Research Article - (2025) Volume 9, Issue 2
Unlocking the Potential The Emergence of Nanobots in Future of Oncology
Received Date: Aug 08, 2025 / Accepted Date: Sep 08, 2025 / Published Date: Sep 15, 2025
Copyright: ©2025 Dr. Kanchan Karmakar, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Karmakar, K., Paul, S. (2025). Unlocking the Potential: The Emergence of Nanobots in Future of Oncology. Adv Nanoscie Nanotec, 9(2), 01-07.
Abstract
The treatment of cancer in the recent era has undergone quite a lot of developments, one of such is the emergence of Nanobots. Until its emergence, the traditional treatment of cancer primarily relied on the use of chemotherapeutic drugs and surgery, which in majority of the cases resulted in catastrophic side effects and in some cases into Multi drug resistance. Nanobots over the years have proved to be the most reliable means of cancer treatment thanks to its high specificity towards carcinogenic cells, exceptional efficiency and minimal side effects. These Nanobots consist of an outer shell, a propeller, power source, Sensors, payloads, nano camera, nano lasers and communicators. Extensive research in this field suggests that these nanobots can be easily converted into completely autonomous bio computing agents that are capable of executing complex Boolean logic gates. So, these nanobots exist in two states: Off and On. Initially they remain in off state, completely incapable of releasing drugs. Upon reaching the target organs through the bloodstream, they get converted into on state by receiving external signals and release the payloads (drugs) to perform the specific functions as discussed below before reverting back to their Off state. Eventually, they get excreted out. Despite their promise, nanobots are not without challenges. Issues such as biocompatibility, precise navigation and targeting, potential immune responses, and the ethical implications of their use present significant hurdles. We herein also discuss their potential challenges and future prospect of them being used as a successful medium of cancer treatment.
Keywords
Cancer, Drug Delivery, Efficiency, Nanobots, Specificity
Introduction
With the rapid advancements in biotechnology, nanorobotics has become a focal point of discussion due to its remarkable potential as an anti-cancer agent. These tiny robots promise high efficacy with minimal side effects, revolutionizing how we approach cancer treatment. The concept of robots originated in the early 1920s through the visionary work of Czech author Karel Capek. Moving to 1959, it was the renowned physicist Richard Feynman who laid the foundational ideas for nanotechnology.
A pivotal advancement came with the invention of the scanning tunnelling microscope by Gerd Binnig and Heinrich Rohrer, which enabled the visualization and manipulation of individual atoms. This groundbreaking tool earned them the Nobel Prize in Physics in 1986. The concept of nanobots was first articulated by Eric Drexler in his influential book "Engines of Creation: The Coming Era of Nanotechnology," where he envisioned nanoscale machines capable of self-replication and intricate manufacturing.
The year 2000 marked the beginning of significant efforts to produce nanobots on a large scale, particularly for targeted drug delivery. By 2004, the first DNA nanobot was created, and subsequent years have seen continuous refinement, making these nanobots increasingly precise and reducing their side effects.
Currently, clinical trials are underway to explore the diverse applications of nanobots in cancer treatment. Anotable development in 2018 involved the creation of bacteria-powered nanobots that utilize the natural chemotaxis movement of bacteria, especially E. coli, for propulsion [1]. These nanobots can autonomously navigate the body, seeking out cancer cells with unprecedented precision.
Nanobots offer several advantages over nanoparticles. They can autonomously detect and target carcinogenic cells, releasing drugs directly where needed. Unlike nanoparticles, nanobots can adapt to changes in physical and chemical conditions such as temperature, pressure, and pH. They can be programmed with sophisticated algorithms to perform complex functions and monitored in real- time through embedded sensors, capabilities beyond the reach of traditional nanoparticles (Figure 1).
Figure 1: A Generative Image of the Types of Nanobots
One of the most significant benefits of nanobots is their ability to be actively transported, enabling them to penetrate deeper and more accurately into tissues. Additionally, their theragnostic properties allow them to simultaneously deliver drugs and diagnose diseases, providing a comprehensive approach to treatment.
In summary, nanobots represent a revolutionary leap in medical technology, particularly in cancer therapy. Their ability to combine diagnostic and therapeutic functions, adapt to the body's environment, and precisely target diseases underscores their potential to transform modern medicine. This new era of nanotechnology holds the promise of more effective and personalized treatments, marking a significant advancement over traditional approaches.
Structure
For the proper functionality of a particular nanobot, it should have all of these following structural characteristics mentioned below for high efficiency, precision, specificity and proper functionality.
• Shells: This comprises the outer component of a nanobot composed of several kinds of particles that primarily include carbon, diamond and silica. They are made in such a way that their absorption rate in our body becomes quite high. One of the major factors to be taken care of while making it is biosafety and health. Shells directly come into contact with the external environment (Body) so any lapse in its preparation may lead to severe toxicity.
• Propeller: The major component of nanobots is the propeller mainly consisting of a swimming tail. This primarily triggers the movement of nanobots inside of our body mainly through the fluid medium like bloodstream or cerebrospinal fluid into the target organs. The major propulsion mechanisms involve magnetic propulsion, and chemical propulsion. The most discussed among them is the magnetically propelled [2].
In the cases of magnetic propulsion, the particles are driven forward by the external magnetic field. The magnetic energy is converted into mechanical energy in the form of magnetic field gradient and torque. The magnetic field vector B is made to rotate along a particular axis to make a rotating magnetic field through which the nanobot is propelled [3].
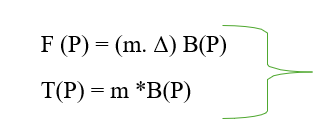
F = Magnetic Force B = Magnetic Induction
T= Magnetic Torque P = Point of Action
The other class of chemical nanoparticles has also quite a lot of applications. Initially these particles were mainly propelled by
• Power sources: These sources involve the external sources providing power to the nanobots for its functionality. They involve nanomotors, nano engines, hydrogen fuels, batteries etc. Although, these power sources are used very selectively keeping in mind the safety of an individual.
• Nano camera: In some of the cases of treatment, a certain camera (Nanoscale) is attached to the nanobots to provide live streaming. This provides live footage of the navigational route and helps in precise control of the nanobots. They act as the navigational guide simplifying the route of propulsion inside the body.
• Payloads: This primarily comprises the drugs carried by the nanobots that perform the specific function on reaching the target sites. They involve the chemotherapeutic agents, genetic materials, proteins, enzymes, immunotherapeutic agents and even sometimes their combination.
• Sensors: Inside the shells are present the nano sensors performing diverse functions starting from monitoring change in physical and chemical factors including temperature and pH to analysing the extent of damage to a cell by performing biological reactions.
• Laser: In some cases, these nanobots are equipped with Nano lasers capable of destroying harmful substances coming their way including cancerous cells, blood clots etc.
• Actuators: They are certain classes of particles involved in navigation, physical interaction with the body and most importantly aid in simplification of drug release in the target site.
• Communicators: Finally, they consist of communicators to be able to communicate information like changes in conditions, extent of cellular damage etc. externally. The major mode of communication involves the electromagnetic one [2].
• Radio Frequency (RF) Communication: Uses electromagnetic waves to exchange data over longer distances within the body
• Infrared (IR) Communication: Suitable for short-range communication, utilizing light waves for precise data transmission.
• Magnetic Fields: Guide and control the movement of nanobots, also used for transmitting signals through deep tissue [5].
One of the most intriguing facts to be noted is that these nanobots can easily be controlled by Logic gates. Several researches in this field suggest that these nanobots can be easily converted into completely autonomous bio computing agents that are capable of executing complex Boolean logic gates. Through sophisticated programming, Complex logic gate functions are integrated into the DNA as a result of which, when the Nanobots are created from the DNA origami a method of making tiny components of DNA, can be easily made to exist in two states, the off and on. As directed by the operator, the bots consisting of 2 halves (Off and on) open or close by a hinge mechanism. The 2 halves are bound together by molecular locks formed by DNA base pair interconnections.
The off state is their natural state when they are incapable of performing any function, but on reaching their target organs, and on encountering requisite physical and chemical conditions, they get converted to on state thereby performing the specifically assigned tasks with high precision and efficiency. Most importantly this transition is once again a reversible one as the nanobots get back to their native off state on completion [2].
Types
Based on their composition and functionality, the Nanobots used in medical purposes can be broadly classified into the following types. Firstly, based on their structures they may be of 2 types [6].
1. Assemblers: Their shape resembles the cell shape, and are quite useful in interpretation of various kinds of atoms or molecules
2. Replicators: They comparatively are more advanced forms capable of self-replicating inside the body. They generally have the ability to collect raw materials and use them in producing exact copies of themselves.
Now comes the major basis of division, the functionality; [7]
1. Pharmacyte: This is the first class of Nanobots, consisting of size range 1 to 2 micron. Its ability to carry drugs limits upto 1 μM. They are quite precisely controlled by the mechanical system sorting out and pumping drugs. To ensure specific targeting and high efficiency, they are equipped with chemotactic sensors or molecular markers. These types of nanobots derive power by extracting glucose and oxygen from the blood, intestinal fluid and cytosol. A process called centrifuge nanapheresis effectively separates and collects them once their task is complete in our body.
2. Diagnosing and imaging: These are a low priced and easily modifiable group of nanobots, consisting of microchips overlayed on human biomolecules. They are primarily used for disease detection. The specialized chips are programmed specifically to send signals once a disease is detected. Besides, they are also used in detection of diabetes.
3. Respirocytes: They involve the highly developed class of nanobots capable of transporting high levels of oxygen. Their shape and functionality more or less resemble the RBC but are highly modified and are able to provide 236 times more oxygen to tissues per unit volume than RBC, making it highly effective in treatment of anoxia. Endogenous serum glucose provides them with the required power.
4. Microbivores: This class comprises particles quite useful in digestion of entrapped bacteria in our body through continual consumption of upto 200pW. Along the major and minor axis respectively their diameter is 3.4 and 2.0 μM. They closely resemble the phagocytes, but are 80 times more efficient than time considering the vol/sec digested per unit volume of phagocytic agents.
5. Clottocytes: These are instant haemostatic agents also termed as artificial platelets. Once directed and programmed, they move inside our body to the site of excessive bleeding and rapidly form clumps leading to blood clotting.
6. Chromallocyte: This involves nanobots capable of replacing an entire chromosome. Their potential uses lie in treatment of genetic disorders. Examining the cellular contents and activities they first assess the cellular condition and functionality, before starting their repair works by genetic repair and restoration of entire cells.
In Cancer Treatment
Nanobots have proved to play quite a significant role in the treatment of cancer through various mechanisms associated with high levels of specificity, efficacy and safety. Programmed nanobots, calibrated for specific quantity and quality, are introduced into the body and directed to the target area. Initially when they are injected, they remain in the off state as mentioned above. This is done to ensure that they don’t affect the healthy cells. They roam throughout the body in this state until they reach the target organs [2].
On reaching the target organ, they are converted to on state by exogenous signals through the communicators that include electromagnetic radiation, ultrasound or light, or by endogenous signals like chemotaxis [8]. Following this, they release their payloads (Drugs) into the target organs before again transitioning back to the off state. Although most of these concepts are still hypothetical, once the drugs enter the target site they perform anti- cancer mechanisms through the following mechanisms;
Mechanisms of Nanobots in Cancer Treatment
Nanobots release certain drugs that demonstrate a range of mechanisms that contribute to their potential as effective agents in cancer therapy. These mechanisms include inducing oxidative stress, causing DNA cleavage, upregulating pro-apoptotic proteins, arresting the cell cycle, inducing mitochondrial fission, and regulating the hypoxia-inducible factor (HIF) pathway. Nanobots based on their modes of propulsion in our body can be broadly classified into the following types;
• External magnet Driven Nanobots: One of the most intriguing models of Magnet driven nanobots was shown by Andhari et al, 2020 [9]. They developed a multi walled carbon nanotube loaded with anticancer antibodies, and doxorubicin. They were specifically directed towards the spheroidal tumours including colorectal carcinoma through a torque generated through a strong external magnetic field leading to a rotational to translational movement. The triggering force was the pH changes or the changes in intracellular H2O2 concentration. Its implication on cervical cancer was seen by Wang et al, 2012 [10] developing an external magnetic driven nickel silver nano-swimmer, capable of reaching speeds exceeding 10 micron per second. The payloads consisted of poly (D, l-lactic-co–glycolic acid microspheres inside which was present doxorubicin.
• External Ultrasound Driven Nanobots: One of the recent and most important developments has been the ultrasound driven nanobots, where a local acoustic streaming strain is produced upon by ultrasonic waves providing the driving force. One of the greatest advantages has been the speed of movement and delivery. The High intensity forced ultrasound results into rapid evaporation of chemical fuels thus increasing the penetrability into even the most impermeable membranes. One of its important uses has been in HeLa cancerous cells by Garcia et al, 2014 [11]. They showed that 30% of payload (DOX) was released into the cancer cells after 15 minutes of trigger (NIR light irradiation). This also ensures quite fewer side effects as the time span of exposure is quite less.
• Biologically Driven Nanobots: This class is less common but equally effective if not more than the other classes. This type is commonly referred to as the biohybrid nanobots as beside constituting an artificial unit they consist of a bacterial subunit capable of propelling with its flagella. Another commonly used component is sperm. The major advantage of sperm hybridized nanobots is that they can not only carry high concentrations of drugs but can protect them from being diluted in the various body fluids and affected by enzymes across its pathway to the target site [12].
• Hybrid Models: The models discussed above were pure models, but keeping in mind bio safety and efficacy certain hybrid models haven been created combining these models. For instance, Victor et al, 2013 discovered a magnetic field guided three-segment Au–Ni–Au nanowire motor, which surprisingly can be guided by ultrasound. The change in magnetic field resulted in achievability of omni directional movement of the particle propelled by Ultrasound waves. Other models include Au half Nano shells that can produce a thermal gradient under NIR radiation, to provide a self- heating effect, overcoming the Brownian motion [13].
Production of Oxidative Stress
Superoxide dismutase (SOD) is an enzyme that converts superoxide radicals into hydrogen peroxide (H2O2). Nanobots can be engineered to upregulate the release of SOD within the cerebrospinal fluid. This upregulation leads to a substantial increase in H2O2 concentration, which induces oxidative stress. Cancer cells, being less equipped to handle oxidative damage compared to normal cells, suffer significant damage. This oxidative stress results in cellular damage and escalates apoptotic rates in cancer cells, effectively reducing the tumour burden [14,15].
DNA Cleavage
The small size of nanobots allows them to navigate through the nuclear pore complex and enter the nucleus. Once inside, nanobots can be designed to catalyse enzymatic reactions that convert hydrogen peroxide into highly reactive hydroxyl radicals. These hydroxyl radicals cause significant DNA damage by cleaving the DNA strands. The resulting DNA fragmentation prevents cancer cells from replicating and repairing their genetic material, ultimately leading to cell death through apoptosis [16].
Bax Upregulation
Nanobots can be programmed to upregulate the expression of Bax, a pro-apoptotic protein. The increased expression of Bax leads to its oligomerization and insertion into the mitochondrial membrane. This insertion causes the formation of glycolipid compounds that disrupt mitochondrial integrity, leading to the release of cytochrome C into the cytoplasm. Cytochrome C binds to apoptotic protease activating factor-1 (Apaf-1) and ATP, forming the apoptosome, which activates caspase 9. Caspase 9 then activates caspase 3, a crucial executioner caspase, leading to apoptosis and ensuring the systematic elimination of cancer cells [17].
Cell Cycle Arrest
Prolonged exposure to nanobots affects key neurotransmitters and enzymes, reducing levels of gamma-aminobutyric acid (GABA) and acetylcholinesterase, which are important for neuronal function. This disruption can impair cancer cell signalling and survival. Additionally, nanobots can inhibit the survival- promoting effects of brain-derived neurotrophic factor (BDNF) by increasing the secretion of lactate dehydrogenase (LDH), an enzyme indicative of cell damage. This process leads to the arrest of the cell cycle at the G2/M phase, preventing the division and proliferation of cancerous cells [17,18].
Mitochondrial Fission
Nanobots can induce mitochondrial fission by disrupting the TOM20 interconnection, a component crucial for mitochondrial protein import. They can also phosphorylate dynamin-related protein 1 (Drp1), a key regulator of mitochondrial fission. The phosphorylation of Drp1 facilitates its translocation to the mitochondrial outer membrane, where it promotes the division of mitochondria. This disruption of normal mitochondrial function leads to a loss of mitochondrial integrity and subsequent cell death. By targeting mitochondrial dynamics, nanobots impair the energy production and metabolic functions of cancer cells, making them more susceptible to apoptosis [19].
Regulation of Hypoxia-Inducible Factor (HIF) Pathway
The HIF pathway is essential for cellular responses to low oxygen conditions, often found in tumour microenvironments. Nanobots can inhibit hypoxia-inducible factor 1 (HIF-1), a key transcription factor that regulates genes involved in response to hypoxia, including those promoting angiogenesis, metabolism, and cell survival. By inhibiting HIF-1, nanobots prevent cancer cells from adapting to hypoxic conditions, reducing their ability to survive, proliferate, and metastasize. This inhibition also sensitizes cancer cells to further treatments, enhancing the overall therapeutic efficacy [20].
Nanobots offer a multifaceted approach to cancer treatment by exploiting various cellular mechanisms to induce cancer cell death. Their ability to induce oxidative stress, cause DNA damage, upregulate pro-apoptotic proteins, arrest the cell cycle, disrupt mitochondrial function, and inhibit critical survival pathways makes them potent agents against cancer. The precise targeting and multifactorial actions of nanobots provide a promising therapeutic strategy for effectively combating cancer while minimizing damage to healthy tissues.
Advantages
Nanorobotics is quite a growing subject and has quite a large number of advantages over the conventional treatment method involving administration of chemotherapy agents or use of gamma radiation.
Nanorobot drug delivery systems offer numerous advantages in medical treatments:
• Computer-Controlled Precision: Nanorobots can be programmed to control the dosage, frequency, and timing of drug release with high precision.
• Targeted Therapy: They precisely target malignant cells, reducing the impact on healthy cells and improving treatment efficacy over other chemotherapy agents.
• Small Size: With an upper size limit of 3 microns, nanorobots can navigate through the bloodstream without obstructing capillary flow.
• Enhanced Mass Transfer: The large surface area of nanorobots facilitates efficient mass transfer, allowing for optimal drug delivery.
• Non-Invasive Technique: This method is non-invasive, reducing the need for surgical procedures.
• Access to Remote Areas: Nanorobots can reach parts of the body that are inaccessible to traditional surgical methods and this proves to be a remarkable success.
• Computer-Controlled Precision: Nanorobots can be programmed to control the dosage, frequency, and timing of drug release with high precision.
• Improved Accuracy: The precise operation of nanorobots ensures accurate delivery of medication.
• Minimized Side Effects: Drugs are released only where needed, minimizing side effects and preventing the drug from acting on unintended areas.
• Increased Bioavailability: Nanorobots highly enhance the bioavailability of drugs, ensuring that more of the active ingredient reaches the target area in our body.
• These attributes collectively make nanorobot drug delivery systems a promising approach for improving the effectiveness and safety of medical treatments [7].
Challenges
As it’s known to all, no scientific methods are flawless, the application of nanobots in therapeutics also faces certain challenges that need to be overcome.
One of the most important conditions is biosafety and biopiracy. Nanobots technologies can easily be developed with ill intentions to target people of a certain country, ethnicity or race by programming nanobots to inject toxins without even being discovered by the immune system.
Nanobots consist of foreign DNA, and when they enter our body has a high possibility to trigger inflammatory and immunologic responses. While on the other hand several particles with which the outer shell is made may prove to be severely cytotoxic. For instance, Nickel used is highly allergenic and even carcinogenic [21]. On the other hand, the potentially inert silver used to produce hinges may prove to be neurotoxic [22].
In some Nanobots UV light is used as a potential source of propulsion, however, exposure to UV radiation over the time leads to skin damage and in the worst-case scenario, skin cancer. Another serious issue that arises is that, especially for self-multiplying nanobots, if somehow, we fail to communicate with it once inside the body, it may start malfunctioning. This malfunction may involve migration and release of drugs in healthy cells and most importantly uncontrolled self-multiplication that may even lead to death. Once excreted by humans, it may get transmitted to another human body unintentionally leading to uncontrolled spread of these nanobots that may even take the form of an epidemic, almost impossible to control due to its nanoscale size [21].
Future prospects & Conclusion
Nanobots, currently employed in specific functions such as diabetes diagnosis and cancer treatment, hold immense prospects for broader applications, particularly in treating neurodegenerative disorders like Alzheimer’s or Parkinson’s disease. Their potential to act as implants, replacing degenerated or inefficient neurons, is quite substantial. Moreover, nanobots could be pivotal in addressing amyloid plaques in the brain, a primary factor in Alzheimer's disease. In cardiovascular treatments, nanobots may replace traditional surgeries for arterial blockages and atherosclerosis, offering a minimally invasive alternative (Figure 2).
One of the most exciting advancements in nanotechnology is the application of swarm intelligence, where numerous nanobots are programmed to communicate and act collectively as a colony. This approach enhances their efficiency and precision, paving the way for breakthroughs in various medical fields. Nanobots on the other hand have a high potential to revolutionize delicate microsurgeries, such as retinal procedures, by significantly reducing risks associated with human error.
The trajectory of nanobots in cancer treatment heralds a future of unparalleled sophistication. Advances in biotechnology have enabled nanobots to target carcinogenic cells with unprecedented specificity and efficiency, potentially providing a flawless alternative to chemotherapy and surgery. Despite existing challenges, we can’t downplay the role of nanobots in cancer treatment. ongoing research and development efforts are likely to overcome these obstacles, making nanobots a cornerstone in the future of medical treatment [23-26].
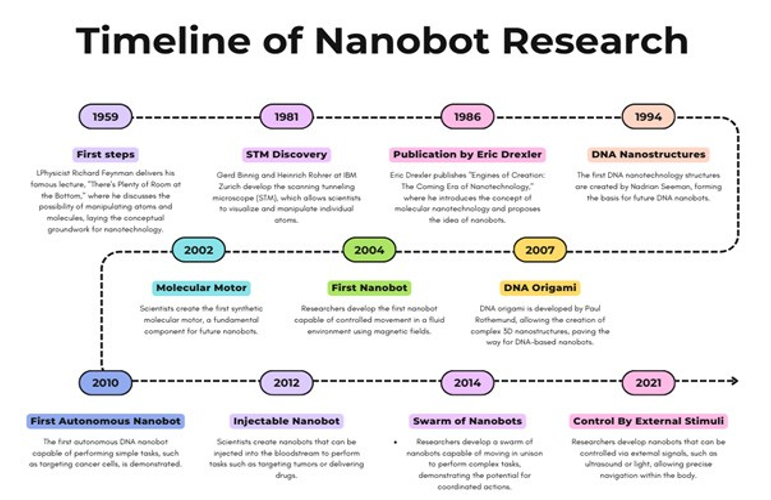
Figure 2: Timeline of Nanobot Research
References
- Schauer, O., Mostaghaci, B., Colin, R., Hürtgen, D., Kraus, D., Sitti, M., & Sourjik, V. (2018). Motility and chemotaxis of bacteria-driven microswimmers fabricated using antigen 43-mediated biotin display. Scientific reports, 8(1), 9801.
- Pradeep, P., Sonia, M. M. L., & Kizhakkepeedika, R. D. (2024). Nanobots in Cancer Treatment–Framing Lung Cancer. Journal of Advanced Lung Health, 4(2), 57-69.
- Liu, D., Guo, R., Wang, B., Hu, J., & Lu, Y. (2022). Magneticmicro/nanorobots: a new age in biomedicines. Advanced Intelligent Systems, 4(12), 2200208.
- Feng, Y., An, M., Liu, Y., Sarwar, M. T., & Yang, H. (2023). Advances in chemically powered micro/nanorobots for biological applications: a review. Advanced functional materials, 33(1), 2209883.
- Kabir, M. H., Islam, S. R., Shrestha, A. P., Ali, F., Badsha, M. A., Piran, M. J., & Do, D. T. (2021). Electromagnetic nanocommunication networks: Principles, applications, andchallenges. IEEE Access, 9, 166147-166165.
- Nanorobots: A Tiny Robot for Diagnosis and Treatment 2022- 03-10 , by Rosemary Salin.
- Kumar, S. S., Nasim, B. P., & Abraham, E. (2018). Nanorobots a future device for diagnosis and treatment. Journal of Pharmacy and Pharmaceutics, 5(1), 44-49.
- Zhang, D., Liu, S., Guan, J., & Mou, F. (2022). “Motile- targeting” drug delivery platforms based on micro/nanorobots for tumor therapy. Frontiers in Bioengineering and Biotechnology, 10, 1002171.
- Andhari, S. S., Wavhale, R. D., Dhobale, K. D., Tawade,B. V., Chate, G. P., Patil, Y. N., ... & Banerjee, S. S. (2020). Self-propelling targeted magneto-nanobots for deep tumor penetration and pH-responsive intracellular drug delivery. Scientific reports, 10(1), 4703.
- Gao, W., Kagan, D., Pak, O. S., Clawson, C., Campuzano, S., ChuluunErdene, E., ... & Wang, J. (2012). Cargotowing fuel free magnetic nanoswimmers for targeted drug delivery. small, 8(3), 460-467.
- GarciaGradilla, V., Sattayasamitsathit, S., Soto, F., Kuralay, F., Yardimci., Wiitala, D., ... & Wang, J. (2014). Ultrasound propelled nanoporous gold wire for efficient drug loading and release. Small, 10(20), 4154-4159.
- Xu, H., Medina-Sánchez, M., Magdanz, V., Schwarz, L., Hebenstreit, F., & Schmidt, O. G. (2018). Sperm-hybrid micromotor for targeted drug delivery. ACS nano, 12(1), 327- 337.
- Xuan, M., Shao, J., Gao, C., Wang, W., Dai, L., & He, Q. (2018). Selfpropelled nanomotors for thermomechanically percolating cell membranes. Angewandte Chemie International Edition, 57(38), 12463-12467.
- Liu, W., Worms, I., & Slaveykova, V. I. (2020). Interaction of silver nanoparticles with antioxidant enzymes. Environmental Science: Nano, 7(5), 1507-1517.
- Zhao, Y., Xiong, H., Li, Y., Gao, W., Hua, C., Wu, J., ... &Zheng, Y. (2022). Magnetically actuated reactive oxygen species scavenging nanorobots for targeted treatment. Advanced Intelligent Systems, 4(7), 2200061.
- DNA damage and Nanoparticles; Fenella Saunders, American Scientist, 2016.
- Seyedi, S. M. R., Asoodeh, A., & Darroudi, M. (2022). The human immune cell simulated anti-breast cancer nanorobot: the efficient, traceable, and dirigible anticancer bio-bot. Cancer Nanotechnology, 13(1), 44.
- Park, J. H., Gurunathan, S., Choi, Y. J., Han, J. W., Song, H., & Kim, J. H. (2017). Silver nanoparticles suppresses brain- derived neurotrophic factor-induced cell survival in the human neuroblastoma cell line SH-SY5Y. Journal of industrial and engineering chemistry, 47, 62-73.
- Dey, S., Fageria, L., Sharma, A., Mukherjee, S., Pande, S., Chowdhury, R., & Chowdhury, S. (2022). Silver nanoparticle- induced alteration of mitochondrial and ER homeostasis affects human breast cancer cell fate. Toxicology Reports, 9, 1977-1984.
- Xu.2020. Silver Nanoparticles: synthesis, medical application and bio-safety, Xu et al, 2020. Theragnostic.
- Arvidsson, R., & Hansen, S. F. (2020). Environmental and health risks of nanorobots: an early review. Environmental Science: Nano, 7(10), 2875-2886.
- Lin, T. W., Chang, C. F., Chang, Y. J., Liao, Y. H., Yu, H. M.,& Chen, Y. R. (2017). Alzheimer’s amyloid-β A2T variant and its N-terminal peptides inhibit amyloid-β fibrillization and rescue the induced cytotoxicity. PLoS one, 12(3), e0174561.
- Padshala, R., Rajan, V., & Patani, P. (2022). Nanobots: Future and Development. Journal of Pharmaceutical Negative Results, 13.
- DNA Nanobots Set to Seek and Destroy Cancer Cells in Human Trial – Janet Fang; 2016.
- Smriti Mathur, Anshul Agarwal, Nanorobotics, A hope for future, National Conference on Synergetic Trends in engineering and Technology (STET-2014) International Journal of Engineering and Technical Research ISSN: 2321- 0869, Special Issue.
- Liu, D., Guo, R., Wang, B., Hu, J., & Lu, Y. (2022). Magnetic micro/nanorobots: a new age in biomedicines. Advanced Intelligent Systems, 4(12), 2200208.


