Research Article - (2025) Volume 10, Issue 2
A Cross Sectional Retrospective Study on Seroprevalence of Chikungunya in Patients Visiting a Tertiary Health Care Centre in Middle Assam, India
2Research Assistant, Department of Microbiology, Tezpur Medical College, Tumuki, Tezpur, Sonitpur, Assam, India
3Professor, Department of Microbiology, Pragjyotish Medical College, Guwahati, India
4Research Scientist (Medical), Department of Microbiology, Tezpur Medical College, Tumuki, Tezpur, Sonitpur Assam, India
5Research Scientist (Non-Medical), Department of Microbiology, Tezpur Medical College, Tumuki Tezpur, India
Received Date: Sep 09, 2025 / Accepted Date: Oct 03, 2025 / Published Date: Oct 10, 2025
Copyright: ©Â©2025 Kaushik Das, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Biswas, S. Das, K., Alam, S. T., Begum, S., Pathak, M. (2025). A Cross Sectional Retrospective Study on Seroprevalence of Chikungunya in Patients Visiting a Tertiary Health Care Centre in Middle Assam, India. J Clin Exp Immunol, 10(2), 01-05.
Abstract
Background: The arthropod-borne Chikungunya virus (CHIKV) has the potential to cause widespread outbreaks. Although the presence of CHIKV and its circulation among the Indian population have been documented in many Indian states since its re-emergence in 2005. Testing facilities for CHIKV were scarce in the majority of locations during the COVID-19 outbreak. Chikungunya fever is still thought to be a significant public health concern that results in inflammatory polyarthropathy as a morbidity. For this reason, particularly in the wake of the COVID-19 pandemic, we must comprehend and assess the seroprevalence of chikungunya in relation to its research period, seasonal tendency, hospitalization needs, and age-sex distribution.
Methods: This study was done to evaluate the seroprevalence, clinical presentation of CHIKV in the districts of Middle Assam. The samples were tested by CHIKV –IgM Elisa Assay which is developed in-house by NIV Pune.
Results: Our study found that Chikungunya virus (CHIKV) affects all age groups, with no mortality but high morbidity. Neurological symptoms were present in 6.81% of positive cases. The most affected group was 21-40 years old. Most cases were from rural settings, likely due to poor economic and sanitary conditions, and areas suitable for Aedes mosquitoes.
Conclusions: No prior study of ChikV data from the region of middle Assam, India. found that ChikV affects humans of all age group but the most affected age group in our study was 21-40 years. Though no mortality has been reported in our study, but there was high moribidity. we also report the presence of Neurological symptoms in 6.81% of total positive cases.
Keywords
Elisa, Igm, Chikungunya, Seropositivity, Loss of Public Health, Middle Assam
List Of Abbreviations
NIV- National Institue of Virology,
ICMR- Indian Council of Medical Research,
JE- Japanese Encephalitis,
CHIKV- Chkiungunya virus,
IgM- Immunoglobulin M,
PCR- Polymerase Chain Reaction,
ELISA- Enzyme Linked Immuno Sorbent Essay.
Introduction
Togoviridae, Chikungunya virus causes the CHIK fever disease in Humans and Animals. ChikV is epidemiologically the most prevalent alphavirus that is transmitted to humans by Aedes Mosquitoes Athralgia, fever, chills, headache, nausea, vomiting, low back pain and also sometimes rash that lasts upto 7 days are its usual symptoms. Rarely fatal, the disease is often self-limiting [1]. The incubation period is usually 2-3 days. Although Chikungunya was first reported in Assam in 2008 [2]. The presence of vector mosquitoe in abundance carries the risk of its circulation to multiple areas Hence this study was conducted to detect its prevalence pattern in the districts of Middle Assam [2,3].
The enzyme-linked immunosorbent assay (ELISA) test used in this study was developed in-house by NIV, Pune. Laboratory diagnosis depends on the quality of sample and time of collection in the course of illness. In first 5 days, viremia is present and can be confirmed by viral culture, Polymerase chain reaction (PCR) or antigen detection. CHIK IgM becomes detectable around after 5 days of fever and persists for several months and Immunoglobin G (IgG) is present by 10-14 days [4]. Serological diagnosis of CHIK by detecting IgM or IgG seroconversion is widely used because it is cheaper and easier to perform [4]. The enzyme- linked immunosorbent assay (ELISA) test used in this study was developed in-house by NIV, Pune. Laboratory diagnosis depends on the quality of sample and time of collection in the course of illness.. Serological diagnosis of CHIK by detecting IgM or IgG seroconversion is widely used because it is cheaper and easier to perform. But the disadvantage of antibody testing is that as IgM persists for months a single-raised IgM may not indicate acute infection and there is a possibility of cross-reactivity with other alphaviruses, using ELISA and Immunochromatography Assay (ICA) to detect antigen antigen is a better alternative [4-9].
Materials and Methods
A total of 432 Serum samples received in the department of microbiology that tested negative for Dengue and JE IgM ELISA from various districts of Middle Assam were included in the study from the month of October 2018 till May 2022.
The samples were tested for dengue and JE IgM antibody using IgM antibody capture ELISA produced by NIV first and those who tested negative were subsequently tested for CHIKV using the same IgM antibody capture ELISA kit by NIV (Arbovirus Diagnostic NIV, Pune, India). The sensitivity and specificity for the CHIK IgM antibody capture ELISA is 95.00% and 97.22%, respectively, and for dengue IgM antibody capture ELISA is 98.53% and 98.84%, respectively. The tests were carried out following the manufacturer instruction.
Principle of IgM Capture ELISA for CHIK: IgM antibodies in the patient's blood are captured by anti-human IgM (μ chain specific) that are coated on to the solid surface (wells). In the next step, CHIK antigen is added, which binds to captured IgM, if the IgM and antigen are homologous. Unbound antigen is removed during the washing step. In the subsequent steps Biotinylated anti-CHIK monoclonal antibody (CHIK-B) is added followed by Avidin- Histidine rich protein (HRP). Subsequently, substrate/chromogen is added and monitored for development of color. The reaction is stopped by 1NH2SO4. The intensity of color/optical density (OD) is monitored at 450 nm. OD values are directly proportional to the amount of CHIK virus specific IgM antibodies present in the sample. The sample was considered positive for IgM antibody if the OD of the sample exceeds OD of negative control by a factor 4.0 (sample OD ≥ negative OD × 4.0). Both positive and negative controls were used to validate the test.
Study Area
This was a retrospective study done with participants, mainly patients visiting tertiary medical college, Tezpur Medical College & Hospital, Assam from the districts of Sonitpur, Biswanath Chariali, Nagaon and Udalguri (Middle Assam).
The four districts geographically located in the middle part of assam, experiences hot and humid climate during summers (33- 38 degree). Vast numbers of paddy cultivation takes place across these districts, which makes the land a suitable breeding ground for mosquitoes during summers.
Participants
In this study patients with acute febrile illness visiting the tertiary medical care centre, Tezpur Medical College & Hospital, showing symptoms ranging from mild to severe were included. Patients with symptoms of CHIKV infection such as fever, body pain, vomiting, arthralgia, headache, haemorrhagic fever etc were included in this study. Patients were randomly selected into six groups, (6-10), (11-20), (21-30), (31-40), (41-50), (>50) to study which age group was affected the most. Patients who were <5 years of age and pregnant woman were excluded from the study. To study the distribution of cases across the year seasonally, we divided the months into 4 quarters across a year. January-March, April-June, July- September, October- December.
Results
A total of 432 samples were tested for CHIK V from the month of May 2018 to December 2022. Out of which 44 tested positive (10.185%) and 388 tested negatives. (Table 1) An attempt was made to study any correlation between age groups and positivity and severity of CHIK V samples as mentioned in table 1 as to find out which age groups were affected the most. Majority of the positive cases were from the age group of (21-30) & (31-40), 49.99% from the total no. of positives (Table 1).
Out of the the 44 total tested positive, 61.36% (27) of cases were males, and females accounted for 38.63% (17) of the cases (Figure 1).
|
Age group |
Samples |
Positive |
Negative |
%positivity |
% positive from total |
|
6-10 Years |
22 |
2 |
20 |
9.09 |
4.54 |
|
11-20 Years |
59 |
5 |
55 |
6.779 |
11.36 |
|
21-30 Years |
139 |
13 |
125 |
9.35 |
29.54 |
|
31-40 Years |
73 |
9 |
64 |
12.32 |
20.45 |
|
41-50 Years |
58 |
6 |
52 |
10.34 |
13.63 |
|
>50 |
81 |
9 |
72 |
11.11 |
20.45 |
|
Total |
432 |
44 |
388 |
|
|
Table 1: Weighted Distribution of Seroprevalence of CHIKV Igm Antibodies
In our study the areas, rural areas accounted for the maximum number of positive cases at 26, followed by urban at 12, Tea estates at 2, and semi urban areas at 1, and if Tea estates are to be included in rural areas, which is mostly the case in districts of Assam and in our study, rural areas accounted for 28 cases out of the 44 total positive cases. All of the samples tested negative for Dengue virus and Japanese encephalitis IgM Antibody. Fever was present in 100% the positive cases, while 36.36% had headache, and 22.72% cases had athralgia as symptoms. Neck rigidty and change in mental status was seen in 6.81% of cases, while abdominal discomfort was seen in 6.81 % of the cases.
While the seasonal distribution of seropositive cases showed the period of July- September had the greatest number of cases from 2018-2022, at 56.81 % (Figure 2).
|
Symptoms |
Total cases |
Percentage |
|
Fever |
44 |
100 |
|
Cough |
4 |
9.09 |
|
Bodyache |
10 |
22.72 |
|
Headache |
16 |
36.36 |
|
Abdominal Pain |
3 |
6.81 |
|
Neck Rigidity |
3 |
6.81 |
|
Change in Mental Status |
3 |
6.81 |
Table 2: Clinical Manifestations Chik-Igm Positive Cases.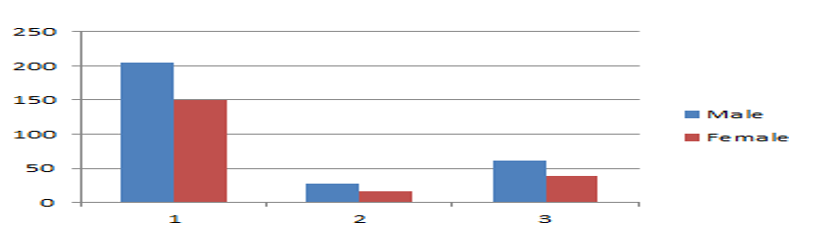
Figure 1: Gender Wise Distribution
Gender Wise Distribution, 1) Total Sample tested 2) Total positive 3) Percentage Positivity w.r.t total number of positive samples.
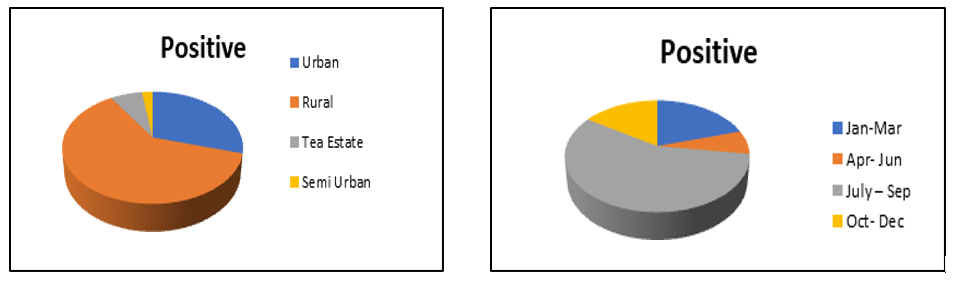
Figure 2: Location and Month Wise Distribution
Location and Month wise distribution of Positive cases.
Discussion
Humans can contract CHIKV, a flaviviral infection, by being bitten by an infected female Aedes mosquito. Since the virus was initially discovered in 1952–1953 isolated cases and significant epidemics of CHIKV infection have been documented in Africa12, India13, Southeast Asia, and the Western Pacific14. Following more than three decades of resurgence, CHIKV infection resurfaced in India in 2006. Assam is affected by various vector borne diseases like malarial, Japanese encephalitis, Dengue. Both Dengue and Chikungunya shows similar symptoms upon infection and Chikungunya diagnosis was often lacking. Since Assam is abundant in Aedes mosquitoes, the possibility of Chikungunya virus circulating in Assam from a very long time cannot be ignored. The first case of CHIKV from Assam was reported back in 2008 as reported by Dutta and colleagues [2].
In our present study, patients visiting a tertiary medical centre were surveyed who belonged to the districts of Sonitpur, Biswanath, Udalguri, and Nagaon of middle Assam. It was observed that the age group that was most affected was found to be 21-30, (29.54%), followed by 31-40, (20.45%). Similar patterns were also reported by Dutta, and Mohanty Since P-value 0.9749 is, there is no statistical significance of CHIKV seropositivity and correlation with age [3,4].
The reason behind the high prevalence of positivity might come down to the fact that most of the people involved in outdoor activ- ities belong to the aforementioned age groups. We also observed in our study that the males were affected more at 61.36 %. Also, the percentage of seropositivity in males was observed to be higher in males (61.36 %) than females (38.63%) Although there were no statistical significance for the outcome in differences across gender, the above disparity may exist due to frequent exposure to males while working and travelling hinting us about the idea of community specific habits, behaviours, and customs.
It was observed that fever was present in all the cases, while Head- ache was present in 36.36 % cases, while arthralgia inspite of be- ing a typical symptom of chikungunya was present only in 22.72% of cases (Table 2). 6.81% of cases also showed Neck rigidity and Change in mental status (each) as symptoms. Although neurological Manifestations of Chikungunya are rare and its presence of neurological symptoms doesn’t conclusively establish the virus being etiological agent affecting the central nervous system, but in some cases of Chikungunya infection, neurological symptoms have been reported and its reporting have increased in the past years [10,11]. One limitation of our study is that it’s also common sometimes for the neurological symptoms to manifest because of co-infections with other viruses such as West Nile [10-12].
Also, the highest prevalence of seropositive cases was found in the rural areas of the 4 districts, at 63.63%. While the highest cases came during the July-September period at 56.81%, which is also the period when Assam receives heavy rainfall. resulting in the increase of the vector density. Again, it may be indicated to the presence of large number of paddy cultivation and swampy lands in the rural areas which acts as a breeding ground for the mosquitoes during the rainy season resulting in the increase of the mosquitoe population [3].
Conclusion
In conclusion, our objective was to study the seroprevalence of chikungunya virus infection, its age sex distribution and seasonal tendencies in districts of middle assam and to observe if there is any pattern in the distribution of cases. It is found that ChikV affects humans of all age group but the most affected age group in our study was 21-40 years. Though no mortality has been reported in our study, but there was high moribidity. Though we also report the presence of Neurological symptoms in 6.81% of total positive cases, one of the limitations of our study is that only those samples were taken into consideration which tested negative for Dengue and JE Igm Antibody, but not other AES causing agents eg, West Nile HSV etc.
As the most affected group is 21-40, which constitutes most of the working population, there is loss of work associated with it. As there is no herd immunity to the virus, there is a risk of the virus spreading to new areas in the districts, followed by further spread in nearby districts as well. While most of the cases came from rural settings, the possible reason could be low economic conditions, poor sanitary conditions and availibility of areas suitable for the presence of Aedes mosquitoes. Therefore, screening of CHIKV and other arboviruses is highly necessary, for awareness and prevention of possible future outbreaks, to assess disease burden of Chikungunya on economy and loss and damage of Public health and quality of life
Author Contributions
All the authors substantially contributed to the conception, design, analysis and interpretation of data, checking and approving the final version of the manuscript and agree to be accountable for its contents.
Conceptualization: Kaushik Das, Sonuwara Begum
Data curation: Kaushik Das.
Formal analysis: Kaushik Das, Mihir Jyoti Pathak.
Investigation: Kaushik Das.
Methodology: Kaushik Das, Sonuwara Begum.
Supervision: Samrat Biswas, Syed Tanwir Alam, Sonuwara
Begum.
Writing – original draft: Kaushik Das.
Writing – review & editing: Kaushik Das, Samrat Biswas, Syed
Tanwir Alam.
Acknowledgement
The authors would like to express gratitude to Department of Health Research, New Delhi for their continuous support and assistance during the study period. We are also thankful to the Principal Cum Chief superintendent of Tezpur Medical College, the faculties of Department of Microbiology, Tezpur Medical college and hospital, to laboratory Technicians, Data entry operators and multi-tasking staff working in VRDL, Department of Microbiology, Tezpur Medical College & Hospital for their kind cooperation throughout the period.
Ethical Statement
This study was approved by the institutional ethical clearance committee of Tezpur Medical College & Hospital.
Funding
This study was funded by DHR- I.C.M.R.
Informed Consent
As it was a retrospective study involving analysis of existing secondary data which were already collected with strict adherence to confidentiality protocols. Informed consent was not required.
Data Availability
The datasets present in the current study are available from the corresponding author upon reasonable request.AI Disclosure
Artificial Intelligence was not used in any part of this study.
References
1.Chotaliya, G., Parmar, R., Gadhavi, H. M., & Shingala, H. (2024). Seroprevalence of Chikungunya Fever at a Tertiary Care Hospital. GAIMS Journal of Medical Sciences, 4(2 (Jul-Dec)), 68-73.
2.Dutta, P., Khan, S. A., Khan, A. M., Borah, J., Chowdhury,P., & Mahanta, J. (2011). First evidence of chikungunya virus infection in Assam, Northeast India. Transactions of the Royal Society of Tropical Medicine and Hygiene, 105(6), 355-357.
3.Dutta, P., Khan, S. A., Phukan, A. C., Hazarika, S., Hazarika,N. K., Chetry, S., ... & Kaur, H. (2019). Surveillance of Chikungunya virus activity in some North-eastern states of India. Asian Pacific Journal of Tropical Medicine, 12(1), 19- 25.
4.Mohanty, I., Dash, M., Sahu, S., Narasimham, M. V., Panda, P., & Padhi, S. (2013). Seroprevalence of chikungunya in southern Odisha. Journal of family medicine and primary care, 2(1), 33-36.
5.Arankalle, V. A., Shrivastava, S., Cherian, S., Gunjikar, R. S., Walimbe, A. M., Jadhav, S. M., ... & Mishra, A. C. (2007). Genetic divergence of Chikungunya viruses in India (1963– 2006) with special reference to the 2005–2006 explosive epidemic. Journal of General Virology, 88(7), 1967-1976.
6.Tomar, S. J., Alagarasu, K., More, A., Nadkarni, M., Bachal, R., Bote, M., ... & Tandale, B. V. (2022). Decadal change in seroprevalence of chikungunya virus infection in Pune city, India. Viruses, 14(5), 998.
7.Mavalankar, D., Shastri, P., Bandyopadhyay, T., Parmar, J., & Ramani, K. V. (2008). Increased mortality rate associated with chikungunya epidemic, Ahmedabad, India. Emerging infectious diseases, 14(3), 412.
8.Kawle, A. P., Nayak, A. R., Bhullar, S. S., Borkar, S. R.,Patankar, S. D., Daginawala, H. F., ... & Kashyap, R. S. (2017). Seroprevalence and clinical manifestations of chikungunya virus infection in rural areas of Chandrapur, Maharashtra, India. Journal of Vector Borne Diseases, 54(1), 35-43.
9.Mehta, R., Gerardin, P., de Brito, C. A. A., Soares, C. N., Ferreira, M. L. B., & Solomon, T. (2018). The neurological complications of chikungunya virus: A systematic review. Reviews in medical virology, 28(3), e1978.
10.Das, T., Jaffar-Bandjee, M. C., Hoarau, J. J., Trotot, P. K.,Denizot, M., Lee-Pat-Yuen, G., ... & Gasque, P. (2010). Chikungunya fever: CNS infection and pathologies of a re- emerging arbovirus. Progress in neurobiology, 91(2), 121- 129.
11.Azami, N. A. M., Salleh, S. A., Shah, S. A., Neoh, H. M., Othman, Z., Zakaria, S. Z. S., & Jamal, R. (2013). Emergence of chikungunya seropositivity in healthy Malaysian adults residing in outbreak-free locations: chikungunya seroprevalence results from the Malaysian Cohort. BMC infectious diseases, 13(1), 67.
12.Sam, I. C., Chua, C. L., & Chan, Y. F. (2011). Chikungunya virus diagnosis in the developing world: a pressing need. Expert review of anti-infective therapy, 9(12), 1089-1091.


