Research Article - (2022) Volume 7, Issue 1
A Clinical Research of Ceritinib Treating Patients with ALK-Positive Advanced Non-small Cell Lung Cancer with Brain Metastasis
1The Second Department of Thoracic Medical Oncology, The Second Hospital, of Dalian Medical University, Dalian, Liaoning, China
Received Date: Mar 02, 2022 / Accepted Date: Mar 07, 2022 / Published Date: Mar 15, 2022
Copyright: ©Zhaoxia Dai, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Saiqiong Cui, Xin Liu, Dan Li, Dafu Yang, Zhaoxia Dai. (2022). A Clinical Research of Ceritinib Treating Patients with ALK-Positive Advanced Non-small Cell Lung Cancer with Brain Metastasis. Int J Women's Health Care, 7(1), 42-49.
Abstract
Objective: As a second-generation oral anaplastic lymphoma kinase (ALK) inhibitor, ceritinib was recommended as posterior line therapy in patients with crizotinib resistance or intolerance. While in the real world, the clinical efficacy and safety of ceritinib, 450mg/d, taken with a low-fat meal in crizotinib resistant patients, especially with brain metastases, are still to be further investigated.
Methods: This retrospective analysis was conducted on patients with ALK- positive advanced non-small cell lung cancer (NSCLC) between May 2017 and December 2020 in our hospital, who were crizotinib resistant or intolerant. The aim was to analyze the clinical efficacy and safety of ceritinib as posterior line therapy in patients, especially in those with brain metastases, and to further explore the efficacy against rare fusion of ALK and the option after ALK inhibitor resistance. RECIST (1.1) criteria were adopted, and for relevant statistical analysis, RStudio was used.
Results: Of the 13 cases treated with ceritinib, 8 were treated as second-line therapy, 5 as third-line or fourth-line treatment. Of the total, 12 had brain metastases, 3 had received brain radiotherapy previously, and 1 had received surgical treatment for brain metastases previously. There were 8 cases evaluated as partial remission (PR), 2 evaluated as stable disease (SD), and 3 unevaluable for the response. There were 1 case of PKNOX2-ALK fusion and 1 case of IGR-ALK fusion observed as rare ALK fusion, and intracranial lesions were evaluated as PR for both. There were 6 cases received subsequent targeted therapy after progression of ceritinib, including 5 with alectinib and 1 with ensartinib, all of which showed response. In 13 cases, by the terminal time of follow-up, median progression-free survival (PFS) was 7.4 m, disease control rate (DCR) was 76.92%, overall response rate (ORR) was 61.54%, and median overall survival (OS) was 25.4m. In 12 cases with brain metastasis, 9 were suitable for intracranial response evaluation, of which, 1 case achieved CR for intracranial lesions, 7 achieved PR and 1 SD was observed, ORR was 88.89%, and DCR was 100%. The adverse events rate of any grade was 91.67%, the common adverse events were diarrhea (46.15%), AST level increased (41.67%), ALT level increased (33.33%), increased GGT (33.33%), increased ALP (25.00%), nausea (16.67%), increased creatinine (16.67%). Other reported AEs were cough, vomiting, dizziness, arthralgia and fatigue, 1 case (8.33%) each.
Conclusion: Ceritinib 450mg taken with taken with low-fat meal showed good efficacy and safety in crizotinib resistant patients with central nervous system metastasis, and it is effective in some cases with rare fusion of ALK.
Keywords
Ceritinib; brain metastasis; ALK fusion; Non-small Cell Lung Cancer
Introduction
Account for about 1% to 3% of patients with advanced non-small cell lung cancer (NSCLC), anaplastic lymphoma kinase (ALK) rearrangements are usually seen in younger, non-smoking or light smoking patients with adenocarcinoma [1,2]. Patients with ALK-rearranged NSCLC are more prone to central nervous sys¬tem (CNS) metastasis, about 20% - 30% them had brain metas¬tases when diagnosed. After administration of crizotinib as first line treatment to CNS metastasis-free patients with ALK-rear-ranged advanced NSCLC, about 20% developed brain metastases, and 70% had intracranial progression in those with baseline brain metastases [3]. Obviously, CNS progression is the main problem of disease progression in ALK-rearranged NSCLC. Ceritinib, a next-generation oral ALK inhibitor, may cross the blood-brain barrier more easily than crizotinib, the first-generation ALK inhib-itor, in animal models [4]. In the ASCEND-4 study, 130 untreat-ed patients without brain metastases at baseline received ceritinib treatment. By the data cutoff date for follow-up, there had been 81 patients with disease progression, of whom 24 (18.5%) had intra¬cranial progression, and 1 (0.8%) had both intracranial and extra cranial progression. Among the 59 cases with brain metastases at baseline, 31 patients had disease progression, of whom only 15 (25.4%) had intracranial progression, and 3 (5%) had both intra¬cranial and extra cranial progression. The intracranial progression rate was lower than among those who received crizotinib [5]. For patients resistant to crizotinib, ceritinib showed better efficacy than chemotherapy as the posterior-line treatment [6]. In previous studies, ceritinib, 750mg/d fasted, had adverse events, especially gastrointestinal (GI) toxicities diarrhea, nausea and vomiting as most frequent AEs, and high incidence rate of elevated alanine aminotransferase, aspartate aminotransferase, and γ-trans pepti-dase [5-7]. In the ASCEND-8 study [5-7], the dose was adjusted to ceritinib, 450 mg with meal, which showed more favorable GI and liver function safety profile than ceritinib, 750mg fasted, whereas the pharmacokinetics in two arms showed no difference. Given better tolerance of patients in 450 mg daily dose of ceritinib taken with food, it seems that patients in this group may demonstrate improvement in survival [8].While, the efficacy and safety of Cer¬itinib 450mg taken with meal for crizotinib resistant patients, es¬pecially for patients with brain metastases still need to be studied in the real world. This study analyzed the efficacy and safety of ceritinib 450mg taken with meal among Chinese crizotinib resis¬tant patients retrospectively.
Materials and Methods
General Information
This retrospective analysis enrolled 13 patients with ALK-rear-ranged advanced NSCLC who got progression disease from crizo-tinib and took ceritinib as posterior line therapy between May 2017 and December 2020. The patients’ general information, treatment, and adverse events after oral administration of ceritinib were col¬lected through their medical records, telephone and WeChat com¬munication.
Inclusion and Exclusion Criteria
Inclusion Criteria
With histologically confirmed NSCLC; At Stage â?¢B to IV NS-CLC according to the eighth edition of AJCC; ALK rearrangement was determined by Ventana immunohistochemistry (IHC) assay, reverse transcription-polymerase chain reaction (RT-PCR), fluo¬rescence in situ hybridization (FISH) or next generation sequenc¬ing (NGS). With disease progression or intolerance after crizotinib treatment, and then ceritinib 450 mg orally taken with meal for at least 1 week as posterior line treatment; with complete medical records. Exclusion criteria: Cases who could not be followed up.
Analytical Methods
Measurable disease as per Response Evaluation Criteria in Solid Tumors (RECIST) 1.1 criteria, and RStudio for statistical analysis. Adverse events were evaluated according to common criteria of the National Cancer Institute (NCI-CTC AE5.0).
Usage and Dosage
For all of the cases, ceritinib 450mg was taken with low-fat meal, once a day, until disease progression, drug intolerance, or death occurs.
Results
Patient Demographics and Disease Characteristics
A total of 13 patients were enrolled in the study. Of these 13 cases, there were 9 females and 4 males, including 3 smoking cases and 10 non-smoking cases. The median age was 54 years (range 44-70 years). For all, the pathological subtype was stage IV lung adeno¬carcinoma. ALK rearrangement status was detected by immuno¬histochemistry in 5 cases, NGS in 6 cases, RT-PCR in 1 case, and unknown method in 1 case. There were 5 cases with EML4 - ALK fusion, 1 case with fusion of PKNOX2 gene of exon 3 and ALK gene of exon 20, and 1 case with IGR-ALK fusion (Table 1).
|
Total |
13 |
|
Median age, y |
|
|
Male |
54 |
|
Female |
51 |
|
Range |
(44, 70) |
|
Gender, n (%) |
|
|
Male |
4 (30.77 %) |
|
Female |
9 (69.23 %) |
|
Smoking history, n (%) |
|
|
Yes |
3 (23.08 %) |
|
No |
10 (76.92 %) |
|
ECOG, n (%) |
|
|
0 |
2 (15.38 %) |
|
1 |
10 (76.92%) |
|
2 |
1 (7.69 %) |
|
Genetic mutation, n (%) |
|
|
ALK-rearrangement |
13 (100%) |
|
EML4-ALK fusion |
5 (38.46%) |
|
rare ALK fusion |
2 (15.38%) |
|
unkown |
6 (46.15%) |
|
Histologic/cytologic subtype, n (%) |
|
|
Adnocacinoma |
13 (100.00 %) |
|
Stage, n (%) |
|
|
Stage IV |
13 (100.00%) |
|
Brain metastases, n (%) |
|
|
Brain metastasis alone |
5 (38.46 %) |
|
Brain and vital organs metastases |
7 (53.85%) |
|
Bone metastasis alone |
1 (7.69 %) |
Among 13 patients, 9 got new brain metastasis during prior crizo-tinib, 1 had intracranial progression, and 1 had extra cranial pro-gression during prior crizotinib and developed brain metastases after switching to chemotherapy. At the initiation of ceritinib treat-ment, 12 patients had brain metastases, 7 of whom had brain me¬tastases accompanied with other vital organs metastases. Eight pa- tients received one prior line of anticancer regimens; five received two or more prior lines of anticancer regimens. Three patients had received brain radiotherapy previously, one case had received sur¬gery for brain metastasis previously, and one case received ceri¬tinib combined with bevacizumab (Table 2).
Table 2: Treatment History and Outcome - Overall Population
|
Total |
13 |
|
Prior lines of anticancer tegimens, n (%) |
|
|
1 |
8 (61.54 %) |
|
≥2 |
5 (38.46%) |
|
Prior crizotinib, n (%) |
13 (100.00 %) |
|
Crizotinib terminated during ceritinib, n (%) |
13(100.00%) |
|
Site of crizotinib progression, n (%) |
|
|
CNS progression |
10 (76.92 %) |
|
Extra cranial progression |
1 (7.69%) |
|
No progression |
2 (15.38 %) |
|
Previous brain radiotherapy, n (%) |
|
|
Yes |
3 (23.08 %) |
|
No |
10 (76.92 %) |
|
Previous surgery of brain metastases, n (%) |
|
|
Yes |
1 (7.69 %) |
|
No |
12 (92.31 %) |
|
Combination therapy, n (%) |
|
|
Yes |
1 (7.69%) |
|
No |
12 (92.31 %) |
Short-Term Efficacy
By data cutoff date, 9 patients had stopped taking ceritinib, and 4 were still receiving oral ceritinib. There were 8 cases evaluated as PR for the best overall response, 2 evaluated as SD, and 3 unable to be evaluated (Table 3). Of these 3 cases, 1 refused imaging since continuous relief of dizziness and headache caused by brain metas¬tases, 1 had not reach the time for response evaluation, and 1 died 1.8 months after administration, which may be caused by disease progression. Median progression-free survival for all patients was 7.4 months (Fig. 1), median overall survival was 25.4 months (Fig. 2), disease control rate was 76.92%, and overall response rate was 61.54%. In the 12 patients with measurable brain metastases at baseline, 9 could be performed response evaluation for intracranial lesions, 1 was evaluated as complete remission (CR), 7 as partial remission (PR), and 1 as stable disease (SD), overall response rate was 88.89%, disease control rate was 100% (Fig. 3).
Table 3: Summary of Overall Response Rate (ORR)
|
Total |
13 |
|
Best response evaluation, n (%) |
|
|
PR |
8 (61.54 %) |
|
SD |
2 (15.38 %) |
|
Unable to evaluate |
3(23.08 %) |
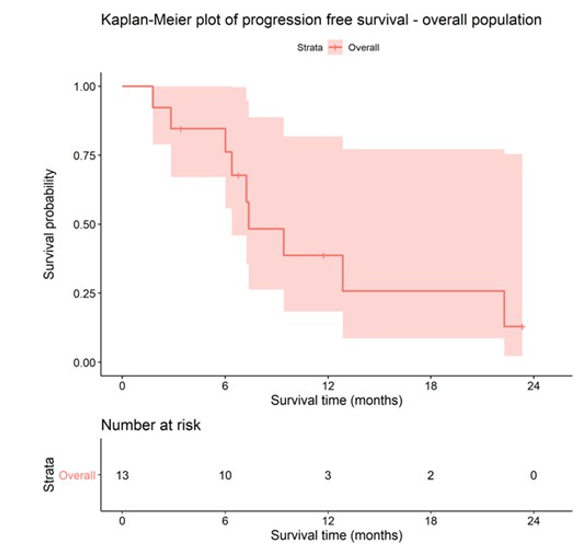
Figure 1: Kaplan-Meier plot of progression free survival-overall population
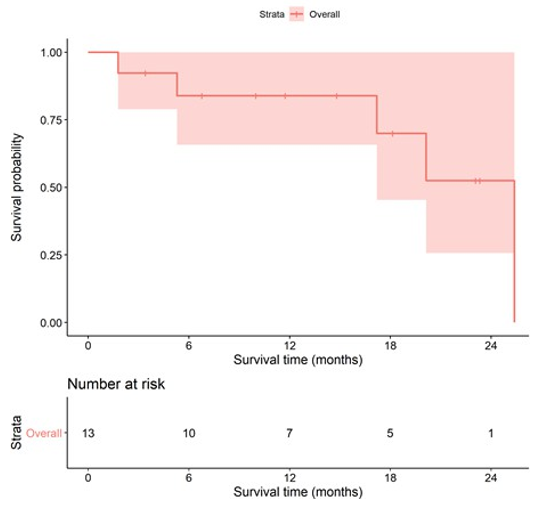
Figure 2: Kaplan-Meier plot of overall survival – overall population
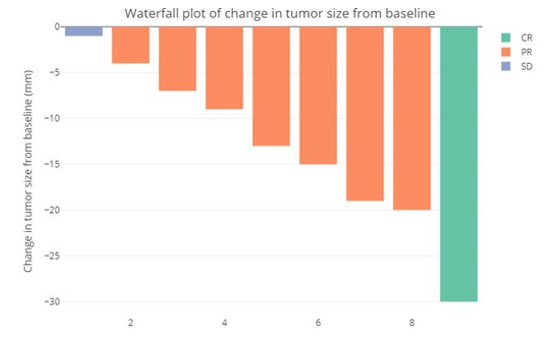
Figure 3: Waterfall plot of change in intracranial tumor size from baseline
Adverse Events
12 patients were followed up for adverse events, except for 1 deceased patient whose family members could not describe the adverse events. 11 patients had adverse events of any grade. The most common adverse event was diarrhea in 6(46.15%) out of 12 patients, including 2 of grade 1 and 4 of grade 2. Other common adverse events with ceritinib were an increase in alanine amino¬transferase (ALT, 4 (33.33%) of 12), aspartate aminotransferase (AST, 5 (41.67%) of 12), alkaline phosphatese (ALP, 3 (25.00%) of 12), and gamma-glutamyl transpeptidase (GGT, 4 (33.33%) of 12). Other adverse events such as nausea (16.67%), increased se-rum creatinine (16.67%), cough, vomiting, dizziness, arthralgia, and fatigue (8.33%) were also seen in this trail (Table 4).
Rare ALK Mutations
In this study, 2 cases with rare ALK fusion were seen: 1 patient with PKNOX2-ALK fusion progressed after second-line chemotherapy, and crizotinib as the third-line regimen, PFS: 11.5months; ceritinib was used for the fourth-line therapy, and was documented partial remission, PFS: 2.8 months. For the fifth-line therapy, ensartinib was adopted, PFS: 4.5 months. For another patient with IGR-ALK fusion was treated with crizotinib for the first-line therapy, PFS was 5 months, and ceritinib was used as second-line regimen, and was documented partial remission, PFS was 12.7 months.
Genetic Testing After Drug Resistance
In this study, a total of 4 patients underwent genetic testing after ALK inhibitor resistance, and among them, the patients with IGR-ALK fusion and with PKNOX2-ALK fusion underwent multiple blood NGS after ALK inhibitor resistance, in which secondary ALK mutation was not seen. One patient diagnosed as ALK-re-arrangement by IHC previously underwent blood NGS after first-line crizotinib and second-line ceritinib resistance, and the results showed no secondary ALK mutation.
Follow-Up Therapy
In this study, 6 cases received follow-up targeted therapy after dis-ease progression from ceritinib, and among them, 5 received alec¬tinib and 1 received ensartinib. Both showed efficacy.
Discussion
At present, for patients with advanced ALK-positive NSCLC who are resistant to the first-generation ALK inhibitor crizotinib, the second-generation ALK inhibitors ceritinib, alectinib, and briga-tinib or the third-generation ALK inhibitor lorlatinib are recom-mended [9]. Central nervous system metastasis was often inevi-table for patients with crizotinib resistance, and the prognosis of which was relatively poor [10]. In this study, 81.82% of patients without baseline brain metastases had developed brain metastases during treatment of crizotinib, indicating that central nervous sys¬tem progression is a major challenge faced in the application of the first-generation ALK inhibitor. In previous studies [6,11,12], the incidence of central nervous system metastasis was 15%, 6.8% and 1% respectively in ceritinib, alectinib and brigatinib in treat-ment-naïve patients with ALK-positive NSCLC without baseline brain metastasis, which were relatively low, showing better con¬trol in intracranial lesions. Then, do second-generation ALK in¬hibitors also show favorite efficacy in crizotinib resistant patients with brain metastases? In this study, for 12 patients with brain metastases, ceritinib was used in posterior line, and the response for intracranial lesions was documented CR for 1 case, PR for 7 cases, and SD for 1 case, ORR was 88.89%, and DCR was 100%,indicating that the second-line therapy with ceritinib for patients with ALK-positive brain metastases had a good efficacy. Brain ra¬diotherapy is also one of the most effective means for treatment of brain metastases [13], and whether radiotherapy will affect the efficacy of ALK inhibitors is also what we concerned. In this study, a total of 3 patients had received brain radiotherapy previously, all of whom had documented partial remission, suggesting that for those who had received brain radiotherapy, ceritinib may also be a good choice, i.e., the radiotherapy may not affect the efficacy of ceritinib. In this study, for the patients who had received at least one or more regimens previously, the median PFS was 7.4 months, and the median OS 25.4 months. The PFS is much longer than that for the second-line therapy in the ASCEND-5 study (5.4 months) [6], and the reason may be that the 450mg daily dose of ceritinib taken with meal improved the safety and tolerability of the pa¬tients, which is yet to be verified by large sample, randomized con¬trolled trials.
In terms of dose selection, based on the ASCEND-8 study [8], cer¬itinib, 450mg taken with meal reduced the frequency and severity of toxicities, and increase the patients’ tolerance without compro¬mising efficacy, and so, the ceritinib 450mg taken with meal was adopted. In our study, common adverse events were elevation of liver enzyme, diarrhea, and nausea, which was consistent with the previous report [14]. Although the incidence of adverse events was high (91.67%), all of the adverse events were of grade 1 or 2, i.e., no grade 3 or 4 adverse events occurred, and no dose adjustment or drug withdrawal occurred due to adverse reactions, showing good safety and tolerability. Therefore, ceritinib 450mg taken with meals is a good choice worthy of clinical application.
At present, there is still no clinical reports about the combina-tion of ALK inhibitor with antiangiogenic drug. Bevacizumab is a monoclonal antibody against vascular endothelial growth factor (VEGF), and it was demonstrated by previous phase II trials that the combination of bevacizumab with erlotinib could prolong the PFS of patients with advanced EGFR-mutation NSCLC [15,16], And the addition of bevacizumab to EGFR-TKIs also showed ef¬ficacy in progression-free survival of patients with EGFR-TKIs resistance [17]. For patients with brain metastases or brain edema, bevacizumab also showed a good efficacy, and the response rate in cerebral edema was 84.74% [18]. In this study, we tried to use ceritinib in combination with bevacizumab in the third-line ther¬apy for 1 patient, unfortunately, the patient died only 1.8 months after medication since the patient’s had been in poor conditions after multiple lines of treatment, unable to assess the efficacy. Of course, for the treatment in combination with bevacizumab, we need to pay more attention to relevant toxic and side effects, espe¬cially hypertension, proteinuria, etc. [19,20]. Whether the combi¬nation of ALK inhibitors with antiangiogenic drugs, e.g. bevaci¬zumab, anlotinib, could bring survival benefits for patients is still to be explored by studies that are more clinical in the future.
In non-small cell lung cancer, the most common site for ALK fusion is the fusion of echinoderm microtubule-associated protein-like 4 (EML4) with ALK [21], other rare fusion sites include the fusion of ALK with TFG, KLC1, KIF5B, STRN, TNFAIP3, TNIP2, etc. [22-24]. There were also case reports concerning efficacy of ALK inhibitors on rare ALK fusion previously [22-25]. The two cases of rare ALK fusion found in this study were PKNOX-ALK fusion and IGR-ALK fusion cases, and both cases had been treated by crizotinib effectively, with PFS 11.5 months and 5 months respec¬tively. The ceritinib treatment after crizotinib resistance was still effective, with intracranial lesions significantly reduced. The two cases received ensartinib and alectinib respectively as posterior line therapy, both of which showed efficacy. With the wide appli¬cation of next-generation sequencing, more and more rare ALK mutations have been found in clinical practice. For both cases of rare ALK fusion in this study, the first-generation ALK inhibitor and the two second-generation ALK inhibitors were used effec¬tively, which has brought us a great deal of confidence in these two kinds of rare ALK fusion, but can all of the patients with rare ALK fusion benefit from the first generation to the third generation of ALK inhibitors? There are only some individual cases for now, and evidence from clinical trials is to be supplemented.
It is well known that all targeted therapies may be faced with drug resistance at last. The resistance mechanism of the first-generation ALK inhibitor crizotinib mainly involves the following aspects: secondary mutation of ALK, ALK bypass activation, and NSCLC transformation. Secondary mutation of ALK includes L1196M, C1156Y, G1269A, S1206Y, V1180L, G1202R, I1171T, D1203N, and 1151Tins, F1174C/L, L1198P, L1152R/P and E1210K [26]. I1171 mutation indicates resistance to crizotinib and alectinib [27], and G1202R or I1171N mutation often occurs after alectinib resis-tance [28], while I1171N site mutation was sensitive to ceritinib [29]. ALK secondary mutation was found in about half of patients with ceritinib mutation, of which, G1202R (21%) andF1174C/L (16.7%) are most common [29]. Furthermore, some patients had more than two kinds of ALK secondary mutation. The T1151R mutation indicates sensitivity to brigatinib [30]. G1202R mutation often occurs after brigatinib resistance as well and it suggests re¬sistance to both the first generation and second-generation ALK inhibitors [31,32]. The third-generation ALK inhibitor is sensitive to not only F1174, C1156Y, G1269A, L1196M, D1203N muta- tions, but also G1202R mutation, therefore, for such patients; the third-generation ALK inhibitor lorlatinib may be recommended [33]. The mechanism of lorlatinib resistance is more complex and variable, including L1196M/D1203N, F1174L/G1202R, and C1156Y/G1269A mutations, etc. Patients with a single mutation of ALK-L1256F may be sensitive to ceritinib. The ALK second-ary mutation rate is approximately 20-30%, while the response rate of the second-generation ALK inhibitors was (48-71%) [28], indicating that the second-generation ALK TKIs may be also ef-fective in crizotinib-resistant patients without ALK secondary mutation. In this study, 4 patients underwent genetic testing after first-generation ALK inhibitor resistance, and no ALK secondary mutation was found among them, but the second-generation ALK inhibitor ceritinib was still effective. Therefore, for cases resis¬tant to the first-generation ALK inhibitor, the treatment with the second-generation ALK inhibitors should be considered even in irst-generation ALK inhibitor resistant patients without genetic testing. It could also maybe the sensitivity of the NGS detection method using blood samples is not as good as using tissue samples. If genetic testing can be performed again on new lesion’s biop¬sy sample to determine the presence of ALK secondary mutation and specific mutation site, the subsequent treatment may be guid¬ed more accurately. Besides ALK second mutation, other kinas¬es mutations and bypass signal abnormal activations (e.g., TP53 mutation, MET mutation, BRAF mutation and MAPK, PI3K/Akt pathway and HER pathway activation) are important mechanisms of resistance for ALK positive patients [26,34], reflecting the di¬versity and complexity of resistance to ALK inhibitors, which has not been fully studied yet. However, it has been shown that the combination of proteasome inhibitors and alectinib is effective in patients with TP53 mutation after alectinib resistance [35]. Some studies have found that a bypass mechanism of resistance caused by NF2 loss-of-function mutations is sensitive to mTOR inhibitor [33]. Another mechanism of resistance is the small cell lung can¬cer transformation. For patients with dramatically elevated NSE or rapid progressive disease or cases with a mutation for dual deletion of RB1 and TP53 during treatment, a second pathological biopsy is recommended for detecting whether there is transformation of small cell lung cancer (32). For patients with rapidly progressive small cell lung cancer transformation, chemotherapy with etopo-side combined with cisplatin could be considered, for patients with less progressive disease or local progression, chemotherapy in combination with original TKI targeted therapy may be appro¬priate, and local treatment may be supplemented if necessary [36].
Declarations
Ethics Approval and Consent to Participate
This research was approved by Ethics Committee of The Second Hospital of Dalian Medical University, and written informed con¬sent for publication was obtained from the participates.
Authors’ Contributions
Conceptualization, Methodology, Funding acquisition: Saiqiong Cui. Data curation, Writing-Original draft preparation: Xin Liu. Visualization, Investigation: Dan Li. Supervision: Dafu Yang. Project administration, Resources: Zhaoxia Dai.
References
- Fallet, V., Cadranel, J., Doubre, H., Toper, C., Monnet, I., Chinet, T., ... & Wislez, M. (2014). Prospective screening for ALK: clinical features and outcome according to ALK status. European journal of cancer, 50(7), 1239-1246.
- Shaw, A. T., Yeap, B. Y., Mino-Kenudson, M., Digumarthy, S. R., Costa, D. B., Heist, R. S., ... & Iafrate, A. J. (2009). Clini-cal features and outcome of patients with non–small-cell lung cancer who harbor EML4-ALK. Journal of clinical oncology, 27(26), 4247.
- Costa, D. B., Shaw, A. T., Ou, S. H. I., Solomon, B. J., Riely,G. J., Ahn, M. J., ... & Crinò, L. (2015). Clinical experience with crizotinib in patients with advanced ALK-rearranged non–small-cell lung cancer and brain metastases. Journal of Clinical Oncology, 33(17), 1881.
- Friboulet, L., Li, N., Katayama, R., Lee, C. C., Gainor, J. F., Crystal, A. S., ... & Engelman, J. A. (2014). The ALK inhibitor ceritinib overcomes crizotinib resistance in non–small cell lung cancer. Cancer discovery, 4(6), 662-673.
- Soria, J. C., Tan, D. S., Chiari, R., Wu, Y. L., Paz-Ares, L., Wolf, J., ... & de Castro Jr, G. (2017). First-line ceritinib versus platinum-based chemotherapy in advanced ALK-rearranged non-small-cell lung cancer (ASCEND-4): a randomised, open-label, phase 3 study. The Lancet, 389(10072), 917-929.
- Shaw, A. T., Kim, T. M., Crinò, L., Gridelli, C., Kiura, K., Liu, G., ... & Felip, E. (2017). Ceritinib versus chemotherapy in patients with ALK-rearranged non-small-cell lung cancer previously given chemotherapy and crizotinib (ASCEND-5): a randomised, controlled, open-label, phase 3 trial. The Lancet Oncology, 18(7), 874-886.
- Kim, D. W., Mehra, R., Tan, D. S., Felip, E., Chow, L. Q., Camidge, D. R., ... & Shaw, A. T. (2016). Activity and safety of ceritinib in patients with ALK-rearranged non-small-cell lung cancer (ASCEND-1): updated results from the multicentre, open-label, phase 1 trial. The lancet oncology, 17(4), 452-463.
- Cho, B. C., Kim, D. W., Bearz, A., Laurie, S. A., McKeage,M., Borra, G., ... & Wrona, A. (2017). ASCEND-8: a randomized phase 1 study of ceritinib, 450 mg or 600 mg, taken with a low-fat meal versus 750 mg in fasted state in patients with anaplastic lymphoma kinase (ALK)-rearranged metastatic non–small cell lung cancer (NSCLC). Journal of Thoracic Oncology, 12(9), 1357-1367.
- Melosky, B., Cheema, P., Agulnik, J., Albadine, R., Bebb, D. G., Blais, N., ... & Liu, G. (2018). Canadian perspectives: update on inhibition of ALK-positive tumours in advanced non-small-cell lung cancer. Current Oncology, 25(5), 317-328.
- Zhang, I., Zaorsky, N. G., Palmer, J. D., Mehra, R., & Lu,B. (2015). Targeting brain metastases in ALK-rearranged non-small-cell lung cancer. The lancet oncology, 16(13), e510-e521.
- Gadgeel, S., Peters, S., Mok, T., Shaw, A. T., Kim, D. W., Ou, S. I., ... & Camidge, D. R. (2018). Alectinib versus crizotinib in treatment-naive anaplastic lymphoma kinase-positive (ALK+) non-small-cell lung cancer: CNS efficacy results from the ALEX study. Annals of oncology, 29(11), 2214-2222.
- Camidge, D. R., Kim, H. R., Ahn, M. J., Yang, J. C. H., Han,J. Y., Lee, J. S., ... & Popat, S. (2018). Brigatinib versus crizotinib in ALK-positive non–small-cell lung cancer. New England Journal of Medicine, 379(21), 2027-2039.
- Gemici, C., & Yaprak, G. (2015). Whole-brain radiation therapy for brain metastases: detrimental or beneficial?. Radiation Oncology, 10(1), 1-3.
- Santarpia, M., Daffinà, M. G., D’Aveni, A., Marabello, G., Li-guori, A., Giovannetti, E., ... & Altavilla, G. (2017). Spotlight on ceritinib in the treatment of ALK+ NSCLC: design, development and place in therapy. Drug design, development and therapy, 11, 2047.
- Yoshida, K., & Yamada, Y. (2015). Erlotinib alone or with bevacizumab as first-line therapy in patients with advanced non-squamous non-small-cell lung cancer harboring EGFR mutations (JO25567): an open-label, randomized, multi-center, phase II study. Translational Lung Cancer Research, 4(3), 217.
- Rosell, R., Dafni, U., Felip, E., Curioni-Fontecedro, A., Gautschi, O., Peters, S., ... & Stahel, R. A. (2017). Erlotinib and bevacizumab in patients with advanced non-small-cell lung cancer and activating EGFR mutations (BELIEF): an international, multicentre, single-arm, phase 2 trial. The Lancet Respiratory Medicine, 5(5), 435-444.
- Yu, Y., Wang, Y., Wu, L., Xu, X., Zhou, H., Wang, Q., &Zhou, J. (2021). Efficacy of epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs) combined with bevacizumab for advanced non-squamous non-small-cell lung cancer patients with gradual progression on EGFR-TKI treatment: a cohort study. Medicine, 100(5).
- Meng, X., Zhao, R., Shen, G., Dong, D., Ding, L., & Wu, S. (2017). Efficacy and safety of bevacizumab treatment for refractory brain edema: Case report. Medicine, 96(44).
- Zhou, C. C., Bai, C. X., Guan, Z. Z., Jiang, G. L., Shi, Y. K.,Wang, M. Z., ... & Zhu, Y. Z. (2014). Safety and efficacy of first-line bevacizumab combination therapy in Chinese population with advanced non-squamous NSCLC: data of subgroup analyses from MO19390 (SAiL) study. Clinical and Translational Oncology, 16(5), 463-468.
- Chen, Z., Zhong, B., Lun, X., Lai, Y., Bella, A. E., Yang, W., & Wu, J. (2015). Specific safety profile of bevacizumab in Asian patients with advanced NSCLC: a meta-analysis. Medicine, 94(24).
- Kazandjian, D., Blumenthal, G. M., Chen, H. Y., He, K., Patel, M., Justice, R., ... & Pazdur, R. (2014). FDA approval summary: Crizotinib for the treatment of metastatic nonâ?ÂÂsmall cell lung cancer with anaplastic lymphoma kinase rearrangements. The oncologist, 19(10), e5-e11.
- Zeng, H., Liu, Y., Wang, W., Tang, Y., Tian, P., & Li, W. (2021). A rare KIF5B-ALK fusion variant in a lung adenocarcinoma patient who responded to crizotinib and acquired the ALK L1196M mutation after resistance: a case report. Annals of Palliative Medicine.
- Wang, P., Xiao, P., Ye, Y., Liu, P., Han, L., Dong, L., ... & Yu,J. (2017). Rapid response of brain metastasis to crizotinib in a patient with KLC1-ALK fusion and MET gene amplification positive non-small cell lung cancer: a case report. Cancer bi-ology & medicine, 14(2), 183.
- Kang, J., Zhang, X. C., Chen, H. J., Zhong, W. Z., Xu, Y., Su, J., ... & Yang, J. J. (2020). Complex ALK fusions are associated with better prognosis in advanced non-small cell lung cancer. Frontiers in Oncology, 2793.
- Sun, K., Nie, L., Nong, L., & Cheng, Y. (2021). Primary resistance to alectinib in a patient with STRN-ALK-positive non-small cell lung cancer: A case report. Thoracic Cancer, 12(12), 1927-1930.
- Golding, B., Luu, A., Jones, R., & Viloria-Petit, A. M. (2018). The function and therapeutic targeting of anaplastic lymphoma kinase (ALK) in non-small cell lung cancer (NSCLC). Molecular cancer, 17(1), 1-15.
- Toyokawa, G., Hirai, F., Inamasu, E., Yoshida, T., Nosaki, K., Takenaka, T., ... & Ichinose, Y. (2014). Secondary mutations at I1171 in the ALK gene confer resistance to both Crizotinib and Alectinib. Journal of Thoracic Oncology, 9(12), e86-e87.
- Gainor, J. F., Dardaei, L., Yoda, S., Friboulet, L., Leshchiner, I., Katayama, R., ... & Shaw, A. T. (2016). Molecular mechanisms of resistance to first-and second-generation ALK inhibitors in ALK-rearranged lung cancer. Cancer discovery, 6(10), 1118-1133.
- Ou, S. H. I., Greenbowe, J., Khan, Z. U., Azada, M. C., Ross,J. S., Stevens, P. J., ... & Gitlitz, B. (2015). I1171 missense mutation (particularly I1171N) is a common resistance mutation in ALK-positive NSCLC patients who have progressive disease while on alectinib and is sensitive to ceritinib. Lung cancer, 88(2), 231-234.
- Mehlman, C., Chaabane, N., Lacave, R., Kerrou, K., Rup-pert, A. M., Cadranel, J., & Fallet, V. (2019). Ceritinib ALK T1151R resistance mutation in lung cancer with initial response to brigatinib. Journal of Thoracic Oncology, 14(5), e95-e96.
- Xiao, Z., Huang, X., Xie, B., Xie, W., Huang, M., & Lin,L. (2020). Primary resistance to brigatinib in a patient with lung adenocarcinoma harboring alk G1202R mutation and li-pi-ntrk1 rearrangement. OncoTargets and therapy, 13, 4591.
- Ou, S. H. I., Lee, T. K., Young, L., Fernandez-Rocha, M. Y.,Pavlick, D., Schrock, A. B., ... & Gitlitz, B. J. (2017). Dual occurrence of ALK G1202R solvent front mutation and small cell lung cancer transformation as resistance mechanisms to second generation ALK inhibitors without prior exposure to crizotinib. Pitfall of solely relying on liquid re-biopsy?. Lung Cancer, 106, 110-114.
- Recondo, G., Mezquita, L., Facchinetti, F., Planchard, D., Gazzah, A., Bigot, L., ... & Friboulet, L. (2020). Diverse resistance mechanisms to the third-generation ALK inhibitor lorlatinib in ALK-rearranged lung cancer. Clinical Cancer Research, 26(1), 242-255.
- Shi, R., Martins Filho, S. N., Li, M., Fares, A., Weiss, J., Pham, N. A., ... & Liu, G. (2020). BRAF V600E mutation and MET amplification as resistance pathways of the second-gen-eration anaplastic lymphoma kinase (ALK) inhibitor alectinib in lung cancer. Lung Cancer, 146, 78-85.
- Tanimoto, A., Matsumoto, S., Takeuchi, S., Arai, S., Fukuda, K., Nishiyama, A., ... & Yano, S. (2021). Proteasome inhibition overcomes ALK-TKI resistance in ALK-rearranged/ TP53-mutant NSCLC via noxa expression. Clinical Cancer Research, 27(5), 1410-1420.
- Marcoux, N., Gettinger, S. N., O’Kane, G., Arbour, K. C.,Neal, J. W., Husain, H., ... & Sequist, L. V. (2019). EGFR-mu-tant adenocarcinomas that transform to small-cell lung cancer and other neuroendocrine carcinomas: clinical outcomes. Journal of Clinical Oncology, 37(4), 278.


