Research Article - (2021) Volume 5, Issue 1
Development of shoe insert for Diabetic Foot Ulcer Patients in case of Ethiopia
2Department of Chemical Engineering, Bahir Dar Institute of Technology, Bahir Dar University, Ethiopia
Received Date: Dec 13, 2021 / Accepted Date: Dec 21, 2021 / Published Date: Dec 24, 2021
Copyright: ©Copyright: ©2021 Awoke Fenta Wodag, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Awoke Fenta Wodag, Adane Adugna Ayalew. (2021). Development of shoe insert for Diabetic Foot Ulcer Patients in case of Ethiopia, Stem Cell Res Int, 5(1), 24-28.
Abstract
Foot ulcer is mainly developing due to diabetic disease. It is an open sore on the foot. The aim of this research is to develop polyurethane memory foam in sock/shoe insert in Ethiopia by replacing costly material silicon surfactant by locally available and affordable input materials castor oil as surfactant and tap water instead of distilled water for the diabetic foot ulcer patients. Under this research, it was able to prove the severity of foot ulcer in Ethiopia and enhance the high demand of orthotic appliance. Three memory foams in sock with different surfactant ratios were manufacture at AMAGA P.L.C foam factory, Bahir Dar Ethiopia using standard Polyols and diisocynates. The shoe insert /in sock was prepared by using materials used in the factory to prepare polyurethane (PU) memory foam mattress with 5g, 9.5 g and 10.36g of castor oil surfactant amount as a new replaced material instead of costly silicon surfactant from the three the first (castor oil 5g is not form a foam and hence no further investigation is conducted on it). Physical properties such as thickness, density, hardness, compression set, water absorption and abrasion resistance of developed in sock were tested. Further the new developed in sock was compared with that of existed commercial polyurethane in sock and the SATRA standards. The new developed in sock has showed an equivalent mechanical property with the standard
Keywords
Shoe Insert, Memory Foam; Foot Ulcer; Diabetic Patient; Polyurethane, Castor Oil.
Introduction
Foot ulcer is a term that references an open sore that forms on or near one or both of our feet. It may form on the bottom of the foot, the toes, and the top of the foot or even the lower region of the leg mainly caused by diabetic disease [2]. According to research result shows about 15% of diabetic patients develop foot ulcers in their lifetime enhancement [3]. Diabetic foot ulcer is becoming major concern of diabetic patients. People with a diabetic foot ul- cer (DFU) have a 40% greater 10-year death rate than people with diabetes alone. Foot ulcer problems with diabetic patients account for up to 15% of healthcare resources in developed countries and 40% in developing countries and one in every six people with dia- betes will develop ulcer [5].
Poverty and unhygienic conditions associated with foot ulceration are major factors contributing to development of the diabetic foot in developing countries like Ethiopia and for diabetic patients liv- ing at or below poverty line, the purchase of appropriate footwear (orthopedic footwear) might not be affordable, feasible or of high priority [1]. For diabetic foot ulcer patients and people who are overweight, extra pressure may be the cause of ongoing foot pain/ ulcer but by using /wearing orthopedic footwear appliance can be redistribute evenly and the development of ongoing foot pain/ ul- ceration development retards [4].
The, knees, and back are just some of the body parts that can be ad- versely affected by improper posture or gait. Orthotics can amend this by spreading the load uniformly around the feet, and then as- sure that they hit the ground perpendicularly, that is why it is so important that cushion memory foam shoe insert is a need to redis- tributes our pressure by absorbing shock. Inappropriate footwear and inappropriate insert can have a large share in contributing fac- tor to the development of foot ulceration [8]. Researches implied, footwear with cushion shoe insert has been implemented to reduce pressure on the foot of diabetic patients [2].
Today Memory foam shoe inserts is available commercially at high cost and its source for foot ulcer purpose is import from developed Countries at high cost. This leads low access, and unable to be use by the low-income patients due to higher cost. Hence, replacement of castor oil surfactant and tap water in the place of silicon sur- factant and distilled water had respectively better option. Polymer elastomers and foams are mostly use to fit the material properties of shoe appliances, like insole and in sock. Mainly ethylene vinyl acetate (EVA), polyurethane (PU) and rubber based materials are mostly used in footwear [8], [10]. PU foams and elastomers are demonstrated, as the best effective materials for shock absorption in footwear [6].
Different types of foam are available commercially at high cost. But the available materials are not scientifically characterized and optimized for application as in sock for foot ulcer patients treat- ment. PU viscoelastic cushion in sock will reduce the foot plantar pressure and give additional cushion support to the foot arches. Therefore, new PU memory foams were developed for application as shoe insert in therapeutic footwear for patients with diabetes. The developed materials were characterized for physical and me- chanical properties and PU foam composition was also optimized for application as cushion in sock.
Materials and Methods
In this study the PU Memory Foam in sock was prepared by using standard polyols, HT, and isocyanates-toluene di isocyante (TDI), castor oil and methyl chloride were obtained from AMAGA, PLC. Bahir Dar bed foam manufacturing and Ethiopian Institute of Textile and Fashion Technology, Bahir Dar University, Bahir Dar Ethiopia. Other additive and auxiliary such as tin, thertialy cross linker; catalyst, inorganic filler and cell opener which are physi- cochemical and environmentally safe were used. The following chemicals specification present in Table1 were also derived from AMAGA, P.L.C. Bahir Dar bed foam manufacturing used for PU memory foam formation at different concentration of castor oil.
Table 1: Quantity of Raw Materials for Three Pair of In Sock / Shoe Inserts Sample Production
|
Component |
Sample -1 |
Sample-2 |
Sample -3 |
|
|
No. |
ingredients used |
Weight (g) |
Weight (g) |
Weight (g) |
|
1 |
Polyols HT,762 |
93.3 |
93.3 |
93.3 |
|
2 |
Tap water |
2*2.6 |
2*2.6 |
2*2.6 |
|
3 |
Castor oïl surfactant |
5 |
9.5 |
10.36 |
|
4 |
Tertiary Amine Catalyst |
1.7 |
1.7 |
1.7 |
|
5 |
Tin Catalyst |
1.37 |
1.37 |
1.37 |
|
6 |
Cross linkers |
8.4 |
8.4 |
8.4 |
|
7 |
Isocynates, TDI |
75.5 |
75.5 |
75.5 |
|
8 |
Auxiliary blowing agents |
4.7 |
4.7 |
4.7 |
|
Total consumption |
----------- |
199.67g |
199.67g |
200.5g |
Experimental Procedure for Foam Manufacturing
In this research work the PU memory foam in sock samples were developed using the following procedural steps.
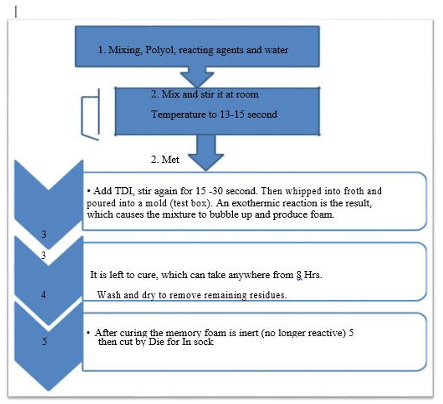
Figure 1: PU Memory Foam in Sock Sample-Manufacturing Steps
According to Figure: 1 Polyol, reacting agents and tap water were mixed, and stirrer at 30oC temperature for 14 seconds. Isocya- nates-toluene Di Isocyante (TDI) was added and again stirrer for 15 seconds to make sure the reactant was well reacted. After all, the product was whipped into froth and poured into a mold (test box). An exothermic reaction was resulted which caused the mix- ture to bubble up and produce foam. The final product foam was no longer reactive (cured) and stored for 8 hours and then the foam was cut by die for next application.
Physiochemical Characterization
Type of foams with varying density and cushion ability were de- veloped by varying the concentration of surfactant (castor oil). The developed foams were characterized for thickness (mm), density (g/cc) hardness (mg/L), water absorption (%) compression set (%) abrasion resistance and pressure redistributing properties of the in sock were.
Plantar Pressure Distribution Test
The plantar pressure reduction and distribution properties of the developed insert/in sock, foam was measured using carbon copy pressure measurement system, and then related with that of ethyl vinyl acetate (EVA) in sock/ shoe insert which was conducted at Leather Industry development institute (LIDI) and Ethiopian insti- tute of textile and Fashion Technology (EiTEX) research laborato- ry with ELETTROTECNIC A.B.C machine at 7 bar pressure and were analyzed the materials effect on the foam.
Results and Discussion
The physiomechanical properties of the PU memory foam are dis- cussed based on the data presented in Table 1& 2. It has shown that the initial formulation of PU memory foam in (sample 1) is not foam but as a thick liquid this is due to very low amount (5g) of castor oil as a surfactant is used due to this no further study is done on it and (sample 2) is not suitable for application as cushion in sock / shoe insert for Diabetic foot ulcer due to low hardness, density and low compassion but sample three shows a very good properties.
Density: By taking similar thickness of PU memory foam in sock and size. The density test resulted of samples are presented in Table 1. The test result value for sample 2 was 0.194 g/cm3and 0.22g/cm3 for sample 3.
If the density is below standard requirements the shoe insert will compressed and sticky during use and if it is too high, it consume more material and become costly and less comfort. Material with higher density the higher the load bearing capacity, this implies that the sample 3 has remarkable load bearing capacity and sup- port. Therefore, the replacement of materials silicon surfactant by castor oil surfactant and distilled water by tap water shows a posi- tive effect in this particular property
Table2: Physical Test Results of PU Memory Foams]
|
No. |
Properties |
Obtained value |
|
||
|
|
|
Sample 2 |
Sample 3 |
Commercial insert |
SATRA standard |
|
1 |
Density (g/cc) |
0.194 |
0.22 |
0.253 |
0.2-0.3 |
|
2 |
Hardness (mg/L) |
19.25 |
21.09 |
5-7 |
17-22 |
|
3 |
Thickness (mm) |
11 |
11.5 |
8 |
13-14 |
|
4 |
Abrasion resistance (%) |
2.98 |
2.62 |
6.9 |
- |
|
5 |
Compression (%) |
3.82 |
4.2 |
6.1 |
5 |
|
6 |
Water absorption (%) |
63 |
52.5 |
39 |
30 |
Hardness: The mean value of hardness was shown is 19.25 % of the new developed PU memory foam in sock of sample 2 and 21.09 for sample 3 is obtained within the standard range in SATRA test method, and greater than that of existing PU foam in sock. The failing of the test result below standard requirement leads the product strength somewhat poor. On the other side, if this prop- erty is above standard it will be less flexible. The only difference between in sample 2 and 3 is due to using of different amount of surfactant (castor oil). Therefore, this research finding fulfills stan- dard requirement.
Thickness: The recipe of the chemicals/materials including the new surfactant material, castor oil and tap water is formulated based on SATRA standards and Amaga foam factory in Ethio- pia materials to produce 11mm foam in sock thickness and then manufactured it. Mean thickness of sample 2 and 3 are uniform. Therefore, research result shows it is possible to manufacture these products with uniform thickness.
Compression Set: Elastic properties of the in sock were conduct- ed for both sample 2 and 3 each in average is 3.82%, which is within standard requirement. In case of sample 2 is 4.2% which is also between the standard but its value is greater than that of sample 1.Its commercial insert value is 6.1, which is not fit with standard requirements, this shows that just commercial in sock is not good for diabetic foot ulcer patients foot ulcer treatment due to lower elastic property than that of standards. This leads the product to have lower comfort particularly in this property because ortho- pedic shoe appliance need to be more flexible and comfort for foot ulcer patients and with good compression set has good comfort.
Abrasion Test: The purpose of testing of this property is to know its resistance to abrasion because during wearing the in sock will rub between our feet and the shoe sole/insole, so it needs to have very good abrasion resistance. To know this property, the experi- mental test in abrasion resistance of normal PU foam and that of the newly developed foam in sock were conducted. From these as result shown the PU memory foam in sock abrasion resistance resulted is very good that commercial foam because it has less amount of weight loses due to abrasion than that of normal PU foam. This implies that the newly developed memory foam in sock abrasion property.
Pressure Redistribution Test of the Shoe Insert
The pressure reduction and distribution properties of the devel- oped shoe insert was measured at leather industry development institute (LIDI) Addis Ababa, Ethiopia. The ethyl vinyl acetate, (EVA) and the newly developed memory foam shoe insert carbon test results is presented in Figure 2.
Figure 2: Comparison of (A) Carbon Copy Test Result of Eva and (B) Memory Foam Shoe Insert
The contact area (points) of carbon on in sock were almost the same but, the figure of test results of memory foam shoe insert slightly greater than that of EVA shoe insert, this implies that, our foot contact to shoe insert during wear is also higher. Therefore, if the contact area is higher it reduces our body pressure and redis- tributes it to the ground in addition the insert can retard the pain of the ulcer by increasing contact areas of our feet.
Conclusion
Foot ulcer is an open sore on the bottom of our feet mainly caused by diabetic disease. Therapeutic shoe with appliance (in socks) like PU memory foam in sock plays a great role in retarding foot ulcer. In this study the insert is developed with 5g, 9.5 g and 10.36g of castor oil surfactant. The in sock with 10.36g of castor amount has shown better physical and comfort properties and that of which with 5g castor oil quantity were unable to be foam but it remains as a thick liquid. Generally the in sock is produced, at low cost by including less cost and locally available surfactant materials (cas- tor oil) instead of costly silicon surfactant and tap water instead of distilled water. In the field test at Adina’s hospital, Bahir Dar Ethi- opia implies a very good comfort, and pressure reduction ability.
Over all, from the result of the research the insert has good futurity for foot ulcer patients and can solve limited access problem by affordable price.
Abbreviation
EVA: ethyl vinyl acetate; PU: Polyurethane; P.L.C: private limited company; SATRA: South African Telecommunications Regulato- ry Authority; LIDI: leather Industry development Institute; TDI: toluene Di –Isocyante; MM: millimeter/CC: gram per centimeter cubic ;G/CM3: gram per centimeter; G/L: gram per liter; HT; spe- cific code polyurethane
Acknowledgment
Our sincere gratefulness goes to the staff of Ethiopian Institute of Textile and Fashion Technology, Lather Technology Bahir Univer- sity for their help and the institute for the financial funding, as well as providing access to all necessary materials for the successful completion of the research.
Author’s Contribution
Awoke Fenta Wodag: Conceptualization the idea, Methodology, Data analysis, writing the first draft preparation, Visualization, In- vestigation, Final reviewing, and editing. Adane Adugna Ayalew: Reviewing the manuscript formats, paper fonts and structure
Funding
This work was supported by research and Postgraduate graduated office, Ethiopian Institute of Textile and Fashion Technology Bahir Dar, University
Availability of Data and Materials
All the datasets are available from corresponding author if re- quired.
Ethics Approval and Consent to Participate.
All the participants, from Adinas Hospital patients in Bahir Dar, Ethiopia, are asked and had showed their voluntariness’ (consent) prior to their participation and then the products are checked on them.
Consent for Publication
Not applicable.
Conflict of Interest
It is declared that they have not conflict of interest.
References
- Eginton, M. T., Brown, K. R., Seabrook, G. R., Towne, J. B.,& Cambria, R. A. (2003). A prospective randomized evalu-ation of negative-pressure wound dressings for diabetic foot wounds. Annals of vascular surgery, 17(6), 645-649.
- Gebre, M. W. (2013). Diabetes mellitus and associated diseas- es from Ethiopian perspective: systematic review. Ethiopian Journal of Health Development, 27(3), 249-253.
- Armstrong, D. G. (2005). Diabetic foot study consortium. Negatrve pressure wound therapy after partial diabetic foot amputation: a multicentre, randomised controlled trial. Lan- cet, 366(9498), 1704-1710.
- Jeon, C. Y., Harries, A. D., Baker, M. A., Hart, J. E., Kapur, A., Lönnroth, K., ... & Murray, M. B. (2010). Biâ?directional screening for tuberculosis and diabetes: a systematic review. Tropical Medicine & International Health, 15(11), 1300-1314.
- Mamo, T., Yifter, H., & Lemessa, T. (2015). Risk factors as- sessment of diabetic foot ulcer using the sixty seconds screen- ing tool: a hospital based cross-sectional study at Tikur An- bessa specialized Hospital. Ethiop Med J, 2, 45-9.
- Khan, H., Khan, Z., Khan, I., Din, J., Rehman, S., & Khan,B. (2011). Factors contributing to the development of diabetic foot ulcers and role of health literacy. Rawal Medical Journal, 36(1), 34-37.
- Gnanasundaram, S., Durairaj, D., Gopalakrishna, G., & Das,B. (2013). PU viscoelastic memory foam for application as cushion insole/insock in shoes. Footwear Science, 5(sup1), S22-S23.
- Zhang, P., Lu, J., Jing, Y., Tang, S., Zhu, D., & Bi, Y. (2017).Global epidemiology of diabetic foot ulceration: a systematic review and meta-analysis. Annals of medicine, 49(2), 106- 116.
- Dhanapal, D., & Raja, K. (2016). PU memory foam for ap- plication as insole in therapeutic footwear for patients with diabetes. IJESC, 6(6), 7784-7785.
- R.Jai Vinod kumar, Sathyaseelan, Jeyakumar 2016. Foam dressings in the Topical Management of Diabetic Foot. J. of Dental and Medic. Sciences. 15, 32-35.
- Saraswathy, G., Gopalakrishna, G., Das, B. N., Mohan, R., Radhakrishnan, G., & Pal, S. (2009). Development of poly- urethane-based sheets by coagulation method and study of mechanical and cushion properties for therapeutic footwear applications. Polymer-Plastics Technology and Engineering, 48(3), 239-250.
- Deribe, B., Woldemichael, K., & Nemera, G. (2014). Preva- lence and factors influencing diabetic foot ulcer among dia- betic patients attending Arbaminch Hospital, South Ethiopia. J Diabetes Metab, 5(1), 1-7.
- Worku, D., Hamza, L., & Woldemichael, K. (2010). Patterns of diabetic complications at jimma university specialized hos- pital, southwest ethiopia. Ethiopian journal of health sciences, 20(1), 33-39.
- Yemane, T., Belachew, T., & Asaminew, B. (2007). Type II di- abetes mellitus in Jimma Town, southwest Ethiopia. Ethiopian Journal of Health Sciences, 17(2), 107-114.


