Research Article - (2024) Volume 7, Issue 1
Ameliorative Effect of Ferulic Acid against Cyclophosphamide Induced Behavioural and Biochemical Changes in Albino Mice
Received Date: Dec 11, 2023 / Accepted Date: Dec 29, 2023 / Published Date: Jan 10, 2024
Copyright: ©Â©2024 Vinay Kumar, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Kumar. V., Tyagi, H., Goyal, G., Varshney, P., Mishra, S. K. (2024). Ameliorative Effect of Ferulic Acid against Cyclophosphamide Induced Behavioural and Biochemical Changes in Albino Mice. Toxi App Phar Insig, 7(1), 01-08.
Abstract
Background: Cyclophosphamide (CP) is a distinguished anticancer and immune subduing medicament but it has neurotoxicity as a limitation.
Objective: The primary objective of this study was to describe the neuroprotective effects of ferulic acid in contradiction to cyclophosphamide-prompted neurotoxicity in the Swiss Albino mice.
Methodology: Mice were alienated into five groups (n=7) and treated with FA for fourteen days and a single dose of CP was administered on the seventh day. Animals were imperilled to neurobehavioral tests such as Rotarod test, Tail suspension test, and Y-maze test. On day fifteenth, animals were forgone and brain was detached and used for biochemical analysis.
Result: Cyclophosphamide administration likewise condensed the activity of antioxidant enzyme (GSH), and increased lipid peroxidation i.e., malondialdehyde (MDA) and pro-inflammatory cytokines (TNF-α and IL-1β). Furthermore, CP administration amplified the level of acetylcholine esterase (AChE).
Conclusion: The study showed a significant neuroprotective effect of ferulic acid and describe that it can be used as an adjuvant among chemotherapeutically treated patients.
Keywords
Cyclophosphamide, Ferulic Acid, Neurotoxicity, Blood-Brain Barrier, Neurobehavioral Test, Antioxidant.
Graphical Abstarct
Introduction
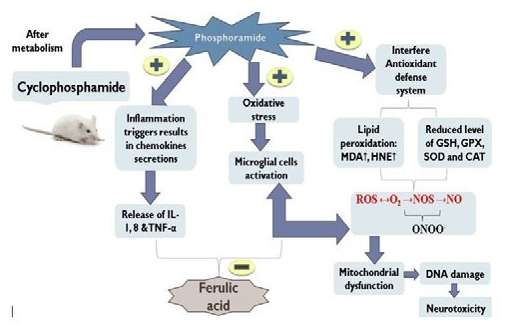
Neurotoxicity is one of the globally leading challenges in clinical science nowadays. In recent studies, it is found that toxicity in neurons is not only restricted to old age patients but it is also affecting children that are either prone to their lifestyle or any kind of chemical exposure. These inflammatory responses not only affect the brain but also affect the whole- body systemic circulation. Although neurotoxicity is self- limiting after the exposure stops but it can be progressive like other neurodegenerative diseases if the exposure continues [1, 2]. Cyclophosphamide (CP) is an immunosuppressant and anti-cancer drug that has alkylating chemical nature type and possesses a cytotoxic action mainly via adjoining the strands of DNA and RNA to inhibit abnormal protein synthesis. There are various adverse effects of cyclophosphamide including nephrotoxicity, hepatotoxicity, urotoxicity, cardiotoxicity, immune toxicity, mutagenicity, Geno toxicity, carcinogenicity, and teratogenicity [3]. Cyclophosphamide (CP) being an inactive pro-drug, its active metabolite (acrolein) has been proposed to be highly lipophilic moiety, crosses the blood-brain barrier, and exhibits significant neurotoxicity [4]. The signs and symptoms of a person affected by neuronal toxicity include loss of memory, weakness in the body, blurred vision, headache, mental confusion; most importantly cognitive and behavioural problems [5].
Ferulic acid (FA) is a well-known natural bioactive agent, which has hydroxyl cinnamic group as an active constituent, is extensively present in seeds, leaves and cereals possesses significant antioxidant as well as anti-inflammatory properties [6]. It is a compound of the phenolic acids category; has a chemical structural resemblance with curcumin which is indeed a potent neuroprotective molecule. Being a phenolic acid by nature it shows its various effects such as neuroinflammation, apoptosis, glutamate-tempted toxicity, seizures, unevenness after traumatic brain grievance, diabetic-induced memory impairment, hearing and vision disturbances, Huntington’s disease, anti-amyotrophic lateral sclerosis, and other fewer strewn disorders. Moreover, Ferulic acid has also been explored against Parkinson's, Alzheimer’s disease, and Ischemia-reperfusion [7]. As per recent studies; FA is known as a free radical scavenger due to its ability to inhibit free radical-generating enzymes. Therefore, in the current study, we have explored ferulic acid's neuroprotective role against CP-induced neurotoxicity and studied its neuroprotective potential against increased oxidative, neuroinflammation, and cognitive dysfunction [8, 9].
Materials and Methods
Drugs and chemicals
Cyclophosphamide (Endoxan®) was obtained from Baxter, oncology Department. Ferulic acid was procured from Sigma Aldrich, USA. ELISA kits for interleukins and cytokines (TNF-α, IL-10, IL-6, and IL-1β) were purchased from KRISHGEN Biosystems (Worli, Mumbai, India). All other chemicals and reagents used in the experiment were of analytical grade.
Animals
In this experimental protocol, Swiss Albino Mice (25-30 g) of either sex was secured from the Animal House Facility of KIET School of Pharmacy (Reg. No: 824/GO/BC/2004/ CPSCEA), Ghaziabad (UP). The experimental practice (IAEC/ KSOP/2021/07) was permitted by the Institutional Animal Ethics Committee (IAEC), KIET School of Pharmacy, Ghaziabad (UP). In harmony with typical laboratory circumstances, mice were retained in optimum temperature (22 ± 2ºC) with 50-70% relative humidity.
Experimental Design
Albino mice were divided in the 5 groups comprises of 7 mice/ group. Group 1 (i.e., control group) received normal saline for 14 days; animals in group 2, 3, and 4 were treated with single dose of cyclophosphamide (100 mg/kg; intraperitoneal [i.p.]) on 7th day of experiment for the induction of neurotoxicity. Animals of group 3 and group 4 were treated with FA (40 mg/ kg, 80 mg/kg; p.o.), for 14 days. Animals in group 5 (i.e., per se group) received only FA, 80 mg/kg; p.o. for 14 days.
Behavioural Studies
Rotarod Test, Tail Suspension Test (TST) and Y-Maze Test:-
After acclimatization, mice were incorporated into the rotarod apparatus. The falling time of mice was noted and compared. The comparison of dormancy was done between the control and test drug (Ferulic acid) [10-12].
Mice were individually hanged withhold tails, at a height of 40 cm with the help of adhesive tape. The Tail was on hold for a time of 6 minutes. During this time stillness of mice was evaluated. The time at which the mice became immobile was recorded [13, 14]. In Y-maze test, a floor with two open arms in dimensions of 35×5 cm2 and one another called a novel arm, mice were placed. The placement of mice was done in the middle of the maze and the head was confronted with an open arm. Firstly, mice were familiarized for around 5 minutes, after which overall number of entries was recorded [15, 16].
Biochemical estimations:- After the mice were sacrificed, brain was weighed and washed with the normal saline to remove the presence of blood and attached debris. Before proceeding for the biochemical estimation, brain tissues were mixed with phosphate buffered saline, homogenized and centrifugation at 8000 rpm for 5 minutes. The markers of oxidative stress, inflammation and neurotransmitters [17]. The lipid peroxidation was performed as per the method of where the level of MDA was estimated. Glutathione activity was evaluated as per the previous reported method and absorbance was recorded at 412 nm and values were expressed as µmol/mg of protein [18, 19].
Inflammatory Biomarkers and Acetylcholinesterase (AChE) Estimation
Commercially available ELISA kits were used the estimation of proinflammatory cytokines. Manufacturer’s instruction was followed for the experiment [20, 21]. To the mixture of the sectioned brain tissue, acetylcholine iodide along with DTNB (5,5-dithio-bis-(2-nitrobenzoic acid) was added. This whole mixture was kept in phosphate buffer with maintained pH. Incubation of 10 min at a temperature of 37ºC was done. Add serine hemi sulphate (500 μl) to it. Check absorbance at 412 nm [22].
Statistical analysis:- One-way ANOVA (one-way analysis test) followed by Turkey’s post hoc test (SPSS 20 version) was used for the statistical analysis and P < 0.05 was considered as significant. Values were presented as Mean ± SEM of different group.
Results
Effect of Ferulic Acid on Cyclophosphamide-Induced Altered Grip Strength
Upon treatment with cyclophosphamide in mice, there was a huge alteration in muscle grip strength as compared to normal saline-treated mice. An i.p. administration of cyclophosphamide considerably (###p<0.001) declined grip strength as compared to normal mice. However, a dose of 40 mg/kg FA has failed to exhibit any protective effect on grip strength against CP. Conversely, a dosage of 80 mg/kg has been found significant (*p<0.01) against CP. A measured quantity of 80 mg/kg was found as important (**p<0.008) as that of 40 mg/kg of FA as shown in fig.1.
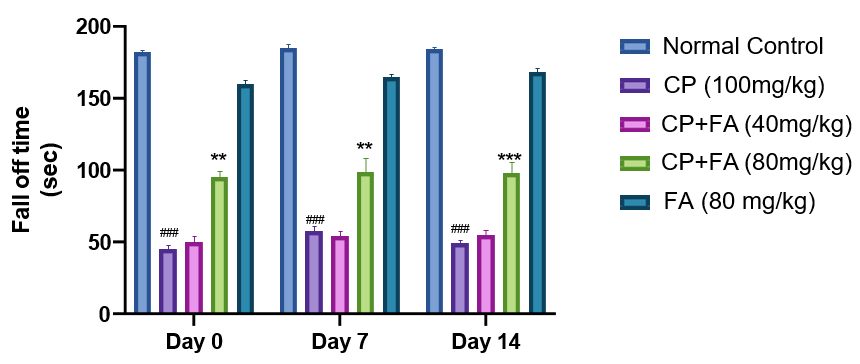
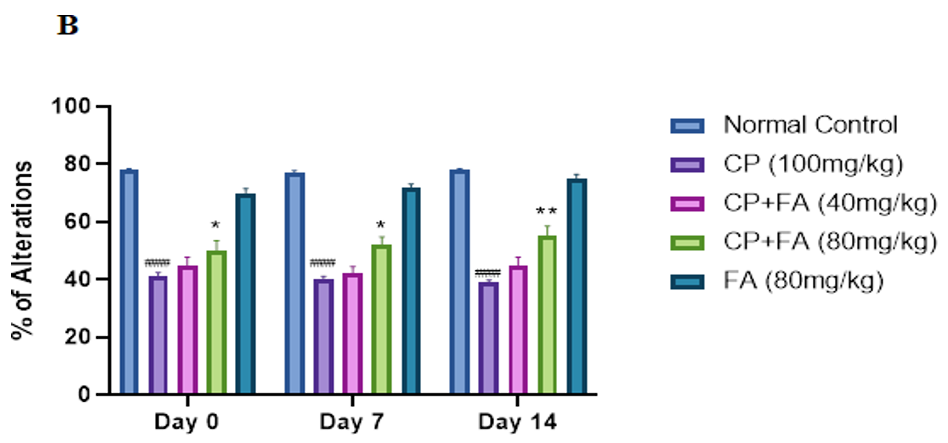
Figure 2: Role of FA on CP-Tempted Changes in Arm Entries
(A) and Percentage of Alterations
(B) During Y-Maze Test.
Graph denotes mean ± SEM (n=7). ####p<0.001 related to control group; ***p<0.001 related to cyclophosphamide brought neurotoxic group.
Effect of Ferulic Acid on Cyclophosphamide-Induced Immobility
The duration of immobility was significantly increased in mice when treated with cyclophosphamide (####p<0.001) from initial days onwards in comparison to normal control. Administration of FA (40 mg/kg and 80 mg/kg) effectively reduced (**p<0.001) the duration of immobility when administered along with CP (Fig 3).
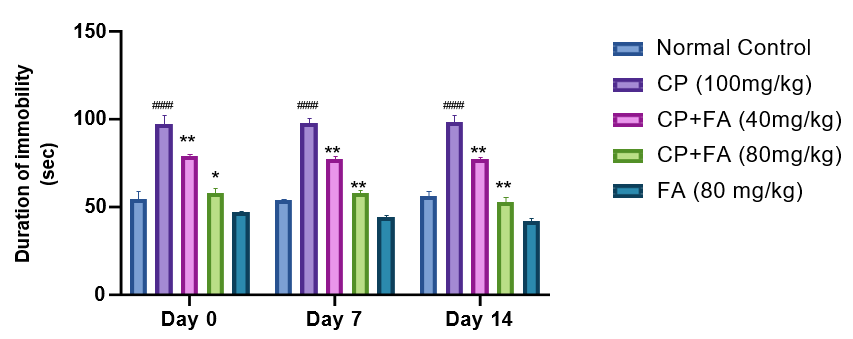
Figure 3: Consequence of FA on CP-Persuaded Changes in Immobility of Mice via TST.
Data depicts mean ± SEM (n=7). ####p<0.001 as opposed to normal group; (**p<0.001) associated to Cyclophosphamide-induced group.
Effect of Ferulic Acid on Cyclophosphamide-Induced Differences in Lipid Peroxidation Levels
After i.p. administration, cyclophosphamide significantly (##p<0.001) increased the MDA side by side in both hippocampus and frontal cortex of cyclophosphamide-treated mice that of the normal group.In contrast, the level of MDA was decreased (*p<0.03 and **p<0.001) in the ferulic acid (40 mg/ kg and 80mg/kg) treated group in brain sections. As revealed in (Fig. 4), no significant effect was observed in MDA levels in any of the brain regions in mice treated with the highest dose of ferulic acid in comparison to normal control mice.
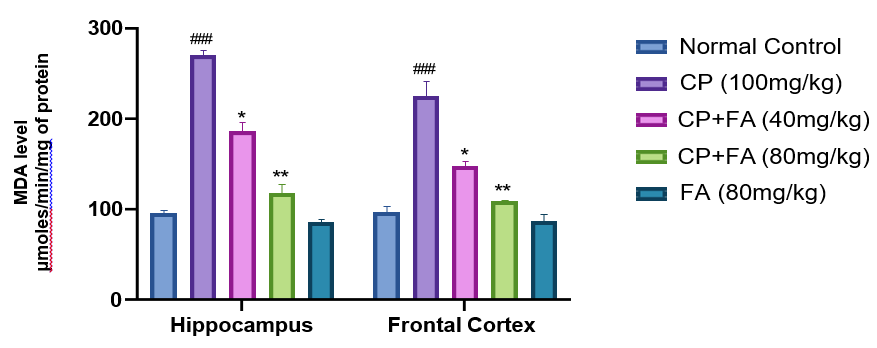
Figure 4: Consequence of FA on CP-Persuaded Changes in Levels of MDA for Evaluation of MDA Scores in the Hippocampus and Frontal Cortex of Mice Brain. Data depicts mean ± SEM (n=7). ##p<0.001 as opposed to normal group; *p<0.03, **p<0.001 associated to Cyclophosphamide- induced group.
Effect of Ferulic Acid on Cyclophosphamide-Induced Differences in Gsh Levels
Cyclophosphamide treated group signifies the major (##p<0.001) diminution in the GSH in both hippocampus and frontal cortex of mice. Together with this ferulic acid (80 mg/ kg) group shown a slight (*p<0.01) upturn in the level of GSH both sections in contrast to the cyclophosphamide-treated group (Fig. 5). In distinction, no weighty effect was noted in ferulic acid (40 mg/kg) in comparison to the cyclophosphamide treated group. Similarly, when matched to the cyclophosphamide group 80 mg/kg of FA was able to elevate GSH levels significantly (**p<0.001).
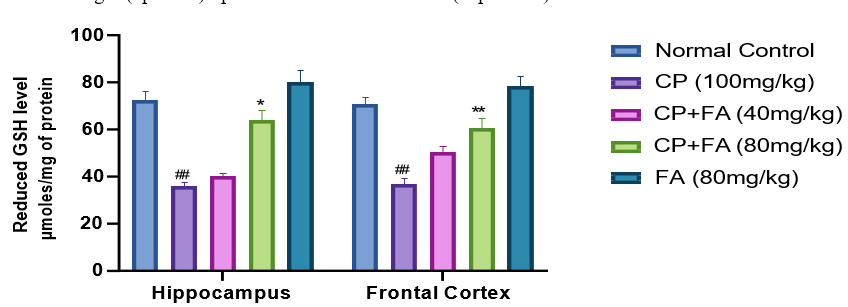
Figure 5: Result of FA on CP-Persuaded Variations in the Level of GSH for Determination of Alteration in Antioxidant Activity in Hippocampus and Frontal Cortex of Mice Brain.
Figures characterize mean ± SEM (n=7). ##p<0.001 related to normal group; *p<0.01 and **p<0.004 related to Cyclophosphamide- induced group.
Effect of Ferulic Acid on Cyclophosphamide-Induced Differences in Ache Levels
A substantial (###p<0.0006) decrease in the activity of AChE was perceived in the hippocampus and frontal of mice on i.p. administration of cyclophosphamide as compared to normal control. A quantity of FA (40 mg/kg) indicated significant (**p<0.001) enhancement in AChE activity. Simultaneously, the ferulic acid treated (80 mg/kg) group showed a noteworthy growth (****p<0.0001) in activity of AChE in both sections (Fig. 6). As a result, it is clear that acetylcholine helps in memory formation but its destruction causes cognitive deficits by AChE. Ferulic acid diminishes cyclophosphamide-induced neurotoxicity through AChE inhibition.
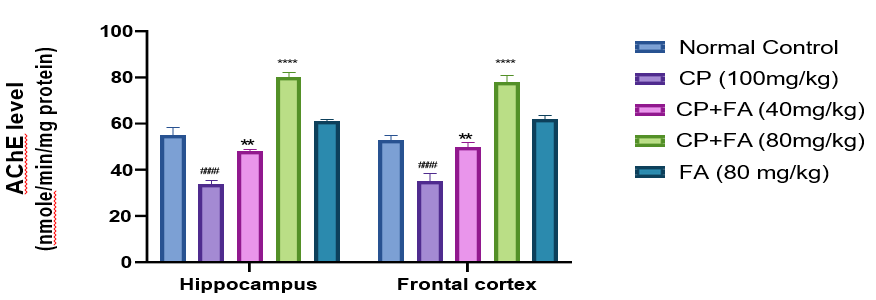
Figure 6: Result of FA on CP-Persuaded Changes in Acetyl Cholinesterase Action in Both Sections.
Records denotes mean ± SEM (n=7). ###p<0.0006 matched to normal group; ****p<0.0001 and **p<0.001compared to cyclophosphamide-induced neurotoxicity group.
Effect of Ferulic Acid on Cyclophosphamide-Induced Differences Inflammatory Biomarkers (Il-1β and Tnf-Α) Levels.
A significant upsurge in the level of pro-inflammatory markers (TNF-α & IL-1β) in the brain tissue of mice was found with the administration of cyclophosphamide (##p<0.003) as compared to normal control. Administration of FRA 80 mg/kg successfully abridged the level of TNF-α (*P<0.01), and IL-1β (*p <0.01), respectively. On the other hand, 40 mg/kg of FA was comparatively less effective in reducing inflammation. However, administration of FA 80 mg/kg, exhibits an anti-inflammatory effect (*p<0.003) to reduce neuroinflammation induced by cyclophosphamide (Fig. 7).
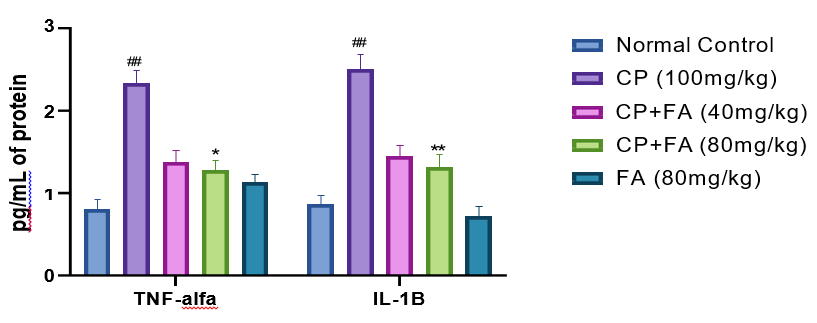
Figure 7: Influence of FA on CP-Induced Changes in Inflammatory Biomarkers for Evaluation of Inflammation in the Frontal Cortex of Mice.
Picture signifies mean ± SEM (n=7). ##p<0.003 related to control group; *p<0.01 and **p<0.003 related to cyclophosphamide- induced neurotoxicity group.
Discussion
Cyclophosphamide is an anti-neoplastic agent that acts against carcinoma cells. Apart from its therapeutic effects, it also shows toxic adverse effects. The neurotoxic effect of cyclophosphamide is due to the formation of aldophosphamide which is easily capable to cross BBB and cause inflammation in the brain [23, 24].
Therefore, the present study results provide substantial evidence to those previous studies of neuroprotection offered by ferulic acid [25]. The epidemiological analysis reported that cyclophosphamide exposure can lead to a deterioration in cognitive tasks [26]. Numerous underlying mechanisms of cyclophosphamide-induced behavioural deficits have been elucidated till now [27-29].
In this instant experiment, the rota rod test demonstrated that administration of cyclophosphamide for 14 days causes a significant decline in motor coordination. Alternatively, concomitant dealing with ferulic acid (80 mg/kg) showed significant improvement in the fall-off time of mice, indicating better motor coordination and grip strength. In addition, ferulic acid-treated mice had a more durable fall-off time. The observed protective action of ferulic acid further supports the previous reports [30]. In Y-maze test, we found that cyclophosphamide administration bases a fall in entries in the arm and the percentage of spontaneous alteration in mice. An enhancement in spontaneous alteration was perceived in mice treated with 80 mg/kg of ferulic acid. Other studies are also in line with some previous findings [31-33].
During TST analysis immobility was done in treated mice as a sign of cognitive impairment. It was found that cyclophosphamide administration leads to immobility in mice whereas ferulic acid reverses it. Accordingly, behavioural upshots of the present study concluded that cyclophosphamide-exposed mice showed decreased grip strength, spatial learning deficits, and cognitive impairment. Ferulic acid treatment significantly ameliorated the cognitive function in cyclophosphamide-exposed mice, suggesting its neuroprotective potential. These study outcomes are corroborated with the earlier reports [34, 35].
The acetyl cholinesterase (AChE) level in the frontal cortex and hippocampus was assessed in the present study to assess the cholinergic function. In this study, it was analysed that the AChE level was condensed in both hippocampus and frontal cortex. However, (80 mg/kg) ferulic acid effectively restored the cyclophosphamide-induced AChE depletion. Previous studies are also analogous to the verdicts of an extant testings had also shown a rise in levels of AChE level after administration of ferulic acid [36, 37]. Thus, the protection conferred by ferulic acid to reverse memory deficits and motor coordination may be mediated through the alleviation of cholinergic dysfunction. Cyclophosphamide induces oxidative stress indirectly via the reduction of endogenous antioxidants such as GSH and other sulfhydryl group-containing enzymes [38]. In the present investigation, cyclophosphamide has shown significant inflammatory biomarkers such as (IL-1β and TNF-α) in the frontal cortex. The direction of ferulic acid (80 mg/kg) was significantly able to reduce inflammatory biomarkers levels. These effects are harmonized with prior experiments [39, 40].
The current experimental study bares the conspicuous engrossment of oxido-nitrosative stress, cholinergic dysfunction, and inflammation in the brain. In addition, it has been found that ferulic acid showed its neuroprotective effect against cholinergic alteration, oxido-nitrosative stress, and lipid peroxidation, inflammation in cyclophosphamide-treated mice.
Conclusion
In conclusion, cyclophosphamide neurotoxicity has arbitrated through cholinergic affliction, oxidative stress, and inflammation in mice brains. A diminution of oxidative stress by curtailing the stages of MDA and inflammatory markers and by cumulative the levels of anti-oxidant markers explicitly GSH was renowned in ferulic acid. The cyclophosphamide-induced neurochemical anomalies were eventually prime to memory deficits. A co- administration of ferulic acid (80 mg/kg) effectively exhibited antioxidant potential, whereas ferulic acid (40 mg/kg) didn’t show a significant antioxidant effect. Further studies are requisite to explicate the more meticulous appliance underlying cyclophosphamide-induced neurotoxicity.
Acknowledgement
Authors are thankful to Director, KIET Group of Institutions (KIET School of Pharmacy) for the moral support to conduct the study.
References
- Zajazkowska, R., Kocot-epska, M., Leppert, W., Wrzosek, A., Mika, J., & Wordliczek, J. (2019). Mechanisms of chemotherapy-induced peripheral neuropathy. International journal of molecular sciences, 20(6), 1451.
- Giordano, G., & Costa, L. G. (2012). Developmental neurotoxicity: some old and new issues. International Scholarly Research Notices, 2012.
- Ponticelli, C. (2012). Tonsillectomy and IgA nephritis. Nephrology Dialysis Transplantation, 27(7), 2610-2613.
- McCarty, M. F. (2021). Nutraceutical, dietary, and lifestyle options for prevention and treatment of ventricular hypertrophy and heart failure. International journal of molecular sciences, 22(7), 3321.
- Ware, M. R., Feller, D. B., & Hall, K. L. (2018). Neuroleptic malignant syndrome: diagnosis and management. The primary care companion for CNS disorders, 20(1), 27030.
- Antonopoulou, I., Sapountzaki, E., Rova, U., & Christakopoulos, P. (2022). Ferulic acid from plant biomass: A phytochemical with promising antiviral properties. Frontiers in Nutrition, 8, 777576.
- Paiva, L. B. D., Goldbeck, R., Santos, W. D. D., & Squina,F. M. (2013). Ferulic acid and derivatives: molecules with potential application in the pharmaceutical field. Brazilian Journal of Pharmaceutical Sciences, 49, 395-411.
- Albuquerque, B. R., Heleno, S. A., Oliveira, M. B. P., Barros, L., & Ferreira, I. C. (2021). Phenolic compounds: Current industrial applications, limitations and future challenges. Food & function, 12(1), 14-29.
- Zdunska, K., Dana, A., Kolodziejczak, A., & Rotsztejn, H. (2018). Antioxidant properties of ferulic acid and its possible application. Skin pharmacology and physiology, 31(6), 332- 336.
- Henry, R. J., Meadows, V. E., Stoica, B. A., Faden, A. I., & Loane, D. J. (2020). Longitudinal assessment of sensorimotor function after controlled cortical impact in mice: comparison of beamwalk, rotarod, and automated gait analysis tests. Journal of neurotrauma, 37(24), 2709-2717.
- Deacon, R. M. (2013). Measuring motor coordination in mice. JoVE (Journal of Visualized Experiments), (75), e2609.
- Rozas, G., Guerra, M. J., & Labandeira-Garcia, J. L. (1997). An automated rotarod method for quantitative drug- free evaluation of overall motor deficits in rat models of parkinsonism. Brain Research Protocols, 2(1), 75-84.
- Bouet, V., Boulouard, M., Toutain, J., Divoux, D., Bernaudin, M., Schumann-Bard, P., & Freret, T. (2009). The adhesive removal test: a sensitive method to assess sensorimotor deficits in mice. Nature protocols, 4(10), 1560-1564.
- Can, A., Dao, D. T., Terrillion, C. E., Piantadosi, S. C., Bhat, S., & Gould, T. D. (2012). The tail suspension test. JoVE (Journal of Visualized Experiments), (59), e3769.
- Gudapati, K., Singh, A., Clarkson-Townsend, D., Phillips,S. Q., Douglass, A., Feola, A. J., & Allen, R. S. (2020). Behavioral assessment of visual function via optomotor response and cognitive function via Y-maze in diabetic rats. JoVE (Journal of Visualized Experiments), (164), e61806.
- Kraeuter, A. K., Guest, P. C., & Sarnyai, Z. (2019). The Y-maze for assessment of spatial working and referencememory in mice. Pre-clinical models: Techniques and protocols, 105-111.
- Adefegha, S. A., Oyeleye, S. I., Akintemi, A., Okeke, B. M., & Oboh, G. (2020). Thyme (Thymus vulgaris) leaf extract modulates purinergic and cholinergic enzyme activities in the brain homogenate of 5-fluorouracil administered rats. Drug and chemical toxicology, 43(1), 43-50.
- Mseddi, M., Ben Mansour, R., Mnif, F., Gargouri, B., Abid, M., Guermazi, F., ... & Lassoued, S. (2015). Lipid peroxidation, proteins modifications, anti-oxidant enzymes activities and selenium deficiency in the plasma of hashitoxicosis patients. Therapeutic advances in endocrinology and metabolism, 6(5), 181-188.
- Alavala, S., Nalban, N., Sangaraju, R., Kuncha, M., Jerald,M. K., Kilari, E. K., & Sistla, R. (2020). Anti-inflammatory effect of stevioside abates Freund’s complete adjuvant (FCA)- induced adjuvant arthritis in rats. Inflammopharmacology, 28, 1579-1597.
- Amsen, D., de Visser, K. E., & Town, T. (2009). Approaches to determine expression of inflammatory cytokines. Inflammation and Cancer: Methods and Protocols: Volume 1: Experimental Models and Practical Approaches, 107-142.
- Benabent, M., Vilanova, E., Sogorb, M. Á., & Estévez,J. (2014). Cholinesterase assay by an efficient fixed time endpoint method. MethodsX, 1, 258-263.
- Iqubal, A., Syed, M. A., Haque, M. M., Najmi, A. K., Ali, J., & Haque, S. E. (2020). Effect of nerolidol on cyclophosphamide-induced bone marrow and hematologic toxicity in Swiss albino mice. Experimental Hematology, 82, 24-32.
- Ogino, M.H., Tadi, P. (2023). Cyclophosphamide. [Updated 2023 Jul 3]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing.
- Gupta, A., Singh, A. K., Loka, M., Pandey, A. K., & Bishayee,A. (2021). Ferulic acid-mediated modulation of apoptotic signaling pathways in cancer. Advances in protein chemistry and structural biology, 125, 215-257.
- Moore, M. J. (1991). Clinical pharmacokinetics of cyclophosphamide. Clinical pharmacokinetics, 20(3), 194-208.
- Brown, T., McElroy, T., Simmons, P., Walters, H., Ntagwabira, F., Wang, J., ... & Allen, A. R. (2021). Cognitive impairment resulting from treatment with docetaxel, doxorubicin, and cyclophosphamide. Brain research, 1760, 147397.
- Alhowail, A. H., Chigurupati, S., Sajid, S., & Mani, V. (2019). Ameliorative effect of metformin on cyclophosphamide- induced memory impairment in mice. European Review for Medical & Pharmacological Sciences, 23(21).
- Tabuchi, H., Kitamura, Y., Ushio, S., Kan, S., Wada, Y., Sumiyoshi, Y., ... & Sendo, T. (2021). Influence of 5-HT 2A receptor function on anxiety-like behavior induced by a combination treatment with doxorubicin and cyclophosphamide in rats. Psychopharmacology, 238, 3607-3614.
- Sohn, Y. T., & Oh, J. H. (2003). Characterization of physicochemical properties of ferulic acid. Archives of pharmacal research, 26, 1002-1008.
- Cheng, Q., Li, C., Yang, C. F., Zhong, Y. J., Wu, D., Shi, L.,... & Li, L. (2019). Methyl ferulic acid attenuates liver fibrosis and hepatic stellate cell activation through the TGF-β1/Smad and NOX4/ROS pathways. Chemico-biological interactions, 299, 131-139.
- Mishra, T., Nagarajan, K., Dixit, P. K., & Kumar, V. (2022). Neuroprotective potential of ferulic acid against cyclophosphamide-induced neuroinflammation and behavioral changes. Journal of Food Biochemistry, 46(12), e14436.
- Chen, J., Wang, Y., Wang, S., Zhao, X., Zhao, L., & Wang,Y. (2022). Salvianolic acid B and ferulic acid synergistically promote angiogenesis in HUVECs and zebrafish via regulating VEGF signaling. Journal of ethnopharmacology, 283, 114667.
- Mori, T., Koyama, N., Tan, J., Segawa, T., Maeda, M., & Town, T. (2019). Combined treatment with the phenolics (−)-epigallocatechin-3-gallate and ferulic acid improves cognition and reduces Alzheimer-like pathology in mice. Journal of Biological Chemistry, 294(8), 2714-5444.
- Michels, B., Zwaka, H., Bartels, R., Lushchak, O., Franke, K., Endres, T., ... & Gerber, B. (2018). Memory enhancement by ferulic acid ester across species. Science advances, 4(10), eaat6994.
- Allegrini, G., Di Desidero, T., Barletta, M. T., Fioravanti, A., Orlandi, P., Canu, B., ... & Bocci, G. (2012). Clinical, pharmacokinetic and pharmacodynamic evaluations of metronomic UFT and cyclophosphamide plus celecoxib in patients with advanced refractory gastrointestinal cancers. Angiogenesis, 15, 275-286.
- Li, B., Li, W., Tian, Y., Guo, S., Qian, L., Xu, D., & Cao,N. (2020). Selenium-alleviated hepatocyte necrosis and DNA damage in cyclophosphamide-treated geese by mitigating oxidative stress. Biological trace element research, 193, 508- 516.
- Ibrahim, H. M., Mohammed-Geba, K., Tawfic, A. A., & El-Magd, M. A. (2019). Camel milk exosomes modulate cyclophosphamide-induced oxidative stress and immuno- toxicity in rats. Food & function, 10(11), 7523-7532.
- Sawaki, A., Miyazaki, K., Yamaguchi, M., Takeuchi, T., Kobayashi, K., Imai, H., ... & Katayama, N. (2020). Genetic polymorphisms and vincristine-induced peripheral neuropathy in patients treated with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone therapy. International journal of hematology, 111, 686-691.
- Liu, Y. M., Shen, J. D., Xu, L. P., Li, H. B., Li, Y. C., & Yi, L.T. (2017). Ferulic acid inhibits neuro-inflammation in mice exposed to chronic unpredictable mild stress. International Immunopharmacology, 45, 128-134.
- Rinwa, P., & Kumar, A. (2017). Quercetin along with piperine prevents cognitive dysfunction, oxidative stress and neuro-inflammation associated with mouse model of chronic unpredictable stress. Archives of pharmacal research, 40, 1166-1175.


