Research Article - (2025) Volume 1, Issue 1
Trends in Mortality Related to Acute Renal Failure in the U.S. from 1999 to 2020: A Retrospective Analysis
2Hayatabad Medical Complex, Pakistan
3Yale School of Medicine, New Haven, USA
Received Date: Oct 03, 2025 / Accepted Date: Nov 04, 2025 / Published Date: Nov 17, 2025
Copyright: ©©2025 Ayesha Aman, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Akram, B., Aman, A., Akram, A., Tariq, E., Javaid, H., et al. (2025). Trends in Mortality Related to Acute Renal Failure in the U.S. from 1999 to 2020: A Retrospective Analysis. Hypertens OA, 1(1), 01-12.
Abstract
Background: Acute renal failure (ARF) indicates a sudden deterioration in renal function. This study analyzes mortality trends related to ARF in the U.S. from 1999 to 2020 to identify disparities.
Methods: We extracted the data from the Centers for Disease Control and Prevention's Wide-Ranging Online Data for Epidemi- ologic Research (CDC WONDER). Age-adjusted mortality rates (AAMRs) per 100,000 population were calculated for variables: year, gender, race and geographic regions. Annual percent changes (APCs) were analyzed using Joinpoint regression software.
Results: From 1999 to 2020, 1,174,156 deaths were reported for ARF-related fatalities in the U.S. Overall AAMR increased from 1999 (17.55, 95% CI 17.36 to 17.75) to 2010 (28.34,95% CI 28.2 to 28.6) with an APC of 4.44 (95% CI 4.17 to 4.7) followed by a decrease from 2010 to 2018 (24.51, 95% CI 24.32 to 24.71) and then an increase till 2020 (30.84,95% CI 30.62 to 31.05) with an APC of 11.85 (95 % CI 7.9 to 14.87). AAMR remained higher in males (30.82) than in females (20.31). Blacks (33.68) had the highest mortality rates, while Asian/Pacific Islanders had lower (16.38). Upon stratification by geographical distribution, the highest AAMR was in the South (26.37). The highest AAMR was observed in Texas and Indiana, with mortality rates being twice as high as states at the lower end of the spectrum, including Alaska, Arizona, and Florida.
Conclusion: High ARF-related mortality rates warrant the need for targeted public health initiatives to address these issues and provide healthcare access that can influence ARF-related outcomes.
Keywords
Acute Renal Failure, Mortality Trends, CDC-WONDER, Epidemiology
Abbreviations
ARF: Acute renal failure
AAMR: Age-adjusted mortality rate APC: Annual percent change
Introduction
Acute renal failure, also known as acute kidney injury, indicates a sudden and usually reversible deterioration in renal function decline in glomerular filtration rate, increased creatinine concentration in serum and plasma, and changes in urine output [1]. The disease burden can be seen from a study conducted in pediatric ICU that indicates that the incidence of ARF was found to be 4.3% among 1145 admissions, and it was the leading cause of mortality, accounting for a 39.1% mortality rate among affected individuals [2].
The causes may be divided into pre-renal, intrinsic and post-renal. Pre-renal causes include hypoperfusion prerenal azotemia that may be due to hypovolemia or low effective arterial blood volume as in heart failure and cirrhosis/hepatorenal syndrome [3]. The low renal perfusion leads to ischemia and necrosis of renal parenchyma and rapid decline of function. The reduced end arterial blood volume accounts for acute renal failure in hepatorenal syndrome, which is characterized by splanchnic vasodilation, leading to low kidney perfusion despite normal or even increased blood volume status [4]. Postrenal cause is urinary obstruction which may be due to stones, BPH, neoplasm or congenital anomalies [3]. All of these factors cause impaired flow of urine and thus lead to pooling of urine in areas proximal to the obstruction. This leads to a decrease in GFR and ultimately also causes a decrease in renal blood flow. These causes are usually reversible and must be treated promptly [5]. Intrinsic causes include acute tubular necrosis, glomerulonephritis and vasculitis [3]. Risk factors are chronic diseases such as heart failure, liver disease, and diabetes, age above 65 years, vascular diseases, drugs, electrolyte abnormalities, sepsis, etc [3].
Complications include electrolyte abnormalities hyperkalemia, acidosis, etc. and cardiovascular events, which include primarily arrhythmias influenced by these electrolyte abnormalities [6]. Other complications may be neurological, such as uremic encephalopathy, characterized by confusion, seizures, or even coma due to the accumulation of toxins in the blood [7]. Cardiopulmonary complications include pulmonary edema caused by fluid overload due to a decline in kidney function and partly due to fluid retention, and this may manifest as tachypnea and tachycardia [8].
The high load and mortality of the condition necessitated the evaluation of demographic mortality trends attributed to acute renal failure. Early diagnosis of this potentially fatal condition may improve clinical outcomes and prevent lifelong complications [9]. This study aims to identify mortality trends pertaining to acute renal failure from 1999 to 2020. The time zone signifies the advancements made in medical and surgical techniques in the two decades between 1999 and 2020. Clinicians will benefit from the findings of this study.
Materials and Methods
Study Design and Population
We extracted the data for this study from the Centers for Disease Control and Prevention’s (CDC) Wide-Ranging Online Data for Epidemiologic Research (WONDER) database. We evaluated the mortality trends due to Acute renal failure in individuals aged 25 years and above in the United States from the year 1999 to 2020. The International Statistical Classification of Diseases and Related Health Problems (ICD-10), 10th Revision codes NI7.0, N17.1, N17.2, N17.8, and N17.9 were used to identify cases of acute renal failure with tubular, cortical, medullary necrosis and other unspecified causes. Prior studies have utilized similar codes for such analysis [10]. Our study focused on certificates of death within the Multiple Cause of Death Public Use dataset to retrieve the mortality data among patients aged ≥25 years with acute renal failure listed as the cause of death. There was no requirement for Institutional review board approval as the data utilized was retrieved from a government-provided, de-identified, publicly available dataset. This study follows the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines for reporting.
Data Extraction
Data from 50 states of the U.S. was retrieved from 1999 to 2020. Extracted data was categorized according to the following variables: year, gender, race, census region and state. Different races included nonâ?Hispanic (NH) White, NH Black or African American, Hispanic or Latino, NH American Indian or Alaskan Native, and NH Asian or Pacific Islander. Consensus regions were divided into four regions: Northeast, Midwest, South and West, as defined by the US Census Bureau [11]. The patients were distributed into 10-year intervals based on whether they were young adults (25–44 years), middle-aged adults (45–64 years), or older adults (65–85+ years).
Statistical Analyses
We calculated the crude and age-adjusted mortality rates (AAMR) per 100,000 individuals for acute renal failure. This enabled us to adequately analyze the mortality rates over the years. AAMR was standardized to the 2000 US population, a method that accounts for the differences in age distribution in the population [12]. While using the statistical software Joint Point Regression Program (Joinpoint V 5.3.0, National Cancer Institute), Annual percent change (APC) values with 95% CI in AAMR were calculated, and temporal trends were evaluated by fitting log-linear regression models to our raw data. The Grid Search method was used to assess all the join points within the data. Permutation test and parametric method were utilized to estimate the annual percent change (APC) and corresponding 95% CIs. Increasing or decreasing trends as indicated by the APC values were assessed by their statistical significance. APC values were considered significant if P≤0.05, according to 2-tailed t-tests.
Results
Annual Trends
A total of 1174156 deaths were recorded between 1999 and 2020 with an AAPC of 2.45 (95% CI 2.11 to 2.97) among adults above 25 years of age caused by acute renal failure. The overall age- adjusted mortality rate (AAMR) in 1999 was 17.55 (95% CI 17.36 to 17.75) which increased till 2010 to 28.34 (95% CI 28.2 to 28.6) with an APC of 4.44 (95% CI 4.17 to 4.7). Conversely, there was a decline in AAMR from 2010 to 2018 (24.51 95% CI 24.32 to 24.71) with an APC of -2.2 (95% CI -2.68 to -1.86) and then there was again an increase in AAMR till 2020 (30.84, 95% CI 30.62 to 31.05) with an APC of 11.85 (95 % CI 7.9 to 14.87) (Figure 1).
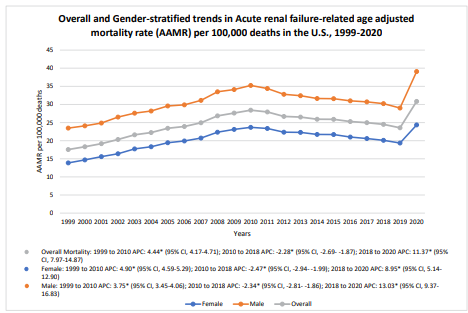
Figure 1: Overall and Gender-stratified Trends in Acute renal failure-related AAMR per 100,000 deaths in adults (> 25 years) in the United States, 1999 to 2020. [* Indicates that Annual Percent Change (APC) is significantly different from zero at α = 0.05; AAMR = Age adjusted mortality rate]
Stratification by Gender
The age-adjusted mortality rate (AAMR) for acute renal failure stratified by gender revealed a higher rate in males (30.82, 95% CI 30.75 to 30.9) than in females (20.31, 95% CI 20.26 to 20.36) throughout the study period, although both reported similar trends in AAMR. In 1999, the AAMR for males was 23.47 (95% CI 23.1 to 23.85), which showed a significant increase to 35.24 (95% CI 34.84 to 35.64) till 2010 with an APC of 3.75 (95% CI 2.7 to 5.22) and then showed a decrease in AAMR to 30.23 (95% CI 29.9 to 30.56) till 2018 with an APC of (-2.33, 95% CI -7.5 to -1.10). A significant rise in AAMR was again reported from 2018 to 2020 (AAMR=39.08, 95% CI 38.71 to 39.44) with an APC of 13.03 (95% CI 3.24 to 18.63). In 1999, the AAMR for females was 13.86 (95% CI 13.64 to 14.09) which showed an increasing trend till 2010 to 23.68 (95% CI 23.41 to 23.95) with an APC of 4.89 (95% CI 4.07 to 5.89) and then a decreasing trend was reported till 2018 to 20.11 (95% CI 19.87 to 20.34) with an APC of -2.4 (95% CI -5.4 to -1.4). A significant rise in AAMR was again reported from 2018 to 2020 (AAMR=24.34, 95% CI 19.87 to 20.34) with an APC of 8.95 (95% CI 1.33 to 12.88) (Figure 1).
Stratification by Race
The age-adjusted mortality rate for acute renal failure stratified by race revealed highest AAMR in African American patients with a rate of 33.68 (95% CI 33.5 to 33.85), followed by American Indian patients (25.28, 95% CI 24.71 to 25.85), White patients (23.97, 95% CI 23.92 to 24.01), Hispanic/Latino patients (22.24, 95% CI 22.08 to 22.41). The lowest AAMR was recorded in Asian or Pacific Islander patients (16.38, 95 % CI 16.19 to 16.57). In 1999, the AAMR in African American patients was 27.61 (95% CI 26.76 to 28.46) which increased till 2009 to 38.02 (95% CI 37.12 to 38.92) with an APC of 3.18 (95% CI 2.5 to 3.7) which then decreased till 2018 to 30.22 (95% CI 29.53 to 30.9) with an APC of -3.2 (95% CI -3.9 to -2.5) and then showed a significant increased till 2020 to 44.67 (95% CI 43.87 to 45.48) with an APC of 21.52 (95% CI 14.95 to 28.47). The White patients reported a similar trend in AAMR.
In 1999, the AAMR was 16.62 (95% CI 16.41 to 16.82) which increased till 2010 to 27.65 (95% CI 27.41 to 27.89) with an APC of 4.71 (95% CI 4.44 to 4.99) which decreased till 2018 to 24.24 (95% CI 24.03 to 24.45) with an APC of -2.06 (95% CI -2.49 to -1.63) and then showed a significant increase till 2020 to 24.63 (95% CI 29.4 to 29.86) with an APC of 9.33 (95% CI 6.02 to 12.74) (Figure 2).
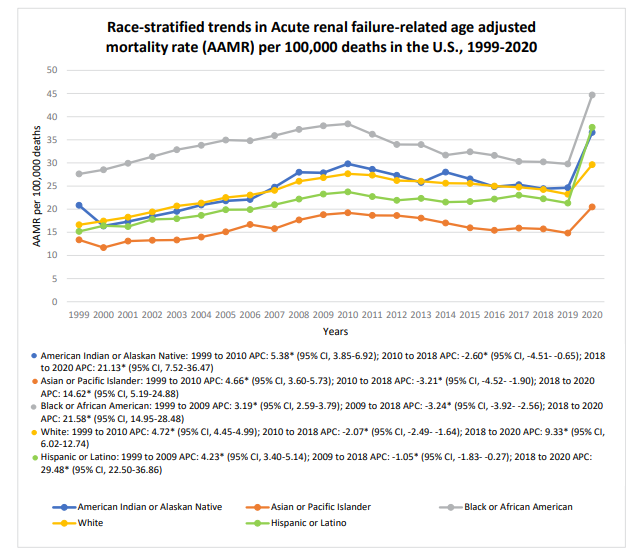
Figure 2: Race-stratified Trends in Acute renal failure-related AAMR per 100,000 deaths in adults (> 25 years) in the United States, 1999 to 2020. [* Indicates that Annual Percent Change (APC) is significantly different from zero at α = 0.05; AAMR = Age adjusted mortality rate]
Stratification by Geographical Variations
The age-adjusted mortality rate for acute renal failure stratified by states revealed the highest AAMR in Texas that was 33.5 (95% CI 33.29 to 33.7), followed by Indiana (33.32, 95% CI 32.96 to 33.68) and the lowest was recorded in Florida with an AAMR of 16.18 (95% CI 16.05 to 16.31). Other states in which the upper 90th percentile of acute renal failure-related mortality were Delaware, Kentucky, South Carolina and West Virginia whereas the states in the lower 10th percentile were Alaska, Arizona, Colorado, Montana, and New York (Figure 3).
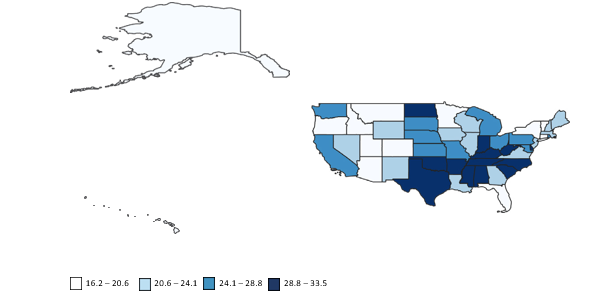
Figure 3: State-wise map showing states with highest and lowest mortality (arranged by state-wise age adjusted mortality rate per 100,000 deaths) in the United States, 1999 to 2020
Among all the regions in the US, the highest AAMR was recorded in the Southern region (26.37, 95% CI 26.29 to 26.44), followed by the Midwestern region (25.37, 95% CI 25.27 to 25.46) and the West (23.37, 95% CI 23.28 to 23.47). The lowest AAMR was recorded in the Northeast region (21.85,95% CI 21.75 to 21.94) (Figure 4).
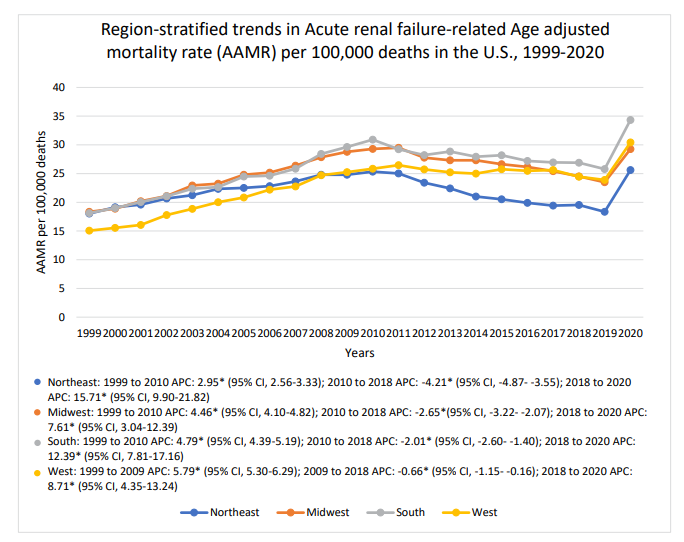
Figure 4: Region-stratified Trends in Acute renal failure-related AAMR per 100,000 deaths in adults (> 25 years) in the United States, 1999 to 2020. [* Indicates that Annual Percent Change (APC) is significantly different from zero at α = 0.05; AAMR = Age adjusted mortality rate]
Discussion
There has been an overall increase in mortality with the observed AAPC of 2.45 over the two decades, suggesting an exacerbating burden of acute renal failure with tubular necrosis. It is associated with three distinct phases: an initial rise from 1999 to 2010, a decline from 2010 to 2018 and a sharp increase from 2018 to 2020. Our study reveals that mortality rates are more consistent in males than in females, although both follow similar trends. Among the racial groups, African American patients have the highest AAMR followed by American Indians, White, Hispanic\Latino and Asian\Pacific islander patients. Geographical variations are also significant as the highest mortality rates are documented in Texas, Indiana and the Southern U.S. while the lowest are in Florida and the Northeast.
The initial increase in mortality is possibly due to the rising prevalence of diabetes and hypertension with advancing age. Diabetic ketoacidosis in type 1 diabetes increases the risk of developing acute renal failure [13]. Sepsis with urinary tract obstruction is a significant cause of acute renal failure in diabetics [14]. Disruption of the signaling pathways and increased vulnerability to hypoxia increases the risk of acute kidney damage [15]. Acute kidney injury is considered a major complication of diabetes and people with diabetes are more likely to develop it. Increased incidence of hypertension also elevates the likelihood of developing acute renal failure with oxidative stress as a key factor linking hypertension and acute renal failure [16]. A decline in the mortality trend from 2010 to 2018 is worth observing which can be contributed to improved clinical management and intervention. Improved treatment focusing on the underlying causes, optimizing hemodynamics and managing electrolyte disturbances can reduce mortality significantly [17]. Early initiation of renal replacement therapy can prolong survival [18].
The spike that is seen in the third phase from 2018 to 2020 can be attributed to disruptions in healthcare systems with the advent of COVID-19 which heavily burdened the ICUs.COVID-19 can cause acute kidney injury, posing challenges in the administration of renal replacement therapy [19]. Kidney is one of the major target organs of SARS-COV-2, directly affecting the renal tubules [20]. People with comorbidities and the elderly are more vulnerable to develop AKI due to COVID-19 which explains the sharp rise in mortality from 2018 to 2020 [21]. Hospital stays and the increased load of ICU patients during the pandemic might have contributed to the sharp rise in mortality in the third phase. The management of COVID-19 requires medicines that can put a load on the kidney [22]. Our study shows gender disparities, with males having higher mortality rates compared to females, although similar trends are followed by both. Males show greater renal impairment as compared to females which can be attributed to the protective role of female hormones and detrimental effects of male hormones. Estrogen is nephroprotective which plays a significant role in reducing oxidative stress as compared to testosterone which hastens kidney damage [23].
So, there is a complex interplay between gender and kidney injury susceptibility. Higher mortality in males can also be attributed to a higher prevalence of comorbidities like diabetes and hypertension in males more than females which can be seen across various ethnic groups like urban Bhutanese and western Nepal [24,25]. Another point worth discussing is the higher incidence of smoking and alcohol consumption in males which is a risk factor for acute kidney disease [26]. Men also tend to delay seeking healthcare due to masculinity norms which results in poorer outcomes and, hence, is one of the reasons for high rates of acute kidney injury in the male population [27].
Racial disparities in acute kidney injury are worth noting, with the black population facing greater risks compared to their White counterparts. This can be attributed to lower socioeconomic status, differences in health care delivery, and genetic factors. The disproportionate burden is greatest among African Americans and American Indians, which can be accredited to the presence of APOL1, a risk variant associated with faster progression to kidney disease [28]. Higher incidence of hypertension, diabetes and obesity in African Americans is another one of the risk factors for higher rates of acute kidney disease in this racial group [29]. Due to better access to healthcare and facilities and socioeconomic factors, the incidence is relatively lower among whites [30].
Geographical variations demonstrate the highest AAMR in Texas and the Southern U.S. and the lowest in the Northeast region. Better healthcare access and dialysis-requiring management in the Northeast region mitigates the burden of acute kidney injury [31]. The incidence of comorbidities like diabetes, hypertension and obesity in the Southern and Midwest US is far greater than in the Northeast owing to the highest AAMR in these areas [32]. Southern U.S. states like Texas and Indiana have a warmer climate compared to the Northeast states which predisposes the population to dehydration and hyperosmolality, heightening the risk of acute kidney injury [33].
Our study encompasses a nationally representative cohort of the U.S. population with detailed stratification like higher mortality rates in males, in the Southern and Midwest states and specific racial groups. It also covers 21 years from 1999 to 2020 with the identification of three major trend phases. It also uses indicators AAMR and APC which reduce the bias and highlight the statistically significant shifts. This allows comprehensive analysis of the data which then guides the targeted interventions in the high-risk groups and regions.
There are some limitations in our study. First, it is retrospective which increases the potential for bias and unreliability. Second, there is a lack of clinical history, treatment plan and lifestyle factors which limits the appropriate analysis of our findings. Third, the results cannot be generalized and the findings are limited by reporting bias and regional differences in healthcare access. Fourth, the death certificates and ICD coding in our study produce misclassification errors, which can lead to data discrepancies and, hence, affect the interpretation of our results.
In conclusion, this study shines light on the disparities in mortality due to acute renal failure. Following a period of decline, there has been a steep rise in mortality in the last decade which indicates the need for expanding the screening programs for early detection, improving healthcare access in vulnerable communities, and establishing climate-related and gender-specific health policies to reduce mortality.
References
- Maund, V. (2023). Care of the acute kidney injury patient.Companion Animal, 28(10), 2-7.
- Pournasiri, Z., Hashemi, S. M., Ahmadizadeh, S. N., Behzad, A., Esfandiar, N., & Hakimhashemi, A. (2024). Acute Renal Failure in the Pediatric Intensive Care Unit: Incidence and Its Effects on Clinical Outcomes. IRANIAN JOURNAL OF PEDIATRICS, 34(4).
- Mercado, M. G., Smith, D. K., & Guard, E. L. (2019). Acute kidney injury: diagnosis and management. American family physician, 100(11), 687-694.
- Simonetto, D. A., Gines, P., & Kamath, P. S. (2020). Hepatorenal syndrome: pathophysiology, diagnosis, and management. bmj, 370.
- Martinez-Maldonado, M., & Kumjian, D. A. (1990). Acute renal failure due to urinary tract obstruction. The Medical clinics of North America, 74(4), 919-932.
- Agrawal, M., Swartz, R. (2025). Acute renal failure [published correction appears in Am Fam Physician 2001 Feb 1;63(3):445]. Am Fam Physician. 2000;61(7):2077-2088.
- Akram, B., Aman, A., Akram, A., Tariq, E., Javed, H., Naveed, H., ... & Gul, M. H. (2025). Trends in Mortality Related to Acute Renal Failure in the US from 1999 to 2020: A CDC- WONDER Retrospective Analysis.
- Boutrus, T.,S. (2025). Basics of the Management of Acute Renal Failure and its Complications for the General Physician. J Intern Med Emerg Res.
- Peerapornratana, S., Manrique-Caballero, C. L., Gomez, H., & Kellum, J. A. (2019). Acute kidney injury from sepsis: current concepts, epidemiology, pathophysiology, prevention and treatment. Kidney international, 96(5), 1083-1099.
- Maqsood, M. H., Talha, K. M., Minhas, A. M. K., Fudim, M., Khan, S. S., Butler, J., & Khan, M. S. (2023). CDC-WONDERdatabase analysis of COVID-19 and cardiovascular disease- related mortality. Journal of the American College of Cardiology, 81(17), 1743-1745.
- Ingram, D. D., & Franco, S. J. (2014). 2013 NCHS urban-rural classification scheme for counties (No. 2014). US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics.
- Anderson, R. N., & Rosenberg, H. M. (1998). Age standardization of death rates; implementation of the year 2000 standard.
- Woodrow, G., Brownjohn, A. M., & Turney, J. H. (1994). Acute renal failure in patients with type 1 diabetes mellitus. Postgraduate Medical Journal, 70(821), 192-194.
- Ramakrishnan, S., GPV. (2025). Acute Renal Failure in Diabetes Mellitus (Prospective Study). J Nephrol Ther. 2013;03(03).
- Infante, B., Conserva, F., Pontrelli, P., Leo, S., Stasi, A., Fiorentino, M., ... & Stallone, G. (2023). Recent advances in molecular mechanisms of acute kidney injury in patients with diabetes mellitus. Frontiers in Endocrinology, 13, 903970.
- Yembarwar, N. K., & Guddetwar, S. G. (2022). To study the biochemical parameter in hypertension and renal failurE. International Journal of Medical and Biomedical Studies, 6(2), 25-29.
- Hertzberg, D., Rydén, L., Pickering, J. W., Sartipy, U., & Holzmann, M. J. (2017). Acute kidney injury—an overview of diagnostic methods and clinical management. Clinical kidney journal, 10(3), 323-331.
- Laake, J. H., & Bugge, J. F. (2010). Akutt nyresvikt hos intensivpasienter. Tidsskrift for Den norske legeforening.
- Adapa, S., Aeddula, N. R., Konala, V. M., Chenna, A.,Naramala, S., Madhira, B. R., ... & Bose, S. (2020). COVID-19 and renal failure: challenges in the delivery of renal replacement therapy. Journal of clinical medicine research, 12(5), 276.
- Diao, B., Wang, C., Wang, R., Feng, Z., Zhang, J., Yang, H., ... & Chen, Y. (2021). Human kidney is a target for novel severe acute respiratory syndrome coronavirus 2 infection. Nature communications, 12(1), 2506.
- Del Giudice, A., Piemontese, M., Valente, G., Prencipe, M., Di Giorgio, C., & Aucella, F. (2012). Acute kidney injury in the elderly: epidemiology, risk factors and outcomes. J Nephrol Therapeut, 2(129), 2161-0959.
- Perico, L., Benigni, A., & Remuzzi, G. (2020). Should COVID-19 concern nephrologists? Why and to what extent? The emerging impasse of angiotensin blockade. Nephron, 144(5), 213-221.
- Hosszu, A., Fekete, A., & Szabo, A. J. (2020). Sex differences in renal ischemia-reperfusion injury. American Journal of Physiology-Renal Physiology, 319(2), F149-F154.
- Sharma, S., & Thapa, B. (2022). Community-Based Screening for Hypertension and Diabetes among Adults: Study of Western Nepal. Asian Journal of Population Sciences, 58-65.
- Giri, B. R., Sharma, K. P., Chapagai, R. N., & Palzom, D. (2013). Diabetes and hypertension in urban bhutanese men and women. Indian Journal of Community Medicine, 38(3), 138-143.
- P P. Cigarette Smoking: Risk Factor for Kidney Injury. Open Access J Urol Nephrol. 2023;8(1):1-4.
- Chikovore, J., Hart, G., Kumwenda, M., Chipungu, G. A., & Corbett, L. (2015). ‘For a mere cough, men must just chew Conjex, gain strength, and continue working’: the provider construction and tuberculosis care-seeking implications in Blantyre, Malawi. Global health action, 8(1), 26292.
- Panico, C., & Thompson, A. (2018). African Americans, kidney disease, and drug development: A regulatory perspective. American Journal of Kidney Diseases, 72(5), S33-S36.
- Turksonâ?Ocran, R. A. N., Nmezi, N. A., Botchway, M. O., Szanton, S. L., Golden, S. H., Cooper, L. A., & Commodoreâ? Mensah, Y. (2020). Comparison of cardiovascular disease riskfactors among African immigrants and African Americans: an analysis of the 2010 to 2016 National Health Interview Surveys. Journal of the American Heart Association, 9(5), e013220.
- Lipworth, L., Mumma, M. T., Cavanaugh, K. L., Edwards,T. L., Ikizler, T. A., E. Tarone, R., ... & Blot, W. J. (2012). Incidence and predictors of end stage renal disease among low-income blacks and whites. PloS one, 7(10), e48407.
- Brown, J. R., Rezaee, M. E., Hisey, W. M., Cox, K. C., Matheny, M. E., & Sarnak, M. J. (2016). Reduced mortality associated with acute kidney injury requiring dialysis in theUnited States. American journal of nephrology, 43(4), 261- 270.
- Jackson, B. E., Oates, G. R., Singh, K. P., Shikany, J. M., Fouad, M. N., Partridge, E. E., & Bae, S. (2017). Disparities in chronic medical conditions in the Mid-South. Ethnicity & health, 22(2), 196-208.
- Johnson, R. J., Sánchez-Lozada, L. G., Newman, L. S., Lanaspa, M. A., Diaz, H. F., Lemery, J., ... & Roncal-Jimenez,C. A. (2019). Climate change and the kidney. Annals of Nutrition and Metabolism, 74(Suppl. 3), 38-44.



