Research Article - (2026) Volume 4, Issue 1
Biomarker- Or Phenotype-Guided Sedation in The Intensive Care Unit: A Systematic Review and Meta-Analysis of Patient-Centered Outcomes
2State of Pará University, Brazil
3Universidad de Costa Rica, Costa Rica. ORCID: 0000-0002-3664-169x, Costa Rica
4Flinders University, Australia
5Frimley Health NHS Foundation Trust, England
6Southwest Kidney Care, USA
7Real Hospital Portugues, Recife, Brasil
8King Edward Memorial Hospital, Pune, India
Received Date: Dec 01, 2025 / Accepted Date: Dec 22, 2025 / Published Date: Jan 15, 2025
Copyright: ©2025 Antonio Andrea Camastra, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Camastra, A. A., Massoud, R.O., Aguilar, P. A., Sriram, K., Amoah, R, et al. (2026). Biomarker- Or Phenotype-Guided Sedation in The Intensive Care Unit: A Systematic Review and Meta-Analysis of Patient-Centered Outcomes. J Future Med Healthcare Innovation, 4(1), 01-08.
Abstract
Introduction: Current practices of sedation in intensive care units (ICUs) are inconsistent, often leading to over- or under-sedation, which are associated with adverse outcomes. Personalized sedation seeks to optimize regimens by incorporating individual characteristics, including pharmacogenomics, electroencephalogram (EEG) patterns, and disease phenotypes. This systematic review and meta-analysis summarizes recent evidence on phenotype- and biomarker- guided sedation in the ICU.
Methods: Eligible studies included randomized controlled trials (RCTs) and observational studies evaluating biomarker- or phenotype-guided sedation with standard care in adult ICU patients. Searches were conducted in PubMed/MEDLINE, Embase, Cochrane CENTRAL, and ClinicalTrials.gov through May 1, 2025. Mean differences (MDs), standardized mean differences (MDs) or risk ratios (RRs) with 95% confidence intervals (CIs) were pooled using random-effects models in RevMan . The I2 statistic was used to calculate heterogeneity.
Results: Five studies met inclusion criteria. Protocolized and biomarker-guided strategies reduced sedative and opioid use, particularly for propofol, midazolam, and morphine. Structured monitoring and analgesia-first approaches were associated with reduced delirium, but no difference for ventilation duration or ICU length of stay. Mortality did not differ significantly, and no increase in adverse events was observed. Conclusion: Biomarker- and phenotype-guided sedation reduces sedative exposure and delirium risk, while remaining safe and without negative impact on mortality.
Keywords
Bispectral Index, Electroencephalography, Sedation and Intensive Care Unit
Introduction
Sedation is a key part of critical care in the intensive care unit (ICU). It helps lower metabolic demands, ease anxiety, make mechanical ventilation easier, and make patients more comfortable. Historically, sedation has depended on generalised dosing and titration informed by clinical scales like the Sedation-Agitation Scale (SAS) or the Richmond Agitation-Sedation Scale (RASS). These tools did help with monitoring, but they don't take into account the wide range of differences in drug metabolism, pharmacodynamics, and underlying conditions. Due to that, patients often get too much or too little sedation, which can lead to bad outcomes. Over-sedation, in particular, has been linked to longer stays in the ICU, a higher risk of delirium, longer ventilation, and a higher death rate.
Precision medicine has become more popular in areas like oncology, cardiology, and infectious diseases. When used in the ICU, this method, called "personalised sedation," takes into account things like pharmacogenomic profiles, electroencephalogram (EEG) patterns, inflammatory states, and disease phenotypes that are unique to each patient. Pharmacogenomics is particularly pertinent, given that numerous sedatives are metabolised by genetically diverse enzymes such as CYP2D6 and CYP3A4. Monitoring based on EEG is another new idea, providing real-time data on brain activity, unlike traditional scales that give subjective and sporadic assessments.
These feedback-driven strategies could improve results by keeping sedation levels more stable. Even though it has potential, challenges make it hard to use it widely. Pharmacogenomic testing has problems with cost, turnaround time, and clinical usefulness. Point-of-care genotyping is not yet common. Many ICUs may not have the infrastructure and expertise needed to do EEG monitoring. Additionally, the diversity within ICU populations and sedation protocols complicates the design and interpretation of studies. This systematic review and meta-analysis seeks to integrate existing evidence regarding phenotype- and biomarker-guided sedation strategies in the ICU.
Methods
This study followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines (17) The protocol was registered in the International Prospective Register of Systematic Reviews (PROSPERO) under registration ID CRD420251061165.
Eligibility Criteria and Outcomes
We included (1) randomized controlled trials (RCTs) and comparative observational studies; (2) enrolling adults (≥18 years) admitted to an ICU and receiving sedation; and (3) comparing sedation strategies informed by phenotypes or biomarkers (e.g., EEG-derived indices, bispectral index [BIS], serum drug concentrations, pharmacogenomic variants) to usual care not guided by phenotypic/biomarker data.
We excluded studies that (1) focused on pediatric populations (<18 years of age), (2) investigated sedation practices unrelated to biomarker or phenotype guidance (e.g., comparisons of sedative agents without personalization), or (3) Focused on or stratified results based on race, ethnicity, or gender-based differences in sedation response, without an accompanying biomarker or phenotypic rationale.
Main outcomes were ICU delirium, invasive mechanical ventilation duration, and ICU length of stay. Additional outcomes included time to awakening, sedation adequacy, adverse events, and hospital mortality.
Search Strategy
We searched PubMed/MEDLINE, Embase, and the Cochrane Central Register of Controlled Trials (CENTRAL) from inception to May 1, 2025. Medical Subject Headings (MeSH) and free-text terms pertaining to biomarker-guided or customised sedation in the intensive care unit (ICU) were combined in the search strategy. No language restrictions were applied during the search, but only studies with full-text available in English were included. Search terms included “personalized sedation,” “pharmacogenomic sedation,” “genotype-guided sedation,” “biomarker-guided sedation,” “phenotype-guided sedation,” “ICU delirium, “sedation,” “EEG biomarkers,” “Bispectral Index,” “BIS monitoring” and “ICU sedation.”
Study Selection and Data Extraction
Following database searches, retrieved records were imported into the Rayyan software (ref to Rayyan). After removal of duplicates, titles and abstracts were screened against eligibility criteria by two independent reviewers (A.A.C., R.O.M.), with consultation with a third reviewer (S.H.) to resolve disagreements. Potentially eligible articles were then reviewed in full-text for final inclusion or exclusion.
Data extracted from included studies included baseline characteristics [patient age, dosing of sedatives and analgesics (propofol, midazoam, fentanyl, and morphine), incidence of delirium, mortality rates, and specific clinical outcomes, including the duration of mechanical ventilation and length of stay in the ICU] and outcomes of interest. Data extraction was conducted in a standardized Excel spreadsheet.
Risk of Bias Assessment
RCTs were assessed with Cochrane Risk of Bias 2 (RoB 2), and non-randomized studies with the Newcastle–Ottawa Scale [1]. RoB 2 evaluates bias across domains related to the randomization process, deviations from intended interventions, missing outcome data, measurement of outcomes, and selection of reported results. The Newcastle–Ottawa Scale (NOS) rates methodological quality based on selection of participants, comparability of study groups, and adequacy of outcome or exposure assessment. Two reviewers (A.A.C. and R.O.M.) assessed risk of bias independently.
Statistical Analysis
All statistical analyses were performed in accordance with PRISMA 2020 recommendations. When at least two studies reported comparable outcomes, data were pooled using a random-effects model to account for anticipated clinical and methodological heterogeneity across ICU populations, sedation protocols, and monitoring strategies. Continuous outcomes (e.g., duration of mechanical ventilation, coma days, propofol infusion rates) were summarized using mean differences (MD) or standardized mean differences (SMD) when measurement scales varied. When studies reported medians and interquartile ranges, values were converted to means and standard deviations using established statistical methods.
Dichotomous outcomes (e.g., incidence of delirium, hospital mortality) were pooled using risk ratios (RRs) with corresponding 95% confidence intervals (CIs). Heterogeneity was assessed using the Cochran Q test and quantified with the I² statistic. Values of 25%, 50%, and 75% were interpreted as low, moderate, and high heterogeneity, respectively. When heterogeneity exceeded 50%, potential sources were explored through sensitivity analyses, including exclusion of outlier studies and restriction to studies at low or moderate risk of bias.
Publication bias was evaluated qualitatively using funnel plot asymmetry when at least 10 studies were available for a given outcome; otherwise, the assessment was considered underpowered. Risk of bias assessments were incorporated into the interpretation of results, and no outcome was pooled when heterogeneity or methodological variability made meta-analysis inappropriate. In such cases, findings were synthesized narratively. All analyses were conducted using Review Manager (RevMan). A two-sided P value < .05 was considered statistically significant for pooled estimates.
Results
Study Selection and Characteristics
Database search retrieved a total of 1,869 records. After removal of duplicates, 1,391 articles were screened by title and abstract, of which 50 were selected for full-text review. Five reports met eligiblity criteria and were included in this systematic review and meta-analysis [2-6]. The study selection process is detailed in Figure 1.
Figure 1: Study Selection Process. PRISMA Flow Diagram
We included a total of 609 patients, of which (50,9%) received personalized sedation and (49,1%) received standard care. Common interventions included minimisation of sedation through individualised titration of propofol, midazolam, and opioids, systematic monitoring using validated instruments such as RASS and SAS, and an analgesia-first strategy that emphasised pain management prior to the escalation of sedation.
Protocols also stressed daily sedation breaks or light sedation goals for easier assessment of neurological function and shortened time spent on mechanical ventilation. Table 1 further describes characteristics of the included studies.
|
Study |
Design |
Population |
Intervention |
Control |
Follow-up |
|
Huespe et al. 2024 |
RCT |
Mechanically ventilated ICU patients |
Protocolized sedation with structured titration of propofol, midazolam, and opioids, guided by sedation scales and multimodal analgesia |
Usual care (clinician-driven sedation and analgesia) |
ICU stay (until extubation or ICU discharge) |
|
Tobar et al. 2022 |
RCT |
Mechanically ventilated ICU patients |
Analgesia-first strategy with protocolized sedation, structured monitoring, and daily sedation interruption |
Standard care without structured protocol |
ICU stay (until extubation or ICU discharge) |
|
Olson et al. 2013 |
RCT |
Critically ill adults requiring mechanical ventilation |
Protocolized sedation minimizing benzodiazepine use and targeting light sedation |
Usual care with clinician-directed sedative administration |
ICU stay (until extubation or ICU discharge) |
|
Weatherburn et al. 2007 |
RCT |
ICU patients requiring sedation |
Protocolized sedation and titration of propofol/ midazolam guided by validated sedation tools |
Conventional sedation practices |
ICU stay (until extubation or ICU discharge) |
|
Alavi et al. 2020 |
Observational cohort |
Adults undergoing elective open-heart surgery |
BIS-guided sedation monitoring |
Sedation guided by GCS and vital signs |
ICU stay (until extubation or ICU discharge) |
Table 1: Characteristics of Included Studies
Sedative and Analgesic Administration
Huespe et al., Tobar et al., and Weatherburn et al. assessed propofol infusion rates, with meta-analysis showing that personalized sedation achieved a significant mean reduction of 0.26 mg/kg/h (95% CI -0.30 to -0.09, p = 0.002, I2 = 20%) in infusion rate compared to standard care (Figure. 2) [7-9].
Figure 2: Propofol Dose (mg/kg/h), Intervention Vs Usual Care
Huespe et al., Olson et al., and Weatherburn et al. assessed midazolam use, finding no significant difference for any midazolam use (RR 0.60, 95% CI 0.28 to 1.30, p = 0.20, I2 = 87%; Figure. 3) or dose (SMD -0.05, 95% CI -1.30 to 1.19, p = 0.93, I2 = 97%; Figure. 4) in pooled analysis [10-12].
Figure 3: Any Midazolam Use, Intervention Vs Usual Care
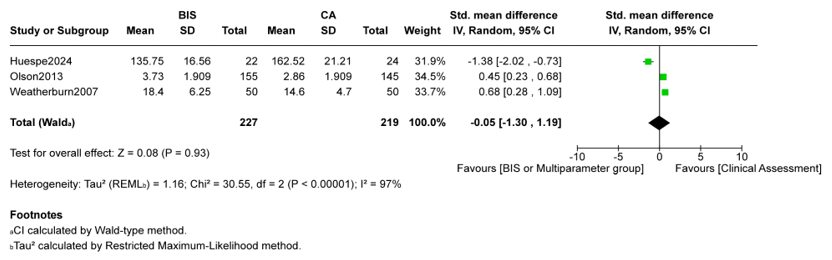
Figure 4: Midazolam Dose (mg/h or mg/day), Intervention Vs Usual Care
Fentanyl dose, as reported in two studies was also not significantly different between groups (MD -0.34 µg/kg/h, 95% CI -1.64 to 0.97,p = 0.61, I2 = 67%; Figure. 5) [13,14].
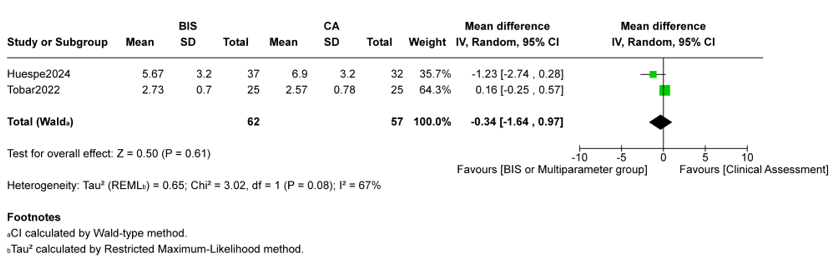
Figure 5: Fentanyl Dose (µg/kg/h), Intervention Vs Usual Carea
Two studies showed that any morphine use was also not significantly different between groups, with mean or median values of 1.79 mg/kg/h to 22.6 mg/day compared to 1.98 mg/kg/h to 25.8 mg/day in controls. This made the pooled difference about−1.3 mg/h. (Figure. 6) [15].
Figure 6: Any Morphine Use, Intervention Vs Usual Care
Delirium
Delirium was reported inconsistently, yet showed consistent decreases in intervention groups. Tobar et al. noted reduced rates of delirium with intervention protocols in contrast to standard care. Likewise, Huespe et al. observed a reduced incidence of delirium in the intervention group, although specific quantitative assessments varied based on diagnostic criteria. Olson et al. and Weatherburn et al. failed to present delirium-specific outcomes, constraining comparability among all studies.
Mortality
Multiple studies reported mortality outcomes. There was no significant difference in all-cause mortality in the ICU or hospital between groups (RR 0.06, 95% CI 0.33 to 1.21, p = 0.17, I2 = 0%; Figure.7) [16].
Figure 7: All-Cause Mortality (ICU or Hospital), Intervention Vs Usual Care
Duration of Mechanical Ventilation
Duration of mechanical ventilation was reported inconsistently. Meta-analysis showed no significant difference between groups for this outcome (MD -0.66 hours, 95% CI -1.92 to 0.61, p = 0.31, I2 = 93%) [17].
ICU Length of Stay
ICU length of stay was not reported in all studies. Tobar et al. noted that patients receiving personalized sedation had shorter ICU length of stay. Conversely, Olson et al. and Weatherburn et al. did not report sufficient detail regarding these endpoints, constraining cross-study comparability (Figure. 8) [18].
Figure 8: Duration of Invasive Mechanical Ventilation (Hours), Intervention Vs Usual Care
Discussion
This systematic review and meta-analysis aimed to assess whether biomarker- or phenotype-guided sedation strategies enhance outcomes in critically ill patients relative to standard care. An examination of five studies, comprising four RCTs and one observational cohort, favoured structured interventions regarding lower exposure to propofol, but showed no significant difference between groups in terms of exposure to other sedatives or opioids, duration of mechanical ventilation or all-cause mortality. Quantitative synthesis was not feasible for ICU length of stay or delirium outcomes, but some studies showed significant results in favor of personalized sedation.
Our findings of reduced propofol exposure suggest that protocol-driven or biomarker-based titration enables clinicians to circumvent unnecessary drug administration. Reducing the sedative burden is clinically significant, as it may mitigate drug accumulation, expedite awakening, and diminish the risk of drug-induced coma. The results concerning delirium may also point to potential benefits of guided sedation, although not all studies included offered comprehensive delirium outcomes. Delirium continues to be a significant complication in critically ill patients, strongly correlated with increased mortality, prolonged hospitalisation, and enduring cognitive impairment. Objective monitoring to keep the right level of sedation seems to stop the cycle of over-sedation and under-sedation, both of which can cause delirium.
Mortality outcomes were consistently neutral across included trials. This finding is not surprising, as mortality in critically ill patients is affected by numerous factors beyond sedation practices. Notably, no harm with structured interventions was reported, suggesting that it is possible to safely lower sedation and aim for lighter levels without putting survival at risk. The outcomes of duration of mechanical ventilation and ICU length of stay exhibited greater variability in reporting, but available evidence indicated that guided sedation strategies may promote earlier extubation.
This study had several limitations. There are still few high-quality RCTs, and it was not feasible to conduct thorough quantitative synthesis for every endpoint due to variations in reporting, patient demographics, sedation techniques, and outcome definitions. The included observational study was naturally susceptible to unmeasured confounding, and although the RCTs were generally of high quality, some did not fully report allocation concealment or handling of missing data. The external validity of the results may be limited because the majority of the included studies were single-center. Furthermore, other potential biomarker- or phenotype-driven strategies, like pharmacogenomics or sophisticated EEG-derived metrics, are understudied because the majority of the evidence base is centered on BIS-guided sedation.
Conclusion
This systematic review and meta-analysis found that biomarker-and phenotype-guided sedation strategies are associated with reduced sedative exposure and a lower incidence of delirium. No significant differences were observed in mortality, ICU length of stay, or duration of mechanical ventilation compared with standard care.
References
- Tobar, E., Farías, J. I., Rojas, V., Penna, A., Egaña, J. I., et al. (2022). Electroencephalography spectral edge frequency and suppression rate–guided sedation in patients with COVID-19: A randomized controlled trial. Frontiers in Medicine, 9, 1013430.
- Huespe, I., Giunta, D., Acosta, K., Avila, D., Prado, E., et al. (2024). Comparing bispectral index monitoring vs clinical assessment for deep sedation in the ICU: Effects on delirium reduction and sedative drug doses—A randomized trial. Chest, 166(4), 733–742.
- Alavi, S. M., Babaee, T., Fard, M. Z., Tirgarfakheri, K., Bourghani-Farahani, E., et al. (2021). Bispectral index monitoring can be an effective method to assess sedation levels after open-heart surgery. Iranian Heart Journal, 22(1).
- Mohamed, M. E., Nguyen, T. T., Larson, J., Schwake, B., Rivers, Z., et al. (2024). Pharmacogenomic variation and sedation outcomes during early intensive care unit admission: A pragmatic study. Clinical and Translational Science, 17(12), e70107.
- Favre, E., Bernini, A., Miroz, J. P., Abed-Maillard, S., Ramelet,A S., et al. (2024). Early processed electroencephalography for the monitoring of deeply sedated mechanically ventilated critically ill patients. Nursing in Critical Care, 29(6), 1781–1787.
- Arbour, R., Waterhouse, J., Seckel, M. A., & Bucher, L. (2009). Correlation between the Sedation–Agitation Scale and the bispectral index in ventilated patients in the intensive care unit. Heart & Lung, 38(4), 336–345.
- Mondello, E., Siliotti, R., Noto, G., Cuzzocrea, E., Scollo, G., et al. (2002). Bispectral index in ICU: Correlation with Ramsay score on assessment of sedation level. Journal of Clinical Monitoring and Computing, 17(5), 271–277.
- Triltsch, A. E., Welte, M., von Homeyer, P., Grosse, J., Genähr, A., et al. (2002). Bispectral index–guided sedation with dexmedetomidine in intensive care: A prospective, randomized, double-blind, placebo-controlled phase II study. Critical Care Medicine, 30(5), 1007–1014
- Türkmen, A., Altan, A., Turgut, N., Vatansever, S., & Gökkaya,S. (2006). The correlation between the Richmond Agitation–Sedation Scale and bispectral index during dexmedetomidine sedation. European Journal of Anaesthesiology, 23(4), 300–304.
- Consales, G., Chelazzi, C., Rinaldi, S., & De Gaudio, A.R. (2006). Bispectral index compared to Ramsay score for sedation monitoring in intensive care units. Minerva Anestesiologica, 72(5), 329–336.
- Tripathi, M., Kumar, V., Kalashetty, M. B., Malviya, D., Bais,P. S., et al. (2017). Comparison of dexmedetomidine and midazolam for sedation in mechanically ventilated patients guided by bispectral index and Sedation–Agitation Scale.Anesthesia: Essays and Researches, 11(4), 828–833.
- Tasaka, C. L., Duby, J. J., Pandya, K., Wilson, M. D., & Hardin, K. A. (2016). Inadequate sedation during therapeutic paralysis: Use of bispectral index in critically ill patients. Drugs – Real World Outcomes, 3(2), 201–208.
- Riess, M. L., Graefe, U. A., Goeters, C., Van Aken, H., & Bone,H. G. (2002). Sedation assessment in critically ill patients with bispectral index. European Journal of Anaesthesiology, 19(1), 18–22.
- Pandharipande, P. P., Pun, B. T., Herr, D. L., et al. (2007). Effect of sedation with dexmedetomidine vs lorazepam on acute brain dysfunction in mechanically ventilated patients: The MENDS randomized controlled trial. JAMA, 298(22), 2644–2653.
- Olson, D. M., Zomorodi, M. G., James, M. L., Cox, C. E., Moretti, E. W., et al. (2014). Exploring the impact of augmenting sedation assessment with physiologic monitors. Australian Critical Care, 27(3), 145–150.
- Weatherburn, C., Endacott, R., Tynan, P., & Bailey, M. (2007). The impact of bispectral index monitoring on sedation administration in mechanically ventilated patients. Anaesthesia and Intensive Care, 35(2), 204–208.
- Page, M. J., McKenzie, J. E., Bossuyt, P. M., Boutron, I., Hoffmann, T. C., et al. (2021). The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ, 372, n71.
- Wells, G. A., Shea, B., O’Connell, D., Peterson, J., Welch, V., et al. (n.d.). The Newcastle–Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ottawa Hospital Research Institute.