Research Article - (2025) Volume 1, Issue 1
BRAF V600E Expression in Papillary Thyroid Carcinoma and its Association with Histological Prognostic Factors
2Associate Professor, Department of Pathology, Bangabandhu Sheikh Mujib Medical University, Dhaka, Bangladesh
3Lecturer, Department of Pathology, Habiganj Medical College, Hobiganj, Sylhet, Bangladesh
4Lecturer, Department of Pathology, Gopalganj Medical College, Gopalganj, Bangladesh
5Medical Officer, Civil Surgeon’s Office, Lakshmipur, Bangladesh
Received Date: Apr 15, 2025 / Accepted Date: May 23, 2025 / Published Date: May 29, 2025
Copyright: ©©2025 Clinical Pathologist, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Chowdhury, F. H., Saba, S., Akhter, S., Rahman, M. M., Paul, E., et al. (2025). BRAF V600E Expression in Papillary Thyroid Carcinoma and its Association with Histological Prognostic Factors. Arch Cienc Investig, 1(1), 01-10.
Abstract
Background: Thyroid cancer is the most common endocrine tumor, accounting for 90% of all endocrine malignancies. Papillary thyroid carcinoma (PTC) is the most common thyroid cancer. Despite its good prognosis, a small number of PTC cases develop lymph node metastasis, extrathyroidal extension, distant metastasis, or recurrent disease. The B-type RAF V600E (BRAF V600E) mutation is the most commonly observed genetic abnormality in PTC and PTC-derived anaplastic carcinoma. This study was done to determine the association between BRAF V600E expression in PTC and tumor size, multifocality, extrathyroidal extension, and chronic lymphocytic thyroiditis.
Method: This was a cross-sectional study conducted with 50 cases of papillary thyroid cancer at the Department of Pathology, BSMMU, Dhaka, Bangladesh from March 2022 to February 2024. Tumor size, tumor focality, extrathyroidal extension, chronic lymphocytic thyroiditis, and PTC histologic subtype are among the histological prognostic variables. The BRAF V600E mutation status was examined with immunohistochemistry on the selected paraffin blocks. BRAF V600E expression was determined by scoring based on signal intensity (0-3) and the proportion of tumor cells (0-100%). Demographic data, histological findings and the BRAF V600E expression status of all the cases were compiled, followed by statistical analysis.
Results: Out of 50 PTC cases, 23 (46%) cases were BRAF V600E expression-positive. No significant association was found between BRAF V600E expression and tumor size, extrathyroidal extension, tumor focality, or chronic lymphocytic thyroiditis.
Discussion: No significant association was found between the expression of BRAF V600E and tumor size, extrathyroidal extension, tumor focality, or chronic lymphocytic thyroiditis in PTC.
Keywords
Papillary Thyroid Carcinoma, BRAF V600E Immune Expression
Introduction
Thyroid cancer is the most common endocrine tumor, accounting for 90% of all endocrine malignancies [1,2]. It is the ninth most common cancer diagnosed globally in 2020, with 5.86 million cases, according to GLOBOCAN. In both adult and pediatric populations, papillary thyroid carcinoma is the most common type of thyroid cancer [3,4]. Papillary thyroid carcinoma was nearly solely responsible for the rise in thyroid cancer incidence. The causes of the rise are complicated, multifactorial, and not fully understood. The most obvious causes include the greater use of diagnostic entities, the changing classification of thyroid neoplasms, environmental risk factors, radiation, obesity, and other issues [5].
Papillary thyroid carcinoma has relatively indolent behavior and a better prognosis compared with other malignant thyroid tumors. But about 25%-30% of PTC cases follow an aggressive clinical course, such as lymph node metastasis, extrathyroidal extension, distant metastasis, or recurrent disease [3,6-8]. Many authors have also reported that the risk of developing PTC increases in those with preexisting Hashimoto’s thyroiditis (HT), an autoimmune disease also known as chronic lymphocytic thyroiditis. However, many investigators have reported that PTC with coexistent HT is associated with lower extrathyroidal extension, advanced stage, lymph node metastasis, and recurrence [9-12].
The revised American Thyroid Association guidelines point out that thyroid cancer should be treated according to risk stratification, assessed on the basis of disease stage and genetic testing [13-16]. Studies have reported that the BRAF mutation in PTC is about 50% to 80%. Point mutations of the BRAF gene are the most common genetic alteration known to occur in thyroid papillary carcinomas [3,17-21]. Some researchers claimed that the BRAF V600E mutation in PTC had been associated with more aggressive histological features, such as older age, male sex, tumor size, multifocality, extrathyroidal extension, capsular invasion, lymph node metastasis, advanced TNM stage, and recurrence [22-24].
Conversely, some studies have shown that the BRAF V600E mutation in PTC has not been fully or partially linked to more aggressive histological features, including advanced TNM stage, recurrence, male sex, larger tumors, multifocality, extrathyroidal extension, capsular invasion and lymph node metastasis [25-28]. It has been reported that PTC patients with the BRAF V600E mutation often lose the ability to uptake radioactive iodine, followed by subsequent resistance to radioactive iodine treatment. Currently, BRAF V600E mutation-specific targeted drugs intended for papillary thyroid carcinoma are under clinical trial [29,30]. If these BRAF V600E inhibitors for papillary thyroid carcinoma are approved by the FDA, they can be used as an alternative therapeutic approach for radioactive iodine-resistant patients.
Within the aforementioned context, the purpose of this study is to investigate the relationship between the BRAFV600E mutation in papillary thyroid carcinoma and histological prognostic factors, such as tumor size, tumor focality, extrathyroidal extension and chronic lymphocytic thyroiditis, that have been linked to aggressive or poor clinical outcomes.
Methods
This was a cross-sectional study conducted with 50 cases of papillary thyroid cancer at the Department of Pathology, BSMMU, Dhaka, Bangladesh from March 2022 to February 2024. Tumor size, tumor focality, extrathyroidal extension, chronic lymphocytic thyroiditis, and PTC histologic subtype are among the histological prognostic variables. A total of 50 papillary thyroid carcinoma cases were selected for the study by matching the inclusion and exclusion criteria. After gross and microscopic examination and clinical correlation, histopathological diagnosis of Papillary thyroid carcinoma is confirmed according to WHO classification. Corresponding slides and paraffin blocks were collected from the Department of Pathology, BSMMU, Dhaka, Bangladesh. Representative sections from paraffin block were reviewed and selected for BRAF V600E immunohistochemical stain. Patient demographic data and clinicopathological features were collected from patient and pathology reports.
If the immunoreactivity of the tumor cells is greater than or equal to the surrounding follicular colloid, or diffuse staining, and is observed at a magnification of 40X, the staining intensity is rated as 3+. If tumor cells displayed distinct cytoplasmic smears, but their intensity was less than that of the surrounding follicular colloids, which were clearly visible at 100X magnification, the staining intensity was rated as moderate (2+). If tumor cells displayed hazy or challenging stain detection, the staining intensity was rated as weak (1+). If there was no staining on any of the tumor cells, the score is 0 [23,27]. The percentage of positive cells was assigned a score of 0 for less than 5%, 1 for between 5 and 25%, 2 for between 26 and 50%, 3 for between 51 and 75%, and >75% for 4 points. Ultimately, the staining degree was ascertained by multiplying the scores denoting the percentage of positive cells and staining intensity, as follows: A score of 1-4 denotes a mildly positive (+), 5-8 a moderately positive (+ +), and a score of ≥9 denotes a strongly positive (+++) [28-31].
Results
Age of the Study Participants
The age range of the study participants was 17–65 years, with a mean age of 38.66 ± (12.637) years. The distribution of age groups by decades between 11 and 70 years has been shown in Table 1.
|
Age (years) |
Frequency (n) |
Percentage (%) |
|
11-20 |
10 |
20 |
|
22-30 |
12 |
24 |
|
31-40 |
11 |
22 |
|
41-50 |
10 |
20 |
|
51-60 |
5 |
10 |
|
61-70 |
2 |
4 |
|
Total |
50 |
100 |
|
Mean age ± (SD) = 38.66 ± (12.637) Minimum=17, Maximum=65 |
||
Table 1: Age of the Study Cases (n = 50)
Table 2 shows the number of patients <55 years of age versus the number of patients ≥55 years of age according to age stratification by AJCC.
|
Age (years) |
Frequency (n) |
Percentage (%) |
|
<55 |
43 |
86 |
|
≥55 |
7 |
14 |
|
Total |
50 |
100 |
|
Mean age ± (SD) = 38.66 ± (12.637) Minimum=17, Maximum=65 |
||
Table-2: Stratification of Study Cases on the Basis of Age (cut off value 55 years) according to AJCC (n= 50) Sex of the patients
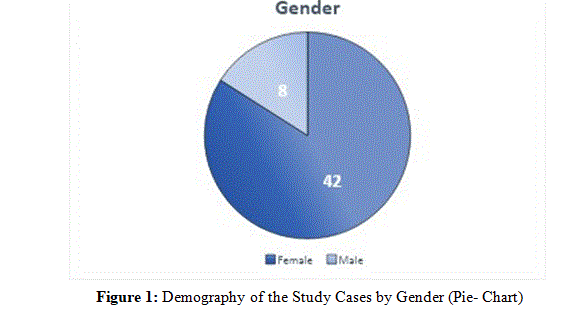
Out of 50 cases, 42 were females and 8 were males, with a male- to-female ratio of 1:5 (figure 1).
Tumor size (pT) of PTC in the study cases
In this current study, 21 (42%) of PTC cases had pT¸?1, whereas 24 (48%) had pT¸?2 and 5 (10%) had pT¸?3 (Table 4).
|
Tumor size (pT) |
Frequency (n) |
Percentage (%) |
|
T1 |
21 |
42 |
|
T2 |
24 |
48 |
|
T3 |
5 |
10 |
|
Total |
50 |
100 |
Table 3: Tumor Size (pT) of PTC in the Study Cases (n= 50)
Characteristics of Study Cases by Histologic Prognostic Factors
Table 5 describes the results of the histological prognostic factors of the PTC cases in this study. It is evident that in 90% of cases, the tumor size was ≤ 4 cm, tumour was unifocal in 68% of cases versus multifocal in 32%, chronic lymphocytic thyroiditis was present only in 30% of cases, and extrathyroidal extension was detected only in 8% of cases.
|
Attributes |
Frequency (n) |
Percentage (%) |
|
|
Tumour Size |
≤ 4cm |
45 |
90 |
|
>4 cm |
5 |
10 |
|
|
Tumour focality |
Unifocal |
34 |
68 |
|
Multifocal |
16 |
32 |
|
|
Chronic lymphocytic thyroiditis |
Absent |
35 |
70 |
|
Present |
15 |
30 |
|
|
Extra thyroidal extension |
Absent |
46 |
92 |
|
Present |
4 |
8 |
|
|
Lymph node metastasis |
Absent |
7 |
14 |
|
Present |
18 |
36 |
|
|
|
LN not found |
25 |
50 |
Table 4: Characteristics of Study Cases by Histologic Prognostic Factors (n=50)
Association of Age, Sex, Tumor Size, Chronic Lymphocyt- ic Thyroiditis, Extrathyroidal Extension, Tumor Focality And Lymph Node Metastasis of Study Cases With BRAF V600e Expression
The association between age and BRAF V600E expression in the PTC cases in this investigation is shown in Table 8. It is evident that 87% of patients in the age group <55 and 13% in the age group ≥ 55 had BRAF V600E expression-positive results (BRAF V600E mutation). This difference in age between BRAF V600E mutation is not statistically significant.
BRAF V600E expression-positive results were found in 20 (87%) female cases and 3 (13%) male cases. The gender difference in the BRAF V600E mutation was not found to be statistically significant. 11 (47.8%) cases had pT¸?1, 10 (43.5%) pT¸2, and 2 (8.7%) had pT3 with positive BRAF V600E mutation expression. The tumour size (pT) and BRAF V600E mutation are statistically non-significant in the Chi-square test (p= 0.183). Chronic lymphocytic thyroiditis is present in 8 (39%) of the BRAF V600E-positive individuals, while it is absent in 15 (65%) of the cases. According to the Chi-square test, the expression of the BRAF V600E mutation in PTC and chronic lymphocytic thyroiditis is statistically not significant (p = 0.496). Extrathyroidal extension was present in four cases. Among the four cases, two show the BRAF V600E mutation. According to the Fisher’s exact test, the expression of the BRAF V600E mutation in PTC and extrathyroidal extension is statistically not significant (p= 1.0). Among available 25 cases with lymph nodes, BRAF V600E expression was present in 9 cases. Therefore, it was not possible to assess the association between lymph node metastases and BRAF V600E expression. In 14 (60.9%) unifocal PTC cases and 9 (39.1%) multifocal PTC cases, BRAF V600E expression was positive. The tumor focality and BRAF V600E mutation expression are statistically non-significant in the chi- square test. (p= 0.318).
In this study, 25 of the 50 histologically confirmed cases of PTC have surgical specimens that include lymph nodes. Among these 25 cases, BRAF V600E expression was present in 9 cases. Therefore, it was not possible to assess the association between lymph node metastases and BRAF V600E expression.
|
Attributes |
BRAF V600E expression |
Significance (p-value) |
|
|
Positive n (%) |
Negative n (%) |
||
|
Age (years) |
Fisher’s exact=2.759 df=4 p= 0.625 |
||
|
<55 |
20 (86.95) |
26 (96.29) |
|
|
≥55 |
3 (13.05) |
1 (3.7) |
|
|
Sex |
Fisher’s exact=0.277 df=1 p= 0.711 |
||
|
Female |
20 (87) |
22 (81.5) |
|
|
Male |
3 (13) |
5 (18.5) |
|
|
Tumour size (pT) |
χ2=3.402 df=2
p= 0.183 |
||
|
T1 |
11 (47.8) |
10 (37.0) |
|
|
T2 |
10 (43.5) |
14 (45.2) |
|
|
T3 |
2 (8.7) |
3 (17.8) |
|
|
Chronic lymphocytic thyroiditis |
χ2=0.464 df=1 p= 0.496 |
||
|
Absent |
15 (65.2%) |
20 (74.1%) |
|
|
Present |
8 (34.8%) |
7 (25.9%) |
|
|
Extrathyroidal extension |
Fisher’s exact =0.028 df=1 p= 1.0 |
||
|
Present |
2 (8.7) |
2 (7.4) |
|
|
Absent |
21 (91.3) |
25 (92.6) |
|
|
Tumour focality |
χ2=0.995 df=1 p= 0.318 |
||
|
Unifocal |
14 (60.9) |
20 (74.1) |
|
|
Multifocal |
9 (39.1) |
7 (25.9) |
|
|
Total |
23 (100) |
27 (100) |
|
Table 5: Association of Age, Sex, Tumour Size, Chronic Lymphocytic Thyroiditis, Extra Thyroidal Extension and Tumour Focality Of Study Cases with BRAF V600E Expression (n=50)
|
Lymph nodes |
Metastasis |
BRAF V600E expression |
Significance (p-value) |
|
|
Positive n (%) |
Negativen (%) |
|||
|
Absent |
7 |
4 |
3 |
Not applicable |
|
Present |
18 |
9 |
9 |
|
|
Total |
25 |
13 |
1 |
|
Table 6: Histopathological Association of Lymph Node Metastasis with BRAF V600E Expression (n= 25)
Discussion
The BRAF V600E mutant in papillary thyroid cancer has been associated with increased age, extra-thyroidal extension, TNM stage, lymph node metastases, recurrence, and a short lifespan [6,7]. Conversely, conflicting findings were found in multiple studies regarding the relationship between the BRAF V600E mutant and unfavorable clinical characteristics in PTC [32]. Several studies showed that there was no significant association between the BRAF V600E mutation and age, which is similar to my study [23,25,33,34]. Many researchers revealed in their studies that most of the patients who were diagnosed with papillary thyroid carcinoma were female. But they did not find any statistically significant association between the BRAF V600E mutation and gender [18,33,34,35]. This result is consistent with my study's findings. Sun et al. (2015) from China, Fraser et al. (2016) from Australia, and Kristiani et al. (2021) from Indonesia have also reported no significant differences in tumor size between positive and negative BRAF V600E expression in cases of papillary thyroid carcinoma, which is consistent with this study [27,33,34,36].
According to several investigations, including comprehensive evaluations, multifocality was not linked to BRAF V600E expression in PTC patients [23,37,38,39,40]. My study's findings are consistent with these outcomes. On the other hand, several studies showed no association between BRAF V600E expression
Illustrations and extrathyroidal extension [41-43]. These results are consistent with the findings of the current study.
There have been investigations reporting that PTCs that coexist with CLT have a lower frequency of the BRAF V600E mutation [25,38,44,45]. This study found only 15 PTC with coexistent lymphocytic thyroiditis, which might have been the reason for not getting any statistically significant association with the BRAF V600E mutation. Some researchers reported that there was no significant association between lymph node metastasis and the BRAF V600E mutation, similar to this study [40,46]. Several researchers reported that there was no association between the BRAF V600E mutation in PTC and tumor size (p�) [47]. This finding is consistent with my study as well.
Conclusion
The results of this current study revealed that 23 (46%) of the PTC cases had the BRAF V600E mutation. However, no significant association was found between the expression of BRAF V600E and tumor size, extrathyroidal extension, tumor focality, or chronic lymphocytic thyroiditis. In the study cases, the BRAF V600E mutant did not exhibit a statistically significant correlation with age or sex; however, the presence of the mutation was six times higher in those under the age of fifty-five than in those beyond the age of fifty-five.
Illustrations
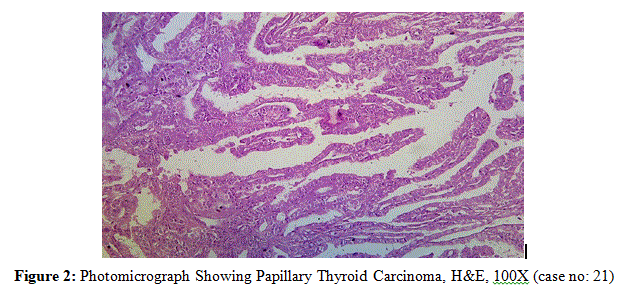
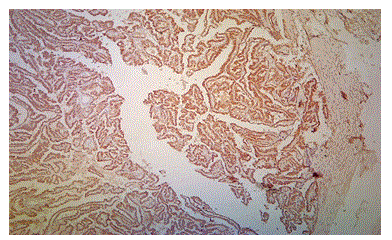
Figure 3: Photomicrograph Showing Papillary Thyroid Carcinoma with Strong and Diffuse Cytoplasm Positivity for BRAF V600E (case no: 21)
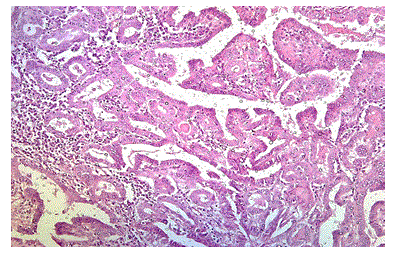
Figure 4: Photomicrograph Showing Papillary Thyroid Carcinoma, H&E, 100X (case no: 3)
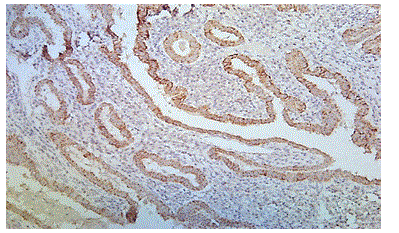
Figure 5: Photomicrograph Showing Papillary Thyroid Carcinoma with Moderate and Diffuse Cytoplasm Positivity for BRAF V600E
(case no: 3)
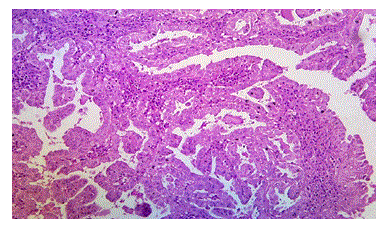
Figure 6: Photomicrograph Showing Papillary Thyroid Carcinoma, H&E, 100X (case no: 50)
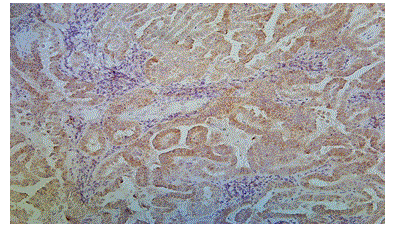
Figure 7: Photomicrograph Showing Papillary Thyroid Carcinoma with Moderate and Diffuse Cytoplasm Positivity for BRAF V600E
(case no: 50)
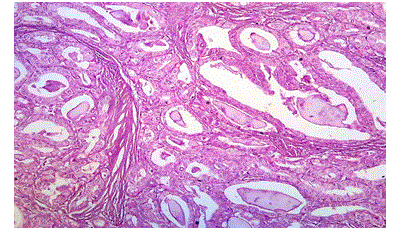
Figure 8: Photomicrograph Showing Papillary Thyroid Carcinoma, H&E, 100X (case no: 20)
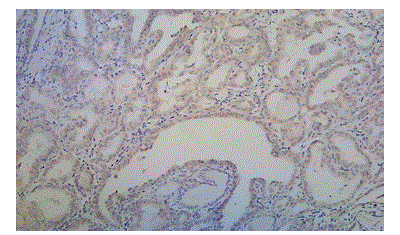
Figure 9: Photomicrograph Showing Papillary Thyroid Carcinoma with Weak and Diffuse Cytoplasm Positivity for BRAF V600E (case
no: 20)
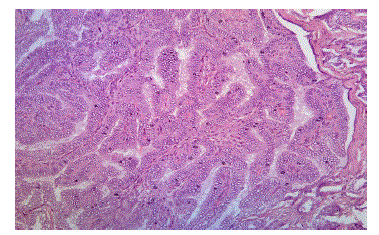
Figure 10: Photomicrograph Showing Papillary Thyroid Carcinoma, H&E, 100X (case no: 31)
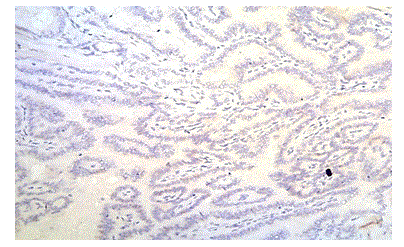
Figure 11: Photomicrograph Showing Papillary Thyroid Carcinoma with No Cytoplasm Positivity for BRAF V600E (case no: 31)
References
1. Baskey, S. C., and Malua, S. (2019). A Cross Section Study of Role of Serum TSH as A Biochemical Predictor of Malignancy in Suspicious Thyroid Swelling In the Tertiary Centre of Jharkhand. 18(3): 46–50.
2. Tang, K. T., and Lee, C. H. (2010) BRAF Mutation in Papillary Thyroid Carcinoma: Pathogenic Role and Clinical Implications. Journal of the Chinese Medical Association 73(3): 113–128.
3. Siddique, F., Ahmad, A. N., Ahmad, A., Ahsan, N., Gauhar, T. M., & Azim, K. M. (2013). PAPILLARY THYROID CARCINOMA. Professional Medical Journal, 20(4).
4. Y Lloyd Ricardo, R. y O., Cloppel, G., and Rosai, J. eds. (2017) World Health Organization Classification of Tumours, 4th Edition.
5. Sung, H., Ferlay, J., Siegel, R. L., Laversanne, M., Soerjomataram, I., Jemal, A., & Bray, F. (2021). Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a cancer journal for clinicians, 71(3), 209-249.
6. Fakhruddin, N., Jabbour, M., Novy, M., Tamim, H., Bahmad, H., Farhat, F., Zaatari, G., Aridi, T., Kriegshauser, G., Oberkanins, C., and Mahfouz, R. (2017) BRAF and NRAS Mutations in Papillary Thyroid Carcinoma and Concordance in BRAF Mutations between Primary and Corresponding Lymph Node Metastases. Scientific Reports 7(1): 1–11.
7. Choden, S., Keelawat, S., Jung, C. K., and Bychkov, A. (2020). VE1 immunohistochemistry improves the limit of genotyping for detecting BRAFV600E mutation in papillary thyroid cancer. Cancers 12(3): 1–13.
8. Abdullah, M. I., Junit, S. M., Ng, K. L., Jayapalan, J. J., Karikalan, B., & Hashim, O. H. (2019). Papillary thyroid cancer: genetic alterations and molecular biomarker investigations. International journal of medical sciences, 16(3), 450.
9. Lee, I., Kim, H. K., Soh, E. Y., & Lee, J. (2020). The association between chronic lymphocytic thyroiditis and the progress of papillary thyroid cancer. World journal of surgery, 44, 1506-1513.
10. Hu, X., Wang, X., Liang, Y., Chen, X., Zhou, S., Fei, W., ... & Que, H. (2022). Cancer risk in Hashimoto’s thyroiditis: a systematic review and meta-analysis. Frontiers in endocrinology, 13, 937871.
11. Babli, S., Payne, R. J., Mitmaker, E., & Rivera, J. (2018). Effects of chronic lymphocytic thyroiditis on the clinicopathological features of papillary thyroid cancer. European thyroid journal, 7(2), 95-101.
12. Yang, L. B., Sun, L. Y., Jiang, Y., Tang, Y., Li, Z. H., Zhang, H. Y., ... & Ye, F. (2015). The clinicopathological features of BRAF mutated papillary thyroid cancers in Chinese patients. International Journal of Endocrinology, 2015(1), 642046.
13. Zhao, J., Liu, P., Yu, Y., Zhi, J., Zheng, X., Yu, J., & Gao, M. (2019). Comparison of diagnostic methods for the detection of a BRAF mutation in papillary thyroid cancer. Oncology Letters, 17(5), 4661-4666.
14. Cipriani, N. A. (2019). Prognostic parameters in differentiated thyroid carcinomas. Surgical Pathology Clinics, 12(4), 883- 900.
15. Haugen, B. R., Alexander, E. K., Bible, K. C., Doherty, G. M., Mandel, S. J., Nikiforov, Y. E., ... & Wartofsky, L. (2016). 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid, 26(1), 1-133.
16. Eilsberger, F., Kreissl, M. C., Reiners, C., Holzgreve, A., Luster, M., & Pfestroff, A. (2023). Application of the American thyroid association risk assessment in patients with differentiated thyroid carcinoma in a German population. Biomedicines, 11(3), 911.
17. Wahid, M. H. A., & Almudhafar, R. H. (2022). Comparative BRAF V600E immunohistochemical expression in differentiated thyroid tumors with papillary features. Journal of Medicine and Life, 15(4), 520.
18. Al-Ankoshy, A. A. M., Alqershi, K. A., & Rabeea, H. W. (2020). The role of BRAF Mutation (V600E) in Papillary Thyroid Carcinoma (PTC). Indian Journal of Forensic Medicine & Toxicology, 14(1), 1377.
19. Kumar, V. (2021). Pathologic Basis of Disease by Robbins and Cotran. Elsevier.
20. Goldblum, J. R., Lamps, L. W., & McKenney, J. K. (2017). Rosai and Ackerman's Surgical Pathology E-Book. Elsevier Health Sciences.
21. Wartofsky, L., Van Nostrand, D., Ridgway, E. C., and Mazzaferri, E. L. (2016) Thyroid cancer (second edition): A comprehensive guide to clinical management. In Thyroid Cancer (Second Edition): A Comprehensive Guide to Clinical Management (3rd ed.).
22. Tabriz, N., Grone, J., Uslar, V., Tannapfel, A., & Weyhe, D. (2020). BRAF V600E mutation correlates with aggressive clinico-pathological features but does not influence tumor recurrence in papillary thyroid carcinoma—10-year single- center results. Gland Surgery, 9(6), 1902.
23. Wikanta, E. R., Prajoko, Y. W., Issakh, B., Istiadi, H., & Puspasari, D. (2022). Relationship between the BRAF V600E and tumor size, lymph node, and distant metastasis in papillary thyroid carcinoma. Russian Open Medical Journal, 11(2), 216.
24. Celik, M., Bulbul, B. Y., Ayturk, S., Durmus, Y., Gurkan, H., Can, N., ... & Guldiken, S. (2019). The relation between BRAFV600E mutation and clinicopathological characteristics of papillary thyroid cancer. Medicinski glasnik, 17(1), 30-34.
25. Yan, C., Huang, M., Li, X., Wang, T., and Ling, R. (2019) Relationship between braf v600e and clinical features in papillary thyroid carcinoma. Endocrine Connections 8(7): 988–996.
26. Alexander, E. K., Pearce, E. N., Brent, G. A., Brown, R. S., Chen, H., Dosiou, C., ... & Sullivan, S. (2017). 2017 Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and the postpartum. Thyroid, 27(3), 315-389.
27. Sun, J., Zhang, J., Lu, J., Gao, J., Lu, T., Ren, X., ... & Liang,
Z. (2015). Immunohistochemistry is highly sensitive and specific for detecting the BRAF V600E mutation in papillary thyroid carcinoma. International journal of clinical and experimental pathology, 8(11), 15072.
28. Dora, J. M., and Maia, A. L. (2020) papillary thyroid carcinoma. 600(4): 751–757.
29. Zaman, A., Wu, W., & Bivona, T. G. (2019). Targeting oncogenic BRAF: past, present, and future. Cancers, 11(8), 1197.
30. Ulisse, S., Baldini, E., Lauro, A., Pironi, D., Tripodi, D., Lori, E., ... & Sorrenti, S. (2021). Papillary thyroid cancer prognosis: An evolving field. Cancers, 13(21), 5567.
31. Zhang, Y., Liu, L., Liu, Y., Cao, N., Wang, L., & Xing, C. (2021). Clinical significance of immunohistochemistry to detect BRAF V600E mutant protein in thyroid tissues. Medicine, 100(16), e25566.
32. Fano, M. P., Olano, A. U., Thomas, E. F., & Alday, A. O. (2017). Immunohistochemical detection of the BRAF V600E mutation in papillary thyroid carcinoma. Evaluation against real-time polymerase chain reaction. Endocrinología, Diabetes y Nutrición (English ed.), 64(2), 75-81.
33. Lai, Y., Gu, Y., Yu, M., & Deng, J. (2023). Younger Than 55 years old and BRAF V600E mutation are risk factors for lymph node metastasis in papillary thyroid carcinomas≤
1.0 cm but not in> 1.0 cm. International Journal of General Medicine, 1403-1414.
34. Kristiani, E., Hardjolukito, E. S., Harahap, A. S., & Makes, B. (2020). BRAF V600E Immunoexpression in papillary thyroid carcinoma and its association with prognostic factors and histopathologic variant. Medicinus, 9(3), 12-21.
35. Van, H. N., Tuan, T. N., Ngoc, M. T., Van, T. P., & Thi, L. D. (2022). Association of BRAF V600E immunoexpression with clinicopathological variant groups in papillary thyroid carcinoma. Vietnam Journal of Science, Technology and Engineering, 64(4), 64-69.
36. Fraser, S., Go, C., Aniss, A., Sidhu, S., Delbridge, L., Learoyd, D., ... & Sywak, M. (2016). BRAF V600E mutation is associated with decreased disease-free survival in papillary thyroid cancer. World journal of surgery, 40, 1618-1624.
37. Rashid, F. A., Tabassum, S., Khan, M. S., Ansari, H. R., Asif, M., Sheikh, A. K., & Sameer Aga, S. (2021). VE1 immunohistochemistry is an adjunct tool for detection of BRAFV600E mutation: Validation in thyroid cancer patients. Journal of Clinical Laboratory Analysis, 35(2), e23628.
38. Celik, M., Bulbul, B. Y., Ayturk, S., Durmus, Y., Gurkan, H., Can, N., ... & Guldiken, S. (2019). The relation between BRAFV600E mutation and clinicopathological characteristics of papillary thyroid cancer. Medicinski glasnik, 17(1), 30-34.
39. Kim, H. I., Hyeon, J., Park, S. Y., Ahn, H. S., Kim, K., Han, J. M., Bae, J. C., Shin, J. H., Kim, J. S., Kim, S. W., Chung, J. H., Kim, T. H., and Oh, Y. L. (2019). Impact of Extranodal Extension on Risk Stratification in Papillary Thyroid Carcinoma. Thyroid 29(7): 963–970.
40. Gouveia, C., Can, N. T., Bostrom, A., Grenert, J. P., van Zante, A., & Orloff, L. A. (2013). Lack of association of BRAF mutation with negative prognostic indicators in papillary thyroid carcinoma: the University of California, San Francisco, experience. JAMA otolaryngology–head & neck surgery, 139(11), 1164-1170.
41. Ito, Y., Yoshida, H., Maruo, R., Morita, S., Takano, T., Hirokawa, M., ... & Miyauchi, A. (2009). BRAF mutation in papillary thyroid carcinoma in a Japanese population: its lack of correlation with high-risk clinicopathological features and disease-free survival of patients. Endocrine journal, 56(1), 89-97.
42. Costa, A. M., Herrero, A., Fresno, M. F., Heymann, J., Alvarez, J. A., Cameselleâ?ÂÂTeijeiro, J., & Garcíaâ?ÂÂRostán, G. (2008). BRAF mutation associated with other genetic events identifies a subset of aggressive papillary thyroid carcinoma. Clinical endocrinology, 68(4), 618-634.
43. Elisei, R., Ugolini, C., Viola, D., Lupi, C., Biagini, A., Giannini, R., ... & Basolo, F. (2008). BRAFV600E mutation and outcome of patients with papillary thyroid carcinoma: a 15-year median follow-up study. The Journal of Clinical Endocrinology & Metabolism, 93(10), 3943-3949.
44. Kim, W. W., Ha, T. K., & Bae, S. K. (2018). Clinical implications of the BRAF mutation in papillary thyroid carcinoma and chronic lymphocytic thyroiditis. Journal of Otolaryngology-Head & Neck Surgery, 47(1), 4.
45. Kim, S. J., Myong, J. P., Jee, H. G., Chai, Y. J., Choi, J. Y., Min, H. S., ... & Youn, Y. K. (2016). Combined effect of
Hashimoto's thyroiditis and BRAFV600E mutation status on aggressiveness in papillary thyroid cancer. Head & neck, 38(1), 95-101.
46. Liu, C., Chen, T., & Liu, Z. (2016). Associations between BRAF V600E and prognostic factors and poor outcomes in papillary thyroid carcinoma: a meta-analysis. World journal of surgical oncology, 14, 1-12.
47. Koperek, O., Kornauth, C., Capper, D., Berghoff, A. S., Asari, R., Niederle, B., ... & Preusser, M. (2012). Immunohistochemical detection of the BRAF V600E-mutated protein in papillary thyroid carcinoma. The American journal of surgical pathology, 36(6), 844-850.