Research Article - (2025) Volume 2, Issue 1
Clinical and Laboratory Rationale for Lymphotropic Defededematic Therapy in Comprehensive Treatment of Patients with Craniobrain Injury and Hemorrhagic Stroke
Received Date: Jul 17, 2025 / Accepted Date: Aug 19, 2025 / Published Date: Aug 25, 2025
Copyright: ©©2025 Eshonov Olim Shoyimkulovich, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Shoyimkulovich, E. O. (2025). Clinical and Laboratory Rationale for Lymphotropic Defededematic Therapy in Comprehensive Treatment of Patients with Craniobrain Injury and Hemorrhagic Stroke. J Path Lab Med, 2(1), 01-09.
Abstract
Introductions: The effectiveness of lymphotropic decongestant therapy in patients with traumatic brain injury and hemorrhagic stroke was studied. Based on the obtained clinical and laboratory data, the effectiveness of lymphotropic decongestant therapy was revealed, which prevents the progression of cerebral edema in this category of patients.
Purpose of the Study: evaluate the effectiveness of lymphotropic decongestant therapy for cerebral edema in patients with severe TBI and hemorrhagic stroke.
Materials and Methods: The scientific study included data from patients who were in the neuroreanimation department of the Bukhara branch of the Russian Research Center for Emergency Medicine. The materials of 75 patients were studied, of which 38 with TBI and 37 patients with HI. The age of the patients ranged from 37 to 68 years (mean age was 58.2±2 years). By gender - there were 43 men (57.3%), women - 32 (42.7%). All patients underwent standard diagnostic methods (assessment of neurological status during a joint examination by a neurologist and neurosurgeon, Multislice-Computed Tomography (MSCT), as well as laboratory tests (leukoformula, neutrophil to lymphocyte ratio index (NLRI)).
Keywords
Cerebral Edema, Traumatic Brain Injury, Hemorrhagic Stroke, Lymphotropic TherapyIntroduction
Relevance
Throughout the history of medicine, the primary goal of protecting the brain after injury has been to minimize neuronal damage, with swelling considered a minor concern. However, recent renewed interest in edema, its molecular precursors, and its significance in Traumatic Brain Injury (TBI) and stroke has changed this paradigm. Current advances in understanding the molecular mechanisms of edema formation suggest that the introduction of new treatments for cerebral edema (CE) may be closer than the introduction of treatments for neuronal death. Treatments for Intracranial Hypertension (ICH), such as decompressive craniectomy and osmotherapy, were developed before any knowledge of the modern pathophysiology of ICH. CE and ICH, as secondary factors of damage, cause death after stroke and TBI. The survival of patients with these diseases largely depends on the effectiveness of treatment for CE. A significant portion of medical and financial resources after traumatic brain injury are directed toward identifying and treating CE, ICH, and associated complications [1-7]. Given the advances and capabilities of neurocritical care, treatment remains exclusively reactive and is initiated only after problems with intracranial pressure arise. Recent reviews have focused on the molecular factors causing brain swelling after injury and the development of therapeutic approaches to combat this problem [8]. According to the Monroe-Kelly doctrine, intracranial contents are in constant dynamic equilibrium as cerebrospinal fluid is constantly produced and absorbed into brain tissue [9,10]. There are two hypotheses about fluid outflow: paravascular and perivascular. According to the first theory, cerebrospinal fluid and intercellular fluid flow from the brain through the middle layers of vein cells, forming the paravascular space of the veins. The second theory states that the outflow occurs in the spaces separating the cerebral arteries and the brain itself, forming the perivascular arterial space. Interestingly, both theories have been experimentally confirmed, so many scientists believe that both theories have a place to be. It is possible that fluid outflow occurs differently in different parts of the brain or at different points in time [11-13].
It has been experimentally confirmed that the tracer, introduced into the ventricles of the brain, where the cerebrospinal fluid is located, is found in the system of lymphatic vessels at the base of the skull, and then in the unchanged lymph nodes. This indicates the existence of a connection between the glymphatic, meningeal and lymphatic systems [16]. The discovery of meningeal lymphatic vessels is a necessary breakthrough in the development of new methods of neuroresuscitation based on the control of lymphatic drainage processes to remove toxins and unnecessary molecules from the central nervous system. This could be an innovative step in developing new ways to deliver drugs to the brain bypassing the blood-brain barrier [17-19]. The available scientific literature contains works that continue to expand our knowledge of the functioning of the lymphatic system of the brain and may have important practical applications in the field of neurology and neurosurgery [21-22]. Indeed, the research finding that brain tumors can metastasize to deep cervical lymph nodes suggests the existence of lymphatic drainage pathways for the central nervous system in humans [23-29]. Reconsideration of traditional concepts has already led to new ideas about brain functioning and may enable the development of new approaches to selected neurodegenerative diseases [30-35]. It is interesting to note that further study of the lymphatic system as a “window” to the central nervous system may lead to innovative methods of delivering drugs to the brain bypassing the blood-brain barrier. Using the lymphatic system may open up new possibilities for drug delivery by bypassing this barrier and reaching the desired brain regions [36,37]. In the studies of Yu.I. Borodin, it was proven that both under physiological conditions and under pathology in the brain, there is a discharge of cerebrospinal fluid into the lymphatic bed of the body [38-40]. Recent studies have also confirmed the circulation of fluid from the meningeal lymphatic system of the brain with macromolecules and immune cells from the central nervous system in the deep cervical lymph nodes. Labeled particles and macromolecules injected into the brain and/or cerebrospinal fluid mainly drain into the cervical lymph nodes [41-48]. Further research is aimed at better understanding the pathophysiology of the brain in order to develop more effective treatments and prevent the pathological process. Discoveries of key molecular pathways involved in the formation of AGM promise the development of effective treatment strategies to further reduce AGM [49,50]. However, none of the available methods for correcting ICH (decompressive craniectomy and osmotherapy) stop the main molecular cascade leading to cerebral edema. Research into the molecular principles of cerebral edema is identifying new targets for treatment, such as exposure factors, vascular permeability, and fluid-electrolyte balance. This opens up opportunities for the development of new drugs and therapeutic approaches that can more effectively target the molecular pathways that cause cerebral edema [51-54]. The current approach to drug treatment of cerebral edema largely revolves around the increasingly artificial distinction between cytotoxic and vasogenic cerebral edema. These off-target interventions, such as hyperosmolar drugs and sedation, have been the mainstay of clinical practice and offer unimpressive solutions to a serious problem. Our current understanding of the underlying molecular mechanisms causing cerebral edema is increasing, with differences between diseases and populations emerging. As our understanding of the underlying molecular mechanisms of neuronal injury continues to expand, so does the list of targeted therapies in development [55,56].
Removal of Fluids from the Central Nervous System
The mechanism of fluid removal from the central nervous system remains not fully understood. Fluid clearance is important to protect against the development of cerebral edema and promote its resolution. Excess water can leave the CNS either by volumetric flow down a perivascular pressure gradient or through the astral-glial or glymphatic system. One important way to remove excess cerebrospinal fluid from the brain is through the fluid pathways. along the basement membrane in the walls of the capillaries to the cervical lymph nodes. GS refers to a dynamic lymphatic network of Aquaporine-4 (APQ4)-expressing astrocytes involved in fluid clearance [57-63]. Glymphatic flow exhibits diurnal fluctuations, with the highest flow occurring during sleep. APQ4 knockout studies have demonstrated differences in the involvement of APQ4 in the formation and clearance of edema between the brain and spinal cord [64-70]. It is important to note that cerebral edema manifests differently in different disease states. For example, edema following cerebral ischemia involves early cellular/cytotoxic edema followed by delayed BBB opening. This differs from the manifestations in TBI, where mechanical disruption of the BBB occurs almost immediately and is subsequently accompanied by cellular, vasogenic and inflammatory factors that promote edema. Moreover, the swelling associated with acute brain injury differs significantly from the swelling that occurs in chronic diseases.
Glymphatic System
First discovered less than a decade ago, HS provides a pathway for the influx of cerebrospinal fluid into the brain (via the periarterial space) and the clearance of interstitial fluid from the brain (perivenous spaces), with a key role being played by aquaporin-4 (AQP4) channels at the vascular ends of astrocytes. Although this system has been widely studied in the context of neurodegenerative diseases (amyloid-b and tau clearance) and sleep pathways, recent efforts have also implicated it in the development of AMS after ischemic stroke. Subsequent studies of subarachnoid hemorrhage (SAH) and TBI also identified GS disruption as a contributing factor to cerebral edema, secondary injury, and BBB dysfunction [71-74]. The discovery of the lymphatic drainage system, which maintains brain homeostasis and is involved in immune surveillance, represents a new therapeutic target for the treatment of neurological dysfunctions. This not only changed the understanding of fluid circulation in the brain, but also contributed to the understanding of the pathology of the central nervous system and the mechanisms of neuroinflammatory diseases. More and more studies highlight the importance of lymphatic function in various neurological diseases. As a widespread clearance system in the brain, GS may influence many neurological disorders, providing a new direction for future research into disease mechanisms and treatments. The main function of the lymphatic system is drainage and detoxification. It has been established that, under physiological conditions, up to 70% of cerebrospinal fluid from the subarachnoid space flows into the venous system of the brain, and up to 30% into the regional lymph nodes of the central nervous system in the cervical and paravertebral regions. Academician Borodin Yu. I. et al. revealed the anatomical and functional relationship of the central nervous system and the lymphatic system, the circulation of cerebrospinal fluid and lymph in the central nervous system, and also proved the effectiveness of lymphotropic therapy in correcting disorders of liquor dynamics in various pathological processes. These prerequisites, as well as the clinical work of many other authors, allow us to consider the use of lymphostimulating technologies in the treatment of TBI as appropriate and pathogenetically justified . Drainage from the CNS to the cervical lymph nodes has been documented in numerous studies using tracers or injection of antigen into the cerebrospinal fluid. Lymphoid-like structures in the meninges have been described in the adult mouse brain, and this pathway may transport extracellular solutes to the cervical ganglia . The discovery of HS opens a new direction in the understanding of brain diseases, which shifts the focus from changes in specific brain structure to the general circulation of fluid in the brain. An example is the crucial role of GS in understanding the occurrence of cerebral edema. However, recent HS studies have shown that CSF is the main source of initial cerebral edema rather than blood leakage. Improving drainage of the brain lymphatic system may have potential clinical implications in neurovascular and neuroinflammatory diseases. Developing strategies to improve the impaired function of the lymphatic drainage system is a priority in the treatment of neurological diseases. Taking into account the anatomical and functional relationship of the central nervous system and the lymphatic system, the circulation of cerebrospinal fluid and lymph, it seems justified and pathogenetically justified to use the method of indirect lymphostimulation in the treatment of patients with cerebral edema due to TBI and hemorrhagic stroke (HI) in the acute period.Materials and Methods
The scientific study included data from patients who were in the neuroreanimation department of the Bukhara branch of the Russian Research Center for Emergency Medicine. The materials of 75 patients were studied, of which 38 with TBI and 37 patients with HI. The age of the patients ranged from 37 to 68 years (mean age was 58.2±2 years). By gender - there were 43 men (57.3%), women - 32 (42.7%). All patients underwent standard diagnostic methods (assessment of neurological status during a joint examination by a neurologist and neurosurgeon, Multislice-Computed Tomography (MSCT), as well as laboratory tests (leukoformula, neutrophil to lymphocyte ratio index (NLRI)). Neurostatus was assessed using the Glasgow Coma Scale (GCS), with the average score upon admission to hospital being 9.3±2.1. On MSCT, in patients with hemorrhagic stroke, hemispheric hematomas accounted for 29 (78.3%), brainstem 4 (10.8%), ventricular 2 (5.4%) and subarachnoid 2 (5.4%), and in patients with TBI Subdural 24 (63.1%), parenchymal 10 (26.3%), as well as brainstem hemorrhage 4 (10.5%) and areas of brain contusion that did not require surgical intervention were identified. All patients with TBI and HI were divided into two groups. The first is a control group that received standard therapy.The second is the main group, whose patients received lymphotropic decongestant therapy as part of an intensive care complex. For the purpose of decongestant therapy, a solution of lidocaine 2%-1 ml, dexamethasone 4 mg, 10% glucose solution 3 ml was injected submastoidally on one side. in one syringe (the method was approved at a meeting of the Ethics Committee of the Ministry of Health of the Republic of Uzbekistan, protocol No. 7 of November 9, 2023). Submastoid lymphotropic injections were carried out for 5 days, along with conservative treatment, including: antibacterial, decongestant, membrane stabilizing, hemorheological, cerebroprotective and symptomatic therapy.A comparison of clinical and laboratory parameters was carried out in three stages: the first stage - upon admission, the second stage: day 5, the third stage - 10 days of intensive therapy.Results and Discussion
Analysis of the results of the control group of patients with TBI (n=20), who received standard treatment methods, showed that the level of impairment of consciousness at the time of admission to the hospital (the first stage of the study) averaged 10.1 points on the GCS. At this stage, in the blood test, segmented neutrophils amounted to 60%, and the number of lymphocytes - 16%, while the ratio index of segmented neutrophils to lymphocytes was 3.7. On the 5th day of treatment (the second stage of the study), the level of consciousness impairment was 11.1 points on the GCS. The number of segmented neutrophils was 62%, lymphocytes 15%, respectively, the NLR indicator was 4.1. On the 10th day of the study, the neurological status remained the same, amounting to 11.2 points on the GCS. At the same time, the number of segmented neutrophils was 65%, lymphocytes 18%, and the ISNL was 3.6 (graph 1).
Table 1: Dynamics of indicators in the control group of patients with TBI.
When analyzing the results of patients in the main group of patients with TBI who underwent lymphotropic decongestant therapy (n=18), the following data were revealed - at the first stage of the study, the level of impairment of consciousness averaged 9.1 points on the Glasgow scale. In laboratory blood tests, the number of segmented neutrophils was 58%, and lymphocytes - 12%, while the ISNL indicator was 4.8. At the second stage of the study, the assessment of the level of consciousness was 11.3 points on the GCS. Segmented neutrophils in the blood accounted for 60%, and lymphocytes 18%, respectively, the ISNL indicator was 3.3. At the third stage of the study, a noticeable increase in all these indicators was observed. There was clearing of consciousness to stupor, the average score of which was 13.1 points. And the indicators of the general blood test were: segmented neutrophils 62%, lymphocytes 20%, and ISNL 3.1 (graph 2).
Table 2: Dynamics of indicators of the main group of patients with TBI who underwent lymphotropic decongestant therapy.
(n=22) showed the following data: at the first stage of the study, the level of impairment of consciousness averaged 8.2 points on the Glasgow scale. In laboratory blood tests, the number of segmented neutrophils was 50%, and lymphocytes - 14%, while the ISNL indicator was 3.6. At the second stage of the study, the assessment of the level of consciousness was 10.1 points on the GCS. Segmented neutrophils accounted for 54%, and lymphocytes 23%, respectively, the ISNL indicator was 2.3. At the third stage of the study, the level of consciousness according to the GCS was 11.3 points. And the indicators of the general blood test were: segmented neutrophils 58%, lymphocytes 22%, and ISNL 3.3 (graph 3).
Table 3: Dynamics of indicators in the control group of patients with HI.
When analyzing the results of patients in the main group with HI (n=15), the following data were revealed: at the first stage of the study, the level of impairment of consciousness averaged 9.1 points on the Glasgow scale. In blood tests, the number of segmented neutrophils was 56%, and lymphocytes - 16%, while the ISNL indicator was 3.5. At the second stage of the study, the assessment of the level of consciousness was 10.2 points on the GCS. Segmented neutrophils accounted for 60%, lymphocytes 22%, the ISNL index was 2.7. At the third stage of the study, a noticeable increase in all these indicators was observed. There was clearing of consciousness to stupor, the average score of which was 13.1 points. And the indicators of the general blood test were: segmented neutrophils 62%, lymphocytes 25%, and ISNL 2.5 (graph 4).
Table 4: Dynamics of indicators of the main group of patients with HI who underwent lymphotropic decongestant therapy
Dynamic neuroimaging control (MSCT of the brain) made it possible to obtain information about the state of the hemorrhage, the bruise zone, and the perifocal zone. Analysis of the obtained data from the MSCT study revealed that all patients in both groups had parenchymal and hemorrhages in the subarachnoid space and ventricles of the brain on the primary MSCT. In addition, congestion of the sulci of the cerebral cortex and narrowing of the basal cistern were signs of intracranial hypertension with symptoms of impaired consciousness. In patients of the main group, against the background of lymphotropic decongestant therapy on the second and third MSCT, regression of cerebral edema was evidenced by the appearance of signs of improvement in the ar chitectonics of the cerebral cortex, restoration of the normal sizes of the basal cisterns and ventricles of the brain (Figures 1 and 3). All these manifestations corresponded to positive neurological changes. And in patients in the control group, the above changes on MSCT of the brain appeared noticeably slowly (Figures 1 and 2).
A) B) C)
Figurer 1: MSCT of the patient. with hemorrhagic stroke of the main group (A – upon admission, B – on the 5th day, C – on the 10th day).
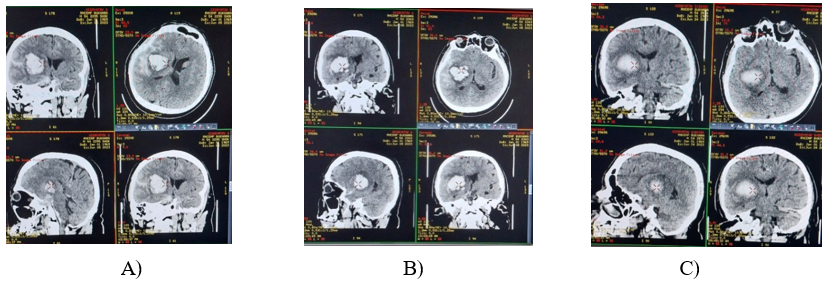
Figure 2: MSCT of a patient with a traumatic brain injury in the control group (A – upon admission, B – on the 5th day, C – on the 10th day).
Dynamic changes in the neurological status and computed tomography in both groups corresponded to changes in laboratory parameters that reflected changes in the leukocyte formula (graph 1,2,3, and 4). Rapid positive dynamics were observed in the main groups that received lymphotropic decongestant therapy compared to data from the control group.
Conclusion
Lymphotropic decongestant therapy increases the effectiveness of intensive basic treatment and prevents the progression of cerebral edema in patients with TBI and HI. Neuromonitoring using MSCT and comparison of clinical data allows dynamic objective monitoring of cerebral edema.References
- Borodin, Yu.I. Brain and body fluids / Yu.I. Borodin, Ya.M. Pesin. - Novosibirsk; Bishkek, 2005. - 184 p.
- Kochetkova M. V., Khabarov D. V., Maksimova O. V., Titova L. V.. Indirect lymphostimulation in the acute period of traumatic brain injury. Journal of Biology and Clinical. Medicine.2011, T.9.Issue 3.pp.156-160.
- Kuldashev K.A., Kuldasheva Y.M., Khakimova Z.K. and others. Diagnosis and surgical treatment of ICH of traumatic origin and intracranial infectious and inflammatory complications in combined traumatic brain injury. Journal of Dentistry and Craniofacial Research. 2021. No. 3, pp. 36-39.
- Morphofunctional characteristics of the pathways for the outflow of cerebrospinal fluid into the lymphatic bed under conditions of prolonged exposure to adrenaline and obsidan/ Ya.M. Pesin, E.A. Chernysheva, A.A. Beisembayev, M.Ya. Velikorodova //Bulletin of Siberian Medicine. - 2008. - No. 5.- P. 36-39.
- Pesin Ya.M., Lopatkina I.N., Imankulova E.M., Minich L.N. Experimental and clinical substantiation of lymphotropic therapy in the clinic of nervous diseases. Bulletin of KRSU. 2012. Volume 12. No. 2. p.133-136
- Semkin, V. A., Nadtochiy, A. G., Vozgoment, O. V., & Ivanova,A. A. (2020). Lymphatic therapy and its importance in the complex treatment of patients. Stomatologiia, 99(5), 116-121.
- Semyachkina-Glushkovskaya, O. V., Postnov, D. E., Khorovodov, A. P., Navolokin, N. A., & Kurthz, J. H. G. (2023). Lymphatic drainage system of the brain: a New player in neuroscience. Journal of Evolutionary Biochemistry and Physiology, 59(1), 1-19.
- Yankova G.S., Bogomyakova O.B. Lymphatic drainage system of the brain: imaging capabilities and the current state of the problem. Complex problems of cardiovascular diseases. 2020; 9 (3): 81-89. DOI: 10.17802/2306-1278-2020-9-3-81-89
- Yarilin A.A. Immunology. - M.: GEOTAR-Media, 2010. P. 381.
- Ahn, J. H., Cho, H., Kim, J. H., Kim, S. H., Ham, J. S., Park,I., ... & Koh, G. Y. (2019). Meningeal lymphatic vessels at the skull base drain cerebrospinal fluid. Nature, 572(7767), 62-66.
- Aspelund, A., Antila, S., Proulx, S. T., Karlsen, T. V., Karaman, S., Detmar, M., ... & Alitalo, K. (2015). A dural lymphatic vascular system that drains brain interstitial fluid and macromolecules. Journal of Experimental Medicine, 212(7), 991-999.
- Bolte, A. C., Dutta, A. B., Hurt, M. E., Smirnov, I., Kovacs,M. A., McKee, C. A., ... & Lukens, J. R. (2020). Meningeal lymphatic dysfunction exacerbates traumatic brain injury pathogenesis. Nature communications, 11(1), 4524.
- Chen, J., Wang, L., Xu, H., Xing, L., Zhuang, Z., Zheng, Y., ... & Wang, Y. (2020). Meningeal lymphatics clear erythrocytes that arise from subarachnoid hemorrhage. Nature communications, 11(1), 3159.
- Christensen, J., Wright, D. K., Yamakawa, G. R., Shultz, S. R., & Mychasiuk, R. (2020). Repetitive mild traumatic brain injury alters glymphatic clearance rates in limbic structures of adolescent female rats. Scientific reports, 10(1), 6254.
- Connections between cerebrospinal fluid and nasal lymphatics in the non-human primate // Neuropathol. Appl. Neurobiol. — 2005. — 31. — P. 632-640.
- Caffes, N., Stokum, J. A., Zhao, R., Jha, R. M., & Simard, J.M. (2022). Post-traumatic cerebral edema: pathophysiology, key contributors, and contemporary management. Medical Research Archives, 10(10).
- Da Mesquita, S., Louveau, A., Vaccari, A., Smirnov, I., Cornelison, R. C., Kingsmore, K. M., ... & Kipnis, J. (2018). Functional aspects of meningeal lymphatics in ageing and Alzheimer’s disease. Nature, 560(7717), 185-191.
- Ding, Z., Fan, X., Zhang, Y., Yao, M., Wang, G., Dong, Y., ... & Song, W. (2023). The glymphatic system: a new perspective on brain diseases. Frontiers in aging neuroscience, 15, 1179988.
- Donkin, J. J., & Vink, R. (2010). Mechanisms of cerebral edema in traumatic brain injury: therapeutic developments. Current opinion in neurology, 23(3), 293-299.
- Hu, X., Deng, Q., Ma, L., Li, Q., Chen, Y., Liao, Y., ... & Luo,J. (2020). Meningeal lymphatic vessels regulate brain tumor drainage and immunity. Cell research, 30(3), 229-243.
- Fang, Y., Shi, H., Ren, R., Huang, L., Okada, T., Lenahan, C., ... & Zhang, J. H. (2020). Pituitary adenylate cyclase-activating polypeptide attenuates brain edema by protecting blood–brain barrier and glymphatic system after subarachnoid hemorrhage in rats. Neurotherapeutics, 17(4), 1954-1972.
- Bohr, T., Hjorth, P. G., Holst, S. C., Hrabetová, S., Kiviniemi, V., Lilius, T., ... & Nedergaard, M. (2022). The glymphatic system: Current understanding and modeling. IScience, 25(9).
- Gao, Y., Liu, K., & Zhu, J. (2023). Glymphatic system: an emerging therapeutic approach for neurological disorders. Frontiers in Molecular Neuroscience, 16, 1138769.
- Graham, M. S., & Mellinghoff, I. K. (2021). Meningeal lymphatics prime tumor immunity in glioblastoma. Cancer Cell, 39(3), 304-306.
- Gu, J., Huang, H., Huang, Y., Sun, H., & Xu, H. (2019).Hypertonic saline or mannitol for treating elevated intracranial pressure in traumatic brain injury: a meta-analysis of randomized controlled trials. Neurosurgical review, 42(2), 499-509.
- Halstead, M.R., Geocadin, R.G. Medical treatment of cerebral edema: treatments past, present and future. Neurotherapy 16, 1133–1148 (2019).
- Hladky, S. B., & Barrand, M. A. (2022). The glymphatic hypothesis: the theory and the evidence. Fluids and Barriers of the CNS, 19(1), 9.
- Hu, X., Deng, Q., Ma, L., Li, Q., Chen, Y., Liao, Y., ... & Luo,J. (2020). Meningeal lymphatic vessels regulate brain tumor drainage and immunity. Cell research, 30(3), 229-243.
- Jha, R. M., Kochanek, P. M., & Simard, J. M. (2019). Pathophysiology and treatment of cerebral edema in traumatic brain injury. Neuropharmacology, 145, 230-246.
- Ji S, Yu X, Xu W, et al. The role of the lymphatic system in the formation of cerebral edema after ischemic stroke. Exp Neurol. 2021 March;113685.
- Johnston, M., Zakharov, A., Papaiconomou, C., Salmasi, G., & Armstrong, D. (2004). Evidence of connections between cerebrospinal fluid and nasal lymphatic vessels in humans, non-human primates and other mammalian species. Cerebrospinal fluid research, 1(1), 2.
- Johnston M. et al. Subarachnoid injection of Microfil reveals
- Kipnis, J., Gadani, S., & Derecki, N. C. (2012). Pro-cognitive properties of T cells. Nature Reviews Immunology, 12(9), 663-669.
- Koenig MA. Brain swelling and increased intracranial pressure. Continuum (Minneap, Minn) 2018; 24(6):1588–602.
- Li, G., Cao, Y., Tang, X., Huang, J., Cai, L., & Zhou, L. (2022).The meningeal lymphatic vessels and the glymphatic system: Potential therapeutic targets in neurological disorders. Journal of Cerebral Blood Flow & Metabolism, 42(8), 1364-1382.
- Liu E, Peng X, Ma X, et al. Involvement of aquaporin-4 in impaired interstitial fluid drainage after subarachnoid hemorrhage. Neurons before aging. 2020;12:611494. [Crossref] [PubMed] [Web of Science ®] , [Google Scholar]
- Liu, E., Sun, L., Zhang, Y., Wang, A., & Yan, J. (2019). Aquaporin4 Knockout Aggravates Early Brain Injury Following Subarachnoid Hemorrhage Through Impairment. Subarachnoid Hemorrhage: Neurological Care and Protection, 127, 59.
- Liu X, Gao C, Yuan, J. Xiang T, Gong Z, Luo H, Jiang W, Song Y, Huang J, Quan W, Wang D, Tian Y, Ge X, Lei P, Zhang J, Jiang R (2020) Subdural haematomas drain into the extracranial lymphatic system through the meningeal lymphatic vessels. Acta Neuropathol Commun 8: 16.
- Liu, K., Zhu, J., Chang, Y., Lin, Z., Shi, Z., Li, X., ... & Huang,K. (2021). Attenuation of cerebral edema facilitates recovery of glymphatic system function after status epilepticus. JCI insight, 6(17), e151835.
- Lohela, T. J., Lilius, T. O., & Nedergaard, M. (2022). The glymphatic system: implications for drugs for central nervous system diseases. Nature reviews Drug discovery, 21(10), 763-779.
- Louveau, A., Da Mesquita, S., & Kipnis, J. (2016). Lymphatics in neurological disorders: a neuro-lympho-vascular component of multiple sclerosis and Alzheimer’s disease?. Neuron, 91(5), 957-973.
- Louveau, A., Smirnov, I., Keyes, T. J., Eccles, J. D., Rouhani,S. J., Peske, J. D., ... & Kipnis, J. (2015). Structural and functional features of central nervous system lymphatic vessels. Nature, 523(7560), 337-341.
- Louveau, A., Smirnov, I., Keyes, T. J., Eccles, J. D., Rouhani,S. J., Peske, J. D., ... & Kipnis, J. (2015). Structural and functional features of central nervous system lymphatic vessels. Nature, 523(7560), 337-341.
- Lv, T., Zhao, B., Hu, Q., & Zhang, X. (2021). The glymphatic system: a novel therapeutic target for stroke treatment. Frontiers in Aging Neuroscience, 13, 689098.
- Mestre, H., Du, T., Sweeney,A. M., Liu, G., Samson,A. J., Peng, W., ... & Nedergaard, M. (2020). Cerebrospinal fluid influx drives acute ischemic tissue swelling. Science, 367(6483), eaax7171.
- Nagra, G., Koh, L., Zakharov, A., Armstrong, D., & Johnston,M. (2006). Quantification of cerebrospinal fluid transport across the cribriform plate into lymphatics in rats. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 291(5), R1383-R1389.
- Nedergaard, M., & Goldman, S. A. (2020). Glymphatic failure as a final common pathway to dementia. Science, 370(6512), 50-56.
- Zusman, B. E., Kochanek, P. M., & Jha, R. M. (2020). Cerebral edema in traumatic brain injury: a historical framework for current therapy. Current treatment options in neurology, 22(3), 9.
- Plog, B. A., & Nedergaard, M. (2018). The glymphatic system in central nervous system health and disease: past, present, and future. Annual Review of Pathology: Mechanisms of Disease, 13(1), 379-394.
- Proulx, S. T. (2021). Cerebrospinal fluid outflow: a review of the historical and contemporary evidence for arachnoid villi, perineural routes, and dural lymphatics. Cellular and Molecular Life Sciences, 78(6), 2429-2457.
- Rasmussen, M. K., Mestre, H., & Nedergaard, M. (2018). The glymphatic pathway in neurological disorders. The Lancet Neurology, 17(11), 1016-1024.
- Regul. Integr. Comp. Physiol. — 2006. — 291. — P. 1383-1389.

- Jha, R. M., Raikwar, S. P., Mihaljevic, S., Casabella, A. M., Catapano, J. S., Rani, A., ... & Simard, J. M. (2021). Emerging therapeutic targets for cerebral edema. Expert opinion on therapeutic targets, 25(11), 917-938.
- Scallan, J., Huxley, V. H., & Korthuis, R. J. (2010). Fluid movement across the endothelial barrier. Morgan & Claypool Life Sciences: San Rafael, CA, USA.
- Semyachkina-Glushkovskaya, O., Esmat, A., Bragin, D., Bragina, O., Shirokov, A. A., Navolokin, N., ... & Kurths, J. (2020). Phenomenon of music-induced opening of the blood-brain barrier in healthy mice. Proceedings of the Royal Society B, 287(1941), 20202337.
- Semyachkina-Glushkovskaya, O., Fedosov, I., Shirokov, A., Vodovozova, E., Alekseeva, A., Khorovodov, A., ... & Kurths, J. (2021). Photomodulation of lymphatic delivery of liposomes to the brain bypassing the blood-brain barrier: new perspectives for glioma therapy. Nanophotonics, 10(12), 3215-3227.
- Semyachkina-Glushkovskaya, O., Penzel, T., Blokhina, I., Khorovodov, A., Fedosov, I., Yu, T., ... & Kurths, J. (2021). Night photostimulation of clearance of beta-amyloid from mouse brain: New strategies in preventing Alzheimer’s disease. Cells, 10(12), 3289.
- Semyachkina-Glushkovskaya, O., Postnov, D., Lavrova,A., Fedosov, I., Borisova, E., Nikolenko, V., ... & Tuchin,V. (2020). Biophotonic strategies of measurement and stimulation of the cranial and the extracranial lymphatic drainage function. IEEE Journal of Selected Topics in Quantum Electronics, 27(4), 1-13.
- Shah, S., & Kimberly, W. T. (2016, December). Today's approach to treating brain swelling in the neuro intensive care unit. In Seminars in neurology (Vol. 36, No. 06, pp. 502-507). Thieme Medical Publishers.
- Simon DW, McGeachie MJ, Bayir H, Clarke RSB, Loane J, Kochanek PM. The far-reaching extent of neuroinflammation following traumatic brain injury. Nat Rev. Neurol. 2017; 13: 171–91.
- Song, E., Mao, T., Dong, H., Boisserand, L. S. B., Antila, S., Bosenberg, M., ... & Iwasaki, A. (2020). VEGF-C-driven lymphatic drainage enables immunosurveillance of brain tumours. Nature, 577(7792), 689-694.
- Stokum J.A., Gerzanich V., Sheth K.N. and others. New pharmacological methods for the treatment of cerebral edema: data from clinical studies. Annu Rev Pharmakol Toksikol. Jan 2020;60(1):291–309. [Crossref] [PubMed] [Web of Science®], [Google Scholar]
- Stokum, J. A., Gerzanich, V., & Simard, J. M. (2016). Molecular pathophysiology of cerebral edema. Journal of Cerebral Blood Flow & Metabolism, 36(3), 513-538.
- Sulhan, S., Lyon, K. A., Shapiro, L. A., & Huang, J.H. (2020). Neuroinflammation and blood–brain barrier disruption following traumatic brain injury: pathophysiology and potential therapeutic targets. Journal of neuroscience research, 98(1), 19-28.
- Sun, B. L., Wang, L. H., Yang, T., Sun, J. Y., Mao, L. L., Yang,M. F., ... & Yang, X. Y. (2018). Lymphatic drainage system of the brain: a novel target for intervention of neurological diseases. Progress in neurobiology, 163, 118-143.
- Dalby, T., Wohl, E., Dinsmore, M., Unger, Z., Chowdhury, T., & Venkatraghavan, L. (2021). Pathophysiology of cerebral edema—a comprehensive review. Journal of neuroanaesthesiology and critical care, 8(03), 163-172.
- Tucker B, Aston J, Dines M, Karaman E, Yatsyshyn M, McCarthy M, et al. Early cerebral edema predicts in-hospital mortality in traumatic brain injury. Jay Emerge Med. 2017; 53:18–29.
- Weller, R. O., Djuanda, E., Yow, H. Y., & Carare, R. O. (2009). Lymphatic drainage of the brain and the pathophysiology of neurological disease. Acta neuropathologica, 117(1), 1-14.
- Weller, R. O., Galea, I., Carare, R. O., & Minagar, A. (2010). Pathophysiology of the lymphatic drainage of the central nervous system: Implications for pathogenesis and therapy of multiple sclerosis. Pathophysiology, 17(4), 295-306.
- Xuan, X., Zhou, G., Chen, C., Shao, A., Zhou, Y., Li, X., & Zhou, J. (2022). [Retracted] Glymphatic System: Emerging Therapeutic Target for Neurological Diseases. Oxidative Medicine and Cellular Longevity, 2022(1), 6189170.
- Yankova, G., Bogomyakova, O., & Tulupov, A. (2021). The glymphatic system and meningeal lymphatics of the brain: new understanding of brain clearance. Reviews in the Neurosciences, 32(7), 693-705.
- Zusman, B. E., Kochanek, P. M., & Jha, R. M. (2020). Cerebral edema in traumatic brain injury: a historical framework for current therapy. Current treatment options in neurology, 22(3), 9.




