Review Article - (2021) Volume 6, Issue 2
Deprivation of Food Increasing Activation of Gene
Received Date: Nov 08, 2021 / Accepted Date: Nov 15, 2021 / Published Date: Nov 24, 2021
Copyright: ©P D Gupta. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: P. D. Gupta. (2021). Deprivation of Food Increasing Activation of Gene. Adv Nutr Food Sci, 6(2), 59-61.
Abstract
Through food we eat DNA every day. The raw material for new DNA comes from food only. The DNA replication is a complex process and many signalling molecules, many enzymes and replication controlling factors ultimately comes from food metabolism only. In this brief review we have described that during fasting metabolic process does not stop and adopted altered pathway while metabolising stored glycogen in liver and muscle. Consuming old stored glycogen from liver and muscles to feed glucose to the brain and synthesizing fresh glycogen benefits other body organs too.
Keywords
Ketogenic diet, Carbohydrate, Fat, Epilepsy
Introduction
All living creatures must show metabolic activity that is a set of chemical reactions which are essential for a living organism to maintain its life. Nutrition is the key to metabolism. Metabolic activity is closely linked to nutrition and the availability of nutri-ents [1]. Bioenergetics is a term that describes the biochemical or metabolic pathways by which the cell ultimately obtains energy. The pathways of metabolism rely upon nutrients that breakdown in order to produce energy. This energy in turn is required by the body to synthesize molecules like new proteins and nucleic acids (DNA, RNA). We get energy from food. Energy formation is one of the vital components of metabolism. However, In the course of evolution, individuals whose bodies functioned well in a fasted state were, enabling their survival well [2, 3].
Body Functions during Fasting
Short and Intermittent fasting brings many structural and function¬al changes in the GI tract. Now it has been shown that changes are beneficial in terms of postponing the onset of Non- communicable diseases (NCDs) prolonging the health age. That might be the rea¬son for Ancient Indian saints to do fasting [4]. These benefits have been described in a series of research papers by us and others [5]. With fasting mainly carbohydrate (liver and muscle, where gly¬cogen id stored and brain where lipids are main components) and lipids are most affected. Liver glycogen stores are depleted and ke¬tones are produced from adipose-cell-derived fatty acids. result in periodic depletion of liver glycogen stores and the associated pro¬duction of ketones from fatty acids. Intermittent metabolic switch¬ing (IMS) occurs rarely or not at all in individuals who eat three or more meals per day and who are fairly sedentary (Figure.1).
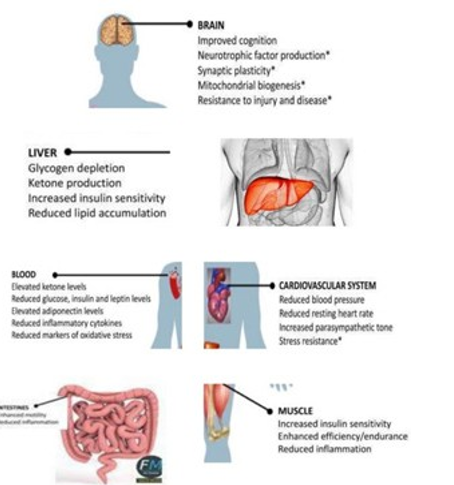
Figure 1: Effect of Fasting on various organs in the body
Liver: Carbohydrate Metabolism
In Liver during short-term fasting periods, the liver produces and releases glucose mainly through glycogenolysis. During pro-longed fasting, glycogen is depleted, and hepatocytes synthesize glucose through gluconeogenesis using lactate, pyruvate, glycerol, and amino acids [6]. Fasting caused parallel changes in plasma glucose and hepatic glycogen concentrations with decreases by 24 hr and subsequent increases, which correlated with increases in hepatic synthase l and decreases in phosphorylase activates. After about 3 days of starvation, the liver forms large amounts of aceto-acetate and d-3-hydroxybutyrate (ketone bodies). Their synthesis from acetyl CoA increases markedly because the citric acid cycle is unable to oxidize all the acetyl units generated by the degrada¬tion of fatty acids. Severe starvation can be associated with acute liver injury that is slowly reversible with careful enteral nutrition [7]. In this clinical situation, profound hepatic glycogen depletion in association with autophagy appears as the leading cause of liver injury.
Glycogenolysis increase during fasting : In that study, increased hepatic glycogen in fa/fa rats correlated with excess hepatic gly-cogenolysis after a 24-h fast Together, these observations suggest that in this animal model after a 24-h fast, hepatic glucose output may be higher because excess glycogen stores permit continued glycogenolysis During starvation, the brain must be supplied with fuel in the form of glucose or ketone bodies. In prolonged starva¬tion, gluconeogenesis provides the glucose oxidised by the brain. The major substrates for gluconeogenesis are amino acids derived from skeletal muscle protein breakdown. Proteolysis also provides carbon skeletons for gluconeogenesis. During starvation, degraded proteins are not replenished and serve as carbon sources for glu¬cose synthesis. Proteolysis of muscle protein provides some of three-carbon precursors of glucose [8].
Brain: Lipid Metabolism.
They found, for example, a very large which creates the protein compromising amyloid plaques often found in the brain of Alzhei-mer’s patients. Conversely, they found a very large upregulation of the CFHR1 gene, which may play a role in lipid metabolism ( i. e: helping you get rid of cholesterol). the long-term protective ben-efits of fasting, both reinforce the notion that intermittent fasting can indeed lead to potentially meaningful physiological changes that warrant further scientific exploration [9, 10].
Brain evolution sustaining high levels of performance in a food-de-prived (fasted) state Brain evolution, including higher cortical functions of humans (imagination, creativity and language), was driven by the necessity of sustaining high levels of performance in a food-deprived (fasted) state Intermittent metabolic switching (IMS) occurs when eating and exercise patterns The ketone β-hy-droxybutyrate (BHB) is transported into the brain and into neuro¬nal mitochondria, where it is used to generate acetyl CoA and ATP. BHB also acts as a signalling molecule in neurons that can induce the expression of brain-derived neurotrophic factor and thereby promote synaptic plasticity and cellular stress resistance IMS can enhance cognition and motor performance and protects neurons against dysfunction and degeneration in animal models of stroke, epilepsy, traumatic brain and spinal cord injury, Alzheimer disease and Parkinson disease [11]. During fasting and extended exercise, adaptive cellular stress-re-sponse signalling pathways are activated and autophagy is stimu¬lated, whereas overall protein synthesis is reduced Upon refeeding, rest and sleep, protein synthesis is upregulated and mitochondrial biogenesis occurs, enabling neurogenesis and synaptogenesis In¬termittent fasting can improve indicators of metabolic and cardio¬vascular health in humans by mechanisms involving reductions in oxidative damage and inflammation. However, it remains to be de¬termined whether and how intermittent fasting impacts the brains of healthy humans and those affected with a neurological disorder [12, 14].
Fasting control gene activity without changing the DNA It has been well established that there is a continuous cross talk between genes and diet. Many signalling pathways cause a cellular response that involves a change in gene expression. Nutrients can induce gene expression thereby altering individual phenotype. The accessibility of large regions of DNA can depend on its chromatin structure, which can be altered as a result of histone modifications directed by DNA methylation, ncRNA, or DNA-binding protein. Hence these modifications may up or down regulate the expres¬sion of a gene. Regulations of gene expression is critical for life, this complex process is not yet fully understood, Gene regulation can occur at any point during gene expression, but most common¬ly occurs at the level of transcription (when the information in a gene’s DNA is passed to mRNA). Signals from the environment including the diet or from other cells activate proteins called tran¬scription factors. These proteins bind to regulatory regions of a gene and increase or decrease the level of transcription. By con¬trolling the level of transcription, this process can determine when and how much protein product is made by a gene [15, 16].
Gene expression
Gene expression is the process in which information from a gene is used by the cell to produce a functional product, typically a pro¬tein. Gene expression is a tightly regulated process that allows a cell to respond to its changing environment. It acts as both an on/ off switch to control when proteins are made and also a volume control that increases or decreases the amount of proteins made. It involves two major steps, transcription and translation. Nutrients can induce gene expression thereby altering individual phenotype. Conversely single nucleotide polymorphisms, in a range of genes important in inflammation and lipid metabolism, alter the bioactiv-ity of important metabolic pathways and mediators and influence the ability of nutrients to interact with them. Gene expression is influenced by numerous factors, including molecules within the cell, mutations causing dominant negative effects and haploinsuf-ficiency, signalling molecules from surrounding cells and the envi¬ronment, and epistasis.
A 2015 study out of the Rejuvenation Research journal found the SIRT3 gene to be upregulated in those who engaged in intermittent fasting [17]. The SIRT3 gene codes for a mitochondrial protein that has been associated with potentially protective “anti-aging” effects the science suggests that, if you can do it, a prolonged fast for 2-3 days will induce your body to clean out some old immune cells and switch on production of new ones [18]. Dietary patterns may impact gene expression through several mechanisms, for ex¬ample certain dietary compounds bind to transcription factors and regulate their activity such as polyunsaturated fatty acids (PUFA) with peroxisome proliferator-activated receptors [19].
References
- Speakman, John R., and Colin Selman. “Physical activity and resting metabolic rate.” Proceedings of the Nutrition Society62.3 (2003): 621-634.
- Solebo, O. R. (2021). The Biochemical Functionality and Cellular Importance of the Vacuolar H+ Translocating Pyro-phosphatase 1, PfVP1, in Plasmodium Falciparum (Doctoral dissertation, Drexel University).
- Mattson, M. P. (2019). An evolutionary perspective on why food overconsumption impairs cognition. Trends in cognitive sciences, 23(3), 200-212.
- Gupta P. D.. Metabolic Depression: A Way to Defying AgeingJ. Cell Tissue Res 21(2): PV 2 (2021).
- Gupta P D. Fasting Gastrointestinal Tract Changes in Structure, Functions and Microbiota Milieu J. Gastroenterology Pancreatology and Hepatobiliary Disorders. 5(3)https://www. researchgate.net/publication/352809761.
- Berg, J. M., Tymoczko, J. L., & Stryer, L. (2002). Biochemistry.
- Sanja KolaÄek and Enteral nutrition World Rev Nutr Diet. 2013;108:86-90.
- Blanco Antonio, Blanco Gustavo. Carbohydrate Metabolism, In Medical Biochemistry Chapter 14 Editor(s): Antonio Blanco, Gustavo Blanco, Academic Press, 2017, Pages 283-323,
- Boules, M., Williams, K., Gollatz, E., Fauq, A., & Richelson,E. (2004). Down-regulation of amyloid precursor protein by peptide nucleic acid in vivo. Journal of Molecular Neuroscience, 24(1), 123-128.
- König, B., Rauer, C., Rosenbaum, S., Brandsch, C., Eder, K., & Stangl, G. I. (2009). Fasting upregulates PPARα target genes in brain and influences pituitary hormone expression in a PPARα dependent manner. PPAR research, 2009.
- Mattson, M. P., Moehl, K., Ghena, N., Schmaedick, M., & Cheng, A. (2018). Intermittent metabolic switching, neuro-plasticity and brain health. Nature Reviews Neuroscience,19(2), 81-94.
- Antunes, F., Erustes, A. G., Costa, A. J., Nascimento, A. C., Bincoletto, C., Ureshino, R. P., ... & Smaili, S. S. (2018). Autophagy and intermittent fasting: the connection for cancer therapy?. Clinics, 73.
- Manzanero, S., Erion, J. R., Santro, T., Steyn, F. J., Chen, C., Arumugam, T. V., & Stranahan, A. M. (2014). Intermittent fasting attenuates increases in neurogenesis after ischemia and reperfusion and improves recovery. Journal of Cerebral Blood Flow & Metabolism, 34(5), 897-905.
- Malinowski, B., Zalewska, K., Wesierska, A., SokoÅ?owska,M. M., Socha, M., & Liczner, G. (2019). Pawlak-Osi nska,K. Wicinski, M. Intermittent fasting in cardiovascular disor-ders—An overview. Nutrients, 11, 673.
- Stover, P. J., & Caudill, M. A. (2008). Genetic and epigen-etic contributions to human nutrition and health: managing genome–diet interactions. Journal of the American Dietetic Association, 108(9), 1480-1487.
- Riethoven, J. J. M. (2010). Regulatory regions in DNA: promoters, enhancers, silencers, and insulators. Computational biology of transcription factor binding, 33-42.
- Liu, Y., Cheng, A., Li, Y. J., Yang, Y., Kishimoto, Y., Zhang, S., ... & Mattson, M. P. (2019). SIRT3 mediates hippocampal synaptic adaptations to intermittent fasting and ameliorates deficits in APP mutant mice. Nature communications, 10(1), 1-11.
- Zhang, J., Xiang, H., Liu, J., Chen, Y., He, R. R., & Liu, B. (2020). Mitochondrial Sirtuin 3: New emerging biological function and therapeutic target. Theranostics, 10(18), 8315.
- Tai, E. S., Corella, D., Demissie, S., Cupples, L. A., Coltell, O., Schaefer, E. J., ... & Ordovas, J. M. (2005). Polyunsaturated fatty acids interact with the PPARA-L162V polymorphism to affect plasma triglyceride and apolipoprotein C-III concentrations in the Framingham Heart Study. The Journal of nutrition, 135(3), 397-403.



