Review Article - (2026) Volume 4, Issue 1
Animal Trials, A Landscape to Gastroenterological Insight
2Department of Ophthalmology, Visual, and Anatomical Sciences.Wayne State University, School of Medic, USA
3Department of Biology, New Mexico State University, New Mexico, USA
4Department of Geography, New Mexico State University, New Mexico, USA
Received Date: Nov 18, 2025 / Accepted Date: Dec 23, 2025 / Published Date: Jan 15, 2026
Copyright: ©2026 Chinyere Mary-Cynthia Ikele. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Ikele, C. M. C., Nnaka, J. A., Agbetsi, J., Atanga, J. A. (2026). Animal Trials, A Landscape to Gastroenterological Insight. Gen Surgery Clin Med, 4(1), 01-13.
Abstract
Animal trials can enhance research quality and efficiency in various fields, but have limited usage for clinical trials due to rights and regulations. This can impact the rate, trend, and outcome of gastro-hepatological insights. How has the trend in finding novel remedies for gastrointestinal diseases evolved over the years up until now? This article comprehensively examines how policies and animal trials have affected the shift and evolution of de novo treatment for gastroenterology. It delves and deepens insights into how resources, accessibility, and availability of unrestricted ethics have positively influenced the gradient with aid and supplementation via interventions, such as the 3D scans and experiments that predict therapeutic benefits and measures.
Keywords
Animal Trials, Ethics, Evolution, 3D, Trends, Gastro-Hepatology, Limitation, Toxicity
Introduction
Gastroenterological research in animals plays a crucial role in understanding digestive diseases and developing internal treatments that can be translated to human medicine. Animal models, particularly large animals like ruminants, offer significant advantages due to their physiological similarities to humans. This research area faces ethical considerations and challenges in ensuring the relevance and applicability of research outcomes to human conditions. Animal trials are being used to guide gastroenterological research, but their ability to predict human outcomes is limited, and improved reporting and evaluation methods are needed. Evidence summaries and ratings of clinical practice and outcome can be used to support decisions on the utility of animals for further experimentation and selection of interventions with the best therapeutic potential to be tested in clinical trials [1]. A study addressed adverse effect of animal trials cum success rate post trials for clinical test and usage. An increase in the failure rate of innovative therapy for drugs and health was attributed to wrong decisions made from the results of animal trials, with 21% toxicity failures attributed to hepatic research [2]. Another study and school of thought found that animal studies did not provide unequivocal evidence to substantiate the decision to conduct clinical trials, judging that the methodological quality of animal studies was poor, not conducted simultaneously rather sequentially [3]. How have animal trials evolved over the years with an impact on gastroenterological research? Randomization has been established as a cornerstone of clinical trial methodology, yet the ethics of randomization continue to generate controversy. Ethical intervention may reduce decisional conflict, improve patient involvement, and enhance quality of life [4]. Take, for instance, the issue of the pandemic; pandemics do not justify lowering scientific standards, as they intensify the responsibility for researchers to maintain high standards to advance health care [5].
The ethics of research in humanitarian crises require carefully considering unique factors such as vulnerable populations, contextual challenges, and community engagement. Moreover, ethical dilemmas faced by healthcare professionals include balancing limited resources, prioritizing treatment, and balancing patient needs. Ethical issues require further investigation and guidance to optimize patient care, reduce financial burden, and protect vulnerable groups.
Figure 1: Diagram illustrating how animal trials impact gastroenterological research
Materials and Methods
Data was retrieved from the National Institute of Health, Sladen Library, and PubMed Database. A comprehensive review of articles, systematic reviews, and data within 10 years (2015 to date) was used for this study. Specific articles (6) cited below the decade gap are instrumental. Any article not within the set standards of the timeline was discarded.
Results
How have animal trials evolved over the years to direct research in gastro-hepatology?
Retracing to the precolonial period when animals were killed and prosecuted for executions of the past centuries ago, we continue to use and punish animals for crimes against human beings [6]. Gergin argued that we do this to maintain order and the achievement of revenge, and concluded that a call for a renewed emphasis on “due process” resulted in what is known as ethics for animal trials. MacGregor explored compelling and specific cases from the so-called “medieval animal trials” in which animals were accused and sentenced for harming persons or property [7]. A close examination revealed that the trials share several features, which not only highlight the importance of following certain legal procedures but also place the animal and its actions into pre-existing legal categories. By treating the animal as a criminal, these records provide a window into the medieval story of legal personhood and fluidity of its borders, while also challenging the history of the human-animal relation as one built on difference and inferiority.
The story of animal trials in gastro-hepatology is one of continuous refinement and adaptation, mirroring the broader evolution of biomedical research. In the early days, animal experimentation was largely exploratory. Researchers used animals to gain foundational insights into anatomy and physiology, often without a clear understanding of how these findings would translate to human health. These early investigations, while crude by modern standards, laid the groundwork for the systematic study of gastrointestinal and liver diseases.
As the 20th century progressed, animal models became more sophisticated and disease-specific. Scientists began to recognize that certain animals could be used to mimic the pathophysiology of human liver and gastrointestinal disorders. Rodents, particularly mice and rats, emerged as the workhorses of gastro-hepatology research. By exposing these animals to specific chemicals, altering their diets, or manipulating their genes, researchers were able to induce conditions such as liver fibrosis, steatohepatitis, and cirrhosis—diseases that closely resemble those seen in human patients. These models allowed for the detailed study of disease mechanisms and the testing of potential therapies in a controlled environment.
However, as our understanding of these diseases deepened, so too did the realization that rodent models, while invaluable, had limitations. Rodents differ from humans in significant anatomical, physiological, and metabolic ways, which can sometimes lead to discrepancies between animal and human responses to disease and treatment. This recognition prompted a shift toward the use of larger animal models, such as pigs, whose gastrointestinal tracts and liver functions more closely mirror those of humans. These large animal models have proven especially useful for developing and refining surgical and endoscopic techniques, as well as for studying complex disease processes in a setting that is more directly relevant to human medicine.
The advent of genetic engineering marked another transformative leap. With the ability to create transgenic and knockout animals, researchers could now investigate the roles of specific genes and molecular pathways in the development and progression of gastro- hepatological diseases. This level of precision has led to important discoveries about the underlying causes of conditions like non- alcoholic fatty liver disease and inflammatory bowel disease, and has opened the door to targeted therapies. In recent years, the development of “humanized” animal models has further bridged the gap between preclinical research and clinical application. By transplanting human cells or tissues into animals, most commonly mice, scientists have created models that more accurately predict human responses to drugs and disease. This approach has been particularly valuable in identifying potential toxicities that might not be apparent in traditional animal models, as was the case with the antiviral drug fialuridine, which caused fatal liver failure in humans despite appearing safe in conventional animal testing. Alongside these scientific advances, there has been a growing emphasis on standardization and reproducibility in animal research. Efforts are underway to harmonize the nomenclature, dietary protocols, and experimental designs used in gastro-hepatology studies, ensuring that findings are robust and comparable across different laboratories and research groups.
Reynolds, 2012, in his article “Twenty Years After: Do animal trials inform clinical resuscitation research? Stated that although impact has culminated from the use of animals to direct research, due to physiological differences and certain limitations associated with the use of animal trials, there are high translational failure rates [8]. Attributed factors pointed out were a lack of standardization, poor reporting, and inadequate modelling of human diseases.
Despite these challenges, animal trials have greatly influenced clinical trials for instance in Pig models showing potential benefit for liver cancer research due to its similarity to human beings [9]. Equally, inflammatory bowel diseases have rapidly evolved over the last 5 years, focusing on novel therapies, optimizing existing treatment approaches, and population health management [10]. Toxicity caused by specie differences is an attributed factor why most clinical trials are not successful, with 21% of toxicity reported in hepatological research [2,9]. Animal studies are poor predictors of human toxicity in health research, leading to delays, high cost, and potential harm to human subjects.
In summary, the evolution of animal trials in gastro-hepatology reflects a broader trend toward greater precision, relevance, and ethical responsibility in biomedical research. From basic anatomical studies to complex, genetically engineered and humanized models, animal research continues to direct and inform the search for a better understanding and treatment of gastrointestinal and liver diseases, always striving to bring laboratory discoveries closer to real-world clinical solutions.
Figure 2: image illustrating how the toxicity of animal trials can influence clinical trials and the usage of medical innovation
Ethical Considerations in Gastro-Hepatology Animal Research
Animal models are central to gastro-hepatology research, primarily because of their biological, physiological, and genetic similarities to humans, which enable scientists to investigate complex disease mechanisms, simulate human pathological conditions, and evaluate potential therapies in ways that is not feasible in human subjects [11,12]. Mice, for instance, share over 98% of their DNA with humans, making them especially valuable for modeling disease progression and evaluating treatment effects across lifespans and generations [13]. Their relatively short life cycles, ease of genetic manipulation, and cost-effectiveness further enhance their utility for longitudinal studies that track disease progression across developmental stages and generations [13]. In addition to rodents, other monogastric animals such as pigs and rabbits are frequently employed for their anatomical and metabolic similarities to human digestive systems, which allow for more accurate modeling of nutrient absorption, microbial dynamics, and drug metabolism [14,15]. Zebrafish, with their transparent embryos and rapid development, have also become valuable tools for high-throughput screening of hepatotoxic agents and for visualizing real-time cellular responses in digestive tissues [15]. More importantly, animal models serve as safety buffers in translational research by helping researchers identify potential toxicities and therapeutic thresholds before proceeding to human clinical trials [12].
This early-phase testing protects human participants from unforeseen adverse effects and helps to minimize the ethical risks associated with first-in-human studies. Additionally, animal models enhance experimental control and reproducibility by enabling researchers to isolate specific variables, replicate pathological conditions, and systematically refine therapeutic interventions. Such precision would be difficult, if not impossible, to achieve in human populations due to inherent ethical constraints, logistical challenges, and biological heterogeneity [16]. Despite the known benefits of animal models in biomedical research, their use raises significant ethical considerations that have led to the development of frameworks that guide ethical practices. Foremost among these is the “Three Rs” principle—Replacement, Reduction, and Refinement—introduced by Russell and Burch in 1959 [17,18]. Replacement involves substituting animal models with alternative methods such as in vitro cell cultures, organoids, and computer simulations, which offer valuable insights without the ethical burdens associated with live animal use [18]. Reduction focuses on minimizing the number of animals used in research by applying robust experimental designs and appropriate statistical methods [19,20]. Refinement entails modifying procedures to minimize pain, distress, or lasting harm through improved techniques, housing, and care [20,21]. To enforce these principles, Institutional Animal Care and Use Committees (IACUCs) or their equivalents rigorously review all animal research protocols before approval [22].
Certain animal models, such as piglets and non-human primates, face even stricter ethical scrutiny due to their higher cognitive abilities, social complexity, and physiological similarities to humans. Studies involving these animals often require strong scientific justification and are subject to additional oversight, including more frequent welfare checks and post-experiment monitoring [23,24]. Furthermore, some interventions - such as inducing severe disease states or performing invasive surgeries - demand comprehensive pain management plans and humane endpoints to prevent unnecessary suffering [21]. Moreover, ethical concerns are further complicated by scientific uncertainties about the translational relevance of certain animal models. Some researchers argue that animal studies are not always reliable predictors of drug toxicity or efficacy in humans, partly due to species-specific differences in physiology, metabolism, and immune responses [2]. For example, drugs that are well-tolerated in rodents may trigger adverse immune reactions in humans, while compounds that appear toxic in animals may prove safe and effective in clinical settings. Such discrepancies raise critical questions about the necessity and limitations of animal testing in the drug development pipeline. These concerns have prompted increased interest in integrating human-based in vitro methods, computational biology, and ethically sourced human tissues into mainstream research [25]. As science continues to progress, there is a growing focus on aligning ethical responsibility with scientific innovation, as this will help create a future where only models that are both humane and predictive of human health are considered in animal research.
In support of this evolving landscape, ethical frameworks remain foundational to scientific research. They ensure the protection of the rights and welfare of both animal and human participants, while also safeguarding vulnerable populations from potential exploitation [26]. They serve not only as regulatory mechanisms but also as moral compasses that align research practices with broader societal values. By promoting transparency, accountability, and fairness, they reinforce public trust in the scientific enterprise and contribute to the overall integrity of research. Furthermore, scholars have emphasized the intrinsic connection between adherence to ethical principles and the broader concept of research integrity [27]. A failure to comply with established ethical guidelines often signals deeper issues related to a researcher’s commitment to integrity, as ethical compliance is a key component of responsible conduct in research [27]. In this sense, ethical lapses do not merely constitute procedural violations but fundamentally undermine the credibility, reliability, and societal value of scientific inquiry.
Conceptual model by Muthanna et al. illustrating the interrelationship between research ethics and research integrity [27].
However, the additional layers of oversight, documentation, and training can increase the logistical complexity and cost of conducting research, potentially slowing the pace of scientific discovery. National and international regulations often require researchers to justify animal use, minimize suffering, and seek alternatives, which can extend the time and resources needed to initiate and conduct studies [28]. Restrictions on experiment duration or the use of certain procedures may limit the scope of research, sometimes preventing the use of the most clinically relevant models or interventions. These regulatory requirements, enforced by multidisciplinary ethics committees and guided by principles such as the 3Rs, can result in additional reviews and modifications to research protocols, occasionally leading to delays or the need to redesign studies [20]. In some cases, stringent ethical constraints may result in underpowered studies or limit innovation, as researchers are forced to compromise on experimental design to meet regulatory requirements, which can affect the generalizability and applicability of findings. A way forward lies in striking a careful balance between ethical rigor and scientific progress. Ethical review boards should continue to enforce high standards for participant protection and research integrity, but also engage in open dialogue with researchers to ensure that regulations are evidence-based, flexible, and responsive to advances in methodology and technology. Incorporating adaptive review processes, promoting transparency, and encouraging the development of alternative research models can help minimize unnecessary burdens while preserving the core ethical principles that safeguard both research subjects and the credibility of gastro-hepatology research. This balance is particularly needed in preclinical gastro-hepatology studies, where researchers must navigate complex ethical landscapes while producing scientifically valuable data.
Table 1 highlights several recent animal-based studies, including the animal model used, experimental interventions, observed outcomes, translational relevance, and the ethical considerations provided. These examples show the varied approaches researchers employ to investigate complex disease mechanisms, evaluate therapeutic strategies, and balance scientific inquiry with ethical responsibility in preclinical research.
Preclinical studies, their outcome, and ethical considerations.
|
Animal Type |
Drug/Research Administered |
Outcome of the Research (Positive/Negative) |
Clinical Outcome |
Ethical Consideration/Restriction |
|
Kim et al., 2022 C57BL/6 mice (monogastric) |
NAFLD/NASH + (GLP)- 1 and GLP-2 agonist |
Reduced body weight, glucose levels, hepatic triglyceride levels, cellular apoptosis, and fibrosis |
Treatment of patients with NAFLD with the GLP- 1/glucagon receptor agonists (Nevola et al., 2023) |
Study approved by the Animal Care and Use Committee at the Yonsei University |
|
Zebrafish |
High-fat diet + metformin in a |
Altered macrophage polarization, |
Metformin therapy shows |
None provided |
|
|
transgenic |
reduced liver size, |
very limited |
|
|
(monogastric) |
zebrafish |
and reduced |
effect on |
|
|
|
hepatocellular |
micronuclei |
NAFLD or |
|
|
|
carcinoma |
formation in |
nonalcoholic |
|
|
|
model |
NAFLD/NASH- |
steatohepatitis |
|
|
|
|
associated HCC |
(Farah et al., |
|
|
|
|
larvae. |
2019) |
|
|
Wang et al., 2025 Mice (monogastric) |
DSS-induced colitis + probiotic (Lactobacillus vaginalis) |
Reduced colitis symptoms, restored gut microbiota and barrier function, increased IAA levels, reduced inflammation via PPAR signaling pathway |
Ongoing Clinical trials on the efficacy of Probiotics in Inflammatory Bowel Disease |
Study complied with the World Medical Association (WMA) Statement and China National Standard (GB/T 35892-2018) on animal use in biomedical research. |
|
Khalil et al., 2020 Sprague Dawley rats (monogastric) |
CCl4-induced fibrosis + bone marrow mesenchymal stem cells therapy |
Improved liver structure and function, reduced liver damage, oxidative stress, and inflammation, and boosted antioxidant levels. |
Stem cell clinical trials in cirrhosis |
Animals are cared for per the National Institutes of Health Guide for the Care and Use of Laboratory Animals. |
|
Liu et al., |
H. pylori |
Reduced gastric |
The study has |
Animals are handled |
|
2020 |
infection + Oral |
bacterial |
not yet |
following the National |
|
BALB/c mice (monogastric) |
immunization with a multivalent |
colonization, increased antigen- specific IgG and |
progressed to human clinical trials |
Management Regulations for the Care and Use of Experimental Animals |
|
|
subunit vaccine |
mucosal IgA |
|
|
|
|
(NAP, UreA, UreB) plus |
responses, and induction of |
|
|
|
|
dmLT |
Th1/Th17 immune |
|
|
|
|
|
responses |
|
|
|
Javadi et al., 2023 Wistar rats (monogastric) |
Bile duct ligation + Trans-chalcone |
Improved liver architecture and reduced inflammation and fibrosis. |
Study still in the preclinical stage |
Experiments carried in accordance with the Guide for the Care and Use of Laboratory Animals (NIH, USA). |
|
|
|
|
|
Anesthesia was induced in rats before BDL surgery |
|
Pu et al., 2024 Sprague- Dawley (monogastric) |
TAA-induced fibrosis + Lactoferrin |
Reduced fibrosis and improved antioxidant activities |
Study still in the preclinical phase |
Experiments were reportedly in line with the Institutional Animal Care and Use Committee of the University. |
|
Gelen et al., 2024 Wistar Rats (monogastric) |
Indomethacin- induced GI injury + probiotic |
Reduced oxidative stress and inflammation, increased antioxidant activity, and suppressed apoptosis in gastric tissue. |
Study still in the preclinical phase |
The study reportedly followed ethical norms approved by the Kafkas University Animal Experiments Local Ethics Committee. |
Table 1: Showing Preclinical Studies, Ethical Considerations, and Outcomes
Ethical Limits and Effects
Ethical limits in gastroenterology and hepatological research often arise from the tension between medical innovation and the foundational principles of clinical ethics, such as autonomy, beneficence, non-maleficence, justice, and utility. These constraints and restrictions are brought by significant ethical dilemmas, including challenges around informed consent, the ethics of deferring treatment, sobriety restrictions imposed by payors, and the high costs of treatments for patients. These issues highlight the need to balance patient autonomy and access to care with broader considerations of fairness and resource allocation.
Ethical considerations are woven into every stage of gastrohepatology research, shaping both the questions scientists ask and the methods they use to answer them. As this field often involves studies on both human participants and animal models, researchers must navigate a complex landscape of ethical responsibilities, balancing scientific progress with the imperative to protect the welfare and rights of all research subjects.
When it comes to human research, ethical oversight is paramount. Investigators are required to obtain approval from institutional review boards or ethics committees before commencing any study involving patients or volunteers. This process ensures that the research design respects the dignity, autonomy, and privacy of participants. Informed consent is a cornerstone of ethical human research, requiring that individuals are fully aware of the study’s aims, methods, potential risks, and benefits before agreeing to participate. In gastrohepatology, where studies may involve invasive procedures or the use of sensitive health data, these safeguards are particularly critical. Furthermore, when publishing results, researchers must ensure that patient identities are protected, and any images or data are anonymized unless explicit consent has been granted.
Animal research in gastrohepatology is similarly governed by rigorous ethical standards. Researchers must obtain approval from animal ethics committees and comply with national or institutional guidelines for the care and use of laboratory animals. The principle of the “3Rs”—Replacement, Reduction, and Refinement—serves as the ethical foundation for animal studies. Scientists are encouraged to replace animal models with alternatives whenever possible, reduce the number of animals used to the minimum necessary for statistical validity, and refine experimental procedures to minimize pain and distress. These requirements have led to the adoption of improved animal housing, better pain management protocols, and the use of advanced technologies that reduce the need for animal testing.
The effects of these ethical frameworks are profound. On a practical level, they have improved the welfare of animals used in research and enhanced the safety and rights of human participants. Ethically mandated rigor in study design and reporting has also elevated the quality and reproducibility of scientific findings. Moreover, the demand for alternatives to animal testing has spurred innovation, leading to the development of organoids, in vitro systems, and computational models that can complement or even replace animal studies in some contexts. Importantly, adherence to ethical standards fosters public trust in gastrohepatology research, ensuring continued support from patients, advocacy groups, and funding agencies. In sum, the ethical limits placed on gastrohepatology research are not simply bureaucratic hurdles; they are essential safeguards that uphold the integrity of science and the welfare of those—human or animal—who make scientific progress possible. These ethical commitments continue to shape the evolution of the field, driving both methodological innovation and a deeper respect for the subjects at the heart of research.
Addressing such dilemmas requires applying ethical frameworks to ensure that new therapies are introduced in ways that respect patient rights and promote equitable access, while also considering the societal impact and sustainability of healthcare innovations. The experience with DAA (Direct-Acting Antiviral) therapy in hepatology serves as a paradigm for navigating similar ethical challenges as new treatments emerge in the field, emphasizing the importance of ongoing ethical reflection and adaptation in research and clinical practice [29].
These limits also extend to biomedical research publication and can be influenced by political concerns and security concerns. A balance is required between academic pursuits and the protection of international interests [30]. Political interests significantly shape medical ethics by influencing both the content of ethical codes and the practical dilemmas faced by healthcare professionals [31]. The increasing complexity of medical practice, advances in science, and public demands for healthcare have brought ethical questions into the public and political domain, shifting the focus from individuals' doctor-patient relationships to broader issues of public policy and regulation.
Ethical frameworks have profoundly influenced hepatological research, shaping not only the conduct of studies but also the broader priorities and methodologies within the field. The increasing emphasis on ethics has led to more rigorous protections for research subjects, both human and animal, ensuring that informed consent, autonomy, and the welfare of participants are at the forefront of study design and execution [26]. For example, researchers are now required to obtain explicit informed consent, allow participants to withdraw at any time, and maintain strict confidentiality, all of which foster greater trust and participation in clinical trials [26].
Ethical scrutiny has also brought attention to potential conflicts of interest, the influence of industry funding, and issues such as ghostwriting and scientific misconduct, prompting the development of clearer codes of conduct and more transparent reporting practices. This has improved the credibility and reproducibility of hepatological research, as journals and funding bodies increasingly demand adherence to established ethical guidelines.In clinical practice, particularly in areas like non-alcoholic fatty liver disease (NAFLD), ethical considerations have shaped how diagnoses are communicated and managed. There is heightened awareness of the potential harms of overdiagnosis, the psychological impact of labeling, and the challenges of counseling patients with diverse backgrounds and risk factors [32]. The need for voluntary, well- informed consent is especially critical in procedures like liver biopsy, which carries inherent risks and must be justified ethically and clinically. Overall, the influence of ethical standards has led to more patient-centered research, greater methodological rigor, and enhanced public trust in hepatological studies, ultimately advancing both scientific knowledge and patient care in the field.
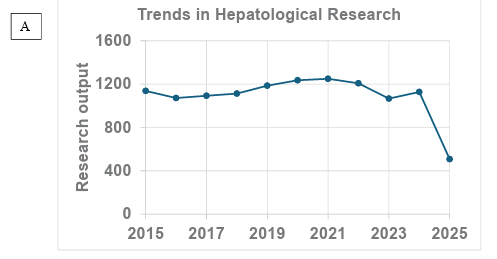
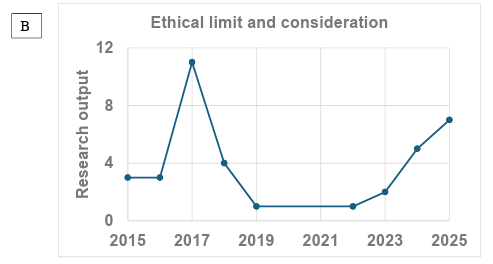
Figure 3: Graph showing ten (10) years trends in (a)research output for hepatology (b) ethical limit and consideration
Supplementary intervention through 3D scans, patient-reported outcome, and experiments
Recent research in hepatology highlights the growing role of 3D imaging and patient-reported outcomes in improving surgical planning and monitoring interventions. In liver surgery, both 3D laparoscopic and robotic approaches for hepatocellular carcinoma have shown comparable short- and long-term outcomes, suggesting that 3D visualization can match the precision and safety of advanced robotic systems while aiding in surgical decision-making [33]. Meta-analyses indicate that preoperative 3D vascular reconstruction and navigation can reduce operation time and intraoperative blood loss compared to standard 2D procedures, although the impact on hospital stay and complication rates remains less clear [34].
Three-dimensional (3D) printing has significantly influenced drug innovation in hepatology by enabling the precise construction of dosage forms and supporting the development of patient-centric treatments. This technology allows for personalized dosing, which is especially important for addressing the unique needs of patients with specific liver diseases or varying metabolic profiles. Additionally, 3D printing offers versatile drug release modes, making it possible to tailor therapies to meet diverse clinical requirements. The adoption of 3D printing has also accelerated early-stage drug development, including pre-clinical research and early human studies, and has facilitated the transition to large-scale pharmaceutical manufacturing. Overall, 3D printing is advancing both the speed and flexibility of drug development and manufacturing, ultimately providing innovative solutions for patients and the pharmaceutical industry alike [35].
For interventional treatments, such as those for hepatocellular carcinoma, 3D digital subtraction angiography (3D-DSA) has been shown to improve therapeutic effectiveness, increase the success rate of targeted procedures, and reduce complication rates, enhancing overall patient safety [36]. In pediatric hepatology, 2D shear wave elastography offers a noninvasive way to monitor liver and spleen stiffness, providing valuable feedback on the effectiveness of radiological interventions and correlating with improved clinical outcomes over time [37]. Collectively, these advances in 3D imaging and patient-centered outcome tracking are shaping more precise, safer, and responsive interventions in hepatology, though further high-quality studies are needed to standardize their use and fully realize their benefits [38-57].
Figure 4: Alternative interventions through 3D technology that impact health and research in hepatolog
References
- Hooijmans, C. R., De Vries, R. B., Ritskes-Hoitinga, M., Rovers, M. M., Leeflang, M. M., IntHout, J., ... & GRADE Working Group. (2018). Facilitating healthcare decisions by assessing the certainty in the evidence from preclinical animal studies. PloS one, 13(1), e0187271.
- Van Norman, G. A. (2019). Limitations of animal studies for predicting toxicity in clinical trials: is it time to rethink our current approach?. JACC: Basic to Translational Science, 4(7), 845-854.
- Pound, P., Ebrahim, S., Sandercock, P., Bracken, M. B., & Roberts, I. (2004). Where is the evidence that animal research benefits humans?. Bmj, 328(7438), 514-517.
- Schildmann, J., Nadolny, S., Haltaufderheide, J., Gysels, M., Vollmann, J., & Bausewein, C. (2019). Ethical case interventions for adult patients. Cochrane Database of Systematic Reviews, (7).
- London, A. J., & Kimmelman, J. (2020). Against pandemic research exceptionalism. Science, 368(6490), 476-477.
- Girgen, J. (2003). The historical and contemporary prosecution and punishment of animals. Animal L., 9, 97.
- MacGregor, L. B. (2019). Criminalising animals in medieval France: Insights from records of executions. Open Library of Humanities, 5(1).
- Reynolds, P. S. (2012). Twenty years after: do animal trials inform clinical resuscitation research?. Resuscitation, 83(1), 16-17.
- Wang, L., Piao, Y., Guo, F., Wei, J., Chen, Y., Dai, X., & Zhang,X. (2023). Current progress of pig models for liver cancer research. Biomedicine & Pharmacotherapy, 165, 115256.
- Singh, S. (2018). Evolution of clinical trials in inflammatory bowel diseases. Current gastroenterology reports, 20(9), 41.
- Perlman, R. L. (2016). Mouse models of human disease: an evolutionary perspective. Evolution, medicine, and public health, 2016(1), 170-176.
- Hylander, B. L., Repasky, E. A., & Sexton, S. (2022). Using mice to model human disease: understanding the roles of baseline housing-induced and experimentally imposed stresses in animal welfare and experimental reproducibility. Animals, 12(3), 371.
- Breschi, A., Gingeras, T. R., & Guigó, R. (2017). Comparative transcriptomics in human and mouse. Nature Reviews Genetics, 18(7), 425-440.
- Choi, T. Y., Choi, T. I., Lee, Y. R., Choe, S. K., & Kim, C. H.(2021). Zebrafish as an animal model for biomedical research.Experimental & Molecular Medicine, 53(3), 310-317.
- Adhish, M., & Manjubala, I. (2023). Effectiveness of zebrafish models in understanding human diseases—A review of models. Heliyon, 9(3).
- Belma, P., Dina, F., Emina, A., Nermina, Z, & Fahir, B. (2019). Animal models in modern biomedical research. European Journal of Pharmaceutical and Medical Research, 6, 35-38.
- Russell, W. M. S., & Burch, R. L. (1959). The principles of humane experimental technique.
- Singh, J. (2012). The national centre for the replacement,refinement, and reduction of animals in research. Journal of Pharmacology and Pharmacotherapeutics, 3(1), 87-89.
- Council, N. H. M. R. (2013). Australian code of practice for the care and use of animals for scientific purposes. Australian Government [Abstract][Google Scholar].
- Smith, A. J. (2020). Guidelines for planning and conducting high-quality research and testing on animals. Laboratory Animal Research, 36(1), 21.
- Lauwereyns, J., Bajramovic, J., Bert, B., Camenzind, S., De Kock, J., Elezovic, A., ... & Ahluwalia, A. (2024). Toward a common interpretation of the 3Rs principles in animal research. Lab animal, 53(12), 347-350.
- Lauwereyns, J., Bajramovic, J., Bert, B., Camenzind, S., De Kock, J., Elezovic, A., ... & Ahluwalia, A. (2024). Toward a common interpretation of the 3Rs principles in animal research. Lab animal, 53(12), 347-350.
- Basel Declaration. (2010). The Basel Declaration: A call for more trust, transparency, and communication on animal research.
- Hubrecht, R. C., & Carter, E. (2019). The 3Rs and humane experimental technique: implementing change. Animals, 9(10), 754.
- Cheluvappa, R., Scowen, P., & Eri, R. (2017). Ethics of animal research in human disease remediation, its institutional teaching; and alternatives to animal experimentation. Pharmacology research & perspectives, 5(4), e00332.
- Eastwood, G. L. (2015). Ethical issues in gastroenterology research. Journal of Gastroenterology and Hepatology, 30(S1), 8–11.
- Muthanna, A., Chaaban, Y., & Qadhi, S. (2024). A model of the interrelationship between research ethics and research integrity. International journal of qualitative studies on health and well-being, 19(1), 2295151.
- Fondi, I. (2023). Animal research’s ethical dimensions and scientific advancements. Animal Nutrition, 8(4), 93.
- Couri, T., & Aronsohn, A. (2021). You Can’t Have One Without the Other: Innovation and Ethical Dilemmas in Gastroenterology and Hepatology. Clinical Gastroenterology and Hepatology, 19(10), 2015-2019.
- Charlier, P., Deo, S., Mamzer-Bruneel, M. F., & Hervé, C. (2016). Ethical limits to biomedical publications?. European journal of internal medicine, 33, e30-e31.
- Essex, R., Mainey, L., Dillard-Wright, J., & Richardson, S. (2024). Political action in nursing and medical codes of ethics. Nursing Inquiry, 31(4), e12658.
- Berry, P., & Kotha, S. (2022, August). The challenging ethical landscape of non-alcoholic fatty liver disease. In ILC Congress 2022 (p. 50).
- Lim, C., Goumard, C., Salloum, C., Tudisco, A., Napoli, N., Boggi, U., ... & Scatton, O. (2021). Outcomes after 3D laparoscopic and robotic liver resection for hepatocellular carcinoma: a multicenter comparative study. Surgical Endoscopy, 35(7), 3258-3266.
- Li, H., Chen, X., & Feng, Q. (2023). The clinical efficacy and safety of 3D vascular reconstruction combined with 3D navigation in laparoscopic hepatectomy. Journal of Gastrointestinal Oncology, 14(1), 476.
- Tracy, T., Wu, L., Liu, X., Cheng, S., & Li, X. (2023). 3D printing: Innovative solutions for patients and pharmaceutical industry. International Journal of Pharmaceutics, 631, 122480.
- Li, R., & Zhang, F. (2021). Three-dimensional DSA guidance reduces complications and enhances the safety during interventional treatment for patients with hepatocellular carcinoma. group, 25, 0-05.
- Samanta, A., Srivastava, A., Yadav, R., Kapoor, A., Ghosh, A., Mishra, P., ... & Poddar, U. (2024). Buddâ?ÂÂChiari syndrome in children: Radiological intervention and role of shear wave elastography in monitoring response. Journal of Pediatric Gastroenterology and Nutrition, 78(1), 17-26.
- de Oliveira, S., Houseright, R. A., Graves, A. L., Golenberg, N., Korte, B. G., Miskolci, V., & Huttenlocher, A. (2019). Metformin modulates innate immune-mediated inflammation and early progression of NAFLD-associated hepatocellular carcinoma in zebrafish. Journal of hepatology, 70(4), 710- 721.
- Maluf-Filho, F., Meyer, A., Martins, P. P. M., Galvão, F. H. F., & D’Albuquerque, L. A. C. (2022). Experimental model of portal hypertension and esophagogastric varices in minipigs: pressure and endoscopic pilot study. Acta cirurgica brasileira, 37, e370103.
- Gralnek, I. M., Hassan, C., Beilenhoff, U., Antonelli, G., Ebigbo, A., Pellisè, M., ... & Dinis-Ribeiro, M. (2020). ESGE and ESGENA Position Statement on gastrointestinal endoscopy and the COVID-19 pandemic. Endoscopy, 52(06), 483-490.
- Farah, S., Nguyen, T., Kelsberg, G., & Safranek, S. (2019). Metformin for nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. American Family Physician, 99(4), 262-263.
- Omar, A. I., Dey, A. R., Alam, M. B. B., Mondal, M. M.H., Khan, M. Y. A., & Faruque, M. O. (2021). Prevalence of common gastrointestinal parasite infection under natural grazing condition in black bengal goat of Bangladesh. International Journal for Asian Contemporary Research, 1(2), 63-72.
- Gelen, V., Gedikli, S., Gelen, S. U., Sengül, E., & Makav,M. (2024). Probiotic bacteria protect against indomethacin- induced gastric ulcers through modulation of oxidative stress, inflammation, and apoptosis. Molecular Biology Reports, 51(1), 684.
- Health Research Library. “National Guidelines: Principles of ethics for research involving Human Subjects.” (2025).
- Health Service Executive (HSE), Ireland. “Research Ethics Review Guideline.” (2022).
- Javadi, F., Ale-Ebrahim, M., Mohseni-Moghaddam, P., Mortazavi, P., Mousavi, Z., & Asghari, A. (2023). Hepatoprotective and antifibrotic effects of trans-chalcone against bile duct ligation-induced liver fibrosis in rats. Iranian Journal of Basic Medical Sciences, 26(10), 1194.
- Johnson, A. C., & Greenwood-Van Meerveld, B. (2017). Critical evaluation of animal models of gastrointestinal disorders. Gastrointestinal Pharmacology, 289-317.
- Khalil, M. R., El-Demerdash, R. S., Elminshawy, H. H., Mehanna, E. T., Mesbah, N. M., & Abo-Elmatty, D. M. (2021). Therapeutic effect of bone marrow mesenchymal stem cells in a rat model of carbon tetrachloride induced liver fibrosis. Biomedical journal, 44(5), 598-610.
- Kim, E. R., Park, J. S., Kim, J. H., Oh, J. Y., Oh, I. J., Choi,D. H., ... & Lee, Y. H. (2022). A GLPâ?ÂÂ1/GLPâ?ÂÂ2 receptor dual agonist to treat NASH: Targeting the gut-liver axis and microbiome. Hepatology, 75(6), 1523-1538.
- Liu, M., Zhong, Y., Chen, J., Liu, Y., Tang, C., Wang, X., ... & Wei, B. (2020). Oral immunization of mice with a multivalent therapeutic subunit vaccine protects against Helicobacter pylori infection. Vaccine, 38(14), 3031-3041.
- National Institutes of Health. “Guiding Principles for Ethical Research.” (Reviewed June 10, 2025).
- Osborne, N., Avey, M. T., Anestidou, L., Ritskes-Hoitinga,M., & Griffin, G. (2018). Improving animal research reporting standards: HARRP, the first step of a unified approach by ICLAS to improve animal research reporting standards worldwide. The EMBO Reports, 19(5), EMBR201846069.
- Nevola, R., Epifani, R., Imbriani, S., Tortorella, G., Aprea, C., Galiero, R., ... & Sasso, F. C. (2023). GLP-1 receptor agonists in non-alcoholic fatty liver disease: current evidence and future perspectives. International Journal of Molecular Sciences, 24(2), 1703.
- Pu, T. Y., Chuang, K. C., Tung, M. C., Yen, C. C., Chen, Y.H., Cidem, A., ... & Chen, C. M. (2024). Lactoferrin as a therapeutic agent for attenuating hepatic stellate cell activation in thioacetamide-induced liver fibrosis. Biomedicine & Pharmacotherapy, 174, 116490.
- Wang, Z., Liu, T., Liu, L., Xie, J., Tang, F., Pi, Y., ... & Zheng,C. (2025). Lactobacillus vaginalis alleviates DSS induced colitis by regulating the gut microbiota and increasing the production of 3-indoleacrylic acid. Pharmacological Research, 213, 107663.