Research Article - (2024) Volume 1, Issue 2
Determining Kinetics Parameters of Oxidase-Like Ce/Fe-Bimetallic Nanozymes Toward 3,3′,5,5′-Tetramethylbenzidine Oxidation
Received Date: Nov 04, 2024 / Accepted Date: Nov 25, 2024 / Published Date: Dec 13, 2024
Copyright: ©Â©2024 Mohammad Reza Hormozi Jangi, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Hormozi Jangi, A. R., Hormozi Jangi, M. R. (2024). Determining Kinetics Parameters of Oxidase-Like Ce/Fe-Bimetallic Nanozymes Toward 3,3?,5,5?-Tetramethylbenzidine Oxidation. Arch of case Rep: Open, 1(2), 01-05.
Abstract
Herein, kinetics parameters, Km and Vmax, of oxidase-like Ce/Fe-bimetallic nanozymes toward 3,3′,5,5′-tetramethylbenzidine oxidation were determined. The Michaelis–Menten kinetic model was used for the evaluation of the kinetic of the nanozyme- mediated oxidation of 3,3′,5,5′-tetramethylbenzidine. To provide the quantitative and accurate values of kinetics parameters, Km and Vmax, the linear plot of Lineweaver–Burk was constructed. The results exhibited a Vmax as high as 67.56 nM min-1 for the oxidase-like Ce/Fe-bimetallic nanozymes. Besides, Km was found to be as low as 0.06 mM for the as-prepared nanozymes, revealing the high affinity of the nanozymes toward 3,3′,5,5′-tetramethylbenzidine. Moreover, the ratio Vmax/Km was estimated as a reliable index of catalytic efficiency of the nanozymes, revealing a high value of 1.0×10-3 min-1.
Keywords
Oxidase-Like Nanozyme, Ce/Fe-Bimetallic Nanozymes, Michaelis–Menten Kinetic Model, Catalytic Efficiency, 3,3′,5,5′-Te-tramethylbenzidine
Introduction
Native enzymes suffer some disadvantages such as instability in harsh reaction conditions, difficult recovery, etc [1-10]. In contrast, nanomaterials with enzyme-like activity reveal high stability against pH and temperature changes along with excellent kinetic performances [11-19]. In fact, with the development of nanoscience, several types of nanomaterials were introduced with unique spectral, optical, catalytic, and stability as a result of the fast development of nanoscience in recent years, some of them reveal significant enzyme-like activity especially peroxidase-like properties with significant advantages over native enzymes for example, high pH and thermal stability, excellent reusability, and high storage stability [20-43]. Up to now, nanozymes have been utilized in organic dye biodegradation, battery development, sensor, and biosensor design, especially after the first report of COVID-19, they applied for its clinical sensing [44-64]. In this field proving the catalytic mechanism of the reaction is an attractive research topic. Hence, herein, kinetics parameters, Km and Vmax, of oxidase-like Ce/Fe-bimetallic nanozymes toward 3,3′,5,5′-tetramethylbenzidine oxidation were determined. The Michaelis–Menten kinetic model was used for the evaluation of the kinetic of the nanozyme-mediated oxidation of 3,3′,5,5′-tetramethylbenzidine. To provide the quantitative and accurate values of kinetics parameters, Km and Vmax, the linear plot of Lineweaver–Burk was constructed.
Experimental
Synthesis of Nanozymes
To synthesize the oxidase-like Ce/Fe-bimetallic nanozymes, 0.4 g fumaric acid was added to 25.0 mL water (solution#1). Besides, 0.3 g (NH4)2[Ce(NO3)6] and 0.3 g Fe(NO3)3.9H2O were introduced to 25 mL water (solution#2) . Afterward, solution#1 was added drop by drop to solution#2, totaling 50 mL, and stirred for 1 hour. The reaction mixture was then heated at 120 ºC for 2 hours to complete the synthesis process.
Kinetic Studies
The Michaelis–Menten kinetic model was used for the evaluation of the kinetic of the nanozyme-mediated oxidation of 3,3′,5,5′-tetramethylbenzidine. To provide the quantitative and accurate values of kinetics parameters, Km and Vmax, the linear plot of Lineweaver–Burk was constructed. It is notable that 3,3′,5,5′-tetramethylbenzidine was used as the standard substrate [65-67].
Results and Discussion
The kinetics of the oxidation process involving TMB chromogenic agents on oxidase-like Ce/Fe-bimetallic nanozymes were investigated. This examination involved analyzing the kinetic components of the Michaelis-Menten equation using the following formula;
V0=(Vmax[S])/(Km+[S]
where V0 represents the initial velocity, Vmax stands for the maximum reaction velocity, [S] denotes the concentration of chromogenic agents, TMB, and Km signifies the Michaelis constant as previously reported in the literature [68-75]. To do this, a series of experiments were carried out for the oxidation of different concentrations of TMB in different concentrations of 0.015-0.2 mM over the developed nanozymes. The oxidation reaction rate was then calculated in nM min-1. Afterward, the Michaelis-Menten curve was plotted by plotting the rate as a function of TMB concentration. The results are shown in Figure 1. As can be seen in this figure, the rate of the oxidation process was increased by increasing the TMB concentration and then leveling off. Based on the Michaelis-Menten curve, a Vmax as high as 55. 5 nM min-1 was provided for the oxidase-like Ce/Fe-bimetallic nanozymes, revealing their high catalytic activity. Besides, the Km value was found to be about 0.05 mM. Moreover, Vmax/Km was calculated as a crucial indicator of enzyme catalytic efficiency, the results revealed a ratio of 1.10 ×10-3 min-1 from the non-linear Michaelis- Menten curve.
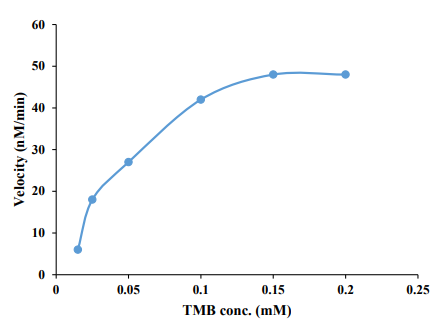
Figure 1: The non-linear Michaelis-Menten curve of the oxidase-like Ce/Fe-bimetallic nanozymes
Besides, the linear Burk diagram was also constructed by plotting the 1/V against 1/[TMB]. The results are shown in Figure 2. As can be seen in this figure, based on the linear-Burk diagram, a Vmax as high as 67.56 nM min-1 was provided for the oxidase-like Ce/ Fe-bimetallic nanozymes, revealing their high catalytic activity.Besides, the Km value was found to be about 0.067 mM, revealing the high affinity of the oxidase-like Ce/Fe-bimetallic nanozymes to TMB. It is notable that the results of the non-linear Michaelis-Menten curve and linear Burk diagram are in good agreement with each other. Moreover, Vmax/Km was calculated as a crucial indicator of enzyme catalytic efficiency, the results revealed a ratio of 1.0×10-3 min-1 from the linear-Burk diagram which is close to that of the results of the non-linear Michaelis-Menten curve. It is notable, that the summary of the kinetic parameters of the oxidase-like Ce/Fe-bimetallic nanozymes provided from linear-Burk diagram and non-linear Michaelis-Menten curve are represented in Table 1.
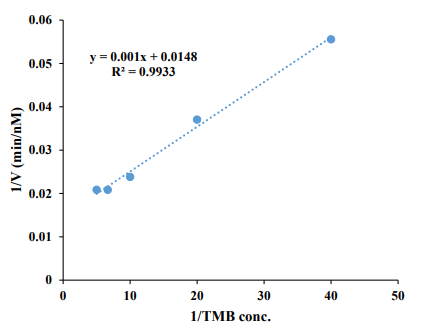
Figure 2: The linear-Burk diagram of the oxidase-like Ce/Fe-bimetallic nanozymes.
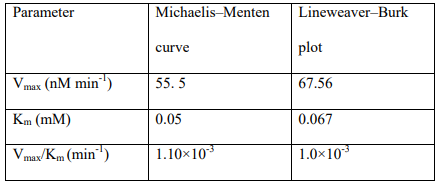
Table 1: Kinetic parameters of the oxidase-like Ce/Fe-bimetallic nanozymes provided from the linear-Burk diagram and non-linear Michaelis-Menten curve
Conclusions
Herein, kinetics parameters, Km and Vmax, of oxidase-like Ce/ Fe-bimetallic nanozymes toward 3,3′,5,5′-tetramethylbenzidine oxidation were determined. The Michaelis–Menten kinetic model was used for the evaluation of the kinetic of the nanozyme-mediated oxidation of 3,3′,5,5′-tetramethylbenzidine. To provide the quantitative and accurate values of kinetics parameters, Km and Vmax, the linear plot of Lineweaver–Burk was constructed. The results exhibited a V max as high as 67.56 nM min-1 for the oxidase-like Ce/Fe-bimetallic nanozymes. Besides, Km was found to be as low as 0.06 mM for the as-prepared nanozymes, revealing the high affinity of the nanozymes toward 3,3′,5,5′-tetramethylbenzidine. Moreover, the ratio Vmax/Km was estimated as a reliable index of catalytic efficiency of the nanozymes, revealing a high value of 1.0×10-3 min-1.
References
- Ghaffari-Moghaddam, M., Eslahi, H., Omay, D., & Zakipour-Rahimabadi, E. (2014). Industrial applications of enzymes. Review Journal of Chemistry, 4, 341-361.
- Basso, A., & Serban, S. (2019). Industrial applications of immobilized enzymes—A review. Molecular Catalysis, 479, 110607.
- Okpara, M. O. (2022). Microbial enzymes and their applications in food industry: a mini-review. Advances in Enzyme Research, 10(1), 23-47.
- Piotrowska-DÅ?ugosz, A. (2019). Significance of enzymes and their application in agriculture. Biocatalysis: Enzymatic Basics and Applications, 277-308.
- Hormozi Jangi, M. R., & Hormozi Jangi, A. R. (2024). Oxygen Vacancies and Hydroxyl Radicals are Contributed in Catalytic Oxidation of 3, 3′, 5, 5′-Tetramethylbenzidine Over NH2-MiL-88 (Fe, Ni) as the Main Reactive Oxygen Species. J Nur Healthcare, 9(4), 01-06.
- Hormozi Jangi, M. R., & Hormozi Jangi, A. R. (2024). Inhibitory and Stability Studies on Graphene Oxide Gold Nanozymes. Arch of case Rep: Open, 1(2), 01-06.
- Anbu, P., Gopinath, S. C., Chaulagain, B. P., & Lakshmipriya,T. (2017). Microbial enzymes and their applications in industries and medicine 2016. BioMed research international, 2017.
- Jangi, A. R. H., & Jangi, M. R. H. (2024). Identification of reactive oxygen species contributed in catalytic oxidation of 3, 3′, 5, 5′-tetramethylbenzidine over NH2-MiL-88 (Fe, Ni). Authorea Preprints.
- Jangi, M. R. H., & Jangi, A. R. H. (2024). Thermal and pH stability of peroxidase-like graphene oxide/gold nanoparticles. Authorea Preprints.
- Bhatia, S., & Bhatia, S. (2018). Introduction to enzymes and their applications. Introduction to pharmaceutical biotechnology, 2, 1-29.
- Jangi, S. R. H. (2023). Time Course Evaluation of Nanozyme-Mediated Reversible/Irreversible Oxidation Reactions over Silver Nanoparticles as Peroxidase Alternatives.
- Meng, Y., Li, W., Pan, X., & Gadd, G. M. (2020). Applications of nanozymes in the environment. Environmental Science: Nano, 7(5), 1305-1318.
- Kaimal, R., Dube, A., Al Souwaileh, A., Wu, J. J., & Anandan,S. (2024). A copper metal–organic framework-based electrochemical sensor for identification of glutathione in pharmaceutical samples. Analyst, 149(3), 947-957.
- Wu, J., Wang, X., Wang, Q., Lou, Z., & Li, S., et al. (2019).Nanomaterials with enzyme-like characteristics (nanozymes): next-generation artificial enzymes (II). Chemical Society Reviews, 48(4), 1004-1076.
- Jangi, S. R. H. (2023). Biochemical characterization of enzyme-like silver nanoparticles toward nanozyme-catalysed oxidation reactions. Micromaterials and Interfaces, 1(1).
- Hormozi Jangi, S. R. (2023). Evaluation of Biochemical Behavior and Stability of Gold Nanoparticles with High Intrinsic Peroxidase-Like Activity. Petro Chem Indus Intern, 6(4), 234-239.
- Jangi, S. R. H. (2023). Effect of daylight and air oxygen on nanozymatic activity of unmodified silver nanoparticles: Shelf-stability. Qeios.
- Huang, Y., Ren, J., & Qu, X. (2019). Nanozymes: classification, catalytic mechanisms, activity regulation, and applications. Chemical reviews, 119(6), 4357-4412.
- Liang, M., & Yan, X. (2019). Nanozymes: from new concepts, mechanisms, and standards to applications. Accounts of chemical research, 52(8), 2190-2200.
- Hormozi Jangi, S. R. (2023). Synthesis and characterization of magnesium-based metal-organic frameworks and investigating the effect of coordination solvent on their biocompatibility. Chemical Research and Nanomaterials, 1(4), 1-9.
- Jangi, S. R. H. (2024). Developing a label-free full-range highly selective pH nanobiosensor using a novel high quantum yield pH-responsive activated-protein-protected gold nanocluster prepared by a novel ultrasonication-protein-assisted procedure. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 322, 124829.
- Jangi, S. R. H. (2024). Developing a novel ultraselective and ultrasensitive label-free direct spectrofluorimetric nanobiosensor for direct highly fast field detection of explosive triacetone triperoxide. Analytica Chimica Acta, 1320, 343016.
- Dehghani, Z., Akhond, M., Jangi, S. R. H., & Absalan, G. (2024). Highly sensitive enantioselective spectrofluorimetric determination of R-/S-mandelic acid using l-tryptophan-modified amino-functional silica-coated N-doped carbon dots as novel high-throughput chiral nanoprobes. Talanta, 266, 124977.
- Hormozi Jangi, S. R. (2023). Low-temperature destructive hydrodechlorination of long-chain chlorinated paraffins to diesel and gasoline range hydrocarbons over a novel low-cost reusable ZSM-5@ Al-MCM nanocatalyst: a new approach toward reuse instead of common mineralization. Chemical Papers, 77(9), 4963-4977.
- Hormozi Jangi, S. R., & Gholamhosseinzadeh, E. (2023). Developing an ultra-reproducible and ultrasensitive label-free nanoassay for L-methionine quantification in biological samples toward application in homocystinuria diagnosis. Chemical Papers, 77(11), 6505-6517.
- Hormozi Jangi, S. R., & Akhond, M. (2023). Evaluating effect of solvent type, metal-core salt and heat-treatment on organic content, yield, and coordination strength in magnesium-based metal-organic frameworks using elemental analysis and Fourier transform infrared spectroscopy. Iranian Journal of Chemistry, 5(2), 225-234.
- Jangi, S. R. H., & Akhond, M. (2021). Ultrasensitive label-free enantioselective quantification of d-/l-leucine enantiomers with a novel detection mechanism using an ultra-small high-quantum yield N-doped CDs prepared by a novel highly fast solvent-free method. Sensors and Actuators B: Chemical, 339, 129901.
- Wang, G. L., Zhu, X. Y., Jiao, H. J., Dong, Y. M., & Li, Z.J. (2012). Ultrasensitive and dual functional colorimetric sensors for mercury (II) ions and hydrogen peroxide based on catalytic reduction property of silver nanoparticles. Biosensors and Bioelectronics, 31(1), 337-342.
- HORMOZI JANGI, S. R., & Akhond, M. (2020). High throughput green reduction of tris (p-nitrophenyl) amine at ambient temperature over homogenous AgNPs as H-transfer catalyst. Journal of Chemical Sciences, 132, 1-8.
- Huang, Y., Ren, J., & Qu, X. (2019). Nanozymes: classification, catalytic mechanisms, activity regulation, and applications. Chemical reviews, 119(6), 4357-4412.
- Jangi, S. R. H. A. (2023). A Mini-Review on Nanozym e Chemistry with Focus on Analytical and Bioanalytica l Sensing Applications.
- Liang, M., & Yan, X. (2019). Nanozymes: from new concepts, mechanisms, and standards to applications. Accounts of chemical research, 52(8), 2190-2200.
- Meng, Y., Li, W., Pan, X., & Gadd, G. M. (2020). Applications of nanozymes in the environment. Environmental Science: Nano, 7(5), 1305-1318.
- Zhang, R., Yan, X., & Fan, K. (2021). Nanozymes inspired by natural enzymes. Accounts of Materials Research, 2(7), 534-547.
- Wei, H., Gao, L., Fan, K., Liu, J., & He, J., et al. (2021).Nanozymes: A clear definition with fuzzy edges. Nano Today, 40, 101269.
- Jiangjiexing, W., & Hui, W. (2021). Efficient design strategies for nanozymes. Progress in Chemistry, 33(1), 42.
- Ahmed, S. R., Cardoso, A. G., Kumar, S., Ortega, G. A., & Srinivasan, S., et al. (2021). Nanozymes in biosensing and bioimaging. In Nanozymes (pp. 115-143). CRC Press.
- Lyu, Y., & Scrimin, P. (2021). Mimicking Enzymes: The quest for powerful catalysts from simple molecules to nanozymes. ACS Catalysis, 11(18), 11501-11509.
- Hormozi Jangi, S. R., & Dehghani, Z. (2024). Captopril Molecules Reveal Strong pH-, Temperature-, And Concentration-Dependent Inhibitory Effect on Nanozymatic Activity of Peroxidase-Like Nitrogen-Doped Carbon Dots. Petro Chem Indus Intern, 7(1), 01-07.
- Jangi, S. R. H. (2023). Experimental evaluation of kinetics and biochemical characteristics of MnO2 nanoparticles as high throughput peroxidase-mimetic nanomaterials. Micromaterials and Interfaces, 1(1).
- Zhang, X., Wu, D., Zhou, X., Yu, Y., & Liu, J., et al. (2019). Recent progress in the construction of nanozyme-based biosensors and their applications to food safety assay. TrAC Trends in Analytical Chemistry, 121, 115668.
- Jangi, A. R. H., Jangi, M. R. H., & Jangi, S. R. H. (2020).Detection mechanism and classification of design principles of peroxidase mimic based colorimetric sensors: A brief overview. Chinese Journal of Chemical Engineering, 28(6), 1492-1503.
- Hu, J., Zheng, Y., Yin, C., Wang, L., & Huang, H., et al. (2024). A novel and facile oxygen-activated time-temperature indicator with wide temperature monitoring range and good stability based on the laccase-like nanozyme. Analytica Chimica Acta, 1330, 343272.
- Hormozi Jangi, S. R. (2023). Detection mechanism and principles of the multinanozyme systems as the new generation of nanozyme-mediated sensing assays: A critical review. Petro Chem Indus Intern, 6(5), 349-357.
- Akhond, M., Hormozi Jangi, S. R., Barzegar, S., & Absalan,G. (2020). Introducing a nanozyme-based sensor for selective and sensitive detection of mercury (II) using its inhibiting effect on production of an indamine polymer through a stable n-electron irreversible system. Chemical Papers, 74, 1321-1330.
- Wang, J., Li, W., & Zheng, Y. Q. (2019). Nitro-functionalized metal–organic frameworks with catalase mimic properties for glutathione detection. Analyst, 144(20), 6041-6047.
- Jangi, S. R. H., Akhond, M., & Absalan, G. (2020). A novel selective and sensitive multinanozyme colorimetric method for glutathione detection by using an indamine polymer. Analytica Chimica Acta, 1127, 1-8.
- Jangi, S. R. H., & Akhond, M. (2020). Synthesis and characterization of a novel metal-organic framework called nanosized electroactive quasi-coral-340 (NEQC-340) and its application for constructing a reusable nanozyme-based sensor for selective and sensitive glutathione quantification. Microchemical Journal, 158, 105328.
- Hormozi Jangi, S. R., Akhond, M., & Absalan, G. (2020). A field-applicable colorimetric assay for notorious explosive triacetone triperoxide through nanozyme-catalyzed irreversible oxidation of 3, 3′-diaminobenzidine. Microchimica Acta, 187, 1-10.
- Khoshsafar, H., Karimian, N., Nguyen, T. A., Fakhri, H., & Khanmohammadi, A., et al. (2022). Enzymeless voltammetric sensor for simultaneous determination of parathion and paraoxon based on Nd-based metal-organic framework. Chemosphere, 292, 133440.
- Diao, Q., Chen, X., Tang, Z., Li, S., & Tian, Q., et al. (2024). Nanozymes: powerful catalytic materials for environmental pollutant detection and degradation. Environmental Science: Nano.
- Hormozi Jangi, S. R., & Dehghani, Z. (2023). Spectrophotometric quantification of hydrogen peroxide utilizing silver nanozyme. Chemical Research and Nanomaterials, 2(1), 15-23.
- Ahmadi-Leilakouhi, B., Hormozi Jangi, S. R., & Khorshidi,A. (2023). Introducing a novel photo-induced nanozymatic method for high throughput reusable biodegradation of organic dyes. Chemical Papers, 77(2), 1033-1046.
- Jangi, S. R. H., Davoudli, H. K., Delshad, Y., Jangi, M. R. H., & Jangi, A. R. H. (2020). A novel and reusable multinanozyme system for sensitive and selective quantification of hydrogen peroxide and highly efficient degradation of organic dye. Surfaces and Interfaces, 21, 100771.
- Hormozi Jangi, S. R. (2024). Lithium-electroactive peroxidase-like MnO2 nanomaterials as an ultrasensitive and selective sensing platform for carcinogenic 3, 3′-diaminobenzidine and high-capacity lithium-ion batteries cathode materials. Chemical Papers, 1-13.
- Jangi, S. R. H. (2023). Introducing a high throughput nanozymatic method for eco-friendly nanozyme-mediated degradation of methylene blue in real water media. Sustainable Chemical Engineering, 90-99.
- Jangi SR, H. (2023). Natural Polyphenols of Pomegranate and Black Tea Juices can Combat COVID-19 through their SARS-CoV-2 3C-like Protease-inhibitory Activity.
- Hormozi Jangi, S. R. (2023). A brief overview on clinical and epidemiological features, mechanism of action, and diagnosis of novel global pandemic infectious disease, Covid-19, and its comparison with Sars, Mers, And H1n1. World J Clin Med Img, 2(1), 45-52.
- Wang, J., Xie, Q., Song, H., Chen, X., & Zhang, X., et al. (2023). Utilizing nanozymes for combating COVID-19: advancements in diagnostics, treatments, and preventative measures. Journal of Nanobiotechnology, 21(1), 200.
- Hormozi Jangi, S. R. (2023). Naked-Eye Sensing of SARS-CoV-2 Utilizing Nanozymatic Nanoassays. J Pediatr Neonatal Biol, 8(4), 283-289.
- Ali, J., Elahi, S. N., Ali, A., Waseem, H., & Abid, R., et al. (2021). Unveiling the potential role of nanozymes in combating the COVID-19 outbreak. Nanomaterials, 11(5), 1328.
- Hormozi Jangi, S. R. (2023). A Brief Overview of Nanozyme-Based Colorimetric and Fluorometric Sensors for Early Diagnosis of COVID-19. Trans Med OA, 1(2), 76-84.
- Liang, C., Liu, B., Li, J., Lu, J., & Zhang, E., et al. (2021). A nanoenzyme linked immunochromatographic sensor for rapid and quantitative detection of SARS-CoV-2 nucleocapsid protein in human blood. Sensors and Actuators B: Chemical, 349, 130718.
- Kumawat, M., Umapathi, A., Lichtfouse, E., & Daima, H.K. (2021). Nanozymes to fight the COVID-19 and future pandemics. Environmental Chemistry Letters, 19(6), 3951-3957.
- Jangi, S. R. H., Akhond, M., & Dehghani, Z. (2020). High throughput covalent immobilization process for improvement of shelf-life, operational cycles, relative activity in organic media and enzymatic kinetics of urease and its application for urea removal from water samples. Process Biochemistry, 90, 102-112.
- Jangi, S. R. H., & Akhond, M. (2022). Introducing a covalent thiol-based protected immobilized acetylcholinesterase with enhanced enzymatic performances for biosynthesis of esters. Process Biochemistry, 120, 138-155.
- Jangi, S. R. H., & Akhond, M. (2021). High throughput urease immobilization onto a new metal-organic framework called nanosized electroactive quasi-coral-340 (NEQC-340) for water treatment and safe blood cleaning. Process Biochemistry, 105, 79-90.
- Hormozi Jangi, S. R. (2023). BSA-Stabilized Gold-Nanozymes Reveal 4-Order Higher Catalytic Efficiency and 2-Fold Higher Substrate Affinity than Mno2-Nanozymes. J App Mat Sci & Engg Res, 7(2), 166-171.
- Hormozi Jangi, S. R., & Dehghani, Z. (2023). Kinetics and biochemical characterization of silver nanozymes and investigating impact of storage conditions on their activity and shelf-life. Chemical Research and Nanomaterials, 1(4), 25-33.
- Jangi, S. R. H. (2023). A comparative study on kinetics performances of BSA-gold nanozymes for nanozyme-mediated oxidation of 3, 3’, 5, 5’-Tetramethylbenzidine and 3, 3’-Diaminobenzidine.
- Hormozi Jangi, S. R. (2023). Experimental Evaluation of Kinetic Characteristics of SiO2@ AuNPs Nanocom-posite and BSA-stabilized gold Nanoparticles toward Peroxidase-Mediated Reactions. Adv Nanoscie Nanotec, 7(1), 01-11.
- Jangi, S. R. H. (2023). Determining kinetics parameters of bovine serum albumin-protected gold nanozymes toward different substrates. Qeios.
- Johnson, K. A., & Goody, R. S. (2011). The original Michaelis constant: translation of the 1913 Michaelis–Menten paper. Biochemistry, 50(39), 8264-8269.
- Srinivasan, B. (2022). A guide to the Michaelis–Menten equation: steady state and beyond. The FEBS journal, 289(20), 6086-6098.
- Cornish-Bowden, A. (2015). One hundred years of Michaelis–Menten kinetics. Perspectives in Science, 4, 3-9.



