Research Article - (2025) Volume 8, Issue 2
Difficulty in Terminating Posture of Three-Dimensional Gait Bradykinesia in Parkinson's Disease
2Department of Neurology, Shinshu University, School of Medicine, Matsumoto 390-8621, Japan
Received Date: Jul 02, 2025 / Accepted Date: Aug 08, 2025 / Published Date: Sep 05, 2025
Copyright: ©Tateo Warabi, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Warabi, T., Furuyama, H., Okada, M., Yanagisawa, N. (2025). Difficulty in Terminating Posture of Three-Dimensional Gait Bradykinesia in Parkinson
Abstract
This study investigated whether difficulty in initiating volitional gait in patients with Parkinson’s disease stems from impaired termination of the preceding posture or impaired initiation of new movement. Forty-one patients with Parkinson’s disease and 15 age-matched controls participated. While participants fixated on a conditioning visual target, a visual cue test stimulus prompted participants to initiate volitional gait. Three-dimensional sole-floor reaction forces during coordinated gait movement, antagonist/ agonist electromyography (EMG), and levodopa therapy were recorded. From the visual cue, gait initiation was measured by tibialis anterior EMG burst onset (EMG-on latency) and coordinated gait movement onset, whereas preceding postural termination latency was measured by the pause in antagonist soleus tonic EMG activity (EMG-off latency). Three visual fixation paradigms were used: no-gap (fixation and test targets turned off simultaneously), gap (fixation turned off 200 ms before test stimulus), and overlap (fixation turned off 200 ms after test stimulus). In all three tasks, antagonist EMG-off latencies were prolonged in the patients. Agonist EMG-on latencies remained within physiological range in the gap and no-gap tasks but were prolonged for the overlap task in the patients. In the three tasks, all coordinated gait movement latencies and overlap task EMG-on latencies were prolonged in the patients and correlated with prolonged EMG-off latencies of the preceding movement/posture termination. Differences in unsynchronized EMG-off/on latency correlated with reduced overall gait movement (slow speed, short steps, low amplitude of movement) and levodopa therapy, suggesting that initiation difficulty in Parkinson’s disease is related to difficulty in terminating the preceding movement/posture rather than difficulty in generating new movement.
Keywords
Parkinson’s Disease, Three-Dimensional Bradykinesia, Bipedal Motor Plan, Prolonged Posture, Levodopa Therapy
Introduction
Parkinson’s disease is characterized by a slow, shuffling gait, short steps, and various clinical symptoms, such as difficulty ininitiating volitional gait and freezing of gait (FoG). These impairments are related to dysfunction in the basal ganglia and cerebral cortex [1-4] Many studies have quantified delayed and slow spatiotemporal parameters in volitional movement as functional deficiencies in the basal ganglia in patients with Parkinson’s disease [5-8]. Recent research suggests that the basal ganglia do not directly generate movement but play a key role in switching between sequential motor actions to enable smooth, continuous motion [9-13]. Further , previous findings indicate that sequential sensorimotor control of gait depends on terminating the preceding posture and initiating new movement [14-18]. This process requires a whole-body coordination motor plan that integrates multiple motor systems, including the cerebral cortex, basal ganglia, and brainstem systems [19-24,11].
In this study, we investigated whether difficulty in initiating volitional gait of bradykinesia in Parkinson’s disease arises from impaired initiation of a new movement or from difficulty in terminating the preceding movement/posture. We investigated sensorimotor switching reaction times of posture/gait triggered by a visual cue in the three-dimensional posture termination (electromyography [EMG]-off) and gait initiation (EMG-on) under different visual conditions and assessed sequential movements in relation to levodopa (L-DOPA) therapy and clinical bradykinesia that were suggested by [1].
Materials and Methods
Participants
We selected 41 patients with idiopathic Parkinson’s disease (11 men, 30 women) who responded to L-DOPA treatments and 15 age-matched older healthy subjects as controls (9 men, 6 women). The two groups were similar in age (Parkinson’s disease: [mean ± standard deviation (SD)], 73.6 ± 8.5 years; controls: 72.1 ± 9.6 years, P = 0.628). Idiopathic Parkinson’s disease was diagnosed based on the criteria established by the UK Parkinson’s Disease Society Brain Bank [25], 3T magnetic resonance imaging, computed tomography, and radioisotope (dopamine transporter) images. Following the Hoehn-Yahr (H-Y) staging system and our previous study, we included only patients with bradykinesia and excluded those with hemiparesis, camptocormia, ataxia, or aphasia. All patients scored above the Mini-Mental State Examination cutoff of 23 [1].
We categorized the 41 patients into three groups based on bradykinesia severity according to the progression of Parkinson’s disease as graded by H-Y stages: stage II (moderate, n = 10), stage III (severe, n = 15), and stage IV (advanced maximum, n = 16). Patients in H-Y stage I (unilateral symptoms) and stage V (non- ambulatory) were excluded. All 41 patients showed a response to L-DOPA combined with either benserazide or carbidopa, along with dopamine agonists, in accordance with a previous study.
In 14 of the 41 patients, the no-gap task was used to assess H-Y stage clinical symptoms and electrophysiological bradykinesia before and after L-DOPA therapy. Improvements in gait bradykinesia following additional L-DOPA therapy were evaluated in seven patients twice within 1 month (four with H-Y stage III and three with stage IV). Conversely, progression of gait bradykinesia was assessed in another seven patients (four with H-Y stage II and 3 with stage III) before and after a >1-year interval without additional L-DOPA therapy. The effects of L-DOPA therapy were compared between these two test periods. All participants were fully informed of the purpose and nature of the procedures, and written informed consent was obtained voluntarily to participate in the experiments. This study complied with the Declaration of Helsinki (1991) and was approved by the Sapporo Yamanoue Hospital Ethics Committee.
Experimental Design
All participants stood with their heels on the starting line on a level floor under normal room lighting while gazing at a panel with two vertically arranged light-emitting diodes (LEDs, each 0.2° in diameter [4 mm]). To have subjects perform successive movements of fixation posture/gait, we used two stimuli presented by the LEDs: a conditional stimulus and a test stimulus. One LED (red) served as the fixation target, and the other (green and yellow) functioned as the test stimulus of the gait trigger target. Participants were first instructed to gaze at the fixation target to assess visually triggered movement latency. Participants were instructed to wait for one of the two test stimuli (go or no-go) to appear. Of the two test stimuli, the green double LED (on-stimulus) signaled participants to reach their comfortable walking speed quickly (go), whereas the yellow LED indicated no-go and was included randomly at irregular intervals [12]. The time interval between target triggering gait and posture offset was defined as soleus EMG-off latency of posture (termination). The onset of the tibialis anterior EMG burst was defined as the EMG-on latency of gait (initiation).
Volitional bipedal locomotion was assessed using three visual cue tasks: no-gap, gap, and overlap. In the no-gap task, the fixation target was turned off at the same time the movement-triggering stimulus was turned on. In the gap task, the fixation target was turned off 200 ms before the triggering stimulus, disengaging visual fixation before movement initiation. In the overlap task, the triggering stimulus was presented while the fixation target remained visible for 200 ms before turning off, thereby simulating natural visual fixation [26,18]. The overlap task simulated the visually fixated posture engagement of natural conditions, whereas the gap task tested movement initiation without a fixation engagement period. The no-gap task served as a baseline to assess standard spatiotemporal gait parameters and reaction times.
These tasks were performed by the 15 healthy elderly controls and 41 patients with Parkinson’s disease. The trials were conducted in three sequential blocks, including the no-gap task, and were completed within 30 min. Each participant performed 7 to 10 walking trials of 5–10 m per task. Most participants did not notice differences among the three tasks. To further examine the effect of visual fixation conditioning, 21 of the patients with Parkinson’s disease also performed an unconditioned vocal cue-triggered task without a fixation target for assessment of gait initiation without fixation engagement/disengagement of the visual target. For each trial, 10–15 steps were recorded.
Measurements of Leg EMG and Three-Dimensional Bipedal Sole-Floor Reaction Forces
Measurements of leg EMG and three-dimensional bipedal sole- floor reaction forces as the three-dimensional temporospatial profile during standing posture and gait locomotion were recorded in this study. The antagonist and agonist EMG activities of the soleus and tibialis anterior muscles were recorded using bipolar surface electrodes placed on both the left and right muscles, respectively, with an interelectrode distance of 5 cm. Participants stood with both heels on the starting line, and the tonic EMG activity of the soleus transitioned into the tibialis anterior EMG burst, in accordance with signaled gait initiation [14,15].
Early-stage sole-floor reaction forces were recorded from five anatomically distinct points in the unilateral foot, reflecting normal human locomotion [27,28].
In this study, three-dimensional body shifts during coordinated gait movements were recorded by measuring bipedal sole-floor reaction forces at distinct anatomical points on both soles. The stance and swing phases were determined by calculating the force difference between the calcaneus and third metatarsal in each foot. These heterogeneous gait movements were defined as the overall synergistic motion of the limbs, trunk, and head, represented as x-, y-, and z-axis vectors of bipedal locomotion forces. This method directly measures force vectors between two defined sole-floor reaction forces, allowing anatomical and physiological analyses of human bipedal locomotion [27,28]. Using this approach, data from at least a dozen individual steps can be obtained for quantitative analysis of human gait [27,28,18,29].
Strain-gauge load cells fixed over the sole (Type 9E01-L42-500N; 14-mm diameter, 4-mm thick, 0–500 N range, NEC San-Ei, Tokyo, Japan) were used to measure sole-floor reaction forces, capturing y-axis forces from each sole and x-axis bipedal vectors during gait. The cells were secured with double-sided adhesive tape on anatomically precise skin points: the medial process of the calcaneus and the head of the third metatarsal on both soles. The x-, y-, and z-axis vectors were computed to represent bipedal locomotion (Fig. 1).
The swing phase of raising the leg (z-axis) was analyzed in relation to the contralateral body shift stance phase (y-axis), thus defining the bipedal motor plan (x-axis) of coordinated gait movement. The sole-floor reaction forces served as key indicators of synergistic gait movements, reflecting body sway involving the limbs, trunk, and head [27,28,18]. A force plate (SAM, NITTA, Osaka, Japan) was used to confirm the center of mass in several patients, tracking the dynamic x-axis vector of gait initiation and initial backward body shift (IBS) on the y-axis [15,9].
Outputs from the strain gauges and EMG electrodes were recorded on a multichannel oscillograph (Recti-Horiz 8K20, NEC, Japan) through four-channel EMG amplifiers (Type Medi-104M, Media- Area-Support Co., Okayama, Japan) and simultaneously stored in a recording unit (LX-10, TEAC, Tokyo, Japan). Data stored on magnetic tapes were digitized at 500 Hz and processed using a microcomputer (Signal Processor 7T18T, NEC San-Ei).
Data Analysis
Electrophysiological data were analyzed using Microsoft Excel (Microsoft Corp. Redmond, WA, USA). The EMG-off latency of the primary posture was defined as the time between cue onset and the pause following tonic soleus EMG activity (leg antagonist). The EMG-on latency of voluntary gait initiation was measured as the time from cue onset to onset of tibialis anterior EMG activity.
The difference between antagonist EMG-off latency and agonist EMG-on latency was correlated with the interaction interval between postural termination and gait initiation. Loss of primary postural balance due to IBS (y-axis) triggered the transition to bipedal locomotion [14,15]. The initiation of coordinated bipedal locomotion was marked by the onset of the initial swing phase (z-axis) and tibialis anterior EMG activity (Fig. 1). The step gain of the bipedal motor plan (x-axis) in three-dimensional gait was calculated as the ratio of the swing phase (z-axis) to the corresponding contralateral body-shift stance phase (y-axis). The mean step gain over six consecutive steps, starting from the third step, was defined as the motor plan for the three-dimensional locomotion and bradykinesia.
Figure 1: Three-dimensional gait performance in the no-gap task for an elderly control and a patient with Parkinson’s disease. The visual cue-triggered bipedal locomotion is superimposed in an elderly control (a) and a parkinsonian patient (b) with Hoehn-Yahr (H-Y) stage IV. Six trials for the control and three for the patient were superimposed, illustrating the x-, y-, and z-axis vectors of gait performance and EMG activity of the soleus and tibialis anterior muscles in the left (L) and right (R) legs. EMG signals were aligned with the onset of tibialis anterior EMG activity for each participant. The dynamic body sway during bipedal sole-floor reaction forces, from heel to toe (y-axis vector), was recorded at the calcaneus (heel) and third metatarsal (toe). The unilateral initial backward body shift (IBS; y-axis body shift) switched the primary posture to the subsequent bipedal locomotion. The inset figure in (a) and (b) illustrates the three- dimensional sole-floor reaction forces, with the force plate monitor capturing the primary posture switching to gait via IBS (y-/x-axis body shift).
Statistical Analysis
One-way analysis of variance (ANOVA) was conducted to assess group differences in measured parameters. A P-value of 0.05 was considered to indicate statistical significance. Statistical analyses were performed with R 4.2.2 (The R Foundation for Statistical Computing, Vienna, Austria).
Results
All participants performed volitional bipedal locomotion on the testing floor stimuli mentioned above.
Three-Dimensional Bipedal Locomotion and Leg EMG Activity
The typical patterns of three-dimensional bipedal locomotion and antagonist/agonist EMG activity (soleus/tibialis anterior) were recorded under the no-gap task (Fig. 1). Bipedal locomotion was aligned with the onset of tibialis anterior EMG activity. Six superimposed gait cycles from a 70-year-old control participant (gait speed: 52 m/s) and 3 from a 73-year-old patient with Parkinson’s disease (H-Y stage IV, gait speed: 34 m/s) are shown in Figs. 1a and b, respectively.
Sole-floor reaction forces captured the three-dimensional x-, y-, and z-axis vectors representing coordinated postural and locomotor movements (Fig. 1, inset). Dynamic force changes in sole- floor reaction forces between the heel and toe of each foot were calculated, and the posterior-anterior force shift (third metatarsal to calcaneus) was defined along the y-axis. During triggered gait, postural stability was initially switched via the IBS (y-axis), which was linked to the swing phase (z-axis) and transitioned into the three-dimensional motor plan for bipedal locomotion (x-axis). In the elderly controls, IBS durations exceeded 300 ms, whereas in the patients with advanced Parkinson’s disease, they were abnormally reduced to under 250 ms in all three tasks.
The visual cue test stimulus triggered the transition from posture to gait and corresponded with the EMG-off/on latencies of the soleus and tibialis anterior muscles. The difference between antagonist (soleus) and agonist (tibialis anterior) EMG latencies reflected the transition from primary posture to the three-dimensional bipedal locomotion in all participants (Fig. 1). In the patients with Parkinson’s disease, the visual cue stimulus prolonged soleus EMG-off latencies across all three paradigms (gap, no-gap, and overlap tasks), whereas tibialis anterior EMG-on latencies remained within the normal range in the gap and no-gap tasks.
In advanced stages of the disease, differences in EMG-off/on latency were abnormally short or even negative, primarily due to prolonged soleus EMG-off latencies, indicating impaired posture termination before gait initiation. In the elderly controls, the difference in EMG-off/on latency was approximately 55 ms (Fig. 1a), whereas in patients with advanced disease, it was reduced to less than 45 ms (Fig. 1b).
Figure 2: Posture EMG-off latencies correlated with the onset of gait movement in the three tasks. Posture EMG-off latencies correlated with the onset of gait movement (ordinate) in the three tasks (a, gap task; b, no-gap task; c, overlap task). Soleus EMG-off latencies (abscissa), reflecting the termination of conditioned posture, were significantly prolonged in all three tasks in Parkinson’s disease. In contrast, EMG-on latencies (ordinate) remained within the normal range in the gap and no-gap tasks across all participants. Motor-on latencies of heterogeneous coordinated movement were prolonged (ordinate) and correlated with EMG-off latencies associated with postural termination in Parkinson’s disease. In (d, no-gap task), the mean values and standard deviations (SDs) of EMG-off/on latency differences were reduced, resulting in smaller SDs in each participant group for all tasks.
|
Gap, no-gap, overlap task |
|
Elderly n = 15 |
PD II n = 10 |
PD III n = 15 |
PD IV n = 16 |
ANOVA |
|
EMG-off |
||||||
|
Gap task |
Mean |
159.01 |
148.50 |
179.79 |
209.26 |
F = 6.7999 |
|
(msec) |
SD |
40.95 |
29.27 |
32.51 |
37.94 |
P = 0.0007 |
|
No-gap task |
Mean |
165.91 |
180.48 |
213.49 |
259.57 |
F = 11.788 |
|
(msec) |
SD |
37.24 |
34.39 |
54.35 |
52.93 |
P < 0.0001 |
|
Overlap task |
Mean |
207.03 |
221.09 |
277.62 |
345.75 |
F = 21.4024 |
|
(msec) |
SD |
49.59 |
31.77 |
47.30 |
60.84 |
P < 0.0001 |
|
EMG-on |
||||||
|
Gap task |
Mean |
213.18 |
196.13 |
198.46 |
208.24 |
F = 0.642 |
|
(msec) |
SD |
40.23 |
34.86 |
31.36 |
37.03 |
P = 0.592 |
|
No-gap task |
Mean |
221.27 |
226.31 |
236.03 |
248.62 |
F = 1.012 |
|
(msec) |
SD |
39.15 |
33.33 |
54.03 |
51.02 |
P = 0.834 |
|
Overlap task |
Mean |
256.94 |
260.25 |
299.00 |
343.09 |
F = 9.7832 |
|
(msec) |
SD |
53.71 |
30.04 |
39.96 |
53.05 |
P = 0.0001 |
|
EMG-off/on difference |
||||||
|
Gap task |
Mean |
54.17 |
47.63 |
18.68 |
-1.01 |
F = 60.623 |
|
(msec) |
SD |
5.98 |
3.80 |
3.22 |
3.34 |
P < 0.0001 |
|
No-gap task |
Mean |
55.96 |
45.52 |
23.47 |
-6.45 |
F = 57.831 |
|
(msec) |
SD |
8.08 |
8.83 |
7.97 |
17.28 |
P < 0.0001 |
|
Overlap task |
Mean |
49.91 |
39.16 |
21.38 |
-2.95 |
F = 63.0773 |
|
(msec) |
SD |
7.10 |
3.35 |
2.83 |
2.94 |
P < 0.0001 |
|
Motor-on |
||||||
|
Gap task |
Mean |
753.19 |
788.65 |
817.67 |
919.67 |
F = 5.1926 |
|
(msec) |
SD |
90.94 |
108.39 |
133.41 |
123.51 |
P = 0.0035 |
|
No-gap task |
Mean |
778.47 |
866.55 |
891.84 |
1086.53 |
F = 7.681 |
|
(msec) |
SD |
107.35 |
147.39 |
155.65 |
266.78 |
P = 0.0002 |
|
Overlap task |
Mean |
799.53 |
858.36 |
935.11 |
1158.59 |
F = 20.6925 |
|
(msec) |
SD |
92.30 |
157.98 |
92.31 |
159.67 |
P < 0.0001 |
|
The mean ± standard deviation (SD) of electromyography (EMG)-off/on latency and EMG-off/on latency differences are summarized for the gap, no-gap, and overlap tasks and different participant groups. Hoehn and Yahr stages II, III, and IV of Parkinson’s disease (PD) show a significant difference (P < 0.0001) |
||||||
Table 1: ANOVA analysis of EMG-off and -on latency, off/on difference, and motor-on latency in the gap, no-gap, and overlap tasks
EMG-Off/On Latency and Heterogeneous Coordinated Gait Movement
For the three different paradigms, we evaluated whether difficulties in prolonged coordinated gait initiation in Parkinson’s disease correlated with EMG-off or EMG-on latencies. Table 1 summarizes the mean values and SDs for the EMG-off/on laten- cies, differences in EMG-off/on latency, and motor-on latencies in heterogeneous coordinated gait movement. The data include re- sponses from elderly controls and patients in advanced H-Y stages of Parkinson’s disease. In all three tasks, patients with advanced Parkinson’s disease exhibited prolonged soleus EMG-off laten- cies, indicating delayed postural termination. However, visually triggered EMG-on latencies of the tibialis anterior for volitional gait initiation remained within the normal physiological range in the gap and no-gap tasks but were prolonged in the overlap task. Motor-on latencies for heterogenous coordinated gait movement were prolonged in all three tasks.
Figure 2a-c shows correlations between posture termination EMG- off latencies (abscissa) and both EMG-on latencies and motor- on latencies of gait initiation (ordinate) across all participants, including those with advanced Parkinson’s disease. Coordinated motor-on latencies were prolonged in the three tasks and correlated with EMG-off latencies (Fig. 2a-c). In the overlap task simulating natural prolonged visual fixation, EMG-on and motor-on latencies were notably prolonged and correlated with EMG-off latencies (Fig. 2c).
Figure 2d illustrates the changes in soleus EMG-off latencies/SD (red line) and tibialis anterior EMG-on latencies/SD (blue line) as the disease progressed, with a decreasing EMG-off/on latency difference/SD observed across all stages and tasks. It is clear from the small SDs of the EMG-off/on latency differences that the large SD shown for each EMG-off/on latency of the antagonist/agonist represents the consistent interacting changes that occurred between EMG-off/on motor systems of the individual participants (Fig. 2d).
Figure 3: Three-dimensional motor plan is correlated with the EMG-off/on latency difference and levodopa therapy. The mean value of the three-dimensional bipedal locomotion ratio of step gain was defined as the bipedal motor plan, calculated by the ratio of the swing phase to the corresponding contralateral stance phase as the step gain. (a) Bipedal motor plan levels correlate with EMG-off/on latency differences across all participants. (b) (white circles): EMG-off/on latency difference levels were evaluated before and after L-DOPA therapy; (b) (black triangles): EMG-off/on latency difference levels were reevaluated after one year of disease progression without additional L-DOPA therapy. The two regression lines illustrate the relationship between L-DOPA therapy and disease progression. (c, d) Gait speed and step length correlate with EMG-off/on latency differences. (e, f) Gait speed and step length correlate with the step gain of the motor plan levels in the reduced overall gait movement.
|
No-gap task |
|
Elderly (15) |
PD II (10) |
PD III (15) |
PD IV (16) |
ANOVA |
|
EMG-off latency (msec) |
Mean |
165.91 |
180.48 |
213.49 |
259.57 |
F = 11.788 |
|
Soleus |
SD |
37.24 |
34.39 |
54.35 |
52.93 |
P < 0.0001 |
|
EMG-on latency (msec) |
Mean |
221.27 |
226.31 |
236.03 |
248.62 |
F = 1.012 |
|
Tibialis anterior |
SD |
39.15 |
33.33 |
54.03 |
51.02 |
P = 0.834 |
|
EMG-off/on latency difference (msec) |
Mean |
55.96 |
45.52 |
23.47 |
-10.95 |
F = 57.831 |
|
|
SD |
8.08 |
8.83 |
7.97 |
23.25 |
P < 0.0001 |
|
Motor-on latency (msec) |
Mean |
778.47 |
866.55 |
891.84 |
1086.53 |
F = 7.681 |
|
|
SD |
107.35 |
147.39 |
155.65 |
266.78 |
P = 0.0002 |
|
Swing phase (msec) |
Mean |
355.34 |
335.87 |
283.29 |
234.94 |
F = 57.831 |
|
(z-axis vector) |
SD |
42.85 |
55.03 |
52.49 |
38.03 |
P < 0.0001 |
|
Step gain |
Mean |
0.60 |
0.56 |
0.44 |
0.36 |
F = 36.725 |
|
(x-axis vector; z/y) |
SD |
0.07 |
0.07 |
0.07 |
0.06 |
P < 0.0001 |
|
Step length (cm) |
Mean |
47.31 |
39.18 |
29.31 |
19.53 |
F = 34.258 |
|
|
SD |
8.36 |
7.45 |
9.16 |
6.66 |
P < 0.0001 |
|
Speed (m/min) |
Mean |
3.48 |
3.11 |
2.39 |
1.53 |
F = 28.226 |
|
|
SD |
0.86 |
0.60 |
0.66 |
0.54 |
P < 0.0001 |
|
Initial backward body shift (msec) |
Mean |
309.49 |
282.75 |
247.80 |
199.49 |
F = 8.557 |
|
|
SD |
50.53 |
82.18 |
64.68 |
58.75 |
P < 0.0001 |
|
ANOVA of the mean ± SD of the EMG-off latency of the soleus muscle, EMG on-latency of the tibialis anterior muscle, EMG-off/on latency difference, motor-on latency, step gain (Fig. 4a), step length, gait speed, and IBS duration in an older control participant (Elderly) and patients with H-Y stages II (PD II), III (PD III), and IV (PD IV) of Parkinson’s disease. EMG electromyography, F, F value; P, probability value, PD Parkinson’s disease, SD standard deviation |
||||||
Table 2: ANOVA analysis of spatiotemporal parameters of gait bradykinesia in patients with Parkinson’s disease
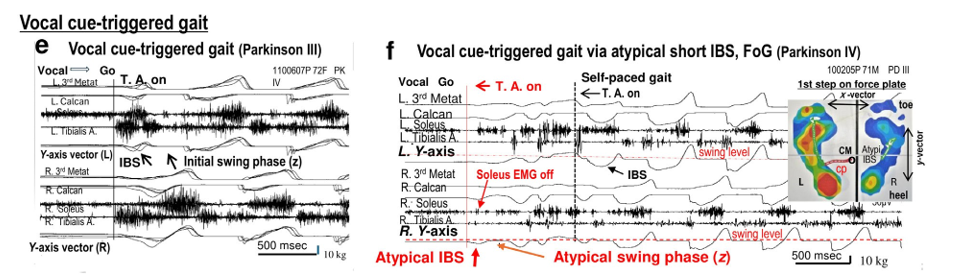
Figure 4: Atypical short IBS switched posture to two-dimensional gait. In visually triggered gait, posture transitions to locomotion through IBS, which links with the initial swing phase and corresponding changes in the x-/y-/z-axis of the motor plan. Ordinary IBS durations (a, b) in conjunction with initial swing phase, EMG-off/on latency difference, and motor plan switch the posture to gait (c, d), but short IBS durations cause difficult in switching the posture to gait in parkinsonian patients (f). In the vocal cue (go)-triggered task (e), which lacks a visual fixation target, gait initiation is superimposed, illustrating the transition from primary posture to typical locomotion in patients. In the vocal cue task (f), IBS is atypically short, producing an insufficient y-axis shift to properly elevate the leg along the z-axis, resulting in two-dimensional bipedal locomotion with freezing of gait (FoG) primarily limited to the x/z-axis.
EMG-Off/On Latency Difference in the Gait Motor Plan and L-DOPA Therapy
The small SD of the EMG-off/on latency difference (Table 1) reflects a consistent relationship between EMG-off/on latency motor systems in each patient. Figure 3a-d shows that EMG-off/ on latency differences correlate both with step gain in the bipedal motor plan and with the motor plan of the small three-dimensional locomotion bradykinesia of all 41 patients undergoing L-DOPA therapy. In patients with advanced Parkinson’s disease, step gains in the bipedal motor plan were below 0.4, whereas elderly controls exhibited step gains of approximately 0.6. As shown in Fig. 3b, the severity of bradykinesia was evaluated twice in 14 of the 41 patients, before and after L-DOPA therapy, in the no-gap task. Figure 3b highlights data from seven patients (four with H-Y stage III and three with stage IV) who recorded additional L-DOPA therapy. Each arrow indicates changes in step gain before and after one month of treatment.
Following L-DOPA therapy, these patients showed recovery from bradykinesia. On average, EMG-off/on latency differences increased by 27.9 ± 7.6 ms, and step gain improved by 0.1 ± 0.08 (Fig. 3b). Symptom improvement corresponded with an average L-DOPA dose increase of 81.4 ± 24.5 mg.
Figure 3b also includes data from another seven patients (four with H-Y stage II and three with stage III) who did not receive additional L-DOPA therapy. These patients were examined twice, once before and once after more than one year. The closed triangles and corresponding arrows indicate changes in their lessened step gain over this period. In this group, the mean EMG-off/on latency difference decreased by 35.9 ± 18.1 ms, whereas step gain declined by 0.09 ± 0.01. Their average L-DOPA dose was at 350 ± 126 mg without additional L-DOPA.
The clinical progression of Parkinson’s disease, as measured by changes in the H-Y stage, was evaluated based on significant differences (P < 0.05) in step gain at each stage of the motor plan. The regression lines and correlation coefficients for the no-gap task are presented in the inset of Figs. 3a-b. These regression analyses highlight a strong relationship between L-DOPA therapy effects and disease progression. In all 14 patients, changes in the bipedal motor plan during bradykinesia were statistically correlated with EMG-off/on latency differences, L-DOPA therapy effects, and deficiency levels (P < 0.0001).
Figure 3c-f and Table 2 show that the mean values of gait step length and gait speed correlate with the step gain of the motor plan, which correlates with the progression of the disease stages. EMG-off/on latency difference represents the degree of abnormal transition between the prolonged posture balance and the subsequent gait movement in patients with Parkinson’s disease. Figures 3d and f show that the slow gait speed in heterogeneous coordinated movements was associated with abnormal transition from the prolonged preceding movement/posture to subsequent gait across all participants. Across Fig. 3a-f, the mean EMG-off/ on latency differences were closely correlated with the sequential bipedal motor plan, step length, and gait speed, further emphasizing the functional link between L-DOPA therapy and deficiency.
IBS and Initial Swing Phase Switching from Posture to Gait Transition
In the visually triggered gait task, posture balance switched to gait through IBS, linking with the initial swing phase (z-axis) of three- dimensional bipedal locomotion in all participants. Durations of IBS were correlated with EMG-off/on latency differences that correlated with EMG-off latency of posture termination, which were also closely associated with visual target fixation disengagement of EMG-off latency (Fig. 4a, b). The IBS durations were more than 100 ms. The initial swing phases (z-axis) were correlated with EMG-off/on latency differences in gait initiation, which were closely associated with execution of the three-dimensional bipedal locomotion motor plan.
A simple vocal cue (go) without fixation engagement/ disengagement triggered bipedal locomotion commonly via a short IBS (y-axis), linking with the initial swing phase (z-axis) in the patients with Parkinson’s disease (Fig. 4e). In the simple vocal cue task, the patients occasionally had difficulty in terminating the preceding posture balance via the atypical short IBS as FoG, which was compensated by a normal IBS in the following volitional motor onset (Fig. 4f). On the force plate, an atypical short y-axis of the short IBS altered the posture balance to the two-dimensional x/z axis of the two-dimensional bipedal movements as FoG (Fig. 4f, inset figure). The atypically short IBS durations were less than 100 ms. In the vocal cue task, this form of FoG occurred in three of 18 trials in one patient, and FoG was observed once each in seven of 21 patients with advanced Parkinson’s disease. In the consistent three visual target tasks (gap, no-gap, and overlap tasks) with typical IBS, FoG was not observed in this study.
Discussion
Parkinson’s disease is characterized by a slow, shuffling gait, short steps, and various clinical symptoms as difficulty in initiating volitional gait. Furthermore, difficulties in gait initiation of Parkinson’s disease are generally more conspicuous when executing coordinated volitional movements compared to simple ones and when performing complex conditioned tasks rather than simpler ones [30,3]. However, these differences are not always significant when comparing patients with Parkinson’s disease to age-matched controls. Evarts et al. emphasized the need for carefully designed studies that consider multiple movement-related factors [6]. In this study, we investigated whether difficulties in initiating the three- dimensional coordinated gait movement in Parkinson’s disease stems from impaired termination of the preceding posture or impaired initiation of new movement.
Prolonged Posture EMG-Off Latency and Three- Dimensional Gait Bradykinesia
Gait bradykinesia in Parkinson’s disease was characterized by difficulty in terminating the preceding sensorimotor program, as described in our previous study [31]. In the present study, we assessed changes in three-dimensional bipedal motor transitions and EMG activities from the fixating posture to gait, which were switched by objective visual conditioning/test stimuli (Fig. 1).
A key finding was the significant prolongation of EMG-off latencies of posture in all three tasks, indicating the important role in terminating the visually fixating posture in patients with Parkinson’s disease (Fig. 2).
Characteristic results of gait bradykinesia in Parkinson’s disease were that the prolonged reaction times of heterogeneous coordinated gait movement are consistently involved in the termination of preceding postures.
However, EMG-on latencies for gait initiation remained within the physiological range in the gap and no-gap tasks but not the overlap task. In the overlap task, for which the visual fixation target presentation was extended by 200 ms, the overlap visual cue test stimulus prolonged both EMG-on and heterogeneous gait reaction times, correlating with the prolonged fixation target and EMG- off latencies. The characteristic results of gait onset triggered by the visual cue test stimulus in Parkinson’s disease were that the delayed motor reaction times and EMG-on latencies are involved in the termination of preceding postures and visual fixation target engagement and that they correlate with preceding posture EMG- off latencies consistently in all three tasks.
Poizner et al. found that in Parkinson’s disease, vision-dependent, arm-trunk-coordinated reaching movements involved prolonged intervals between successive motions [42]. Our results also supported the idea that delayed initiation of three-dimensional, heterogeneous movements is linked to prolonged EMG-off latencies reflected as the difficulty in terminating prior posture balance and disengaging from the visual fixation. These findings suggested that the prolonged reaction times in gait initiation may stem from difficulty in releasing visual fixation on environmental targets, such as doorways or narrow passages, which contributes to impairments in gait initiation as the difficulty in terminating the visual fixation in patients with Parkinson’s disease. The essential quality of delayed gait bradykinesia as EMG-off latency in Parkinson’s disease indicated that the delay in volitional movement impairment is related not only to difficulty in terminating the primary posture balance but also to difficulty in disengaging from visually fixating circumstances.
EMG-Off/On Latency Differences, Motor Planning, and Dopamine Therapy
Clinical studies in Parkinson’s disease indicated that the slow gait speed of bradykinesia is involved in unsynchronized EMG-off/on latency differences between antagonist and agonist leg muscles [29]. A key finding in the parkinsonian patients of the present study was the significant prolongation of EMG-off latencies, whereas EMG-on latency remained within the normal physiological range in the gap and no-gap tasks. In the healthy control individuals, the mean value between the EMG-off/on latency differences of the normal set synchronization motor plan was 55.9 ms in the no-gap task.
In contrast, the values of the unsynchronized EMG-off/on latency differences of the motor plan in Parkinson’s disease were below 45.5 ms or were negative, reflecting difficulty in terminating the preceding movement/posture associating with prolonged EMG- off latencies (Fig. 4a). The present study, however, has shown that EMG-off/on latency differences of overall motor plans are involved in the consistent interaction between the preceding movement/posture and gait from the small SDs of the EMG-off/ on latency differences in contrast to the large SD shown in each EMG-off and on latency (Fig. 2d, Table 1).
These EMG-off/on latency differences were also linked to the three-dimensional bipedal motor plan and correlated with gait speed and step length of bradykinesia in the parkinsonian patients with L-DOPA therapy (Fig. 3). Accordingly, this study showed that the values between EMG-off/on latency differences of the motor plan were associated with the degree of L-DOPA therapy and deficiency during disease progression.
DeLong proposed that the switching activity imbalance model interacting between direct and indirect pathways changes the mean firing rate of the basal ganglia output by dopamine depletion [33]. Our results suggested that L-DOPA deficiency and therapy are involved in sequential motor function in gait bradykinesia. Recent neurophysiological studies have indicated that globus pallidus pars interna (GPi)/substantia nigra pars reticulata (SNr) neurons in the basal ganglia not only control motor programs but also facilitate transitions between sequential sensorimotor processes through the direct, indirect, and hyperdirect pathways [34-38].
Recent studies also suggested that the hyperdirect and indirect pathways regulate motor program selection by suppressing competing or irrelevant motor programs, and the direct pathway facilitates the release of the selected motor program in sequential sensorimotor processing [35,37,31]. Accordingly, it is hypothesized that the hyperdirect and indirect pathways contribute to termination of the overall competing coordinated movement or preceding movement/posture as EMG-off latency. Meanwhile, the direct pathway inhibits GPi/SNr neurons leading to the disinhibition of thalamic neurons, thereby facilitating the initiation of the selected motor program, as indicated by EMG-on latency [39,17,35,37,40,31].
An important finding in preceding studies in the association of sensorimotor processes with the clinical grade of Parkinson’s disease was that EMG-on latency plays a role in the three- dimensional motor plan of bradykinesia and changes to the sequential transitions of postural muscle tone and locomotion through the basal ganglia brainstem systems [19,41].
The present study has shown that EMG-off/on latency differences of overall motor plans are involved in the consistent sequential interaction between the preceding movement/posture and multiple motor systems, which is involved in either posture termination or gait initiation through the cerebral cortex and basal ganglia brainstem systems. The deterioration of EMG-off/on latency differences of the three-dimensional motor plan was represented at a later stage of bradykinesia and was demonstrated by a prolonged EMG-off latency, indicating the difficulty in terminating preceding coordinated movements or postures in all three tasks at advanced disease stages.
Termination Difficulty of the Posture in Sequential Three- Dimensional Gait Bradykinesia
Parkinson noted that walking often requires considerable effort and attention to raise the legs properly in the patients [42]. Moreover, difficulty and effortful attempts to raise the legs (z-axis) to avoid the sensation of being “glued” to the floor have been described as FoG or gait festination [42-45]. Furthermore, in the simple vocal cue task without a visual target, patients with Parkinson’s disease had to struggle occasionally in efforts to lift their leg up (z-axis) from the floor as FoG when switching to the gait with an atypical short y-axis IBS. However, in association with typical y-axis IBS in the consistent three visual tasks, FoG was not observed in this study.
In visually triggered three-dimensional gait bradykinesia, the duration of IBS (y-axis) switching was commonly more than 100 ms (Fig. 4b). In the simple vocal cue task, FoG was occasionally found in association with a short IBS of less than 100 ms, but sometimes, the atypical short y-axis IBS changed to a transition from the preceding movement/posture to the two-dimensional x/z- axis as FoG (Fig. 4f).
However, a volitional backward heel kick in the y-axis has been shown to terminate the posture. This suggested that the prolonged reaction times in gait initiation may stem from balance and serve as a compensatory motor strategy for FoG in Parkinson’s disease [29]. These findings and our results suggested that FoG is not only associated with difficulty in lifting the foot (z-axis) but also difficulty in terminating the preceding movement/posture via an atypical short y-axis IBS in the initiation of sequential three- dimensional gait bradykinesia in Parkinson’s disease. Basmajian described human gait onset as consisting of two phases: (1) the stance phase, which begins when the heel strikes the ground, and (2) the swing phase, which begins with toe-off (i.e., when the toes push off the ground) [15,14]. In addition, Thatch suggested that bradykinesia reflects a deficiency in the ability to switch between sequential motor programs within an overall motor plan involving different motor systems in Parkinson’s disease [46].
However, Marsden and Rothwell proposed a mechanism for the automatic sequence of motor programs by which the basal ganglia associate with the supplementary motor area to regulate smooth automatic sequential movement between the extremities and trunk controlling the posture as a complex clinical symptom in Parkinson’s disease [47]. The following studies, furthermore, suggested that the volitional switching process involving the cerebral cortex and the basal ganglia plays an important role in smoothing the sequential transition from a preceding posture balance to three-dimensional bipedal locomotion via the switching body shift of y-axis IBS and the initial swing phase in Parkinson’s disease [17,40,48].
In the present study, the difficulty in initiating the three-dimensional sequential movement in Parkinson’s disease was influenced by the termination of natural visual context, L-DOPA deficiency/therapy, and y-axis IBS. Furthermore, we found it important to evaluate the gait bradykinesia and FoG of the three-dimensional bipedal locomotion, which are involved in the difficulty of a smooth automatic sequence via y-axis IBS terminating the preceding movement/posture. These factors were involved in the termination difficulty of the natural visual context, L-DOPA deficiency, and short y-axis IBS as the motor plan for reduced overall gait movement (comprising slow speed, short steps, and low amplitude of movement) in the bradykinesia framework.
Conclusion
The present study has shown that the primary issue in volitional gait initiation is not the difficulty in initiating a new movement, but rather the difficulty in terminating the preceding movement/ posture and visual context in Parkinson’s disease. Electrodiagnostic assessments comparing difficulty in initiating gait revealed that prolonged EMG-off latency, IBS, and EMG-off/on latency differences in the three-dimensional motor plan as indicators of difficulty in termination of posture contributed best to an objective appraisal of gait bradykinesia and its response to L-DOPA therapy/ deficiency in patients with Parkinson’s disease [49-52].
Abbreviations

Acknowledgements
The authors wish to express their gratitude to Prof. Masamichi Kato, MD, PhD, for his valuable comments and Prof. Tina Tajima for editing of the English text. The authors would also like to thank the healthy volunteers for participating in the study.
Ethics Approval
Our study complied with the Declaration of Helsinki and was approved by the Sapporo Yamanoue Hospital Ethics Committee.
Consent to Participate
All participants were fully informed of the purpose and nature of the study, and written informed consent was obtained voluntarily from each participant.
Author Contributions
All authors met the criteria for authorship. Warabi designed the study. Warabi and Furuyama collected and analyzed the data. Fu- ruyama performed the statistical analysis. Warabi and Furuyama wrote the manuscript. Okada and Yanagisawa reviewed the manu- script for clarity in English. All authors contributed to the discus- sion, read the manuscript, and consented to take public responsi- bility for its content.
Data Availability
Data supporting our study findings are available upon request from the corresponding author. The data are not publicly available.
References
- Hoehn, M. M., & Yahr, M. D. (1967). Parkinsonism: Onset, Progression, and Mortality. Neurology, 17(5), 427-427.
- Brain, W. R. (1940). Diseases of the Nervous System. InDiseases of the Nervous System (p. 950).
- David, J. D. L. (1973). Sensorimotor Control in Parkinsonism. Journal of Neurology, Neurosurgery & Psychiatry, 36(5), 742-746.
- Yanagisawa, N., Hayashi, R., & Mitoma, H. (2001). Pathophysiology of Frozen Gait in Parkinsonism. Advances in Neurology, 87, 199-207.
- Talland, G. A., & Schwab, R. S. (1964). Performance with Multiple Sets in Parkinson's Disease. Neuropsychologia, 2(1), 45-53.
- Evarts, E. V., Teräväinen, H., & Calne, D. B. (1981). Reaction Time in Parkinson's Disease. Brain: A Journal of Neurology, 104(Pt 1), 167-186.
- Crutcher, M. D., & DeLong, M. R. (1984). Single Cell Studies of the Primate Putamen: I. Functional Organization. Experimental Brain Research, 53(2), 233-243.
- Bloxham, C. A., Mindel, T. A., & Frith, C. D. (1984). Initiation and Execution of Predictable and Unpredictable Movements in Parkinson's Disease. Brain, 107(2), 371-384.
- Kiriyama, K., Warabi, T., Kato, M., Yoshida, T., & Kobayashi,N. (2004). Progression of Human Body Sway During Successive Walking Studied by Recording Sole–Floor Reaction Forces. Neuroscience Letters, 359(1-2), 130-132.
- Tanji, J. (2001). Sequential Organization of Multiple Movements: Involvement of Cortical Motor areas. Annual Review of Neuroscience, 24(1), 631-651.
- Graybiel, A. M. (2008). Habits, Rituals, and the EvaluativeBrain. Annual Review of Neuroscience, 31(1), 359-387.
- Warabi, T., Fukushima, K., Olley, P. M., Chiba, S., & Yanagisawa, N. (2011). Difficulty in Terminating the Preceding Movement/Posture Explains the Impaired Initiation of New Movements in Parkinson's Disease. Neuroscience Letters, 496(2), 84-89.
- Bologna, M., Paparella, G., Fasano, A., Hallett, M., & Berardelli, A. (2020). Evolving Concepts on Bradykinesia. Brain, 143(3), 727-750.
- Carlsöö, S., & Molbech, S. (1966). The Functions of Certain Two-Joint Muscles in a Closed Muscular Chain. Acta Morphologica Neerlando-Scandinavica, 6(4), 377-386.
- Basmajian, J. V. (1962). Muscles Alive. Their Functions Revealed by Electromyography. Academic Medicine, 37(8), 802.
- Harrington, D. L., & Haaland, K. Y. (1991). Sequencing in Parkinson's Disease: Abnormalities in Programming and Controlling Movement. Brain, 114(1), 99-115.
- Crenna, P., & Frigo, C. (1991). A Motor Programme for the Initiation of Forwardâ?ÂÃÂ?ÂÂOriented Movements in Humans. The Journal of Physiology, 437(1), 635-653.
- Warabi, T., Furuyama, H., Sugai, E., Kato, M., & Yanagisawa,N. (2018). Gait Bradykinesia in Parkinson’s Disease: a Change in the Motor Program Which Controls the Synergy of Gait. Experimental Brain Research, 236(1), 43-57.
- Alexander, G. E., DeLong, M. R., & Strick, P. L. (1986). Parallel Organization of Functionally Segregated Circuits Linking Basal Ganglia and Cortex. Annual Review of Neuroscience, 9(1), 357-381.
- Crammond, D. J., & Kalaska, J. F. (1994). Modulation of Preparatory Neuronal Activity in Dorsal Premotor Cortex Due to Stimulus-Response Compatibility. Journal of Neurophysiology, 71(3), 1281-1284.
- Middleton, F. A., & Strick, P. L. (1994). Anatomical Evidence for Cerebellar and Basal Ganglia Involvement in Higher Cognitive Function. Science, 266(5184), 458-461.
- Tanji, J. (1994). The Supplementary Motor area in the CerebralCortex. Neuroscience Research, 19(3), 251-268.
- Sabatini, U., Boulanouar, K., Fabre, N., Martin, F., Carel, C., Colonnese, C., Bozzao, L., Berry, I., Montastrue, J. L., Chollet, F., & Rascol, O. (2000). Cortical Motor Reorganization in Akinetic Patients with Parkinson's Disease: a Functional MRI Study. Brain, 123(2), 394-403.
- Takakusaki, K., Saitoh, K., Harada, H., & Kashiwayanagi,M. (2004). Role of Basal Ganglia–Brainstem Pathways in the Control of Motor Behaviors. Neuroscience Research, 50(2), 137-151.
- Hughes, A. J., Daniel, S. E., Kilford, L., & Lees, A. J. (1992). Accuracy of Clinical Diagnosis of Idiopathic Parkinson's Disease: a Clinico-Pathological Study of 100 Cases. Journal of Neurology, Neurosurgery & Psychiatry, 55(3), 181-184.
- Hikosaka, O., & Wurtz, R. H. (1985). Modification of Saccadic Eye Movements by GABA-Related Substances. I. Effects of Muscimol and Bicuculline in Monkey Superior Colliculus. Journal of Neurophysiology, 53(1), 266-291.
- Warabi, T., Kato, M., Kiriyama, K., Yoshida, T., & Kobayashi,N. (2004). Analysis of Human Locomotion by Recording Sole-Floor Reaction Forces from Anatomically Discrete Points. Neuroscience Research, 50(4), 419-426.
- Warabi, T., & Kato, M. (2009). A New Method to Measure Sole-Floor Reaction Forces from Anatomically Discrete Points of the Sole During Human Locomotion. Recent Research Developments in Neuroscience, 3, 1-23.
- Warabi, T., Furuyama, H., & Kato, M. (2020). Gait Bradykinesia: Difficulty in Switching Posture/Gait Measured by the Anatomical Y-Axis Vector of the Sole in Parkinson’s Disease. Experimental Brain Research, 238(1), 139-151.
- Perret, E., Eggenberger, E., & Siegfried, J. (1970). Simple and Complex Finger Movement Performance of Patients with Parkinsonism Before and After a Unilateral Stereotaxic Thalamotomy. Journal of Neurology, Neurosurgery, and Psychiatry, 33(1), 16.
- Warabi, T., Inoue, K., & Yanagisawa, N. (2020). Motor Programs Interlinking Gait and Bradykinesia in Parkinson's Disease. In C. Martin & V. Preedy (Eds), Genetics, Neurology, Behavior, and Diet in Parkinson's Disease (pp. 521-537). Academic Press.
- Poizner, H., Feldman, A. G., Levin, M. F., Berkinblit, M. B., Hening, W. A., Patel, A., & Adamovich, S. V. (2000). The Timing of Arm-Trunk Coordination is Deficient and Vision-Dependent in Parkinson's Patients During Reaching Movements. Experimental Brain Research, 133(3), 279-292.
- DeLong, M. R. (1990). Primate Models of Movement Disorders of Basal Ganglia Origin. Trends in Neurosciences, 13(7), 281-285.
- Wichmann, T., Bergman, H., & DeLong, M. R. (1994). The Primate Subthalamic Nucleus. I. Functional Properties in Intact Animals. Journal of Neurophysiology, 72(2), 494-506.
- Mink, J. W. (1996). The Basal Ganglia: Focused Selection and Inhibition of Competing Motor Programs. Progress in Neurobiology, 50(4), 381-425.
- Hikosaka, O., Takikawa, Y., & Kawagoe, R. (2000). Role of the Basal Ganglia in the Control of Purposive Saccadic Eye Movements. Physiological Reviews, 80(3), 953-978.
- Nambu, A., Kaneda, K., Tokuno, H., & Takada, M. (2002). Organization of Corticostriatal Motor Inputs in Monkey Putamen. Journal of Neurophysiology, 88(4), 1830-1842.
- Gulberti, A., Wagner, J. R., Horn, M. A., Reuss, J. H., Heise,M., Koeppen, J. A., Pinnschmolt, H. O., Westphal, M., Engel,A. K., Gerloff, C., Shorott, A., Hamel, W., Moll, C. K. E., & Pötter-Nerger, M. (2023). Subthalamic and Nigral Neurons are Differentially Modulated During Parkinsonian Gait. Brain, 146(7), 2766-2779.
- Crutcher, M. D., & Alexander, G. E. (1990). Movement- Related Neuronal Activity Selectively Coding Either Direction or Muscle Pattern in Three Motor Areas of the Monkey. Journal of Neurophysiology, 64(1), 151-163.
- Crenna, P., Carpinella, I., Rabuffetti, M., Rizzone, M., Lopiano, L., Lanotte, M., & Ferrarin, M. (2006). Impact of Subthalamic Nucleus Stimulation on the Initiation of Gait in Parkinson’s Disease. Experimental Brain Research, 172(4), 519-532.
- Takakusaki, K., Tomita, N., & Yano, M. (2008). Substrates for Normal Gait and Pathophysiology of Gait Disturbances with Respect to the Basal Ganglia Dysfunction. Journal of Neurology, 255(Suppl 4), 19-29.
- Parkinson, J. (2002). An Essay on the Shaking Palsy. The Journal of Neuropsychiatry and Clinical Neurosciences, 14(2), 223-236.
- Nutt, J. G., Bloem, B. R., Giladi, N., Hallett, M., Horak, F. B., & Nieuwboer, A. (2011). Freezing of Gait: Moving Forward on a Mysterious Clinical Phenomenon. The Lancet Neurology, 10(8), 734-744.
- Nantel, J., McDonald, J. C., Tan, S., & Bronte-Stewart, H. (2012). Deficits in Visuospatial Processing Contribute to Quantitative Measures of Freezing of Gait in Parkinson’s Disease. Neuroscience, 221, 151-156.
- Delval, A., Rambour, M., Tard, C., Dujardin, K., Devos, D., Bleuse, S., Defebvre, L., & Moreau, C. (2016). Freezing/ Festination During Motor Tasks in Earlyâ?ÂÃÂ?ÂÂStage Parkinson's Disease: a Prospective Study. Movement Disorders, 31(12), 1837-1845.
- Thach, W. T. (1978). Correlation of Neural Discharge with Pattern and Force of Muscular Activity, Joint Position, and Direction of Intended Next Movement in Motor Cortex and Cerebellum. Journal of Neurophysiology, 41(3), 654-676.
- Marsden, C. D., & Rothwell, J. C. (1987). The Physiology of Idiopathic Dystonia. Canadian Journal of Neurological Sciences, 14(S3), 521-527.
- Carpinella, I., Crenna, P., Calabrese, E., Rabuffetti, M., Mazzoleni, P., Nemni, R., & Ferrarin, M. (2007). Locomotor Function in the Early Stage of Parkinson's Disease. IEEE Transactions on Neural Systems and Rehabilitation Engineering, 15(4), 543-551.
- Alexander, G. E., & Crutcher, M. D. (1990). Functional Architecture of Basal Ganglia Circuits: Neural Substrates of Parallel Processing. Trends in Neurosciences, 13(7), 266-271.
- Benecke, R., Rothwell, J. C., Dick, J. P. R., Day, B. L., & Marsden, C. D. (1987). Disturbance of Sequential Movements in Patients with Parkinson's Disease. Brain, 110(2), 361-379.
- Warabi, T., Noda, H., Yanagisawa, N., Tashiro, K., & Shindo,R. (1986). Changes in Sensorimotor Function Associated with the Degree of Bradkinesia of Parkinson's Disease. Brain, 109(6), 1209-1224.
- Yanagisawa, N., Hayashi, R., & Mitoma, H. (2001). Pathophysiology of Frozen Gait in Parkinsonism. Advances in Neurology, 87, 199-207.


