Research Article - (2023) Volume 1, Issue 1
Biochemistry Characterization of Type 2 Diabetes in Tunisia and their Relationship with 4545G/C Polymorphism Located in Adiponectin Gene
2University of Tunis El Manar, Faculty of Medicine of Tunis, LR99ES09 Laboratory of Antimicrobial Res, Tunisia
Received Date: Apr 12, 2023 / Accepted Date: May 15, 2023 / Published Date: Jun 05, 2023
Copyright: ©Â©2023 Hajer Kilani, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Kilani, H., Kallel, A., Jemaa, R. (2023). Biochemistry Characterization of Type 2 Diabetes in Tunisia and their Relation-ship with 4545G/C Polymorphism Located in Adiponectin Gene. Transl Med OA, 1(1), 09-16.
Abstract
Diabetes incidence is increasing worldwide. Many studies demonstrated that polymorphisms within the adiponectin gene could be associated with type 2 diabetes mellitus (T2DM). A case–control study was conducted to find the association between SNP 4545G/C and T2DM in Tunisian population. The study included 561 patients referring to diabetic and 437 healthy con- trols. DNA was extracted from blood and genotyped by PCR–RFLP by using (HinfI) enzyme. Prevalence of obesity (p <0.001), dyslipidemia (p <0.001), hypertension (p <0.001) were significantly increased in cases with report to witnesses. Also for the lipid parameters Urea, CRP and HDL-C were significantly (p <0.001) and HDL / LDL (p = 0.983) are decreased in diabetics compared to controls two types of alleles G (Absence of restriction site) and C (presence of the restriction site) respectively of frequencies (0.76 and 0.23) in the controls and (0.73 and 0.26) in the diabetics, as well as three genotypes GG, GC and CC In comparison with the GG genotype the relative risk (OR [95% CI]) for diabetes was 1.06 [0.86-1.31]; p = 0.275 for the genotypes (GC + CC) . Our results show significant variations for the anthropometric and lipid parameters in the type 2 diabetes mellitus (T2DM) but were not associated with the polymorphisms at site of 4545.
Keywords
Diabetes, Adiponectin, Genetic Variants, LDL, HDL
Introduction
Type 2 diabetes (T2D) involves a complex interaction between genetic variants and environmental factors, and obesity can increase the risk of developing diabetes [1]. Adiponectin(also calledAPM1,ACRP30, ADIPOQ, and GBP28) is an abundant adipocyte-secreted protein in plasma. It can regulate glucose levels, insulin action and lipid metabolism [5-6]. The circulat- ing adiponectin was significantly reduced in patients with type 2 diabetes and obesity [7,8]. Prospective studies showed that subjects with high adiponectin levels were protected against type 2 diabetes [9]. Moreover, administration of recombinant adiponectin decreased glucose and insulin resistance in mice of obesity or diabetes [6-10]. The plasma level of adiponectin is partly influenced by genetic factors which account for about 40–70% [11]. The human adiponectin gene is mapped to the 3q27 region where the metabolic syndrome and T2D loci were reported [12-14].
Some studies have found that the single nucleotide polymor- phisms (SNPs) of this gene might increase the risk of T2D [15- 17], whereas few reported the relationship between its SNPs and type 2 diabetes combined with obesity. As far as we know, there are only two teams who reported these associations with obese T2D. For example, +45 T/G and +276 G/T single nucleo- tide polymorphisms (SNPs) of adiponectin (ADIPOQ, 3q27) are highly involved SNPs as candidate risk variants for T2DM in Asian populations, whereas these same SNPs are not correlated with a risk of T2DM in Europeans, including Italian, French and Swedish individuals [18-21].
The 4545G/C polymorphism was chosen because they might in- fluence the gene expression or change the protein function. cho- lesterol (TC), triglyceride (TG), high density lipoprotein (HDL) and low density lipoprotein (LDL) were determined using an automatic chemistry analyzer (Olympus AU5400, Japan) or an enzymatic kit (Roche Diagnostics GmbH, Basel, Switzerland). Fasting plasma glucose (FPG) was measured by a glucose ox- idase or hexokinase reaction, and fasting serum insulin (Fins) was assayed by means of electro-chemiluminescence immuno- assay (ECLIA, Roche Diagnostics, Rotkreuz, Switzerland). The homeostasis model assessment (the HOMA) indices for insulin resistance (HOMA-IR) and for beta-cell function (HBCI) were subsequently calculated by using the following formulas: HO- MA-IR = Fins (mU/L) _ FPG (mmol/L)/ 22.5 and HBCI = 20 _ Fins (mU/L)/[FPG (mmol/L) _ 3.5], respectively.
The objective of this study is to evaluate the biochemical analy- ses in type 2 diabetic subjects and non-diabetic subjects and re- search the relationship between 4545G/C polymorphism located in adiponectin gene and type 2 diabetes in Tunisia.
Materials and Methods
Study Population In this study, a total of 1034 subjects were recruited, consisting of 561 type 2 diabetic subjects (mean age 55,44±9,15 years) and 473 non-diabetic subjects (mean age 54,35±9,35 years) at the research Unit in the Department of Endocrinology of Rabta Uni- versity Hospital of Tunis. The study population (561 subjects) was recruited from diabetic patients consulting in the endocrinology department of the Rabta hospital.
A group of controls (473 subjects) was recruited as part of a prospective survey on "Biochemical and genetic markers of ath- erosclerosis in the Tunisian population"
-A standardized questionnaire was carried out for all subjects comprising:
- Information relating to the subjects’s personal and family his- tory, their risk factors and their drug consumption.
- Part of the questionnaire is reserved for patients aiming to char- acterize diabetes and complications.
Biochemical Analyses
Assay of Biological Parameters
The blood samples were in the morning after 12 hours on EDTA tubes for the genetic study and lithium heparin for the lipid bal- ance By using a standard enzymatic methods with commercial kits (Roche Diagnostics, Mannheim, Germany) on a Hitachi 912 analyzer. The serum was separated immediately from the pelled centrifugation at 3000 rpm for 15 minutes at 4°C. All the lip- id parameters (total cholesterol,HDL cholesterol, triglycerides were assayed on ARCHITECT ci 8200 (Abbott). It allows the assays tobe carried out by: UV- visible spectrophotometry for TG, CT and HDL. LDL-C concentrations were calculated using Friedwald’s formula : LDL= CT- (TG/ 5 + HDL) [22].
Statistical Analysis
All statistical analyses were conducted using the SPSS 11.5 (SPSS Inc., Chicago, IL, USA) statistical package. For all tests performed, a p value of less than 0.05 was considered as signif- icant. Data are presented as means ± SD. Skewed continuous variables such as triglyceride including age, BMI was evaluated by multiple linear regression analysis, In order to explore the different genetic backgrounds of type 2 diabetes, we tried to gen- otype the SNP (4545G/C) in the adipose most abundant gene transcript-1 (APM1) gene in 561 type 2 diabetes (T2D) patients and 473 non-diabetic subjects by PCR–RFLP.).
Adiponectin Genotyping
DNA Extraction (DNA Concentration and Purity Asses- sement)
A dilution of 1/50 of the DNA extract by adding 50 µl of the DNA solution in 950 µl of double-distilled water is carried out. A first reading of the optical density (OD) at 260mm makes it possible to assess the DNA concentration of the sample, 1 unit OD 260 nm -> 50 µg / ml of double-stranded DNA. DNA concentration µg / ml = OD value * 50 * dilution factor Asecond reading of the OD at 280 nm allows the protein con- tamination to be estimated and the calculation of the OD at 260 / OD at 280 ratio allows the purity of the DNA to be assessed. A DNA is considered pure when this ratio is between 1.8 and 2. The optical density is read by a conventional spectrophotometer. Thus, depending on the value of the report, possible contami- nants such as proteins or RNAs are detected:

Results
Description of the Study Population
• Anthropometric Parameters of the Studied Population Prevalence of obesity (p <0.001), dyslipidemia (p <0.001), hy- pertension (p <0.001) were significantly increased in cases with report to witnesses (table 1).
|
|
Witnesses controls (N =473) |
Case Diab (N =561) |
P |
|
Age (Years) |
55,44±9,15 |
54,35±9,35 |
0,425 |
|
BMI |
27,86±5,55 |
30,24±5,35 |
<0,001* |
|
TAS |
122,04±19,06 |
134,32±20,09 |
<0,001* |
|
TAD |
71,84±12,817 |
78,30±12,22 |
<0,001* |
|
Obesity (%) |
31,8 |
46,6 |
<0,001* |
|
Dyslipidemia (%) |
18,3 |
35,7 |
<0,001* |
|
HTA (%) |
16 |
54,1 |
<0,001* |
|
Smoking (%) |
80,1 |
83,2 |
0,117 |
Comparison of anthropometric characteristics between diabetic and control groups.
* P < 0.05.
Table 1: Anthropometric characteristics of the population
• Lipid Parameters of the Population
Comparison of lipids between the two groups studied did show a statistically significant increase in HDL-C and Urea, CRP are (p <0.001) in diabetics compared to controls (table 2).
|
Settings Organic |
Witnesses CONTROL (N = 473) |
Diab (N=561) |
P |
|
Gly |
0,91±0,12 |
1,77±0,77 |
<0,001* |
|
CT (mmol / l) |
1,89±0,38 |
1,9±0,41 |
0,928 |
|
TG (mmol / l) |
1,22±0,79 |
1,3±0,72 |
0,029 |
|
C-HDL (mmol / l) |
0,49±0,13 |
0,42±0,12 |
<0,001* |
|
LDL-C (mmol / l) |
1,21±0,37 |
1,2±0,36 |
0,895 |
|
Urea (g / l) |
0,299±0,08 |
0,33±0,17 |
<0,001* |
|
CRP (mg / ml) |
3,68±7,19 |
6,25±6,43 |
<0,001* |
|
CREAT (g) |
9,00±1,72 |
9,19±4,60 |
0,419 |
|
CT / C-HDL |
4,19±1,98 |
4,92±3,42 |
<0,001* |
|
HDL / LDL |
0,44±0,212 |
0,44±0,516 |
0,983 |
Comparison of lipid parameters between diabetic and control groups.
* P < 0.05.
Table 2: Average values of lipid parameters in the population studied
• Allelic Frequencies and Distribution of Genotypes
Using the RFLP method (PCR, enzymatic digestion and agarose gel electrophoresis) we were able to determine the genotypes of our subjects, and demonstrate two types of alleles G (Ab- sence of restriction site) and C (presence of the restriction site) respectively of frequencies (0.76 and 0.23) in the controls and (0.73 and 0.26) in the diabetics, as well as three genotypes GG, GC and CC (figure 1), the frequencies of which are given in the table below.
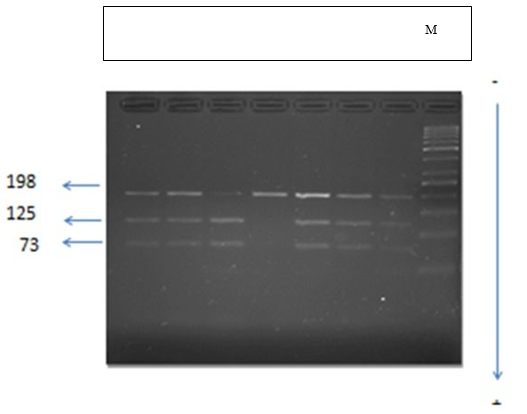
Figure 1: Electrophoretic profile of restriction fragments by the HinfI enzyme in a 3% agarose gel which identify 3 genotypes:
CC: a single 198 bp band. It is the wild homozygous form.
CA: 3 bands of 198bp, 125bp and 73bp. This is the heterozygous form.
AA: 2 bands of 125 bp and 73 bp. This is the homozygous mutated form.
M: mark size (50-1000 bp).
In our population, the genotypic distribution is in Hardy-Weinberg equilibrium. Our results show that the frequency of the G allele is not significantly increased in diabetics compared to controls (table 3).
|
|
WITNESS CONTROL (N= 473) |
Diab (N= 561) |
P |
OR (95% CI) |
||
|
N |
% |
N |
% |
|||
|
Genotypic frequencies |
||||||
|
GG |
286 |
60,5 |
312 |
55,6 |
|
|
|
GC |
154 |
32.6 |
202 |
36 |
|
|
|
CC |
33 |
7,0 |
47 |
8,4 |
|
|
|
GC + CC |
187 |
39,6 |
249 |
44,4 |
0,275 |
1,06(0,86-1,31) |
|
Allelic frequencies |
||||||
|
G |
0,76 |
|
0,73 |
|
0,565 |
|
|
C |
0,23 |
|
0,26 |
|
|
|
GG: Wild homozygous; GC: Heterozygous; CC: Mutated homozygote
Table 3: Allelic frequencies and genotypic distribution of the -4545 G / C polymorphism in cases (diab) and controls.
In comparison with the GG genotype the relative risk (OR [95% CI]) for diabetes was 1.06 [0.86-1.31]; p = 0.275 for the genotypes (GC + CC) (Table 3).
• Lipid and Anthropometric Parameters According to Genotypes
Depending on the genotypes, our results do not show significant variations for the anthropometric and lipid parameters in the con- trols (table 4).
|
Genotypes |
GG |
GC |
CC |
P |
||||
|
Anthro- pometric parameters |
control |
case |
control |
case |
control |
case |
control |
case |
|
Age |
55±9,24 |
55,24±9,30 |
55,87±8,95 |
54,98±9,31 |
55,56±9,60 |
53,28±9,85 |
0,765 |
0,408 |
|
BMI |
27,85±5,57 |
30,17±5,80 |
27,48±4,93 |
30,32±4,70 |
29,66±7,45 |
30,39±4,95 |
0,163 |
0,935 |
|
Heap |
121,11±19,62 |
133,87±21,39 |
122,11±19,17 |
133,36±17,90 |
130 ,00±17,32 |
139,35±19,87 |
0,753 |
0,209 |
|
Tad |
70,00±12,63 |
77,89±12,55 |
73,68±13,82 |
78,27±11,69 |
76,67±5,77 |
81,20±12,16 |
0,512 |
0,232 |
|
Lipid parameters |
||||||||
|
Gly |
0,91±0,11 |
1,67±0,71 |
0,91±0,15 |
1,67±0,73 |
0,96±0,12 |
- |
0,091 |
0,128 |
|
CT |
1,90±0,36 |
2,05±1,64 |
1,90±0,36 |
1,91±0,41 |
1,86±0,44 |
- |
0,873 |
0,132 |
|
TG |
1,20±0,79 |
1,26±0,61 |
1,23±0,77 |
1,36±0,7 |
1,31±0,92 |
- |
0,739 |
0,957 |
|
C-HDL |
0,49±0,14 |
0,45±0,17 |
0,49±0,12 |
0,46±0,28 |
0,52±0,15 |
- |
0,442 |
0,155 |
|
C-LDL |
1,24±0,33 |
1,37±1,6 |
1,20±0,36 |
1,18±0,36 |
1,07±0,59 |
- |
0,359 |
0,295 |
|
Urée |
0,29±0,07 |
0,34±0,13 |
0,30±0,08 |
0,31±0,12 |
0, 31±0,08 |
- |
0,243 |
0,974 |
|
CRP |
3,80±8,56 |
5,64±5,41 |
3,47±4,01 |
6,06±4,65 |
3,57±5,27 |
- |
0,9 |
0,950 |
|
CREAT |
8,95±1,61 |
9,8±6,1 |
9,08±1,74 |
8,7±3,14 |
9,07±2,34 |
- |
0,749 |
0,262 |
|
CT/HDL |
4,15±1,30 |
5,07±4,79 |
4,27±2,74 |
4,71±1,67 |
4,11±2,64 |
- |
0,803 |
0,844 |
|
HDL/LDL |
0,42±0,19 |
0,43±0,62 |
0,45±0,23 |
0,54±0,76 |
0,45±0,22 |
- |
0,202 |
0,535 |
Table 4: Average values of anthropometric and lipid parameters as a function of genotypes in controls (N=473) and in cases (Diab) (N=561)
Similarly, in diabetics, no variation in biological parameters de- pending on genotypes has been observed.
Discussion
The aim of our study was to determine in a Tunisian population the allelic and genotypic frequencies of the 4545G / C polymor- phism of the adiponectin gene and its possible association with biological parameters on the one hand and with type 2 diabetes on the one hand somewhere else. Most previous studies exam- ining the association of ADIPOQ and T2DM were conducted on Caucasian, Asian, and African populations [23-25], The allelic and genotypic frequencies of our population are in Hardy Wein- berg equilibrium. Our results show that the allele frequencies of the G and C alleles of the 4545G / C polymorphism of the ad- iponectin gene are 0.73 and 0.26 in diabetics, respectively, and 0.76 and 0.23 in controls, respectively. This result is contradic- tory with the result of Xizhen et al 2009 (26) with frequencies of the G and C alleles respectively in diabetics 0.68 and 0.31 and in controls 0.65 and 0.35. The 4545G / C polymorphism of the adiponectin gene is not associated with type 2 diabetes in our population. Our results are contradictory to those found in a Chinese population who described variant C as a predisposing marker for type 2 diabetes [26], The frequency of the C allele in our diabetic population (0.26) is comparable to those described in the literature in the Sweden population [27]. Several studies have investigated the effect of the 4545G / C polymorphism of the adiponectin gene on variation in biological parameters and BMI in the population.
Comparison of lipids between the two groups studied did not show a statistically significant increase in CT (p = 0.928) in cases compared to controls. As well as Prevalence of obesity, dyslip- idemia, hypertension were significantly increased in cases with report to witnesses. Urea, CRP and HDL-C were increased in diabetics compared to controls. Our results show no significant variation in biological parameters as a function of the 4545G / C polymorphism in both cases and in diabetics and are in agree- ment with Harvest results in a Sweden population [26,27]. Our study show similar result with the study of Xizhen et al in 2009, which showed that HDL levels are significantly lower in dia- betic subjects compared to controls. This disagreement between the different studies can be explained by the ethnic origin of the populations and / or by gene-gene and gene-environment inter- actions.
Regarding the association of the 4545G / C polymorphism with BMI, our results are consistent with the work published so far in the literature [26]. Variation in adiponectin expression has been reported to be involved in insulin resistance and therefore in the development of type 2 diabetes. In our subjects, adiponectin de- creases insignificantly depending on the mutated C allele. It has been shown that androgens decrease plasma adiponectin levels (Arita et al., 1999) 28. Given the probable role of adiponectin in the regulation of mRNA stability or post-translational modi- fications which are of great importance in the biological activi- ties of adiponectin [26]. exon 3 mutation intervention in type 2 diabetes [26–29],. Xizhen et al suggests that the modification of adiponectin at the β-cell level may be an important factor in the development of insulin resistance and type 2 diabetes [30]. An SNP 4545G / C of exon 3 of the adiponectin gene has been identified, this polymorphism is due to a change that affects the structure of the protein by replacing the following amino ac- ids guanine by cytosine at position 4545 which is found in the variable region of the protein, this is explained by the presence of several uradylates and adenylates in and downstream of the RNA sequences. It potentiates the hypoglycemic effects of in- sulin in peripheral tissues, mainly in the liver and muscle, and modulates food intake in the central nervous system.
The absence of association of the 4545G / C polymorphism of the adiponectin gene with this pathology in our population could be explained as follows: at the level of exon 3, the repetitive se- quences in lover and downstream of mRNA n 'have no direct in- fluence on variable regions [26], therefore could not be involved in the modification of the protein conformation at the level of skeletal muscle and β cells (Chan et al., 2005; Zhou et al., 2004) [31,32] to affect its activity. Such as other study suggest that aer- obic exercise affects adiponectin levels regardless of weight loss and this effect would not be influenced by SNP45 and SNP276 in the adiponectin gene [33].
Conclusion
In summary, the current study provides evidences that prevalence of obesity, dyslipidemia, hypertension and the lipid parameters Urea, CRP and HDL-C were significantly were significantly in- creased in cases with report to witnesses. The polymorphisms at site of 4545 are not associated with type 2 diabetes, respectively. But these should be validated with more comprehensive and in- formative data and their mechanisms how the variants increase the risk of T2D should be clarified in further studies.
Acknowledgments
This work was supported by a grant from “The Ministry of High- er Education, Scientific Research and Technology” of Tunisia.
Funding: This study was funded by the authors.
Author Contributions
Hajer Kilani wrote original draft manuscript and conducted for- mal analysis; AK con-ducted and validated formal analysis; AK gave an idea and conducted data analysis AK conducted data analysis and correction of context; HK corrected in the context and gave an idea; RJ conceptualized and supervised whole man- uscript. All authors have read and approved the final manuscript
Declarations
Competing interests
The authors declare that they have no competing interests.
References
- Mokdad, A. H., Ford, E. S., Bowman, B. A., Dietz, W. H., Vinicor, F., Bales, V. S., & Marks, J. S. (2003). Prevalence of obesity, diabetes, and obesity-related health risk factors, 2001. Jama, 289(1), 76-79.
- Ahima, R. S., Saper, C. B., Flier, J. S., & Elmquist, J. K. (2000). Leptin regulation of neuroendocrine systems. Fron- tiers in neuroendocrinology, 21(3), 263-307.
- Trayhurn, P., & Beattie, J. H. (2001). Physiological role of adipose tissue: white adipose tissue as an endocrine and se- cretory organ. Proceedings of the Nutrition Society, 60(3), 329-339.
- Holst, D., & Grimaldi, P. A. (2002). New factors in the reg- ulation of adipose differentiation and metabolism. Current opinion in lipidology, 13(3), 241-245.
- Berg, A. H., Combs, T. P., Du, X., Brownlee, M., & Scherer,P. E. (2001). The adipocyte-secreted protein Acrp30 enhanc- es hepatic insulin action. Nature medicine, 7(8), 947-953.
- Berg, A. H., Combs, T. P., & Scherer, P. E. (2002). ACRP30/ adiponectin: an adipokine regulating glucose and lipid me- tabolism. Trends in Endocrinology & Metabolism, 13(2), 84-89.
- Hotta, K., Funahashi, T., Arita, Y., Takahashi, M., Matsuda, M., Okamoto, Y., ... & Matsuzawa, Y. (2000). Plasma con- centrations of a novel, adipose-specific protein, adiponec- tin, in type 2 diabetic patients. Arteriosclerosis, thrombosis, and vascular biology, 20(6), 1595-1599.
- Arita, Y., Kihara, S., Ouchi, N., Takahashi, M., Maeda, K., Miyagawa, J. I., ... & Matsuzawa, Y. (1999). Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity. Biochemical and biophysical research communica- tions, 257(1), 79-83.
- Spranger, J., Kroke, A., Möhlig, M., Bergmann, M. M., Ri-stow, M., Boeing, H., & Pfeiffer, A. F. (2003). Adiponectin and protection against type 2 diabetes mellitus. The Lancet, 361(9353), 226-228.
- Tsao, T. S., Lodish, H. F., & Fruebis, J. (2002). ACRP30, a new hormone controlling fat and glucose metabolism. Euro- pean journal of pharmacology, 440(2-3), 213-221.
- Comuzzie, A. G., Funahashi, T., Sonnenberg, G., Martin, L. J., Jacob, H. J., Black, A. E. K., ... & Kissebah, A. (2001). The genetic basis of plasma variation in adiponectin, a glob- al endophenotype for obesity and the metabolic syndrome. The Journal of Clinical Endocrinology & Metabolism, 86(9), 4321-4325.
- Vionnet, N., Dupont, S., Gallina, S., Francke, S., Dotte, S., De Matos, F., ... & Froguel, P. (2000). Genomewide search for type 2 diabetes–susceptibility genes in French Whites: evidence for a novel susceptibility locus for early-onset di- abetes on chromosome 3q27-qter and independent replica- tion of a type 2–diabetes locus on chromosome 1q21–q24. The American Journal of Human Genetics, 67(6), 1470- 1480.
- Kissebah, A. H., Sonnenberg, G. E., Myklebust, J., Gold- stein, M., Broman, K., James, R. G., ... & Comuzzie, A. G. (2000). Quantitative trait loci on chromosomes 3 and 17 in- fluence phenotypes of the metabolic syndrome. Proceedings of the National Academy of Sciences, 97(26), 14478-14483.
- Mori, Y., Otabe, S., Dina, C., Yasuda, K., Populaire, C., Le- coeur, C., ... & Froguel, P. (2002). Genome-wide search for type 2 diabetes in Japanese affected sib-pairs confirms sus- ceptibility genes on 3q, 15q, and 20q and identifies two new candidate Loci on 7p and 11p. Diabetes, 51(4), 1247-1255.
- Kishida, K., Nagaretani, H., Kondo, H., Kobayashi, H., Tanaka, S., Maeda, N., ... & Matsuzawa, Y. (2003). Dis- turbed secretion of mutant adiponectin associated with the metabolic syndrome. Biochemical and Biophysical Re- search Communications, 306(1), 286-292.
- Vasseur, F., Helbecque, N., Dina, C., Lobbens, S., Delan- noy, V., Gaget, S., ... & Froguel, P. (2002). Single-nucleotide polymorphism haplotypes in the both proximal promoter and exon 3 of the APM1 gene modulate adipocyte-secret- ed adiponectin hormone levels and contribute to the genetic risk for type 2 diabetes in French Caucasians. Human mo- lecular genetics, 11(21), 2607-2614.
- Hara, K., Boutin, P., Mori, Y., Tobe, K., Dina, C., Yasuda, K., ... & Kadowaki, T. (2002). Genetic variation in the gene encoding adiponectin is associated with an increased risk of type 2 diabetes in the Japanese population. Diabetes, 51(2), 536-540.
- Mohammadzadeh, G., & Zarghami, N. (2009). Associations between single-nucleotide polymorphisms of the adiponec- tin gene, serum adiponectin levels and increased risk of type 2 diabetes mellitus in Iranian obese individuals. Scan- dinavian Journal of Clinical and Laboratory Investigation, 69(7), 764-771.
- Li, L. L., Kang, X. L., Ran, X. J., Wang, Y., Wang, C. H.,Huang, L., ... & Mao, X. M. (2007). Associations between 45T/G polymorphism of the adiponectin gene and plasma adiponectin levels with type 2 diabetes. Clinical and Exper- imental Pharmacology and Physiology, 34(12), 1287-1290.
- Gu, H. F., Abulaiti, A., Ostenson, C. G., Humphreys, KWahlestedt, C., Brookes, A. J., & Efendic, S. (2004). Single nucleotide polymorphisms in the proximal promoter region of the adiponectin (APM1) gene are associated with type 2 diabetes in Swedish caucasians. Diabetes, 53(suppl_1), S31-S35.
- Kraja, A. T., Rao, D. C., Weder, A. B., Mosley, T. H., Turn-er, S. T., Hsiung, C. A., ... & Province, M. A. (2005). An evaluation of the metabolic syndrome in a large multi-eth- nic study: the Family Blood Pressure Program. Nutrition & metabolism, 2(1), 1-13.
- Gable, D. R., Matin, J., Whittall, R., Cakmak, H., Li, K. W., Cooper, J., ... & HIFMECH investigators. (2007). Common adiponectin gene variants show different effects on risk of cardiovascular disease and type 2 diabetes in European sub- jects. Annals of human genetics, 71(4), 453-466.
- Wang, X., Zhang, S., Chen, Y., Liu, H., Lan, C., Chen, X., ... & Zhang, W. (2009). APM1 gene variants− 11377C/G and 4545G/C are associated respectively with obesity and with non-obesity in Chinese type 2 diabetes. diabetes research and clinical practice, 84(3), 205-210.
- Han, L. Y., Wu, Q. H., Jiao, M. L., Hao, Y. H., Liang, L.B., Gao, L. J., ... & Sun, H. (2011). Associations between single-nucleotide polymorphisms (+ 45T> G,+ 276G> T,− 11377C> G,− 11391G> A) of adiponectin gene and type 2 diabetes mellitus: a systematic review and meta-analysis. Diabetologia, 54, 2303-2314.
- Wang, X., Zhang, S., Chen, Y., Liu, H., Lan, C., Chen, X., ... & Zhang, W. (2009). APM1 gene variants− 11377C/G and 4545G/C are associated respectively with obesity and with non-obesity in Chinese type 2 diabetes. diabetes research and clinical practice, 84(3), 205-210..,
- Gu, H. F. (2009). Biomarkers of adiponectin: plasma pro- tein variation and genomic DNA polymorphisms. Biomark- er insights, 4, BMI-S3453.
- Arita, Y., Kihara, S., Ouchi, N., Takahashi, M., Maeda, K., Miyagawa, J. I., ... & Matsuzawa, Y. (1999). Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity. Biochemical and biophysical research communica- tions, 257(1), 79-83.
- Vasseur, F. (2003, August). The genetics of adiponectin. In International Congress Series (Vol. 1253, pp. 37-44). Else- vier.
- Hotta, K., Funahashi, T., Arita, Y., Takahashi, M., Matsuda, M., Okamoto, Y., ... & Matsuzawa, Y. (2000). Plasma con- centrations of a novel, adipose-specific protein, adiponec- tin, in type 2 diabetic patients. Arteriosclerosis, thrombosis, and vascular biology, 20(6), 1595-1599.
- Chan, D. C., Watts, G. F., Ng, T. W., Uchida, Y., Sakai, N., Yamashita, S., & Barrett, P. H. R. (2005). Adiponectin and other adipocytokines as predictors of markers of triglycer- ide-rich lipoprotein metabolism. Clinical chemistry, 51(3), 578-585.
- Zhou, H., Song, X., Briggs, M., Violand, B., Salsgiver, W., Gulve, E. A., & Luo, Y. (2005). Adiponectin represses gluconeogenesis independent of insulin in hepatocytes. Biochemical and biophysical research communications, 338(2), 793-799.
- Lee, K. Y., Kang, H. S., & Shin, Y. A. (2013). Exercise im- proves adiponectin concentrations irrespective of the adi- ponectin gene polymorphisms SNP45 and the SNP276 in obese Korean women. Gene, 516(2), 271-276.



