Research Article - (2021) Volume 6, Issue 2
Treatment of Iatrogenic Coronary Artery Dissection in Resource Constraint in Cath-Lab, A Retrospective Study
2Demonstrator, Department of Pathology, Bilawal Medical College, LUMHS, Jamshoro, Pakistan
Received Date: Jul 08, 2021 / Accepted Date: Jul 21, 2021 / Published Date: Aug 05, 2021
Copyright: ©Khalida Soomro, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Khalida Soomro, Muhammad Ali Soomro (2021) Treatment of Iatrogenic Coronary Artery Dissection in Resource Constraint in Cath-Lab, A Retrospective Study. Cardio Open, 6(2): 141-145.
Abstract
Introduction: Coronary Artery Dissection is a well-known serious complication of invasive procedures (e.g. coronary angiography, balloon angioplasty and surgical cardioplegia) and is due to mechanical injury of the arterial wall by interventional devices used during procedures needing skilled operator to manage it timely.
Objective: We wanted to look at these large tears that can dramatically affect blood flow and result in severe complications to understand where they happen most and how efficient are we at managing them for our patients in limited resources of the Cath-labs in developing countries.
Methods: We reviewed the record of 28 (3.4%) cases of large dissection found in 806 patients who underwent coronary angioplasty in Cath lab of Cardiology Department of Dow University of Health Sciences Karachi. We analyzed as per American College of Cardiology/American Heart Association (ACC/AHA) criteria of the type of lesion, chronic total occlusion, calcification, intra-procedural dissection, and vessel site, related complications and management strategies.
Results: Among the 806 patients that underwent per-cutaneous coronary intervention during 3 years (2014-2017) period at Cath-lab of Cardiology Department of Dow University of Health Sciences Karachi, 28 (3.4%) patients were found to have extensive dissection. Out of 10 patients who had immediate angioplasty and stenting for closure of dissection 8 patients were successfully treated and 2 (7.1) died during procedures. 8 (28.5%) needed immediate coronary artery bypass surgery. 10 (35.7%) out of the 28 (3.4%) patients were managed by further hospitalization with medical treatment out of them 3 patients needed angioplasty procedures. There were no late deaths.
Conclusion: Coronary artery dissection remains a common occurrence during PCI since pre-stent procedures and clinical sequelae have been minimized by the routine use of coronary stents. Rapid recognition and attention to the angiographic appearance of the dissection is essential to the successful management of this complication. None the less, patients with extensive dissection who are free from the manifestation at the end of the procedure can be managed conservatively. Attempts should be made to stabilize extensive dissection during coronary angioplasty so that surgical intervention can be delayed or avoided altogether if possible.
Keywords
Cath-lab: Catheterization Laboratory, PCI: Per-cutaneous Coronary Intervention, BMS: Bare Metal Stent, DES: Drug-Elut- ing Stents, TIMI: Thrombolysis in Myocardial Infarction, LCX: Left Circumflex Artery, RCA: Right Coronary Artery, LAD: Left An- terior Descending Artery, MI: Myocardial Infarction, CABG: Coronary Artery Bypass Graft, IVUS: Intravascular Ultrasound, PTCAs: Per-cutaneous Transluminal Coronary Angioplasties.
Introduction
A ‘controlled tear’ is the mechanism by which angioplasty over dilates the blocked vessels. A large tear, or spiral dissection, that continues almost entirely down the artery, however is associated with serious complications. When such a dissection occurs, the interior wall of the artery is torn, causing it to fold into the path of blood flow and sometimes block flow of blood in the artery al- together [1,2]. Blockages in the right coronary artery and those in bending areas of the coronary arteries are the most common plac- es for coronary dissection. Coronary dissections occur frequently after balloon dilation of coronary atherosclerotic plaques proce- dures performed in the pre-stent area and now-a-days due to use of coronary bare-metal stents (BMS) and coronary interventional de- vices e.g. de-bulking devices intended to remove or ablate tissue, Trans-luminal extraction coronary atherectomy and Excimer laser coronary angioplasty [3,4]. Indeed, flow-limiting dissections occur during or at the end of the procedure and persisting post-procedur- al area may be a consequence of extensive vessel trauma or due to lesion complexity, can provide a pro-thrombotic milieu leading to abrupt vessel closure, thrombosis, with ensuing myocardial isch- emia and necrosis [5]. However, the adverse role of minor and non-obstructive procedural dissections has been questioned in- cluding target lesion revascularizations during hospitalization and follow up [4,6].
Objectives
“We wanted to witness large tears that can dramatically affect blood flow and result in severe complications to understand where they happen most and how good we are at fixing them for our pa- tients” in a resource constrained Cath-lab of public sector hospital of low socio economical country.
Methods
We reviewed the records of 28 (3.4%) cases of large dissection in 806 patients that underwent coronary angioplasty in Cath lab of Cardiology department of Dow university of Health Sciences Karachi. All the patients had history of administered bolus of un- fractionated heparin at a dose of 70 IU/kg to achieve an activated clotting time ≥250 s at the beginning of the procedure. Patients were administered 300-mg clopidogrel loading dose in non-pre- treated patients. Glycoprotein IIb/IIIa (GpIIb/IIIa) inhibitors were used electively or as bail-out at the discretion of the operator. Cor- onary angioplasty and stenting were performed with the aim to fully cover the diseased segment. In the case of dissection after stent deployment, the recommended management strategy was ei- ther to refrain from further percutaneous maneuvers because the dissection was not associated with impaired epicardial flow or myocardial ischemia or to try managing per-cutaneously. Whenev- er dissections were found resistant to balloon inflations and stent- ing, approaches were not feasible for lesion characteristics (e.g. due to very distal lesion, calcifications, or vessel tortuosity) or for the impossibility of tracking other DES in place (e.g. bifurcation), dissections were left in place and managed conservatively. Only dissections occurring either before or after stent implantation and persists after the last balloon inflation were adjudicated as final dissections [7,8].
These were classified according to the National Heart Lung and Blood Institute (NHLBI) criteria:
(i) minor radiolucent areas within the coronary lumen during con- trast injection, with little or no persistence of contrast after the dye has cleared
(ii) parallel tracts or a double lumen separated by a radiolucent area during contrast injection, with minimal or no persistence after dye clearance
(iii) contrast outside the coronary lumen with persistence of con- trast after dye has cleared from the lumen
(iv) spiral luminal filling defects
(v) appearance of a new, persistent filling defects within the coro- nary lumen, and
(vi) dissections leading to total occlusion of the coronary lumen without distal ante grade flow [9,10].
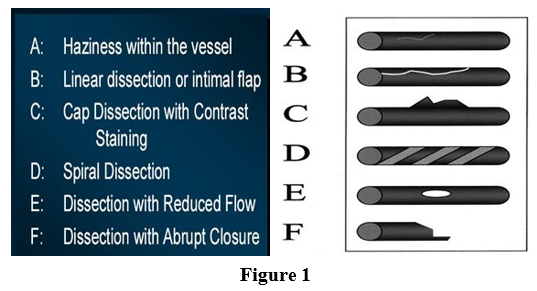
Final distal coronary flow was assessed according to the epicardial thrombolysis in myocardial infarction (TIMI) flow classification (from 0 to 3) in patients without persistent dissections and in those with dissections type A. We saw for as per American College of Cardiology/American Heart Association (ACC/AHA) criteria le- sion type, chronic total occlusion, calcific lesion, intra-procedur- al dissection, bifurcation with balloon-only dilation of the side branch, use of GpIIb/IIIa inhibitors, and vessel site [11].
Results
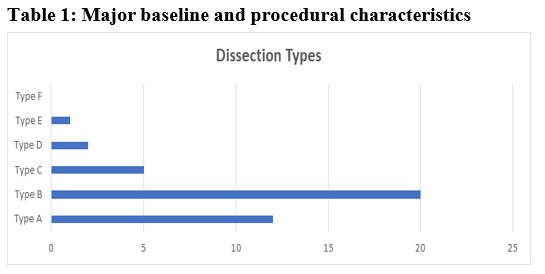
Among the 806 patients that underwent percutaneous coronary intervention during 3 years (2014-2017) period at Cath-Lab of Cardiology Department of Dow University of Health Sciences Karachi, 28 (3.4%) patients had extensive dissection (defined as a dissection extending beyond the limits of the dilated angioplasty balloon) in the coronary artery, Dissection type was found A in 12 (42.8%) cases, B in 20 (71.4%), C in 5 (17.5%), D in 2 (7.1%), E in 1 (3.5%), and F in 0 (0.0%) Table1.
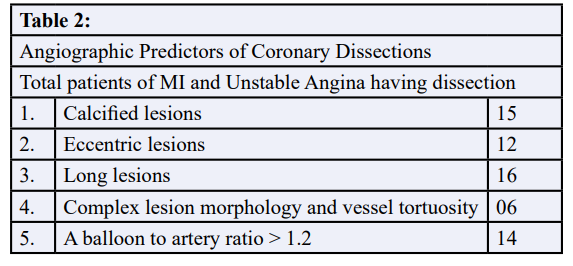
Angiographic Predictors of Coronary Dissections in patients who underwent angioplasty because of Myocardial infarction or unsta- ble angina that was refractory to medical treatment were calcified lesions in 15 (53.5%), eccentric lesions in 12 (42.8%), long lesions in 16 (57.1%), complex lesion morphology and vessel tortuosity in 06 (21.4%), A balloon to artery ratio >1.2 in 14 (50%) patients Table 2.
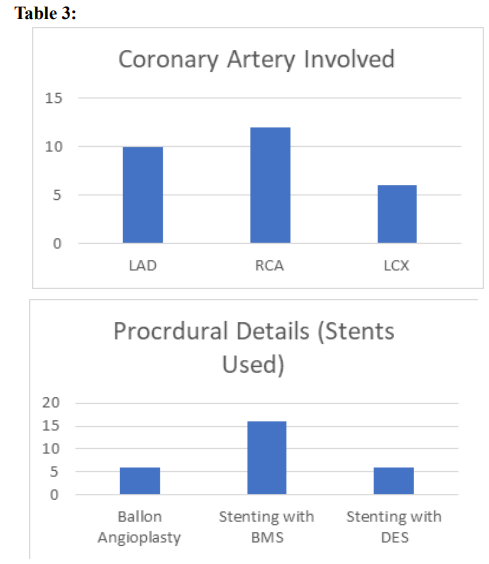
Dissection occurred in 6 patients with balloon angioplasty and in 22 patients during procedure of angioplasty with stenting. Overall, final dissections occurred with increased frequency, as expected in longer and more complex lesions, 10 (35.7%) patients with left anterior descending artery, in 6 (21.4%) patients with side branch- es of the LCX. In 12 (42.8%) patients with RCA complicated by propagating dissection compared to other coronary arteries.
Lesions on a bend of 45 degrees or greater were 12 times more likely to develop a dissection compared to lesions that were not on a bend Table 4 and in patients with normal distal epicardial flow i.e. TIMI 3 present with dissections in 13 (46.4%). 10 patients had immediate angioplasty with stenting, out of them 8 patients successfully treated and 2 (7.1) died during procedures of closure of dissection with stenting. 8 (28.5%) needed immediate coronary artery bypass surgery. Table 4A and Table 4B.
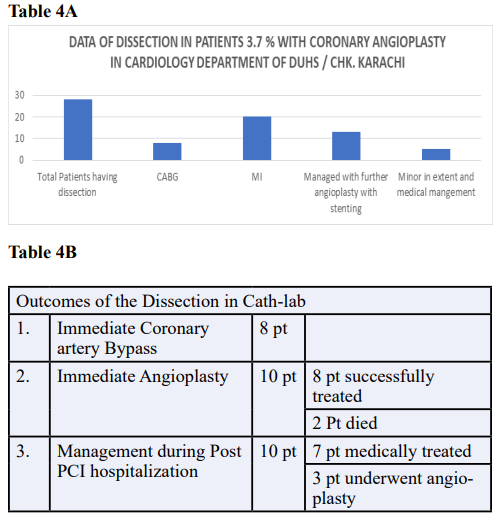
10 (35.7%) out of the 28patients were managed by further hospi- talization with medical treatment; out of them 3 patients needed angioplasty procedures. There were no late deaths
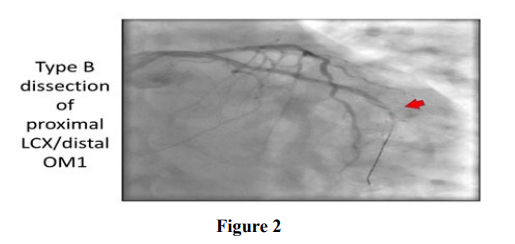
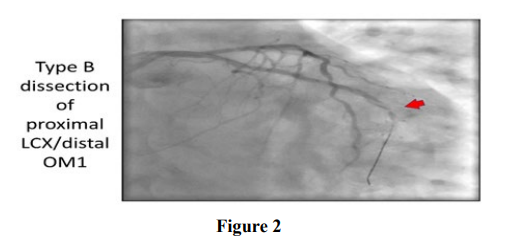
Stenting was successful in treating the dissection in 11 (39.2%) percent of patients. Major in-hospital adverse coronary events (heart attack, need for emergent bypass surgery, stent thrombosis or death) occurred in 18 (53.5%) percent of patients in the large dissection group
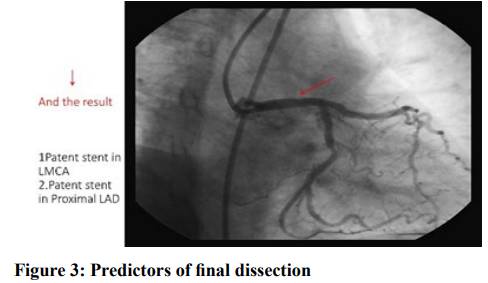
Discussion
Despite its numerous benefits, serious and potentially life-threat- ening complications can occur up to 30% of all conventional bal- loon angioplasties and may result in angiographically significant coronary artery dissection. With the advent of coronary stents, the incidence of acute closure in elective PCI is now less than 1%. But plaque fracture and tearing when occurs are critical in plaque redistribution and reduction of elastic recoil, as well as in later positive coronary remodeling [12-16]. However, plaque disrup- tion with exposure of its prothrombotic milieu and concomitant intimal flaps may promote thrombosis and distal embolization or even impair distal flow directly when large dissections are pres- ent. These pathophysiological events and data underlie in the as- sociation between angiographically evident coronary dissections and adverse clinical outcomes seen with increased frequency in resource constrained Cath-lab. Our study is reporting for the first time on the incidence, occurrence of dissection during procedure, and outcomes of final residual dissections after coronary angio- plasty and stenting. Our study confirms the detrimental clinical impact of this procedural complication dissection in 3.4% patients which is more than Cath-labs of developed countries after the ad- vent of stents, associated with significantly increased rate adverse events. In 10 patients undergoing stenting for closure of dissection. 2 (7.1%) died during procedure. 8 (28.5%) needed immediate cor- onary artery bypass surgery. 11 patients had successful PCI with DES Stenting in patient with dissection. Thus, our study provides compelling data in support of a safe strategy of managing residual dissections and related complications with limited resources.
Cappelletti et al. showed that benign non-occlusive (type A–D) dissections after balloon-only angioplasty were associated with a significantly decreased risk of re-stenosis (P<0.001) and apparent- ly favorable clinical outcomes as seen in our study [4]. However, the detrimental clinical role of final dissections on major adverse thrombotic events such as ST has been confirmed in the BMS era, even in the presence of optimal antiplatelet therapy by producing MI. In our study, the occurrence of dissections after DES implan- tation is incompletely characterized as few DES were implanted in initial procedures, as no study has reported specific data concern- ing their incidence of dissection and its angiographic predictors of Coronary Dissections yet. Calcified lesions 15 (53.5%), eccen- tric lesions 12 (42.8%), long lesions 16 (57.1%), complex lesion morphology and vessel tortuosity 06 (21.4%), A balloon to artery ratio>1.2 14 (50%) as found in our study is impossible to be com- pared with non-availability ofevidence-based data in Pakistan or with other developing countries. Despite the improvements in ad- junctive medical therapy (i.e. GpIIb/IIIa inhibition and long-term thienopyridine use), the pattern of patients undergoing coronary revascularization has dramatically changed. This is verified by the inclusion of complex lesion and patient subsets in the present study, the adverse events were likely to stem from coronary thrombosis, either in-stent or peri-stent, eventually leading to death, non-fatal MI, urgent TVR, or bypass grafting. This detrimental impact of final dissections is probably due to the lesion complexity pattern in delay in approaching the cath lab [17].
However, the inherent risk of confounding, the clustering of several unfavorable risk factors in the same patients experienc- ing dissections cannot be excluded. As, patients with coronary dissections represent a small but with many additional problems and risk-factors, both present and acquired during the procedure, which may contribute to the increased adverse event rate use of bare-metal stent and availability size of balloon likely to compli- cate any mechanistic analysis [18]. However, many authorities have suggested that whenever a dissection cannot be sealed by means of stents or prolonged balloon inflations, maximal medical therapy should be enforced (including GpIIb/IIIa inhibitors and/or anticoagulants) and CABG be considered in the case of impaired TIMI flow in a proximal lesion or refractory myocardial ischemia. Such recommendations probably hold true nowadays in the DES era even [17-19].
Nonetheless, considering the propensity for edge (<5 mm) or more distal re-stenosis near deployed stent and whenever lesions are treated with only balloon angioplasty. Finally, the role of IVUS in better defining the characteristics of lesion and management of dissections should not be dismissed [20]. Although angiography has a very limited discriminatory power in identifying low-risk dissections, an IVUS cross-sectional study should be considered a reliable hint that the index dissection is not likely to bear ad- verse consequences [8,21]. Perforation and acute vessel closure is the most feared complication in equipped modern Cath-labs. The recent registries show perforation occurring in 0.3 0.6% of all pa- tients undergoing PCI and acute vessel closure due to coronary dissection up to 11% of all elective PTCAs. Unfortunately, cost and logistic hurdles obviously limit the scientific testing and clin- ical application of this strategy in Cath-labs of other developing countries may have resulted in such data of dissection in our study [1,22,23].
Limitation of this Study
Drawbacks of this work are inherent to a retrospective study and include selection bias, and confounding. While we tried to address these potential sources of systematic error by systematic data en- try, the risk of persistent underlying bias may be borne in mind. Dissections left untreated after BMS stent implantation, the con- tinuing difficulty for angiography to well evaluate if a dissection is benign or not without IVUS support were the major weak points as, However, dissections are quite common and logistic, techni- cal, but especially ethical issues would likely make unfeasible any randomized controlled trial concerning their optimal management
Conclusion
Coronary artery dissection remains a common occurrence during PCI but clinical sequelae have been minimized by the routine use of coronary stents. Rapid recognition and attention to the angio- graphic appearance of the dissection is essential to the successful management of this complication in resource constrained Cath- labs. None the less, patients with extensive dissection who are free from the manifestations of acute ischemia at the end of the proce- dure can be managed conservatively and have a good immediate output. Attempts should be made to stabilize extensive dissection during coronary angioplasty so that surgical intervention can be delayed or avoided altogether if possible.
References
- Black AJ, Namay DL, Niederman AL, Lembo NJ, Roubin GS, et al. (1989) Tear or dissection after coronary angioplasty. Morphologic correlates of an ischemic complication. Circula- tion 79: 1035-1042.
- Block PC, Myler RK, Stertzer S, Fallon JT (1981) Morphol- ogy after transluminal angioplasty in human beings. N Engl J Med 305: 382-385.
- Guagliumi G, Virmani R, Musumeci G, Motta T, Valsecchi O, et al. (2003) Drug-eluting versus bare-metal coronary stents: long-term human pathology. Findings from different coronary arteries in the same patient. Ital Heart J 4: 713-720.
- Cappelletti A, Margonato A, Rosano G, Mailhac A, Veglia F, et al. (1999) Short-and long-term evolution of unstented nonocclusive coronary dissection after coronary angioplasty. J Am Coll Cardiol 34: 1484-1488.
- Hiehle JF, Bourgelais DB, Shapshay S, Schoen FJ, Kim D, et al. (1985) Nd-YAG laser fusion of human atheromatous plaque-arterial wall separations in vitro. Am J Cardiol 56: 953-957.
- Agarwal R, Kaul U, Dev V, Sharma S, Venugopal P (1991) The morphology of coronary arterial dissection occurring subsequent to angioplasty and its influence on acute compli- cations. Int J Cardiol 31: 59-64.
- Sigwart U, Puel J, Mirkovitch V, Joffre F, Kappenberger L (1987) Intravascular stents to prevent occlusion and restenosis after transluminal angioplasty. N Engl J Med 316: 701-706.
- Colombo A, De Gregorio J, Moussa I, Kobayashi Y, Karvouni E, et al. (2001) Intravascular ultrasound-guided percutane- ous transluminal coronary angioplasty with-provisional spot stenting for treatment of long coronary lesions. J Am Coll Cardiol 38: 1427-1433.
- Huber MS, Mooney JF, Madison J, Mooney MR (1991) Use of a morphologic classification to predict clinical outcome af- ter dissection from coronary angioplasty. Am J Cardiol 68: 467-471.
- Shigeyama J, S Ito, H Kondo, O Ito, T Matsushita, et al. (2001) Angiographic Classification of Coronary Dissections after Plain old balloon Angioplasty for Prediction of Regres- sion at Follow-up. Jpn Heart J 42: 393-408
- Ashkan P, Jacqueline WLS (2019) Spontaneous Coronary Artery Dissection: Clinical Considerations in Diagnosis and Treatment. A Coll Cardiol.
- steffenino G, Meier B, Finci L, Velebit V, von Segesser L, et al. (1988) Acute complications of elective coronary angio- plasty: a review of 500 consecutive procedures. Br Heart J 59: 151-158.
- Hermans WR, Rensing BJ, Foley DP, Deckers JW, Rutsch W, et al. (1992) Therapeutic dissection after successful coronary balloon angioplasty: no influence on restenosis or on clinical outcome in 693 patients. The MERCATOR Study Group. J Am Coll Cardiol 20: 767-780.
- Shiu MF, Silverton NP, Oakley D, Cumberland D (1985) Acute coronary occlusion during percutaneous transluminal coronary angioplasty. Br Heart J 54: 129-133.
- Holmes DR, Holubkov R, Vlietstra RE, Kelsey SF, Reeder GS, et al. (1988) Comparison of complications during percu- taneous transluminal angioplasty from 1977 to 1981 and from 1985 to 1986: The National Heart, Lung and Blood Institute Percutaneous Transluminal Coronary Angioplasty Registry. J Am Coll Cardiol 12: 1149-1155.
- Sharma SK, Israel DH, Kamean JL, Bodian CA, Ambrose JA (1993) Clinical, angiographic, and procedural determinants of major and minor coronary dissection during angioplasty. Am Heart J 126: 39-47.
- Murphy DA, Craver JM, Jones EL, Gruentzig AR, King SB, et al. Surgical revascularization following unsuccessful percu- taneous transluminal coronary angioplasty. J Thorac Cardio- vasc Surg 84: 342-348.
- Phillips SJ, Kongtahworn C, Zeff RH, Skinner JR, Toon RS, et al. (1989) Disrupted coronary artery caused by angioplasty: supportive and surgical considerations. Ann Thorac Surg 47: 880-883.
- McClean DR, Thomas MR (2001) Non-flow limiting dissec- tion leading to late coronary restenosis following intracoro- nary brachytherapy. Cathet Cardiovasc Intervent 54: 355-357.
- Hong MK, Park SW, Lee NH, Nah DY, Lee CW, et al. (2000) Long-term outcomes of minor dissections at the edge of stents detected with intravascular ultrasound. Am J Cardiol 86: 791- 795.
- Nishida T, Colombo A, Briguori C, Stankovic G, Albiero R, et al. (2002) Outcome of nonobstructive residual dissections detected by intravascular ultrasound following percutaneous coronary intervention. Am J Cardiol 89: 1257-1262.
- Hermans WR, Foley DP, Rensing BJ, Rutsch W, Heyndrickx GR, et al. (1993) Usefulness of quantitative and qualitative angiographic lesion morphology, and clinical characteristics in predicting major adverse cardiac events during and after native coronary balloon angioplasty. Am J Cardiol 72: 14-20.
- Kay I, Sabate M, Van Langenhove G, Costa M, Wardeh A, et al. (2000) Outcome from balloon induced coronary dissection after intracoronary β radiation. Heart 83: 332-337




