Research Article - (2025) Volume 10, Issue 3
Assessment of the Impact of Epidural Anesthesia on Postpartum Hemorrhage Risk in Vaginal Delivery: A Retrospective Study
2Anesthesiology, Grand Hôpital de Charleroi, Belgium
Received Date: Jun 10, 2025 / Accepted Date: Aug 11, 2025 / Published Date: Sep 29, 2025
Copyright: ©Â©2025 Thomas Botti, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Botti, T., Zec, T., Lenoir, N., Vermoezen, A., Lerberghe, C. V., et al. (2025). Assessment of the Impact of Epidural Anesthesia on Postpartum Hemorrhage Risk in Vaginal Delivery: A Retrospective Study. J Anesth Pain Med, 10(3), 01-07.
Abstract
Background: Post-partum hemorrhage is a major complication of childbirth. Obstetric epidurals are often implicated in the occurrence of post-partum hemorrhage. Given the paradoxical data in the literature, we aim to retrospectively evaluate the impact of epidural on the risk of post-partum hemorrhage, instrumental delivery and the occurrence of risk factors for post- partum hemorrhage.
Material and Methods: We retrospectively analyzed 5753 records of patients with vaginal deliveries at the Brugmann University Hospital between January 1, 2020, and December 31, 2021. Our primary objective was to evaluate whether the epidural constitutes a risk of post-partum hemorrhage. Secondly, we assessed whether epidurals increase the risk of childbirth instrumentation and the emergence of risk factors for post-partum hemorrhage.
Results: A total of 4143 patients were included in our study. No significant difference was observed between the "epidural" and "no epidural" groups regarding postpartum blood loss (340.72mL ± 410.05 vs 354.14mL ± 371.08; p=0.2996). The "epidural" group had a higher rate of instrumental delivery (11.23% vs 6.99%; p<0.001) and obstetric trauma (33.93% vs 29.41%; p=0.0029). The rate of coagulopathy was higher in the " no epidural" group (1.51% vs 5.79%; p<0.001). No significant difference was observed for uterine atony (5.89% vs 5.79%; p=0.8939) and tissue abnormalities (7.15% vs 8.59%; p=0.1099).
Conclusion: Epidural anesthesia is not associated with an increased risk of post-partum hemorrhage in patients who have undergone vaginal delivery. However, it is associated with a higher rate of instrumental delivery and obstetric trauma.
Keywords
Epidural Anesthesia, Instrumental Delivery, Obstetrical Analgesia, Obstetrical Trauma, Neuraxial Anesthesia, Postpartum Hemorrhage
Introduction
Postpartum hemorrhage (PPH) is one of the leading causes of maternal morbidity and mortality worldwide, complicating approximatively 1 to 10% of deliveries [1,2]. It is one of the complications that most concerns anesthesiologists and obstetricians and is also the leading cause of intensive care unit admission during or after childbirth [3,4]. Since 2017, PPH is defined as cumulative blood loss greater than or equal to 1000mL or blood loss associated with signs and/or symptoms of hypovolemia within 24 hours of delivery according to American College of Obstetricians and Gynecologists (ACOG) guidelines [4]. The main etiologies responsible for PPH can be summarized by the “Four T’s”: atony (Tone), tissue abnormality (Tissue), trauma (Trauma), coagulopathy (Thrombin) [5].
Pain during labor is a factor affecting maternal satisfaction, regardless of the delivery method [6]. Epidural analgesia remains the gold standard for obstetric analgesia [7]. Although available in certain countries, its effectiveness and maternal safety have been solidly established compared to other methods [8]. Many women seek effective pain relief during labor and delivery, and various medical conditions warrant the use of anesthesia during these procedures (for example: preeclampsia, multiple pregnancy, morbid obesity, previous cesarean section) [9,10].
Epidural anesthesia has been proposed as a potential risk factor for PPH [11,12]. This is explained by two mechanisms. First, epidural anesthesia is known to prolong the second stage of labor, potentially leading to an increased risk of instrumentation and cesarean section, although certain randomizes controlled trials and meta-analyses do not confirm this risk [8,9]. Second, epidural anesthesia induces vasodilation of the uteroplacental vessels, suggesting its administration may constitute a potential risk factor for PPH [13].
While the link between anesthesia type and PPH incidence is currently unclear, some studies show no impact of obstetric epidural anesthesia on PPH risk in vaginal delivery, while others indicate an increased incidence [11,14,15]. Considering these paradoxical data, we retrospectively assessed the impact of epidural anesthesia on the risk of postpartum hemorrhage in vaginal delivery. The primary aim of this study was to determine if epidural anesthesia is a risk factor for PPH in patients who delivered vaginally. The secondary aim was to assess if epidural anesthesia is associated with a higher risk of instrumental delivery and the emergence of the PPH risk factors (4Ts).
Additionally, the ongoing prospective Peri-HPP study seeks to provide a more detailed assessment of the association between epidural anesthesia and postpartum hemorrhage risk following vaginal delivery. Comparing our retrospective findings with prospective data from Peri-HPP will help clarify the influence of epidural anesthesia on PPH in this context. While the present study aimed to perform an initial retrospective analysis to identify potential associations and generate hypotheses, the Peri-HPP study will offer more robust evidence for future research.
Materials & Methods
Data Collection
Following approval from the local ethics committee (CE 2023/208), we conducted a retrospective single-center study at the Brugmann University Hospital maternity ward. We reviewed 5,753 records of patients who delivered between January 1, 2020, and December 31, 2021.
Inclusion criteria:
- Patients who delivered vaginally at Brugmann University Hospital, Horta site, between January 1, 2020, and December 31, 2021.
- Adults patients (age ≥ 18 years).
Exclusion criteria:
- Minors (age < 18 years).
- Patients who delivered before 25 weeks GA (Gestational age).
- Patients who delivered outside of the hospital.
- Patients who underwent a medical termination of pregnancy.
- Patients who delivered via cesarean section.
Throughout their obstetric care, patients receive comprehensive information on the various anesthesia options to better prepare for their delivery. In the third trimester of pregnancy, patients are provided with an explanatory sheet and a consent form for epidural use by the medical team. On the day of delivery, patients can choose to accept or decline the epidural option. If they choose to proceed, an anesthesia consultation is scheduled, during which the anesthesiologist conducts a targeted history and clinical examination, especially to identify signs of coagulation disorders. A blood test is systematically performed for all patients at least one month before the day of delivery, along with urine analysis if necessary. Furthermore, a coagulation test is performed within 24 hours before delivery or if the INR is below 1.3 in the following conditions: Von Willebrand disease, coagulation factor deficiency, preeclampsia, intrauterine fetal death, or liver impairment. The anesthesiologist also checks for obstetric history and other contraindications, such as allergies to local anesthetics, skin infections at the injection site, increased intracranial pressure, uncompensated hypovolemia, coagulation disorders or patient refusal. Epidurals were administered according to the hospital's maternity protocol (Appendix A).
We collected demographic data from the patients, delivery data (mode of delivery, number of fetuses, instrumentation vacuum- assisted (V-A), presence of gestational diabetes, presence of polyhydramnios, and Preterm premature rupture of membranes (PPROM)), neuraxial anesthesia parameters (type of local anesthetic used, its concentration, puncture level), blood loss, and recognized PPH risk factors such as uterine atony, placental abnormalities, trauma, and coagulopathy. All vaginal tears, regardless of their degree (D1 to D4), were grouped together during data collection. Therefore, no distinction was made between the different degrees of tears in the analyses presented. The type of uterine atony was identified from the delivery report. Thus, we collected data from pre-delivery blood tests (hemoglobin and hematocrit, platelets, PT, INR, aPTT, and fibrinogen). Our laboratory’s normal aPTT values range from 21.6 to 28.7 seconds. Macrosomia was defined as a birth weight over 4000 grams [16].
At Brugmann University Hospital, blood loss is analyzed according to a standardized protocol. During a vaginal delivery, a calibrated surgical drape and a precision scale for weighing compresses are used to accurately estimate blood loss.
To ensure confidentiality and facilitate centralization, data collection was performed on the RedCap-Brugmann platform.
Statistical Analysis
A propensity score was calculated between two groups: those without epidural anesthesia (n = 1488) and those with epidural anesthesia (n = 2655). After imputing missing values in the database 15 times using the R package "mice," the R package "CBPS" was employed to calculate the propensity score by estimating an Average Treatment Effect (ATE), which requires exact matching between groups [17]. An absolute standardized mean difference below 15% indicates balance between the groups on selected covariates, which allows comparison across variables measured in different units [18]. After propensity score matching, the R "survey" package was used to compare the two groups on primary and secondary outcomes using linear regressions for continuous outcomes and logistic regressions for binary outcomes to obtain a doubly robust treatment effect estimator [19]. The "survey" package includes Huber-White corrected standard errors, which maintain unbiased standard errors even in cases of heteroscedastic residuals [20]. A p-value was considered significant if less than 0.05 for the primary outcome and less than 0.01 for the secondary outcomes following the Bonferroni correction.
The within-imputation approach, which averages treatment effects across the 15 logistic/linear regressions, has been shown to produce less biased estimators compared to the across-imputation approach, which applies logistic/linear regressions on the datasets averaged after multiple imputations [21]. R version 4.1.2 (R Core Team, 2021) was used for statistical analysis.
Results
We examined the records of 5753 patients who delivered at Brugmann University Hospital between January 1, 2020, and December 31, 2021. Out of these, we included 4143 patients in our study based on inclusion and exclusion criteria (Figure 1).
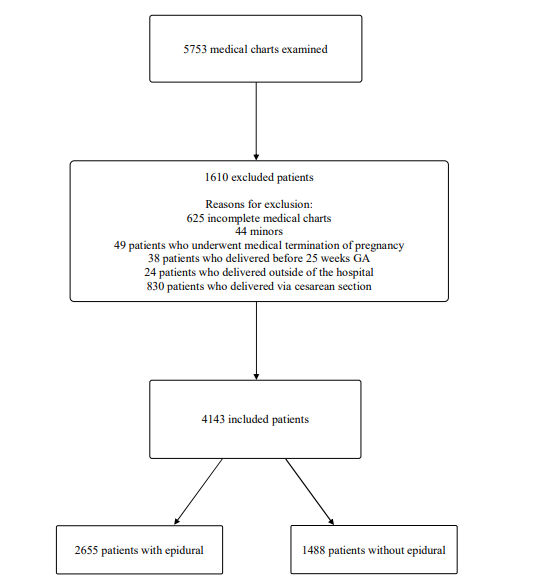
Figure 1: Flow Chart of the Study
Before matching, the two groups (with and without epidural anesthesia) were imbalanced on several covariates, as some SMDs exceeded the 15% threshold regarding demographic data. Compared to women who did not receive epidural anesthesia, those who did had fewer pregnancies and viable births and were more likely to have labor induced. The most significant difference concerned, among other factors, the number of pregnancies (SMD = 34.85%) and viable births (SMD = 42.90%), followed by labor induction (SMD = 17.64%). After propensity score matching, the SMD for all covariates was reduced to well below the 15% threshold, indicating successful group balancing (Table 1).
|
|
Before matching |
After matching |
||||
|
|
No epidural (n = 1488) |
Epidural (n = 2655) |
SMD |
No epidural (n = 1488) |
Epidural (n = 2655) |
SMD |
|
Age (year) |
30.8 (5.6) |
30.1 (5.4) |
12.9% |
30.4 (5.6) |
30.4 (5.4) |
0.01% |
|
Weight (kg) |
71.6 (14.6) |
72.7 (15.3) |
7.7% |
72.37 (15) |
72.4 (15.1) |
0.02% |
|
Height (cm) |
163.7 (6.4) |
163.9 (6.4) |
3.2% |
163.9 (6,4) |
163.9 (6.6) |
0.02% |
|
BMI (kg/m2) |
26.7 (5.2) |
27 (5.4) |
6.6% |
26.9 (5,3) |
26.9 (5.4) |
0.00% |
|
Gravida (number) |
3 (1.8) |
2.5 (1.6) |
34.8% |
2.7 (1,6) |
2.7 (1.8) |
0.02% |
|
Para (number) |
1.6 (1.3) |
1 (1.1) |
42.9% |
1.2 (1,2) |
1.2 (1.3) |
0.03% |
|
Gestational age (GA) |
38.9 (1.8) |
39 (1.5) |
7.8% |
38.9 (1,8) |
38.9 (1.5) |
0.02% |
|
Labor induction |
14.3% |
21% |
17.6% |
18.7% |
18.7% |
0.02% |
|
Parturients with multiple gestation |
0 |
0 (0,1) |
11.4% |
1 (0,08) |
1 (0.1) |
0.03% |
|
Newborn weight (g) |
3317.1 (519.4) |
3344.8 (486) |
5.5% |
3337.2 (503,2) |
3337.2 (488.6) |
0.00% |
|
Preexisting diabetes |
0.7% |
1.2% |
4.4% |
1% |
1% |
0.05% |
|
Gestational diabetes |
21.6% |
23.2% |
4% |
22.4% |
22.4% |
0.00% |
|
Uterine fibroma |
1.1% |
1.4% |
1.9% |
1.2% |
1.2% |
0.00% |
|
History of uterine surgery |
7.2% |
9.2% |
7.4% |
8.5% |
8.5% |
0.04% |
|
History of c-section |
6% |
7.6% |
6.5% |
7% |
7% |
0.02% |
|
C-section number |
0.06 (0.3) |
0.1 (0.3) |
5% |
0.1 (0.3) |
0.1 (0.3) |
0.01% |
|
History of PPH |
4.6% |
4.4% |
0.8% |
4.6% |
4.6% |
0.01% |
|
Preeclampsia |
1.6% |
2% |
3.4% |
1.9% |
1.9% |
0.02% |
|
IVF-ICSI |
1% |
1.4% |
3.5% |
1.2% |
1.2% |
0.02% |
|
Polyhydramnios |
1.3% |
1.6% |
2.6% |
1.5% |
1.3% |
0.00% |
|
PPROM |
1.3% |
1% |
2% |
1.1% |
1.1% |
0.01% |
|
aContinuous variables are expressed as mean ± standard deviation. Dichotomous variables are expressed as percentages. bSMD = Standardized mean difference. An SMD of less than 15% will be considered as indicating balance between groups on the chosen covariates. IVF: In vitro fertilization ICSI: Intracytoplasmic sperm injection |
||||||
Table 1: Comparison of Demographic Data between the "No Epidural" Group and the "Epidural" Group Before and After Propensity Score Matching. (CHU Brugmann, 2020–2021)
In our study population, 2655 patients (64%) received an epidural during vaginal delivery. Our results showed no significant difference between the group with epidural anesthesia and the group without it in terms of postpartum blood loss (Figure 2)
However, we found a statistically significant difference regarding the variables related to our secondary objective, particularly concerning the rates of instrumentation V-A, trauma, and coagulopathy (Table 2). Patients who received an epidural were more likely to undergo instrumental delivery compared to those without an epidural (11.23% vs 6.99%; p < 0.001).
We also observed that the incidence of trauma was higher in patients with an epidural compared to those without (33.93% vs 29.41%; p = 0.0029). Tears were the most common cause of trauma (90.8%) (Table B.1, Appendix B). Conversely, the rate of coagulopathy was lower in the group of patients who received an epidural (1.51% vs 5.79%; p < 0.001). Abnormal aPTT was the most prominent feature of coagulopathy (91.9%) (Table B.3, Appendix B).
Nevertheless, no significant difference was found between the two groups regarding the risk of developing uterine atony or tissue abnormalities (Table 2).
aThe p-value is considered significant if it is less than 0.05
Figure 2: Comparison of Postpartum Blood Loss Between the Group of Patients without Epidural and the Group of Patients with Epidural
|
Variables |
No epidural (n = 1488) |
Epidural (n = 2655) |
P |
|
Uterine atony |
5,8% |
5,9% |
0.9 |
|
Tissue Abnormalities |
8.6% |
7.15% |
0.1 |
|
Trauma |
29.4% |
33.9% |
0.003 |
|
Coagulopathy |
5.8% |
1.5% |
<0.001 |
|
Instrumental Delivery V-A |
7% |
11.2% |
<0.001 |
|
aFive comparisons are performed. We apply the Bonferroni correction by dividing the p-value (0.05) by the number of comparisons: 0.05/5 = 0.01. bThe p-value is considered significant if it is less than 0.01. |
|||
Table 2: Comparison of Variables Analyzed in our Study Population between the Non-Epidural and Epidural Group
Discussion
The objective of this retrospective study was to evaluate whether epidural anesthesia represents a risk factor for postpartum hemorrhage (PPH) in patients who delivered vaginally. We analyzed the impact of epidural anesthesia on postpartum blood loss, as well as the risk of instrumental V-A delivery and the emergence of the main PPH risk factors (“Four T’s”). According to our findings, there is no significant difference between patients with and without epidural anesthesia in terms of postpartum blood loss. However, we observed a significant difference in the rates of instrumental V-A delivery, trauma, and coagulopathy, suggesting a potential role of epidural anesthesia in these obstetric complications.
First, regarding the amount of blood loss, Borovac-Pinheiro et al. found that epidural anesthesia did not increase the risk of bleeding within 24 hours following vaginal delivery, which is consistent with our findings [13]. However, their study included only women in labor beyond 34 weeks of gestation, and coagulopathy was an exclusion criterion. Nevertheless, our findings are also aligned with the study by Rossen et al., spanning 10 years and including 41365 deliveries, which suggests that epidural analgesia does not increase the risk of PPH [12].
Few studies address the relationship between epidural anesthesia and the risk of postpartum bleeding during vaginal delivery. Most of the currently available studies consider cesarean deliveries, which was an exclusion criterion in our study population. The absence of a significant difference in postpartum blood loss between the two groups of our study supports the idea that the use of epidural anesthesia can be considered a safe option for pain management and hemorrhage control during childbirth and vaginal delivery.
Our secondary objective was initially to determine whether epidural anesthesia increased the risk of instrumental V-A delivery. Physiological changes triggered by epidural anesthesia, such as reduced maternal expulsive efforts, are primarily observed during the second stage. The effects of epidural anesthesia depend primarily on the concentration of the anesthetic solution, but also on the type of local anesthetic used and potential adjuncts [22, 23]. Previously, the use of higher doses, such as bupivacaine concentrations >0.1%, was standard practice, which could induce motor block and lead to an increased need for instrumentation [23, 24]. A meta-analysis of 11 studies validated the benefits of using a reduced-concentration local anesthetic solution, demonstrating a decrease in instrumental extractions without compromising the effectiveness of analgesia [24].
A systematic review also showed that early initiation of epidural analgesia is not associated with higher rates of cesarean sections or instrumental vaginal deliveries [25]. However, this review only included women with gestational ages over 36 weeks, spontaneous or induced labor, single pregnancies, and cephalic presentations. In contrast, our study included a broader population, with gestational ages above 25 weeks and varying numbers of fetuses, without specifying presentation type.
In our population, the epidural group had a higher risk of instrumental V-A delivery compared to the non-epidural group (11.23% vs 6.99%). The obstetric team may be more inclined to use instruments for women with epidural anesthesia to prevent maternal fatigue, shorten labor time, and avoid cesarean delivery. Furthermore, ineffective coordination between the patient and medical staff may also play a role, particularly due to language barriers, which are present in a proportion of patients delivering at our institution.
Trauma to the genital tract is the most common complication of obstetric trauma [9]. In our study population, we observed a significant difference between the epidural and non-epidural groups (33.93% vs 29.41%). Myrick et al. reported a negative association between epidural analgesia and vaginal tears, which contradicts our findings [26]. However, their study also noted that instrumental delivery is strongly associated with an increased risk of tears, which aligns with our results [26]. It is reasonable to infer that the rate of trauma, particularly tears which are the most common type in our study increases in the group with higher rates of instrumental V-A delivery.
Coagulopathy is a relatively rare cause of PPH [27]. We observed a significant difference between the epidural and non-epidural groups (1.51% vs 5.79%). This discrepancy can be explained by several factors. First, the threshold values for coagulation tests, such as aPTT, vary between laboratories, potentially leading to an overestimation of incidence. Second, given that coagulopathy is a relative contraindication for epidural anesthesia, it is reasonable to expect higher rates in the non-epidural group [28]. Additionally, some patients may exhibit prolonged aPTT values even without a history suggesting coagulation disorders. In such cases, anesthesiologists may proceed with epidural placement after carefully weighing the risks and benefits in cases of mild to moderate coagulopathy [29].
Finally, the use of epidural anesthesia did not result in a significant difference in the rates of uterine atony or tissue abnormalities in our study population. However, a study by Driessen et al. on a cohort of 4,550 patients with uterine atony after vaginal delivery suggests that the use of epidural anesthesia reduces postpartum bleeding, likely due to more effective management enabled by the epidural catheter, which allows precise pain control, continuous or adjustable anesthetic administration, rapid conversion to surgical anesthesia if needed, and effective intervention for postpartum hemorrhage or invasive procedures without requiring additional anesthesia [30].
Our study has certain limitations. First, selection bias may be present due to the retrospective nature of our analysis. Retrospective data collection exposes the study to information bias stemming from incomplete or inaccurate medical records. While propensity score matching helps adjust for pre-existing differences, it cannot fully account for unmeasured variables that may influence the results, such as anticoagulant use, the duration of anesthesia, or the administration of additional lidocaine boluses, which were challenging to evaluate. All vaginal tears, from D1 to D4, were grouped together during data collection, which limits the analysis as it does not account for differences in clinical severity.
To address these limitations, the ongoing prospective Peri-HPP study aims to provide a more comprehensive evaluation of the relationship between epidural anesthesia and PPH risk following vaginal delivery. We did not analyze epidural anesthesia as an independent risk factor for PPH because PPH risk factors were considered secondary outcomes. Additionally, the diverse population at our hospital may have introduced linguistic barriers, potentially hindering the smooth progression of labor and delivery. Our study also has several notable strengths. First, the large sample size provides high statistical power and reflects a diverse population in terms of age, medical history, and obstetric characteristics. Second, propensity score matching effectively balanced the groups based on demographic and clinical covariates, making the comparisons between the epidural and non-epidural groups more robust. Third, the study addressed multiple objectives, including the impact of epidural anesthesia on PPH in vaginal delivery, the rate of instrumental delivery V-A, and the main risk factors for PPH. Finally, our results align with existing literature while highlighting discrepancies that generate new research hypotheses.
Conclusion
This retrospective study indicates that epidural anesthesia is not associated with an increased risk of PPH in vaginal delivery, although it is associated with an increased risk of instrumental V-A delivery and obstetric trauma. By comparing our retrospective findings with the ongoing prospective data from Peri-HPP, we aim to establish a clearer understanding of the potential impact of epidural anesthesia on postpartum hemorrhage, particularly in the context of vaginal deliveries.
References
- Bláha, J., & Bartošová, T. (2022). Epidemiology and definition of PPH worldwide. Best practice & research Clinical anaesthesiology, 36(3-4), 325-339.
- Feduniw, S., Warzecha, D., Szymusik, I., & Wielgos, M. (2020). Epidemiology, prevention and management of early postpartum hemorrhage—a systematic review. Ginekologia polska, 91(1), 38-44.
- Gonzalez-Brown, V., & Schneider, P. (2020, October). Prevention of postpartum hemorrhage. In Seminars in Fetal and Neonatal Medicine (Vol. 25, No. 5, p. 101129). WB Saunders.
- Hemorrhage, P. (2017). Practice bulletin no. 183. American College of Obstetricians and Gynecologists. Obstet Gynecol, 130, e168-81.
- Bienstock, J. L., Eke, A. C., & Hueppchen, N. A. (2021). Postpartum hemorrhage. New England Journal of Medicine, 384(17), 1635-1645.
- Clivatti, J., Siddiqui, N., Goel, A., Shaw, M., Crisan, I., & Carvalho, J. C. (2013). Quality of labour neuraxial analgesia and maternal satisfaction at a tertiary care teaching hospital: a prospective observational study. Canadian Journal of Anesthesia/Journal canadien d'anesthésie, 60(8), 787-795.
- Halliday, L., Nelson, S. M., & Kearns, R. J. (2022). Epidural analgesia in labor: A narrative review. International Journal of Gynecology & Obstetrics, 159(2), 356-364. https://doi. org/10.1002/ijgo.14175
- Animâ?Somuah, M., Smyth, R. M., Cyna, A. M., & Cuthbert,A. (2018). Epidural versus nonâ?epidural or no analgesia for pain management in labour. Cochrane database of systematic reviews, (5).
- American College of Obstetricians and Gynecologists. (2017). Practice bulletin no. 177: Obstetric analgesia and anesthesia. Obstet Gynecol, 129(4), e73-e89.
- Jones, L., Othman, M., Dowswell, T., Alfirevic, Z., Gates, S., Newburn, M., ... & Neilson, J. P. (2012). Pain management for women in labour: an overview of systematic reviews. Cochrane database of systematic reviews, (3).
- Lisonkova, S., Mehrabadi, A., Allen, V. M., Bujold, E., Crane, J. M., Gaudet, L., ... & Joseph, K. S. (2016). Atonic postpartum hemorrhage: blood loss, risk factors, and third stage management. Journal of Obstetrics and Gynaecology Canada, 38(12), 1081-1090.
- Srebnik, N., Barkan, O., Rottenstreich, M., Ioscovich, A., Farkash, R., Rotshenker-Olshinka, K., ... & Grisaru- Granovsky, S. (2020). The impact of epidural analgesia on the mode of delivery in nulliparous women that attain the second stage of labor. The Journal of Maternal-Fetal & Neonatal Medicine, 33(14), 2451-2458.
- Ginosar, Y., Nadjari, M., Hoffman, A., Firman, N., Davidson,E. M., Weiniger, C. F., ... & ACET study group. (2009). Antepartum continuous epidural ropivacaine therapy reduces uterine artery vascular resistance in pre-eclampsia: a randomized, dose-ranging, placebo-controlled study. British journal of anaesthesia, 102(3), 369-378.
- Rossen, J., Økland, I., Nilsen, O. B., & Eggebø, T. M. (2010). Is there an increase of postpartum hemorrhage, and is severe hemorrhage associated with more frequent use of obstetric interventions?. Acta obstetricia et gynecologica Scandinavica, 89(10), 1248-1255.
- Borovac-Pinheiro, A., Brandão, M. J. N., Argenton, J. L. P., Barbosa, T. D. A., & Pacagnella, R. C. (2022). Anesthesia technique and postpartum hemorrhage: a prospective cohort study. Brazilian Journal of Anesthesiology, 72(3), 338-341.
- Júnior, E. A., Peixoto, A. B., Zamarian, A. C. P., Júnior, J. E., & Tonni, G. (2017). Macrosomia. Best practice & research Clinical obstetrics & gynaecology, 38, 83-96.
- Imai, K., & Ratkovic, M. (2015). Robust estimation of inverse probability weights for marginal structural models. Journal of the American Statistical Association, 110(511), 1013-1023.
- Austin, P. C. (2011). An introduction to propensity score methods for reducingthe effects of confoundinginobservational studies. Multivariate behavioral research, 46(3), 399-424.
- Funk, M. J., Westreich, D., Wiesen, C., Stürmer, T., Brookhart,M. A., & Davidian, M. (2011). Doubly robust estimation of causal effects. American journal of epidemiology, 173(7), 761-767.
- Lin, W. (2013). Agnostic notes on regression adjustments toexperimental data: Reexamining Freedman's critique. TheAnnals of Applied Statistics, 295-318.
- Mitra, R., & Reiter, J. P. (2016). A comparison of two methods of estimating propensity scores after multiple imputation. Statistical methods in medical research, 25(1), 188-204.
- Aveline, C., Bonnet, F. (2001). Influence of Epidural Anesthesia on the Duration and Modes of Delivery. French Annals of Anesthesia and Resuscitation.20(5):471-84.
- Stalla-Bourdillon, A., Moyano, G., Levenet, S., Hoffmann, C., & Mercier, F. J. (2015). Influence of epidural analgesia on obstetric mechanics in long time. ANESTHESIE & REANIMATION, 1(2), 167-172.
- Sultan, P., Murphy, C., Halpern, S., & Carvalho, B. (2013). The effect of low concentrations versus high concentrations of local anesthetics for labour analgesia on obstetric and anesthetic outcomes: a meta-analysis. Canadian Journal of Anesthesia/Journal canadien d'anesthésie, 60(9), 840-854.
- Wassen, M. M., Zuijlen, J., Roumen, F. J., Smits, L. J. M., Marcus, M. A., & Nijhuis, J. G. (2011). Early versus late epidural analgesia and risk of instrumental delivery in nulliparous women: a systematic review. BJOG: An International Journal of Obstetrics & Gynaecology, 118(6), 655-661.
- Myrick, T. G., & Sandri, K. J. (2018). Epidural analgesia and any vaginal laceration. The Journal of the American Board of Family Medicine, 31(5), 768-773.
- James, A. H., Grotegut, C., Ahmadzia, H., Peterson-Layne, C., & Lockhart, E. (2016, October). Management of coagulopathy in postpartum hemorrhage. In Seminars in thrombosis and hemostasis (Vol. 42, No. 07, pp. 724-731). Thieme Medical Publishers.
- Avila Hernandez, A. N., & Singh, P. (2024). Epidural anesthesia. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing.
- Zec, T., Schmartz, D., Temmerman, P., Fils, J. F., Ickx, B., Bonhomme, F., & Van Der Linden, P. (2024). Assessment of haemostasis in pregnant women: A retrospective evaluation of the diagnostic performance of the HEMSTOP standardised questionnaire. European Journal of Anaesthesiology and Intensive Care, 3(2), e0050.
- Driessen, M., Bouvier-Colle, M. H., Dupont, C., Khoshnood, B., Rudigoz, R. C., & Deneux-Tharaux, C. (2011). Postpartum hemorrhage resulting from uterine atony after vaginal delivery: factors associated with severity. Obstetrics & Gynecology, 117(1), 21-31.



