Research Article - (2025) Volume 4, Issue 1
Assessment of Basella Alba on Chemical Induced Hepatocellular Carcinoma in Rats
22Department of Pharmacology, NBRI (CSIR), Ranapratap Marg, India
3Department of Pharmacology, MCOPS, Manipal, India
Received Date: Nov 17, 2025 / Accepted Date: Dec 22, 2025 / Published Date: Dec 31, 2025
Copyright: ©2025 Gaddam Dayanand Reddy, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Reddy, G. D., Karthik, R., Ojha, S. K., Rao, C. V., Unnikrishnan, M., et al. (2025). Assessment of Basella Alba on Chemical Induced Hepatocellular Carcinoma in Rats. Insights Herbal Med, 4(1), 01-08.
Abstract
Chemical hepatotoxins including diethylnitrosamine (NDEA) and carbon tetrachloride (CCl4) frequently induce hepatocellular carcinoma (HCC), which is a leading cause of cancer-related deaths globally. Conventional therapy is still limited due to toxicity and poor prognosis, generating renewed interest in natural products as safer alternatives. Basella alba (B. alba) an edible leafy vegetable that has long been used in folk medicine, includes flavonoids, phenolics, carotenoids, and saponins, all of which have strong antioxidant properties. B. alba extract was administered to rats with NDEA + CCl4-induced HCC for 16 weeks. Biochemical, oxidative stress, and histological markers were assessed. The extract effectively restored blood liver function indices, reduced lipid peroxidation, increased antioxidant enzyme activity, and improved hepatic architecture by lowering necrosis and fibrosis. These data show that B. alba has substantial hepatoprotective and chemopreventive effect against chemically generated HCC, most likely due to its antioxidant and membrane-stabilizing capabilities. The findings support its traditional use and emphasise its potential as a functional food and phytotherapeutic agent for liver cancer, necessitating additional mechanistic and clinical research.
Keywords
Basella alba, Hepatocellular Carcinoma, NDEA, CCl4 and Oxidative Stress
Introduction
Hepatocellular carcinoma (HCC) represents the most common primary malignancy of the liver and is a leading cause of cancer- related deaths globally [1,2]. Its development is multifactorial, often associated with chronic inflammation, oxidative stress, and hepatotoxic exposures, including aflatoxins, viral hepatitis, and chemical carcinogens [3,4]. Experimental hepatocarcinogenesis can be reliably reproduced in rodents using agents such as N-nitrosodiethylamine (NDEA), diethylnitrosamine (DEN), and carbon tetrachloride (CCl4), which mimic the molecular and pathological features of human HCC. The NDEA/DEN models initiate DNA alkylation and mutagenesis, while CCl4 acts as a promoter of fibrosis and cirrhosis through reactive oxygen species (ROS) generation and lipid peroxidation [5,6].
Oxidative stress and chronic inflammation play a crucial role in HCC growth, causing dysregulation of redox-sensitive transcription factors including NF-κB and Nrf2, activation of pro- inflammatory cytokines (TNF-α, IL-6), and suppression of tumour suppressor genes like p53 [7,8]. As a result, chemopreventive techniques targeting these processes using natural antioxidants and phytochemicals are gaining popularity [9].
Experimental studies have shown that B. alba extracts have hepatoprotective, anti-inflammatory, anti-proliferative, and wound-healing activities [10]. Importantly, its antioxidant-rich nature implies that it may be effective against oxidative stress- induced cancers such as hepatocellular carcinoma [11].
Given the scarcity of in vivo research evaluating B. alba against liver cancer, the current study sought to examine its chemopreventive effects in a chemically produced rat model of HCC. The study examined biochemical, antioxidant, histological, and molecular data to determine the hepatoprotective and anti- carcinogenic processes.
Materials and Methods
Plant Material
Preparation of Plant Extract
Basella alba plant material was collected from the Botanical Garden of the National Botanical Research Institute in Lucknow, India (NBRI). Dr.SayeedaKhatoon, a taxonomist, confirmed the plant material, and the voucher specimens (NAB2B04293, 200495, and 200492) were archived in the departmental herbarium and the Institutional Museum for future reference. B. alba fresh plant material was rinsed with distilled water to remove dirt and soil.
B. alba powder (100g) was air-dried (30±20c) and extracted overnight with 10 ml of ethanaol. It was then centrifuged at 10000 rev/min on a Rota evaporator (Bouchi, USA) and dried in a lyophilizer (Labeonco, USA) under reduced pressure. The extract was then subjected to phytochemical and pharmacological testing.
HPTLC Analysis
Reflux 5 g of finely powdered medication with 25 ml of ethanol over a water bath for 25 minutes three times in a row, then filter and extract the solvent under low pressure. Dissolve 25 milligrammes of extractive in 20 millilitres of ethanol. Using an automator applicator (CAMAG Linomat IV), apply 10 l of extract on Merck percoated silica gel 60 F254 plates with a 0.2mm thickness. The plates were then run using a fresh solvent system (ethyl acetate). Formic acid: Acetic acid. Water in a 100:11:11:27 ratio for Basella alba in a CAMAG twin through chamber up to a distance of approximately 9 cm, dry, and scan. UV 254 and visible light were used to view the plates. If necessary, spray the plate with anisaldehyde-sulphuric acid.If necessary, spray the plate with anisaldehyde-sulphuric acid and heat at 110°C for 10 minutes. Record the Rf values and colour of the resolved bands, then use the Desaga video documentation unit to document the movie. The plates were densitometrically scanned with a CAMAG TLC scanner at the relevant wavelength [12].
In Vivo Study
Animals
Studies are carried out on rats weighing between 140 and 160 g. They were obtained from the toxicity control animal house at the Central Drug Research Institute in Lochnow, as well as cattle raised in the departmental animal facility. The rats were housed in polyacrylic cages with no more than 6 animals per cage and kept under typical laboratory conditions (temperature 25±20C, 12h dark/light cycle). They have free access to a standard dry pellet meal (amrut, India) and unrestricted tap water. The institutional committee for the ethical use of animals reviewed and approved all of the disclosed procedures.
Induction of Hepatocellular Carcinoma
The rats were randomly assigned to the experimental and control groups (n = 6). Group 1 rats were given 0.9% normal saline. Group II rats with chemically induced HCC (NDEA+CCl4) were given a single intraperitoneal injection of N-nitroso di ethyl amine (200 +3 mg/kg b.w.s.c). Group III rats received 100 mg/kg b.w.i.p. of B. alba in 50% ethanol (EtoH). Groups IV and V were given i.p injections of 200 and 400 mg/kg/b.w, respectively, whereas Group VI rats received 6 mg/kg b.w of 50% EtoHB. alba.
Laboratory Investigations
Body weights were measured on the day of receipt, before randomisation, on the day of dosing, and weekly thereafter for the treatment and recovery groups, while food and drink consumption were documented daily and reported weekly. All animals had their blood drawn for haematology and clinical biochemistry. Animals were placed in metabolic cages and fasted overnight before blood sample, but they had unlimited access to water. Blood was drawn from the retroorbital plexus using a micro-hematocrit heparinised glass capillary tube.
Potassium EDTA was employed as an anticoagulant during haematology tests. Blood samples were stored in serum tubes at room temperature for around 30 minutes before being aliquoted. After clotting, the blood tubes were centrifuged at 3000 rpm for 15 minutes. The supernatants were decanted and stored at 700°C for subsequent analysis.
Biochemical Marker Estimation
The biochemical markers like SGOT(U/l), SGPT(U/l), SALP(U/l), Bilirubin level (U/l) and Gamma glutamyltranspeptidase, GGT (U/l) were determined for both control and treated groups by using standard biochemical method [13].
Estimation of Free Radical Generation
The liver homogenate (5%) in ice-cold phosphate buffer was centrifuged at 800X g for 10 minutes, followed by centrifugation of the supernatant at 12,000X g for 15 minutes to obtain the mitochondrial fractions, which were used to measure lipid peroxidation (LPO), superoxide dismutase activity (SOD), catalase activity (CAT), reduced glutathione (GSH), and glutathione peroxidase (GPX) [14].
Hematological Estimation
Red blood cell counts, White blood cell counts and hemoglobin were estimated with the help of hematology analyzer (Medonic CA620, Boule, Sweden) [13].
Histopathological Analysis
The livers were taken immediately upon autopsy for histological analysis, and the tissues were preserved in 10% formalin for at least 24 hours. The paraffin sections were subsequently processed (Automatic Tissue Processor, Lipshaw) and sliced into 5 um thick sections using a rotary microtome. The sections were then stained with haematoxilin-eosin dye (Merck) and mounted in Canada balasam. The histopathology slides were inspected and photographed using a picture zoom microscope (3.2X10 and 10X10) [15].
Statistical Analysis
Data are expressed as mean +SEM (standard error of mean). The difference among means has been analyzed by unpaired students t-test.
Results
HPTLC Analysis of 50% EtOH Extract of B. alba B. alba
TLC plates with a 50% EtOH extract were visible at UV 254 nm. The plates were coated with anisaldehyde-sulfuric acid and then heated to 1100 degrees Celsius for 10 minutes. A band (Rf = 0.51), colour of the resolved bands, and video were documented. The plates were densitometrically scanned using a CAMAG TLC scanner at 254 nm (Figure 1).
Formic acid(5:5:1)
Figure 1: HPTLC finger print of ethanolic extract of B. alba - EtOAcToluene: EtOAc: Formic acid (5:5:1)
The plant was extracted with 50% EtOH, producing a percentage yield of 6.26 (Table 1).
|
peak |
Start Rf |
Height % |
End Rf |
Area % |
|
1 |
0.12 |
17.03 |
0.14 |
12.93 |
|
2 |
0.15 |
23.99 |
0.16 |
14.48 |
|
3 |
0.16 |
13.41 |
0.20 |
11.47 |
|
4 |
0.23 |
11.67 |
0.30 |
11.52 |
|
5 |
0.33 |
11.26 |
0.36 |
10.62 |
|
6 |
0.36 |
11.39 |
0.40 |
11.05 |
|
7 |
0.50 |
12.98 |
0.57 |
13.30 |
|
8 |
0.57 |
17.84 |
0.68 |
13.44 |
|
9 |
0.68 |
24.19 |
0.81 |
35.74 |
|
10 |
0.82 |
18.05 |
0.87 |
15.49 |
|
11 |
0.87 |
18.21 |
0.95 |
19.04 |
|
12 |
0.96 |
18.73 |
0.103 |
19.40 |
|
13 |
0.98 |
32.26 |
1.10 |
31.53 |
Table 1: HPTLC Values
Effect of 50% ethanolic extract of B. albaon body weight, liver weight and average liver weight in (NDEA+CCl4) induced HCC rat
50% ethanolic extracts of B. alba, at a dose of 100, 200 mg and 400 mg once daily for 28 days and standard Cisplatin at a dose of 6 mg/kg were subjected for studying the body weight, liver weight and average liver weight in HCC rats. The study showed that the liver weights were significantly increased from 19.3,15.33 to 22.8,15.78 in NDEA+CCl4 group to 21.1,15.36 to 19.8, 10.66 in B. albatreated animal. whereas, standard drug Cisplatin 6 mg/ kg showed significant reduction in liver weight compared to NDEA+CCl4 group figure 2.
*Values are mean of 6 rats in each group
Figure 2: Effect of 50% ethanolic extract of B. albaon body weight, liver weight and average liver weight in (NDEA+CCl4) induced HCC rat
Effect of 50% ethanolic extract of B. albaon SGOT, SGPT, SALP, BL and GGT against NDEA+CCl4 induced HCC
In the NDEA+CCl4 group, the level of SGOT (202.20-364.21, P<0.001), SGPT (93.21-381.53, P<0.001), SALP (244.14-449.31, P<0.01), BL (0.82-1.36, P<0.001) and GGT (42.4-164.2, P<0.001). in contrast, the groups treated with B. albaextract at dose of (100- 400 mg/kg) once daily for 28 days prevented the incidence of cancer of in a dose related manner. The ranges of protection in the serum marker were found to be SGOT (364.21-208.32, P<0.05 to p<0.001), SGPT (381.53-102.34, P<0.05 to p<0.001), SALP (449.31-252.26, P<0.01 to p<0.001), BL (1.36-0.88, p<0.001), and GGT (164.2-62.4, P<0.001) respectively. The protection of cisplatin ranged for SGOT (369.62-206.21, p<0.001), SGPT (391.48-104.28, p<0.001), SALP (447.39-248.29, p<0.001), BL (1.39-0.92, p<0.01) and GGT (166.4-62.3, p<0.001) respectively as shown in Table 2.
|
Groups |
Treatment |
Dose |
SGOT |
SGPT |
SALP |
BL |
GGT |
|
I |
Control |
--- |
202.20±1.64 |
93.21±1.60 |
244.14±10.34 |
0.82±0.02 |
42.4±5.4 |
|
II |
NDEA+CCl4 |
200 mg (NDEA)+3ml/kgbw |
364.21±36.31z |
381.53±42.72z |
449.31±28.31z |
1.36±0.08z |
164.2±12.8z |
|
III |
B.alba |
(CCl4) 100mg/kg |
266.34±22.52a |
256.38±34.29a |
364.24±24.34a |
0.101±0.07b |
158.8±10.8 |
|
IV |
B.alba |
200mg/kg |
228.51±20.39b |
191.14±36.29b |
309.28±21.54b |
0.97±0.06b |
131.4±9.6 |
|
V |
B.alba |
400mg/kg |
212.81±15.71b |
119.06±21.34c |
259.96±18.28c |
0.92±0.04c |
88.1±7.7c |
|
VI |
Cisplatin |
6mg/kg |
208.32±10.78b |
102.34±23.48c |
252.26±18.72c |
0.88±0.03c |
62.4±8.1c |
|
Values are mean ± S.E.M. of 6 rats in each group P values: z<0.001 compared with respective control group P values: a<0.05, b<0.01, c<0.001 compared with group II (NDEA+CCl4) |
|||||||
Table 2: Effect of the 50% ethanolic extract of B. alba on SGOT(U/l), SGPT(U/l), SALP(U/l), Bilirubin level (U/l) and Gamma glutamyltranspeptidase, GGT (U/l) in serum of rat
Effect of 50% ethanolic extract of B. albaon LPO, SOD, CAT, GPX, GST and GSH against NDEA+CCl4 induced HCC
Administration of NDEA+CCl4 led to increase in the levels of LPO (10.46-14.64, p<0.001), and decrease in SOD (124.4- 58.20, p<0.001), CAT (38.8-16.24, p<0.001), GPX (13.54-11.06, p<0.001), GST (11.06-10.47, p<0.001) and GSH (10.36-10.04, P<0.001) levels in the 5% w/v liver homogenate. Treatment of rats with 50% ethanolic extract of B. albaat dose of (100-400 mg/kg b.w) markedly prevented the NDEA+CCl4 induced alterations of various parameters LPO (13.62-11.02, p<0.05 to p<0.01), SOD (88.12-108.52, p<0.05 to p<0.001), CAT (22.19-31.39, p<0.05 to p<0.001), GPX (11.50-13.24, p<0.001), GST (10.60-10.94, p<0.001) and GSH (10.10-10.27, p<0.001) respectively. The protection of Cisplatin ranged for LPO (14.64-10.88, p<0.05 to p<0.01), SOD (58.20-112.31, p<0.05 to p<0.001), CAT (16.24- 34.09, p<0.05 to p<0.001), GPX (11.06-13.42, p<0.001), GST (10.47-10.97, p<0.001) and GSH (10.10-10.32, p<0.001) respectively as shown in Table 3.
|
Groups |
Treatment |
Dose |
SOD |
CAT |
LPO |
GPx |
GST |
GSH |
|
I |
Control |
--- |
124.4±9.1 |
38.8±1.2 |
10.46±0.04 |
13.54±0.04 |
11.06±0.12 |
10.36±0.02 |
|
II |
NDEA+CCl4 |
200mg/kg |
58.20±9.14z |
16.24±2.2z |
14.64±1.21z |
11.42±0.02z |
10.47±0.02z |
10.04± 0.01z |
|
III |
B.alba |
100mg/kg |
88.12±5.31a |
22.19±1.2a |
13.62±1.14 |
11.50±0.02a |
10.60±0.04a |
10.10±0.02a |
|
IV |
B.alba |
200mg/kg |
101.21±6.26b |
26.19±1.30b |
12.16±0.98 |
12.94±0.03c |
10.82±0.05c |
10.18±0.03b |
|
V |
B.alba |
400mg/kg |
108.52±4.21c |
31.39±0.93c |
11.02±0.36a |
13.24±0.02c |
10.94±0.08c |
10.27±0.04c |
|
VI |
Cisplatin |
6mg/kg |
112.31±5.24c |
34.09±2.12c |
10.88±0.22b |
13.42±0.02c |
10.97±0.09c |
10.32±0.05c |
|
Values are mean ± S.E.M. of 6 rats in each group P values: z<0.001compared with respective control group P values: a<0.05, b<0.01, c<0.001compared with group II (NDEA+CCl4) |
||||||||
Table 3: Effect of 50% ethanolic extract of B.alba on LPO, SOD, CAT, GPX, GST and GSH against NDEA+CCl4 induced HCC
Effect of 50% ethanolic extract of B. albaon haematological parameters (RBC, WBC and Hb) of control and NDEA+CCl4 induced HCC
The Table 4 shows the level of Hb, RBC counts, all of which were significantly decreased (21.97-18.52, p<0.001) and (18.12-16.42, p<0.05) and with simultaneous increase in WBC (16.22-18.79, p<0.01) with respect to control. In contrast, the groups treated with B. albaextract at dose of (100-400 mg/kg b.w) once daily for 28 days prevented the cancer in a dose related manner. The range of protection in the Hb, RBC and WBC show (18.91-20.67, p<0.05), (16.91-17.89) and (17.57-16.54, p<0.01). the protection of Cisplatin ranged for Hb (18.52-21.45, p<0.01), RBC (16.42- 17.97, p<0.05) and WBC (18.79-16.24, P<0.01) respectively.
|
Group |
Parameter |
Dose |
RBC (million/ mm3) |
WBC (million/ mm3) |
Hb(g/dl) |
|
I |
Control |
--- |
18.12±0.76 |
16.22±0.06 |
21.97±0.42 |
|
II |
NDEA+CCl4 |
200mg/kg (NDEA)+3ml/ kgbw (CCl4) |
16.42±0.52 |
18.79±0.82x |
18.52±0.32z |
|
III |
B.alba |
100mg/kg |
16.91±0.58 |
17.57±0.71 |
18.91±0.72 |
|
IV |
B.alba |
200mg/kg |
17.12±0.37 |
17.02±0.53 |
19.76±0.4a |
|
V |
B.alba |
400mg/kg |
17.89±0.82 |
16.54±0.43a |
20.67±0.82a |
|
VI |
Cisplatin |
6mg/kg |
17.97±0.58 |
16.24±0.39a |
21.45±0.82b |
|
Values are mean ±S.E.M. of 6 rats in each group P values: x<0.05, y<0.01, z<0.001 compared with respective control group P values: a<0.05, b<0.01 compared with group II (NDEA +CCl4) |
|||||
Table 4: Effects of B.alba on haematological parameter (RBC, WBC and Hb) of control and (NDEA+CCl4) induced HCC in rat
Histopathological Analysis
The histological investigation of liver slices from the control group revealed normal hepatic architecture, with well-preserved hepatocytes arranged in cords extending from the central vein, intact sinusoids, and no necrosis or inflammatory infiltration. Rat livers treated with NDEA + CCl4 exhibited severe pathological alterations linked to hepatocarcinogenesis. These included distorted lobular architecture, extensive infiltration of inflammatory cells, substantial hepatocyte ballooning, cytoplasmic vacuolization, bile duct hyperplasia, and the formation of many preneoplastic nodules. Hepatocytes exhibited nuclear pleomorphism, hyperchromasia, large nucleoli, and increased mitotic activity. Bridging fibrosis and pseudolobule formation were also observed, indicating a potential for malignant change.
Treatment with dosages of B. alba extract (100, 200, and 400 mg/ kg. p.o.) considerably improved these degenerative changes in a dose-dependent fashion. Liver sections showed partial repair of hepatic cords, reduced inflammatory infiltration, ballooning degeneration, and fewer preneoplastic nodules compared to the NDEA + CCl4group. Fibrosis was greatly reduced, resulting in the retention of sinusoidal gaps and decreased pseudolobular formation. Hepatocyte nuclei seemed more homogeneous and exhibited less pleomorphism, indicating that neoplastic growth had been prevented.
Figure 3 demonstrates that the Cisplatin-treated group, which served as the reference control, had nearly normal histoarchitecture, with few lipid changes, rare inflammatory foci, and no nodular lesions.
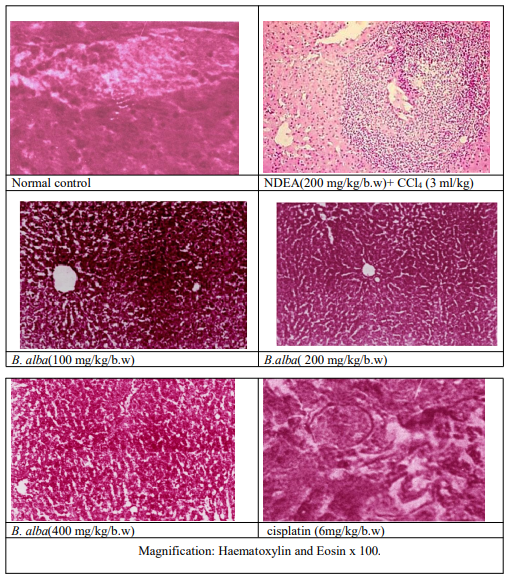
B. alba treatment effectively maintained hepatic tissue integrity, reduced fibrosis, and regulated tumorigenic changes caused by NDEA + CCl4.
Figure 3: Histopathological analysis of B.alba against NDEA+CCl4 induced HCC
Discussion
The present investigation found that B. alba extract greatly reduced liver damage and decreased carcinogenesis in chemically generated HCC. Animals treated with carcinogens showed standard hepatocarcinogenesis features such as higher liver enzymes (ALT, AST, ALP), increased bilirubin, increased lipid peroxidation, and histological evidence of nodular lesions, fibrosis, and nuclear pleomorphism. Previous research suggests that oxidative stress and inflammation play a key role in NDEA/DEN + CClâ??-induced hepatocarcinogenesis.
Treatment with B. alba restored hepatic enzyme profiles, increased antioxidant enzyme activity (SOD, CAT, and GPx), and dramatically reduced malondialdehyde (MDA) levels, showing robust hepatoprotective and free radical scavenging properties. These findings are consistent with prior research indicating the antioxidant and hepatoprotective properties of B. alba in chemically and drug-induced liver damage models [16]. The inclusion of phenolics, flavonoids, and betalains in B. alba is likely responsible for these beneficial effects by enhancing endogenous defence systems and inhibiting lipid peroxidation [17]
. Histopathological study indicated that B. alba treatment reduced preneoplastic nodule formation, decreased fibrosis, and conserved hepatic architecture as compared to untreated HCC mice. These improvements are similar to the hepatoprotective effects described for other antioxidant-rich plants, such as Phyllanthusniruri and Curcuma longa [18].
Mechanistically, B. alba'schemopreventive activity could be related to regulation of molecular pathways involved in hepatocarcinogenesis. B. alba reduces ROS levels, increasing Nrf2-mediated antioxidant defence and suppressing NF-κB- driven inflammation. This leads to decreased DNA damage and proliferative signalling [7]. Furthermore, the extract's flavonoid contents may promote p53-dependent apoptosis while restoring hepatocyte homeostasis [11].
The current findings support the hypothesis that B. alba, an edible leafy vegetable with high nutritional and medicinal value, could be explored as a functional food or adjunct phytomedicine for liver cancer prevention. Given its favorable safety profile and traditional dietary use, it represents a promising candidate for nutraceutical development. However, further studies focusing on bioactive compound isolation, molecular target identification, and pharmacokinetics are essential to establish its translational relevance.
Conclusion
The study found that B. alba extract had strong hepatoprotective and chemopreventive effects on NDEA + CClâ??-induced hepatocellular cancer in rats. Treatment with the extract normalised serum biochemical indicators, increased endogenous antioxidant defences, decreased lipid peroxidation, and conserved liver histoarchitecture. These positive results can be ascribed to its abundant phytoconstituents, which include flavonoids, phenolics, and carotenoids, which work together to reduce oxidative stress and inflammation. The findings scientifically verify the traditional usage of B. alba as a hepatoprotective agent and point to its potential as a functional food or phytotherapeutic candidate in liver cancer treatment. Future research should focus on bioactive molecule isolation, molecular mechanism elucidation, and clinical validation in order to advance translational applications.
References
- El-Serag, H. B. (2020). Epidemiology of hepatocellular carcinoma. The liver: Biology and pathobiology, 758-772.
- Sung, H., Ferlay, J., Siegel, R. L., Laversanne, M., Soerjomataram, I., Jemal, A., & Bray, F. (2021). Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a cancer journal for clinicians, 71(3), 209-249.
- Forner, A., & Reig, M. (2018). carcinoma Bruix JHepatocellular. Lancet, 391(10127), 1301-14.
- Nault, J. C., & Villanueva, A. (2021). Biomarkers for hepatobiliary cancers. Hepatology, 73, 115-127.
- Zheng, Z., Zhang, L., & Hou, X. (2022). Potential roles and molecular mechanisms of phytochemicals against cancer. Food & Function, 13(18), 9208-9225.
- Thapa, B. R., & Walia, A. (2007). Liver function tests and their interpretation. The Indian Journal of Pediatrics, 74(7), 663-671.
- Farazi, P. A., & DePinho, R. A. (2006). Hepatocellular carcinoma pathogenesis: from genes to environment. Nature Reviews Cancer, 6(9), 674-687.
- Khatun, A., Khan, M. A. A., Rahman, M. A., Akter, M.S., Hasan, A., Parvin, W., ... & Rahmatullah, M. (2013). Ethnomedicinal usage of plants and animals by folk medicinal practitioners of three villages in Chuadanga and Jhenaidah districts, Bangladesh. Am-Eur J Sustain Agr, 7, 319-39.
- Alamgir, A. N. M. (2017). Therapeutic use of medicinal plants and their extracts: volume 1 (Vol. 73). Cham: Springer.
- Bosch, F. X., Ribes, J., & Borràs, J. (1999). Epidemiology of primary liver cancer. In Seminars in liver disease (Vol. 19, No. 03, pp. 271-285). © 1999 by Thieme Medical Publishers, Inc.
- Halliwell, B., & Gutteridge, J. M. (2015). Free radicals in biology and medicine. Oxford university press.
- Perumal, P. C., Sophia, D., Raj, C. A., Ragavendran, P., Starlin, T., & Gopalakrishnan, V. K. (2012). In vitro antioxidant activities and HPTLC analysis of ethanolic extract of Cayratia trifolia (L.). Asian Pacific Journal of tropical disease, 2, S952-S956.
- Gnanamani, A., Sudha, M., Deepa, G., Sudha, M., Deivanai, K., & Sadulla, S. (2008). Haematological and biochemical effects of polyphenolics in animal models. Chemosphere, 72(9), 1321-1326.
- Sánchez-Moreno, C. (2002). Methods used to evaluate the free radical scavenging activity in foods and biological systems. Food science and technology international, 8(3), 121-137.
- Greaves, P. (2011). Histopathology of preclinical toxicity studies: interpretation and relevance in drug safety evaluation. Academic Press.
- Surai, P. F., & Surai, A. (2023). Silymarin and NF- κB. Wageningen Academic Publishers: Wageningen, The Netherlands, 151-177.
- CHEN, C. J., YU, M. W., & LIAW, Y. F. (1997).Epidemiological characteristics and risk factors of hepatocellular carcinoma. Journal of gastroenterology and hepatology, 12(9-10), S294-S308.
- Valko, M., Rhodes, C. J. B., Moncol, J., Izakovic, M. M., & Mazur, M. (2006). Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chemico-biological interactions, 160(1), 1-40.