Research Article - (2026) Volume 9, Issue 1
Controlling Colletotrichum sp. Causing Anthracnose on Yellow Horn Pepper Using Chlorine and Oligochitosan: Efficacy under Net House and Storage Conditions
2Faculty of Plant Protection, College of Agriculture, Can Tho University, Vietnam
Received Date: Jan 14, 2026 / Accepted Date: Feb 11, 2026 / Published Date: Feb 24, 2026
Copyright: ©2026 To Ngoc Son, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Xuan, L. T. N., Son, T. N., Nhan, N. T., Xuan, T. T. (2026). Controlling Colletotrichum sp. Causing Anthracnose on Yellow Horn Pepper Using Chlorine and Oligochitosan: Efficacy under Net House and Storage Conditions. J Agri Horti Res, 9(1). 01-06.
Abstract
The potential of chlorine and oligochitosan in controlling the fungus Colletotrichum sp. causing anthracnose disease on yellow horn pepper under net house and post-harvest storage conditions. Research objective (1) is to evaluate the efficacy of Chlorine 0.2% and Oligochitosan 3% in controlling anthracnose disease on yellow horn pepper fruit under net house conditions. Results showed that applying both Chlorine 0.2% and Oligochitosan 3% twice (before and after inoculation) was effective in controlling anthracnose disease on yellow horn pepper fruit. Specifically, Chlorine 0.2% achieved a disease reduction efficacy of 31.2% at 11 Daises After Inoculation (DAI). (2) the application time of chlorine 0.2% and oligochitosan 3% for inhibition against Colletotrichum sp. The Col-8-ÄT strain causing anthracnose disease on horn pepper fruit under laboratory conditions (post-harvest storage), both chlorine 0.2% and oligochitosan 3% were capable of controlling post-harvest anthracnose disease on pepper fruit; soaking pepper fruit in chlorine 0.2% for 5 minutes showed an inhibition efficiency of up to 93% at 6 DAI
Keywords
Pepper Anthracnose Disease, Colletotrichum sp., Chlorine, Oligochitosan
Introduction
Chili pepper (Capsicum spp.) is a popular and long-cultivated spice vegetable in Vietnam. According to Vo Van Chi (2008), chili pepper is an ingredient in traditional medicine that can treat certain ailments such as indigestion, dysentery, rheumatism, and stimulate the stomach . However, pepper cultivation faces difficulties and reduced yields due to diseases such as: fungal anthracnose, viral mosaic disease, bacterial wilt, fungal damping-off… Among these, anthracnose caused by the fungus Colletotrichum sp. is a disease that seriously harms pepper yield globally [1]. Furthermore, this disease not only causes damage in the field but also affects the fruit after harvest [2].
Some studies apply environmentally friendly, rapidly degrading chemicals or biological pesticides in controlling crop diseases. For example: Controlling post-harvest diseases on NÄ?m Roi pomelo, Sành orange, and ÄÆ°á»ng mandarin using Chlorine , applying chitosan to inhibit Colletotrichum gloeosporioides causing anthracnose disease on Hoà Lá»?c mango [3,4]. Chili pepper is a fresh spice vegetable, and the safety of post-harvest pesticides is crucial. Chlorine and oligochitosan are substances with very low toxicity, used in this study to investigate their potential to limit anthracnose disease on yellow horn pepper under net house conditions and post-harvest storage.
Research Results and Discussion
Efficacy of Chlorine and Oligochitosan in Controlling Anthracnose Disease on Yellow Horn Pepper Fruit Under Net House Conditions
Table 3 results show that the treatments chlorine 0.2%-PT, chlorine 0.2%-PS, chlorine 0.2%-PTS, oligochitosan 3%-PTS, Ridomil Gold 68WG-PS, Ridomil Gold 68WG-PTS all showed a gradual decrease in disease reduction efficacy over the surveyed time points (5, 7, 9, 11 DAI). The remaining treatments, oligochitosan 3%-PT, oligochitosan 3%-PS, Ridomil Gold 68WG-PT, showed an increase in disease reduction efficacy up to 7 DAI, but then the efficacy gradually decreased.
|
No |
Treatment |
Disease reduction efficacy (%) over time points |
|||||||
|
|
|
5NSLB |
7 NSLB |
9 NSLB |
11 NSLB |
||||
|
01 |
Chlorine 0,2%-PT |
29,18 |
cde |
20,69 |
c |
14,81 |
b |
16,43 |
bc |
|
02 |
Chlorine 0,2%-PS |
28,42 |
cde |
22,44 |
c |
16,69 |
b |
10,75 |
c |
|
03 |
Chlorine 0,2%-PTS |
52,74 |
a |
46,94 |
a |
33,04 |
a |
31,20 |
a |
|
04 |
Oligochitosan 3%-PT |
34,45 |
bc |
36,83 |
b |
19,68 |
b |
12,25 |
c |
|
05 |
Oligochitosan 3%-PS |
17,53 |
e |
31,97 |
b |
18,26 |
b |
17,16 |
bc |
|
06 |
Oligochitosan 3%-PTS |
42,90 |
ab |
35,68 |
b |
21,69 |
b |
22,55 |
b |
|
07 |
Ridomil Gold 68WG-PT |
19,28 |
de |
21,95 |
c |
12,19 |
b |
10,30 |
c |
|
08 |
Ridomil Gold 68WG-PS |
31,01 |
cd |
20,73 |
c |
18,59 |
b |
15,02 |
bc |
|
09 |
Ridomil Gold 68WG-PTS |
43,72 |
ab |
33,06 |
b |
20,67 |
b |
22,17 |
b |
|
Significance level |
* |
* |
* |
* |
|||||
|
CV (%) |
14,19 |
8,53 |
25,03 |
15,99 |
|||||
|
Note: In the same column, numbers followed by the same letter do not differ significantly according to Duncan's test. *: difference at the 5% significance level. DAI: DAIs After Inoculation. |
|||||||||
Table 3: Disease Reduction Efficacy (%) of Chlorine and Oligochitosan on Yellow Horn Pepper Fruit Over Dais After Inoculation
The disease reduction efficacy of the Chlorine 0.2% treatment when sprayed twice (before and after inoculation) was significantly higher than the Ridomil Gold 68WG treatment and other treatments at the recorded time points. At 5 DAI, the disease reduction efficacy in the chlorine 0.2% treatment sprayed twice, before and after, was over 50% (52.74%); subsequently, the efficacy gradually decreased until 11 DAI, where chlorine 0.2%-PTS maintained an efficacy of 31.20%. According to the study by Buck (2006), Chlorine is a strong oxidant, capable of penetrating cells and disrupting the metabolic processes of microorganisms, such as protein synthesis, DNA [3]. Additionally, chlorine can combine with amine molecules in microbial cells to form new substances, which destroy the cell's vital functions, leading to the death of the microorganism [5]. When testing chlorine 0.2% on horn pepper plants under net house conditions, it showed efficacy in inhibiting the anthracnose pathogen caused by the fungal strain Col-8-DT on the fruit (Figure 1). In particular, chlorine applied twice, before and after spraying (chlorine 0.2%-PTS), yielded the highest disease reduction efficacy, reaching 31.20% at 11 DAI.
Figure 1: Disease reduction efficacy of chlorine, oligochitosan, and Ridomil Gold 68WG against the fungus Col-8-DT at 11 DAY under net house conditions
A: NT chlorine 0,2%-PT B: NT chlorine 0,2%-PS
C: NT chlorine 0,2%-PTS D: NT oligochitosan 3%-PT
E: NT oligochitosan 3%-PS F: NT oligochitosan 3%-PTS
G: NT Ridomil Gold 68WG-PT H: NT Ridomil Gold 68WG-PS
I: NT Ridomil Gold 68WG-PTS J: NT Distilled water control treatment
Determining the Application Time of Chlorine and Oligochitosan for Inhibitory Efficacy Against the Fungus Col- 8-Dt Under Laboratory Conditions
In Table 4, at 4 DAI, the treatments chlorine 0.2%-5p, chlorine 0.2%-7p, and oligochitosan 3%-3p showed the highest disease inhibition efficacy, reaching 100%, and oligochitosan 3%-7p also showed a comparable inhibition efficacy (93.75%), with no significant statistical difference at the 5% level.
|
No. |
Treatment |
Inhibition efficacy (%) over time points |
|||||
|
|
|
4 NSLB |
6 NSLB |
8 NSLB |
|||
|
01 |
Chlorine 0,2%-3p |
85,42 |
b |
69,75 |
d |
0,00 |
c |
|
02 |
Chlorine 0,2%-5p |
100,00 |
a |
93,00 |
a |
15,56 |
a |
|
03 |
Chlorine 0,2%-7p |
100,00 |
a |
86,00 |
b |
2,22 |
b |
|
04 |
Oligochitosan 3%-3p |
100,00 |
a |
74,50 |
cd |
0,00 |
c |
|
05 |
Oligochitosan 3%-5p |
85,42 |
b |
79,00 |
c |
2,22 |
b |
|
06 |
Oligochitosan 3%-7p |
93,75 |
ab |
76,49 |
c |
0,00 |
c |
|
Significance level |
* |
* |
* |
||||
|
CV (%) |
3,67 |
3,49 |
15,75 |
||||
|
Note: In the same column, numbers followed by the same letter do not differ significantly according to Duncan's test. *: difference at the 5% significance level. DAI: DAIs After Inoculation. |
|||||||
Table 4: Inhibition Efficacy (%) of Chlorine and Oligochitosan Against Anthracnose Disease Caused by the Fungus Col-8-DT at 4, 6, and 8 DAI
At 6 and 8 DAI, the treatment chlorine 0.2%-5p showed the highest disease inhibition efficacy and was statistically different (P < 0.05$) from all other treatments, with inhibition efficacy of 93% (6 DAI) and 15.56% (8 DAI). The chlorine 0.2%-7p treatment showed higher disease inhibition efficacy than the chlorine 0.2%-3p treatment (Figure 2). Overall, the inhibition efficacy of treatments with chlorine and oligochitosan gradually decreased over the time points 4, 6, and 8 DAI.
Materials and Research Methods
• Fungal Source: Colletotrichum sp. (Col-8-DT) causing anthracnose disease on pepper (provided by the Faculty of Plant Protection).
• Pepper Variety: Yellow horn pepper from Trung Nong Plant Seed Co., Ltd.
• Pesticide: Ridomil Gold 68WG (a fungicide selected as the positive control) is effective in controlling the fungus Colletotrichum sp.
• PDA medium (Shurleff and Averre III, 1997):
Potato 200 g
Dextrose 20 g
Agar 20 g
Distilled water 1000 ml
pH 6.8 - 7.0
Experiment 1: Evaluating the efficacy of chlorine and oligochitosan in controlling anthracnose disease on yellow horn pepper fruit under net house conditions
• Experimental Objective: Investigate the ability of chlorine and oligochitosan to control anthracnose disease on yellow horn pepper.
• Experimental Method: The experiment was arranged in a completely randomized design, with 4 replications, including 10 treatments (Table 1); each replication was 1 pepper plant with uniform fruits (first flush fruit) artificially inoculated on 4 fruits/plant.
|
No. |
Treatment |
Time of active ingredient application |
|
1 |
Chlorine 0.2%-PT |
Spraying chlorine 0.2%; 2 DAIs before Artificial Inoculation (AI) (PT) |
|
2 |
Chlorine 0.2%-PS |
Spraying chlorine 0.2%; 2 DAIs after AI (PS) |
|
3 |
Chlorine 0.2%-PTS |
Spraying chlorine 0.2%; 2 DAIs before and 2 DAIs after AI (PTS) |
|
4 |
Oligochitosan 3%-PT |
Spraying oligochitosan 3%; 2 DAIs before AI (PT) |
|
5 |
Oligochitosan 3%-PS |
Spraying oligochitosan 3%; 2 DAIs after AI (PS) |
|
6 |
Oligochitosan 3%-PTS |
Spraying oligochitosan 3%; 2 DAIs before and 2 DAIs after AI (PTS) |
|
7 |
Ridomil Gold 68WG-PT |
Spraying Ridomil Gold 68WG; 2 DAIs before AI (PT) |
|
8 |
Ridomil Gold 68WG-PS |
Spraying Ridomil Gold 68WG; 2 DAIs after AI (PS) |
|
9 |
Ridomil Gold 68WG-PTS |
Spraying Ridomil Gold 68WG; 2 DAIs before and 2 DAIs after AI (PTS) |
|
10 |
Control (spraying distilled water) |
Spraying distilled water |
Table 1: Treatments Arranged in the Net House
LBNT: Artificial Inoculation
• Experimental Procedure:
![]() Yellow horn pepper plants were grown in pots until they produced uniform fruits, then artificial inoculation was performed.
Yellow horn pepper plants were grown in pots until they produced uniform fruits, then artificial inoculation was performed.
![]() Fungal Source Preparation: The fungus Col-8-DT was cultured on PDA medium in Petri dishes.
Fungal Source Preparation: The fungus Col-8-DT was cultured on PDA medium in Petri dishes.
![]() Artificial Inoculation: Each pepper fruit was injured in the middle with a sterile needle bundle (5 needles/bundle), and then a 5 mm diameter paper disc, soaked with a spore suspension of the fungus Col-8-DT at a concentration of 10^7 spores/ml, was placed on the wound.
Artificial Inoculation: Each pepper fruit was injured in the middle with a sterile needle bundle (5 needles/bundle), and then a 5 mm diameter paper disc, soaked with a spore suspension of the fungus Col-8-DT at a concentration of 10^7 spores/ml, was placed on the wound.
![]() Monitoring Indicators: Record the length and width of the lesion at 5, 7, 9, 11 DAIs after Inoculation (DAI). The efficacy of the substances (HQT) was calculated according to the formula of Abbott (1925):
Monitoring Indicators: Record the length and width of the lesion at 5, 7, 9, 11 DAIs after Inoculation (DAI). The efficacy of the substances (HQT) was calculated according to the formula of Abbott (1925):
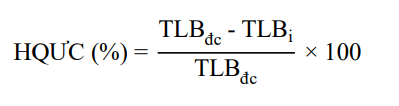
In there: - C: average lesion length in the control treatment.
• T: average lesion length in the treated treatment.
Experiment 2: Determining the application time of chlorine and oligochitosan for inhibitory efficacy against the fungus Col- 8-DT causing anthracnose disease on horn pepper fruit under laboratory conditions.
Experimental objective: Investigate the application time of chlorine and oligochitosan on post-harvest horn pepper fruit to limit anthracnose inoculum [9].
• Experimental Method:
![]() The experiment was arranged in a completely randomized design with 7 treatments (Table 2): 6 treatments were soaked in chlorine 0.2% and oligochitosan 3% at time points 3, 5, 7 minutes, and 1 control treatment. Each treatment had 4 replications, and each replication consisted of 15 pepper fruits.
The experiment was arranged in a completely randomized design with 7 treatments (Table 2): 6 treatments were soaked in chlorine 0.2% and oligochitosan 3% at time points 3, 5, 7 minutes, and 1 control treatment. Each treatment had 4 replications, and each replication consisted of 15 pepper fruits.
|
No. |
Treatment |
|
1 |
Chlorine 0.2%-3p |
|
2 |
Chlorine 0.2%-5p |
|
3 |
Chlorine 0.2%-7p |
|
4 |
Oligochitosan 3%-3p |
|
5 |
Oligochitosan 3%-5p |
|
6 |
Oligochitosan 3%-7p |
|
7 |
Distilled water control |
Table 2: Treatments Arranged in the Laboratory
![]() Fungal Source Preparation: the fungus was cultured on PDA medium.
Fungal Source Preparation: the fungus was cultured on PDA medium.
![]() Procedure for Treating Pepper Fruit with Chlorine and Oligochitosan: Select pepper fruits that have reached physiological ripeness, are uniform in size and color, wash thoroughly with water, allow to air-dry naturally, and wipe the fruit with 70% alcohol. Then, place the pepper fruit in a container with 500 ml of chlorine 0.2% or oligochitosan 3% solution, and soak the fruits completely for 3, 5, or 7 minutes.
Procedure for Treating Pepper Fruit with Chlorine and Oligochitosan: Select pepper fruits that have reached physiological ripeness, are uniform in size and color, wash thoroughly with water, allow to air-dry naturally, and wipe the fruit with 70% alcohol. Then, place the pepper fruit in a container with 500 ml of chlorine 0.2% or oligochitosan 3% solution, and soak the fruits completely for 3, 5, or 7 minutes.
![]() Inoculation Method: Remove the fruits and place them on a plate, then spray 3 ml of fungal spore suspension at a concentration of 10^7 spores/ml. Each plate of pepper, after spraying the fungal spores, was placed in a nylon bag and incubated at a temperature of 25^\circC for 48 hours, then the treatments were moved to laboratory conditions (28 - 30^\ circC).
Inoculation Method: Remove the fruits and place them on a plate, then spray 3 ml of fungal spore suspension at a concentration of 10^7 spores/ml. Each plate of pepper, after spraying the fungal spores, was placed in a nylon bag and incubated at a temperature of 25^\circC for 48 hours, then the treatments were moved to laboratory conditions (28 - 30^\ circC).
![]() Monitoring Indicators: Record the percentage of fruits showing disease symptoms (%), once every 2 DAIs, until the control treatment reaches 100% diseased fruits.
Monitoring Indicators: Record the percentage of fruits showing disease symptoms (%), once every 2 DAIs, until the control treatment reaches 100% diseased fruits.
![]() Disease inhibition efficacy was calculated according to the formula of Yu et al. (2012):
Disease inhibition efficacy was calculated according to the formula of Yu et al. (2012):
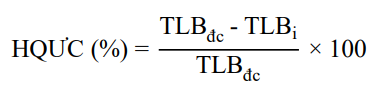
In there: HQUC: Disease inhibition efficacy of the applied chemicals.
TLB_{dc}: Disease rate (number of diseased fruits on the total number of fruits in the control treatment) in the control treatment.
TLB_{i}: Disease rate (number of diseased fruits on the total number of fruits in the treated treatment) in the treated treatment.
Conclusion and Recommendations
Under net house conditions, chlorine 0.2% and oligochitosan 3% both effectively control anthracnose disease on pepper, and the efficacy is higher when sprayed twice—once before inoculation and once after inoculation. The disease reduction efficacy of the Chlorine 0.2%-PTS treatment (31.2%) was higher than that of Oligochitosan 3%-PTS (22.55%) and Ridomil Gold 68WG-PTS (22.17%) at 11 DAI. Under laboratory conditions, chlorine 0.2% and oligochitosan 3% both showed high efficacy in controlling anthracnose disease on pepper fruit after harvest. The disease reduction efficacy when soaking the pepper fruit for 5 minutes was higher than for 3 minutes and 7 minutes. For chlorine 0.2%, the efficacies were 93%, 69.75%, and 86%, respectively; for oligochitosan 3%, the efficacies were 79%, 74.5%, and 76.49%, respectively, at 6 DAI. Further studies on the potential of chlorine and oligochitosan for controlling anthracnose disease on pepper under field conditions are needed. The author wishes to express deep gratitude for the significant assistance from the AI assistant Gemini, including but not limited to the aspects of: suggesting ideas for student survey questions, checking for spelling errors and scientific terminology, ensuring accurate academic translation into English, and consulting on the design of data collection tables.
References
- Kim, S. H., Yoon, J. B., Do, J. W., & Park, H. G. (2007).Resistance to anthracnose caused by Colletotrichum acutatum in chili pepper (Capsicum annuum L.). Journal of Crop Science and Biotechnology, 10(4), 277-280.
- Hadden, J. F., & Black, L. L. (1989). Anthracnose of pepper caused by Colletotrichum spp (No. AVRDC Staff Publication). AVRDC.
- TrU¡ng Thi Hong Tham. (2006). Eficacy of Chlorine and essential oils in managing post-harvest diseases on Nam Roi pomelo, Sành orange,Duong mandarin fruit (Master's thesis). Can Tho University.
- Lê Nguyen Doan Duy, K. TuyEn, L. T. Lan, & N. C. Hà. (2014). Research on the application of Chitosan to inhibit the fungus Colletotrichum gloeosporioides isolated from anthracnose-infected Hoà LOc mango. Can Tho University Journal of Science, 154–161.
- Dào VAn HoAng. (2005). Pesticides and Principles of Pesticide Use. Agriculture Publishing House.
- HuYnh ThI Thúy Leu. (2010). Effects of Chlorine, Naphthyl acetic acid, and Storage Materials on the Quality of Post- harvest Sweet Potato (Ipomoea batatas) (Master's thesis). Can Tho University.
- Badawy, M. E., & Rabea, E. L. (2009). Potential of the biopolymer Chitosan with different molecular weights to control postharvest gray mold of tomato fruit. Postharvest Biology and Technology, 51(1), 110–117.
- Meng, X. H., Qin, G. Z., & Tian, S. P. (2010). Influences of preharvest spraying Cryptococcus laurentii combined with postharvest Chitosan coating on postharvest diseases and quality of table grapes in storage. LWT - Food Science and Technology, 43(3), 596–601.
- Abbott, U. S. (1925). A method of computing the effectiveness of an insecticide. Journal of Economic Entomology, 18(3), 265–267
- Lê Thanh Long. (2006). Research on using Chitosan solution and additives to extend the shelf life of fresh chicken eggs (HYLINE) (Master's thesis). Nha Trang University.
- Vo Van Chi. (2008). Vegetables, Beans used for Food and Medicine. Science and Technical Publishing House.
- Wilson, S. (2002). Institute of Post-harvest Technology. Colombo.


