Research Article - (2025) Volume 10, Issue 2
Translation, Cross-Cultural Adaption, Reliability, Validation of King’s Health Questionnaire (KHQ) in the Arabic Language
Received Date: Jul 21, 2025 / Accepted Date: Aug 06, 2025 / Published Date: Aug 14, 2025
Copyright: ©2025 Najwa Alfarra, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Alfarra, N., Aldosari, H., Alharbi, S., Takroni, M., Assoud, R., et al. (2025). Translation, Cross-Cultural Adaption, Reliability, Validation of King
Abstract
Introduction: The King’s Health Questionnaire (KHQ) is a disease-specific, self- administered questionnaire designed to assess the impact of urinary incontinence on quality of life (QoL) in women [1]. There are some Saudi Obey- gynecology physicians/therapists employ the King’s Health Questionnaire (KHQ), but there has never been an official Arabic translation with its reliability and validity. Therefore, we will translate the KHQ to Arabic language to evaluate incontinence women quality of life and to use it as an outcome measurement in the future researches.
Materials and Methods: cross-cultural study was used. KHQ was translated into the Arabic language using forward and backward translation. The study took a place at King Faisal Specialist Hospital, Riyadh. A study was conducted to assess both the general and specific effects of UI on QoL of 55 patients from physical rehabilitation outpatient clinic. The reliability of the study was assessed by using Cronbach’s alpha coefficient and test- retest reliability, as well as the validity of the study was assessed by using construct validity.
Objectives: The objectives of this study is to translate KHQ to Arabic version for application in the assessment of female urinary incontinence (QoL), and to be used as an outcome measurement in the future researches.
Result: Cronbach’s alpha: ≥ 0.85 across most domains, indicating high internal consistency. ICC: Values > 0.80 confirmed excellent test-retest reliability. Content Validity: High face validity with strong cultural acceptability. Construct Validity: Moderate to high correlation with SF-36 domains (r = 0.60–0.78). The domain structure closely resembled the original KHQ, confirming conceptual consistency.
Conclusions: The Arabic version of the King’s Health Questionnaire (KHQ-AR) is a valid, reliable, and culturally appropriate tool for assessing quality of life in Arabic-speaking patients with urinary incontinence. It can be confidently used in clinical practice and research.
Keywords
Women’s Health, Urinary Incontinence, Validated Questionnaire
Introduction
Urinary incontinence (UI) in women, according to International Continence Society (ICS) refers to an involuntary, uncontrollable, unwitting leakage of urine causing physical discomfort and problems due to maintenance of feminine hygiene [2]. Quality of life refers to the degree to which a person enjoys important possibilities of his or her life, and includes both subjective and objective indicators. It reflects individuals’ sense of well-being and satisfaction with life. Questionnaires or surveys are widely used in the assessment of female urinary incontinence research to collect qualitative information from patients such as incontinence impact, physical and social limitation that affected patient. Although using an existing questionnaire will save time and resources, but sometimes the published questionnaire is not available in the language required for the targeted respondents [3]. As a result, this study Aimed to translate an existing questionnaire (KHQ) into the Arabic language to evaluate the condition-specific QoL using the KHQ for female UI.
King’s Health Questionnaire (KHQ) was formulated by Kelleher and his colleague (1997) Kings College London, they opined that KHQ is useful for the rapid appraisal and follow-up in many clinical trials involving new treatments for urinary incontinence. KHQ is a patient self-administered self-report and has 3 parts consisting of 21 items. Part 1 contains general health perception and incontinence impact. Part 2 contains role limitation, physical limitation, social limitation, personal relationships, emotions, and sleep/energy, severity measures. Part 3 is considered as a single item and contains ten responses in relation to frequency, nocturia, urgency, urge, stress, intercourse incontinence, nocturnal enuresis, infections, pain, and difficulty in voiding. The response in KHQ have four-point rating system. The eight subscales scored between 0 (best) and 100 (worst), the symptoms severity scale is scored from 0 (best) to 30 (worst). Decreases in KHQ domain scores indicate an improvement in quality of life, and higher score mean that the person is severely affected by the UI [4] Appendix 1.
Rationale of the Study
As a senior women’s health physiotherapist working at KFSH&RC, our main concern is to provide a high quality of care, we used different modalities of questionnaires to evaluate the presence and severity of urinary symptoms, as well as the impact of incontinence on daily activities and quality of life. Most of the questions were employed in that field is in English language, and most of our patients are not efficient in English. This concern stimulated our interest in helping these patients and translate the KHQ to Arabic in order to help therapists in the assessment of female urinary incontinence (QoL), and to be used as an outcome measurement in the future researches. However, to date, no research has been translated KHQ to Arabic language.
Methods: Cross-Culture Adaptation Study, Using Purposeful Sampling
Instrument
An Arabic version of KHQ (free of copyrights) was produced to assess both general and specific effects of UI on QoL, using the forward-backward translation method to ensure conceptual equivalence. Patients were referred to the physical Rehabilitation clinic by the senior therapist was recruited patients randomly from the screening clinic at the physical rehabilitation department. The patients were informed about the study and given the information sheet to understand further about the nature of the study. Hard copy of the KHQ was completed anonymously at the clinic and added in envelop and returned to senior therapist who distributed the questionnaires.
Sample Size
To calculate the sample size, a minimum correlation of -0.30 between the two instruments was assumed, with an alpha (type 1 error) of 5% and Beta (type 11 error) of 0.10. A sample of 55 patients will be estimated [5,6].
Inclusions Criteria
• Women who diagnosed with UI by the physician.
• Aged between 25-75 years old.
• Able to read and answer the questionnaire by herself (at least secondary school degree).
Exclusion Criteria
• Illiterate women.
• Aged over 75years or younger than 25 years.
Independent Variables
• Sex
• Age
• Education level.
Translating a Questionnaire
The following section will summarize the translation process of the KING’S HEALTH QUESTIONNAIRE into Arabic language. This process will follow the recommendations of Beaton et al (2007) [7].
Forward Translation
The initial translation from the original language to the Arabic language was made by at least two independent translators [1,3]. Preferably, the bilingual translators were translated the questionnaire into their mother tongue, to better reflect the nuances of the target language [4]. In this study one translator was aware of the concepts the questionnaire intends to measure, to provide a translation that more closely resembles the original instrument. The other translator is suggested to be a naïve translator, who is unaware of the objective of the questionnaire, produce the second translation so that subtle differences in the original questionnaire was detected [1,3]. Discrepancies between the two translators were discussed and resolved between the original translators, and the primary author of the study, who should not have involved in the previous translations.
Backward Translation
The initial translation was independently back-have translated from the target language into the original language to ensure the accuracy of the translation. Misunderstanding or unclear wording in the initial translations were revealed in the back- translation [3]. The back translation was performed by two independent translators, who was not involve in the forward translation. Preferably translating into their mother language (the original Language) to avoid bias, back translators were not aware of the intended concepts the questionnaire measures [1,3].
Expert Committee
Five members of the committee were included: primary author of the study who are familiar with the construct of interest, both the forward (one translator and women’s health therapist) and backward translators (translator and one therapist not aware of the subject). The expert committee were needed to review all versions of the translations and determine whether the translation and original versions achieve semantic, idiomatic, experiential and conceptual equivalence [3,8]. Any discrepancies were resolved, and the author of the study was reached a consensus on all items to produce a final version of the translated questionnaire.
Preliminary Pilot Testing
The draft version of the translated questionnaire was pilot tested on a small sample (about 20 patients) of the intended respondents [8]. The respondent was asked at the screening clinic / existing patient at the women’s health clinic to answer the questionnaire and their corresponding response meant. This approach was allowed the investigator to make sure that the translation items retained the same meaning as the original items, and to ensure there is no confusion regarding the translated questionnaire. This process was repeated a few times to finalize the final translation version of the questionnaire.
Validity
After the translated questionnaire items pass through preliminary pilot testing. Then the final version of the questionnaire was administered to a large representative sample of respondents for whom the questionnaire is intended [7].
Construct Validity
Construct validity will compare KHQ with other urinary symptoms assessment tools (for example SF-36). Spearman’s rank correlation coefficient will be used to test the agreement between two scales). Spearman’s rank correlation coefficient is a technique which can be used to summarize the strength and direction (negative or positive) of a relationship between two variables. Usually, this type of testing is required when the researcher wants to compare the results of intervention using different scales [6].
Reliability
Internal Consistency
Cronbach’s alpha coefficient was used for each subscale. A score of less than 0.50 indicated poor internal consistency, scores ranging from 0.51 to 0.69 will considered suspicious, scores ranging from 0.70 to 0.80 will considered acceptable, scores ranging from 0.81 to 0.90 will considered good, and scores greater than 0.90 indicated excellent internal consistency [9].
Procedure for Reliability Testing
Two experienced women’s health therapists (author and senior women’s health therapist) who previously employed KHQ in their clinic practice conducted reliability testing. Because the conditions were varied if the two raters examined the patients independently, they will only test once. A single rater was administered the commands, observe, and provide a separate rating for each participant. KHQ is a patient- administer self-report that was comprises three parts and 21 items. Raters was determined who would issue commands and switch from one participant to the next.
Participants for Reliability Testing
In this cross-sectional study, 55 women aged 25-75 years who visited the physical rehabilitation department and were diagnosed with urine incontinence. All the females were explained about the study procedure and confidentiality of their information. Participants provided written information sheet. They were asked about demographic details such as age, marital status, working status and duration of complaint. To assess the test-retest reliability, the KHQ was administered twice with an interval of 15 days from the first evaluation to assess the reproducibility of the KHQ. During this period, these patients were asked for no changes in the course of medication or treatment for UI [10]. That was done by the author and one of the co-investigators who had PhD and are familiar in conducting validity and reliability assessment.
Results
In the statistical analysis, 55 women with a mean age of 47.5 (standard deviation +/- 9.1) and a median of 51 years (ranging from 25-75 years) were interviewed with complaints of UI. Two groups were considered: 67% lived with a husband. In terms of the level of education, 55% of this population sample consisted of patients with Bachler degree, and the remaining 45% were educated until higher secondary. In terms of profession, 48% office worker, and homemaker in 52% of cases. Respectively sociodemographic and clinical characteristics are shown in Table 1
|
Subjects (n = 55) |
Means +/- |
|
Age |
47.5+/-9.1 |
|
Type of complaint |
Frequency % |
|
Urge |
23 |
|
Stress |
62 |
|
Mixed |
15 |
Table1: Sociodemographic and Clinical Characteristics of the Patients With Urinary Incontinence
A normality test was done on the data, and it was found that the data were distributed normally (p>0.005); hence, it was considered ordinal data.Therefore,Cronbach’s alpha coefficient was used to assess interrater reliability for the total scores and each item of KHQ. This is done to distinguish serious disagreement between the examiners and to find out the weight of their disagreement.
A value of >0.75 indicates excellent agreement, 0.4-0.75 is fair to good agreement, and <0.4 is suggested as poor agreement. Cronbach’s alpha was calculated with MedCalc [11]. The intraclass correlation (ICC) coefficient was calculated using SPSS 22 to make comparisons of the interrater reliability with the results of others studies. An ICC of 0.80 or above reflects high reliability, 0.60-0.79 is moderate reliability, and <0.60 is indicated as poor reliability. Test-retest reliability was tested in a test –retest design and was evaluated using the ICC. In table 2 the ICC was interpreted as follows:0.90, excellent reliability. The internal consistency was evaluated by Cronbach’s α coefficient, which is considered statistically significant when between 0.70 and 0.95.
|
KHQ |
Mean +/- SD |
Median |
Minimum - Maximum |
|
General health |
46 +/- 9.2 |
50 |
0-100 |
|
Impact of incontinence |
70 +/- 10.4 |
60 |
0-100 |
|
Limitations of daily activities |
55.3 +/- 20.1 |
55.4 |
0-100 |
|
Physical limitations |
56.3 +/- 20.1 |
65.7 |
0-100 |
|
Social limitations |
24.6 +/- 25.4 |
23.3 |
0-100 |
|
Personal relationships |
38.5 +/- 9.3 |
0 |
0-100 |
|
Emotions |
67.7 +/-9.7 |
67.2 |
0-100 |
|
Sleep/ disposition |
53.2 +/- 12.2 |
51 |
0-100 |
|
Urinary symptoms |
59.1 +/- 8.3 |
66.2 |
0-100 |
Table 2: Evaluation of Mean (Standard Deviation) and Median (Range) for each Domain of the King’s Health
Questionnaire
To Assess Test-Retest Reliability
The general standard Cronbach’s a coefficient (overall the eight classical domains, in addition to the scale of urinary symptoms scale) was 0.87. Considering the domains individually, their scores varied between 0.49 (sleep / disposition) and 0.92 (emotions). The ICCs, which assess the test-retest as well as the Cronbach’s a value for each domain, are shown in table 3. The domain of general health perception and impact of UI, composed of single items, did not have internal consistency.
|
KHQ domain |
Alpha Cronbach |
ICC |
CI (95%) |
|
General health perception |
- |
0.53 |
0.38-0.66 |
|
Impact of incontinence |
- |
0.70 |
0.60-0.79 |
|
Limitation of daily activities |
0.83 |
0.77 |
0.66-0.82 |
|
Physical limitations |
0.77 |
0.70 |
0.58-0.78 |
|
Social limitations |
0.68 |
0.74 |
0.65-0.82 |
|
Personal relationship |
0.86 |
0.70 |
0.53-0.77 |
|
Emotions |
0.91 |
0.71 |
0.62-0.80 |
|
Sleep /disposition |
0.48 |
0.65 |
0.53-0.75 |
|
Severity measures |
0.78 |
0.80 |
0.74-0.87 |
|
Severity of symptoms |
0.78 |
- |
- |
Table 3: Internal Consistency (Cronbach’s) and Test –Retest Reliability (Interclass Correlation Coefficient 95%- Minimum-Maximum) for the Kings’ Health Questionnaire Domains
To Assess Validity
Concurrent validity was tested to find the association of the average of the values of scores obtained for each domain of the KHQ and SF-36 and of the categories “yes” and “no” of the variable “use of sanitary napkins” in Table 4. A multidimensional questionnaire, the SF-36, has 36 items divided into physical and mental components. Each component consists of four domains filled with items that evaluate the same aspect of patients’ lives. The physical component includes functional capacity (10 items). Physical aspects (4 items), pain (2 items), general health status (5 items), and the mental component includes vitality (4 items), social aspects (2 items), emotional aspects (3 items), and mental health (5 items). Consider comparing present health situations to those from a year ago. The response options are displayed on a Likert scale.
|
KHQ domain |
P |
SF-36 |
P |
|
General health perception |
0.5509 |
General Health status |
0.6213 |
|
Impact of incontinence |
<0.0001 |
Functional capacity |
0.074 |
|
Limitation of daily activities |
<0.0001 |
Physical aspect |
0.0002 |
|
Physical limitations |
<0.0001 |
Emotional aspects |
0.3854 |
|
Social limitations |
<0.0001 |
Pain |
0.8075 |
|
Personal relationship |
0.63 |
Vitality |
0.2706 |
|
Emotions |
0.0075 |
Mental health |
0.824 |
|
Sleep /disposition |
0.9 |
Social aspects |
0.42 |
Table 4: Testing Validity as Per Average of the Values of the Scores Obtained for Each Domain of the Kings Health Domain and Short Form -36.
In summary, Cronbach’s alpha: ≥ 0.75 across most domains, indicating high internal consistency. ICC: Values > 0.70 confirmed excellent test-retest reliability. Content Validity: High face validity with strong cultural acceptability. Construct Validity: Moderate to high correlation with SF-36 domains (r = 0.60–0.78). The domain structure closely resembled the original KHQ, confirming conceptual consistency.
Discussion
The demographic sample, which was primarily taken from low- and middle –class families, was one of the study’s shortcomings [12]. Patients who presented to the outpatient department, and who were then referred to the researcher, were chosen for the study. The KHQ’s internal consistency, as determined by the standardized Cronbach’s alpha Coefficient, was good, with an overall index of 0.87 above the minimum value often used as a reference in clinical trials of 0.7. The values for the internal consistency of the various domains ranged from 0.49 (sleep/disposition) to 0.92 (emotions). Although Cronbach’s alpha in the sleep/disposition domain was lower than the other values, when this domain was deleted, there was no change in the standardized general Cronbach’s alpha value, even though its value dropped to 0.86. The symptom scale’s internal consistency could not be determined because patients were not anticipated to experience all symptoms simultaneously. Nevertheless, a Cronbach’s alpha value of 0.73 was noted, and similar results were reported by Tamanini et al (2003) [13,14].
As per the study published in the Spanish version ranged from 0.65 (gravity measurements) to 0.92 (personal relationships), while the internal consistency of the original English version 12 ranged from 0.72 (physical constraints) to 0.89 (personal relationships). The ICC examined the reliability of test–retest. The correlation indices found in each specific category, which ranged from 0.53 (overall health perception) to 0.81 (gravity measurements), were regarded as moderate to strong. The only domain to have a moderate correlation coefficient (0.53) was the general perception of health. Since the patients were not being treated, this outcome was already anticipated because it was the only one that could experience some spontaneous change [12]. In all domains of KHQ areas except general health perception, intermittent mixed urinary incidence was associated with lower QoL, which is corroborated by global research. When patients used sanitary pads (difference in all KHQ domains, except general perception of health and sleep/ disposition) or increased the number of pads used daily, QoL was worse.
The assessment of construct validity involved examining the correlation between the two questionnaires administered during the initial consultation. Concurrent validity was evaluated by examining the association between the KHQ and certain clinical measures that are relevant to urine incontinence. There was a moderate degree of correlation observed between the analyzed areas that were connected. It is important to note that the observed associations were negative as a result of the inherent scoring criteria associated with each questionnaire. Overall, it was shown that the QoL was perceived to be lower when the type of complaint was non-continuous mixed urine incidence. This was evident across all domains of the KHQ, except for the domain of overall health perception. The study findings indicated that the SF-36 instrument had limited sensitivity in discerning disparities between the “yes” and “no” groups in relation to the variable “use of tampons.” Nevertheless, in this particular context, it was not anticipated to be as such. The majority of KHQ domains exhibited a notable disparity in relation to the proposed categories, except for the domains of general health perception and sleep/disposition. On the other hand, the SF-36 was only able to indicate significant differences in one of its eight domains, specifically the physical aspects domain. The likely cause of the inadequate content validity shown in SF-36 for UI is the substandard quality of its content.
Data Management and Storage
All the collected data was saved and the confidentiality of the patients were protected. The patient identity was protected by substituting anonymous codes for names. Privacy and confidentiality of the patients protected health information was ensured. There was no link with the patient’s name or identification number. Participants will be given an information sheet to understand the nature of the research Completed questionnaires will be collected, collated and sent to the primary investigator for further analysis and it will be kept in looked drawer at the primary author office.
Ethical Consideration
The consideration of ethics and rules governing research is an important aspect that every researcher must consider to come up with valid data while obtaining a mutual understanding with the people he/she comes in contact with Creswell [15,16]. Ethical approval for the research will be obtained from King Faisal Specialist Hospital and Research Center. We will seek a waiver of written informed consent due to the nature of the study.
Conclusions
Arabic version of KHQs measured a person’s vitality, physical functionality, body pain, general perceptions of health, and physical role functioning. It helped to assess other aspects of life, such as interpersonal and sexual relationships, professions, and psychological welfare, were not projected, though. These instruments were further divided into “generic” and “disease- specific” categories. Hence, it can be advised for use in standard clinical practice among Arabic-speaking patients because it is simple to administer.
References
- Guillemin, F., Bombardier, C., & Beaton, D. (1993). Cross- cultural adaptation of health-related quality of life measures: literature review and proposed guidelines. Journal of clinical epidemiology, 46(12), 1417-1432.
- Abrams, P., Cardozo, L., Fall, M., Griffiths, D., Rosier, P., Ulmsten, U., ... & Wein, A. (2003). The standardisation of terminology in lower urinary tract function: report from the standardisation sub-committee of the International Continence Society. Urology, 61(1), 37-49.
- Boynton, P. M., & Greenhalgh, T. (2004). Selecting, designing, and developing your questionnaire. Bmj, 328(7451), 1312- 1315.
- Kelleher, C. J., Pleil, A. M., Reese, P. R., Burgess, S. M., & Brodish, P. H. (2004). How much is enough and who says so? The case of the King's Health Questionnaire and overactive bladder. BJOG: An International Journal of Obstetrics & Gynaecology, 111(6), 605-612.
- Osborne, J. W., & Costello, A. B. (2004). Sample size and subject to item ratio in principal components analysis. Practical Assessment, Research, and Evaluation, 9(1).
- Alnahhal, A., & May, S. (2012). Validation of the Arabic version of the Quebec back pain disability scale. Spine, 37(26), E1645-E1650.
- Beaton, D., Bombardier, C., Guillemin, F., & Ferraz, M. B. (2007). Recommendations for the cross-cultural adaptation of the DASH & QuickDASH outcome measures. Institute for work & health, 1(1), 1-45.
- Hendricson, W. D., Jon Russell, I., Prihoda, T. J., Jacobson,J. M., Rogan, A., Bishop, G. D., & Castillo, R. (1989). Development and initial validation of a dualâ?language English–Spanish format for the arthritis impact measurement scales. Arthritis & Rheumatism: Oficial Journal of the American College of Rheumatology, 32(9), 1153-1159.
- Perneger, T. V., Courvoisier, D. S., Hudelson, P. M., & Gayet-Ageron, A. (2015). Sample size for pre-tests of questionnaires. Quality of life Research, 24(1), 147-151.
- Pearson, K. (1895). VII. Note on regression and inheritance in the case of two parents. proceedings of the royal society of London, 58(347-352), 240-242.
- Haley, S. M., & Osberg, J. S. (1989). Kappa coefficient calculation using multiple ratings per subject: a special communication. Physical Therapy, 69(11), 970-974.
- Lin YF, Lin YC, Wu IC, Chang YH Urinary incontinence and its association with socioeconomic status among middle- aged and older persons in Taiwan: A population-based study. Geriatr Gerontol Int 2021;21: 245–53. Cited Here | Google Scholar.
- Tamanini, J. T. N., D'Ancona, C. A. L., Botega, N. J., & Rodrigues Netto Jr, N. (2003). Validation of the Portuguese version of the King's Health Questionnaire for urinary incontinent women. Revista de Saúde Pública, 37, 203-211.
- Viana, R., Viana, S., Neto, F., & Mascarenhas, T. (2015). Adaptation and validation of the King’s Health Questionnaire in Portuguese women with urinary incontinence. International urogynecology journal, 26(7), 1027-1033.
- Creswell, J. W. (2002). Educational research: Planning, conducting, and evaluating quantitative (Vol. 7). Prentice Hall Upper Saddle River, NJ.
- Beaton, D. E., Bombardier, C., Guillemin, F., & Ferraz, M. B. (2000). Guidelines for the process of cross-cultural adaptation of self-report measures. Spine, 25(24), 3186-3191.
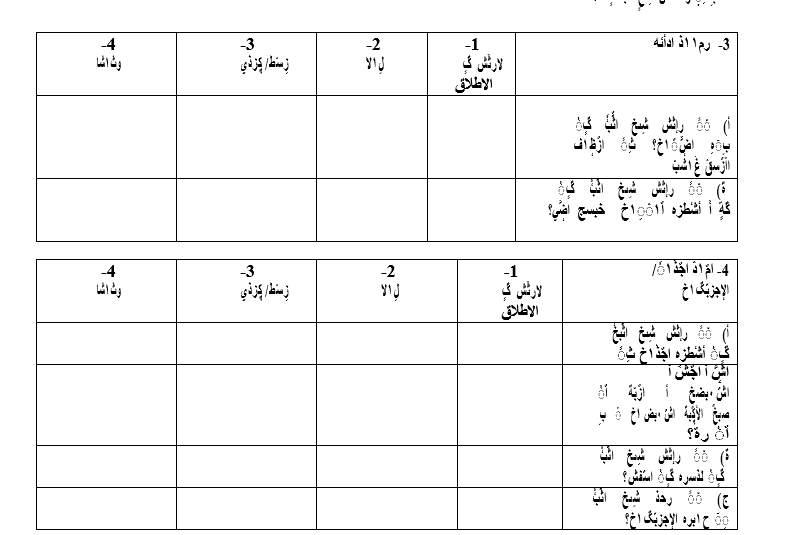
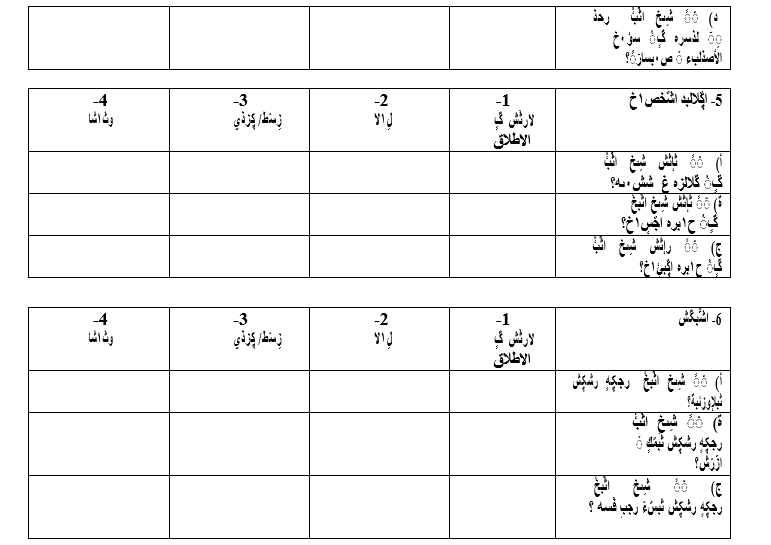
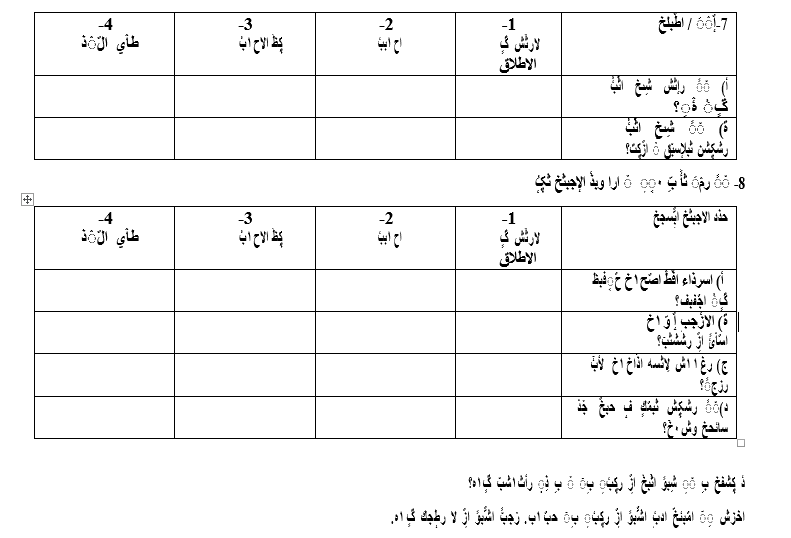
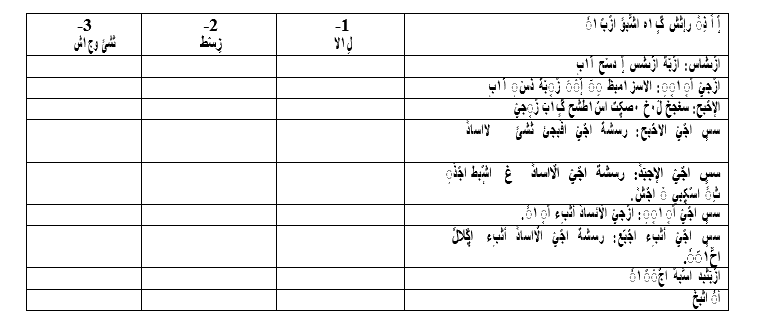
Appendix
Appendix 1: King’s Health Questionnaires (English/Arabic version)
Your Name:
MRN:
How you describe your current health status?
Please select one answer only from the following options:
• Very Good
• Good
• Fair
• Poor
• Very Poor
To what extent does your bladder problem affect your life?
Please select one answer only from the following options:
• It does not affect me at all
• It affects me to a minimal extent
• It affects me to a moderate degree
• It affects me significantly
Some daily activities that are affected due to your bladder problem: To what extent are the following activities affected by your bladder problem?
To what extent does your bladder problem affect the following matter? Please tick the appropriate option:
1. Role Restrictions
|
|
Not at all |
Slightly |
Moderately |
Significantly |
|
Does your bladder problem affect your household chores such as cleaning, shopping… etc. |
|
|
|
|
|
Does your bladder problem affect your work or daily activities outside the home? |
|
|
|
|
2. Physical/Social Limitations
|
|
Not at all |
Slightly |
Moderately |
Significantly |
|
Does your bladder problem affect your physical activities (Like walking, running or sports)? |
|
|
|
|
|
Does your bladder problem affect your ability to travel? |
|
|
|
|
|
Does your bladder problem affect your social life? |
|
|
|
|
|
Does your bladder problem affect your ability to see and visit friends? |
|
|
|
|
3. Personal Relationship:
|
|
Not at all |
Slightly |
Moderately |
Significantly |
|
Does your bladder problem affect your relationship with your partner? |
|
|
|
|
|
Does your bladder problem affect your sexual life? |
|
|
|
|
|
Does your bladder problem affect your family life? |
|
|
|
4. |
4. Emotions
|
|
Not at all |
Slightly |
Moderately |
Significantly |
|
Does your bladder problem make you feel depressed? |
|
|
|
|
|
Does your bladder problem make you feel anxious or stressed? |
|
|
|
|
|
Does your bladder problem make you feel bad about yourself? |
|
|
|
|
5. Sleep/ Energy
|
|
Not at all |
Slightly |
Moderately |
Significantly |
|
Does your bladder problem affect the quality of your sleep? |
|
|
|
|
|
Does your bladder problem make you feel tired or fatigued? |
|
|
|
6. |
6. Do you do any of the following?
|
|
Not at all |
Slightly |
Moderately |
Significantly |
|
Do you wear pads to stay dry? |
|
|
|
|
|
Do you pay attention to the amount of fluids you drink? |
|
|
|
|
|
Do change your underwear because it gets wet? |
|
|
|
|
|
Are you worried about a bad smell? |
|
|
|
|
We would like to know what bladder problems you are suffering from and to what extent they affect you:
Please select your current bladder problems you are experiencing and ignoring everything that does not apply to your situation or condition:
To what extent do the following problems affect you?
1. Frequent visits to bathroom:
• Slightly
• Moderately
• Significantly
2. Waking up to go to the bathroom:
• Slightly • Moderately
• Significantly
3. Difficulty in controlling the urge to urinate:
• Slightly
• Moderately
• Significantly
4. Urgency urinary incontinence: Involuntary leakage of urine with strong urge to urinate:
• Slightly
• Moderately
• Significantly
5. Stress incontinence: Involuntary leakage of urine during movements or physical activities such as coughing and running:
• Slightly
• Moderately
• Significantly
6. Bed wetting during sleep
• Slightly
• Moderately
• Significantly
7. Involuntary urinary leakage during sexual intercourse:
• Slightly
• Moderately
• Significantly
8. Urinary tract infection:
• Slightly
• Moderately
• Significantly
9. Bladder Pain:
• Slightly
• Moderately
• Significantly