cells, 10(4), 925.
- Huang, W., Tourmouzis, K., Perry, H., Honkanen, R. A., & Rigas, B. (2021). Animal models of dry eye disease: Useful, varied and evolving. Experimental and Therapeutic Medicine, 22(6), 1-10.
- Yoon, J. J., Ismail, S., & Sherwin, T. (2014). Limbal stem cells: Central concepts of corneal epithelial homeostasis. World journal of stem cells, 6(4), 391.
- Chahal, K. K., Dhaiwal, K., Kumar, A., Kataria, D., & Singla, N. (2017). Chemical composition of Trachyspermum ammi L. and its biological properties: A review. Journal of Pharmacognosy and Phytochemistry, 6(3), 131-140.
- Morsy, N. F. (2020). Production of thymol rich extracts from ajwain (Carum copticum L.) and thyme (Thymus vulgaris L.) using supercritical CO2. Industrial Crops and Products, 145, 112072
- Ali, M., Khan, S. Y., Jang, Y., Na, C. H., Talbot, C. C., Gottsch,J. D., ... & Riazuddin, S. A. (2021). Cigarette smoke triggersloss of corneal endothelial cells and disruption of Descemet's membrane proteins in mice. Investigative Ophthalmology & Visual Science, 62(3), 3-3.
- Ainsworth, E. A., & Gillespie, K. M. (2007). Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin–Ciocalteu reagent. Nature protocols, 2(4), 875-877.
- Bhadra, P. (2020). An Overview of Ajwain (Trachyspermum ammi). Indian Journal of Natural Sciences, 10(59), 18466- 182474.
- Mehta, R. L., Zayas, J. F., & Yang, S. S. (1994). Ajowan as a source of natural lipid antioxidant. Journal of agricultural and food chemistry, 42(7), 1420-1422.
- Goyal, S., Chaturvedi, V., Dhingra, G., Tanwar, S., Sharma, K., & Singh, S. (2022). Trachyspermum ammi: A review on traditional and modern pharmacological aspects. Biological Sciences, 2(4), 324-337.
- Thangam, C., & Dhananjayan, R. (2003). Antiinflammatory potential of the seeds of Carum copticum Linn. Indian Journal of pharmacology, 35(6), 388-391.



