Research Article - (2026) Volume 7, Issue 1
Transforming the Landscape of Gastrointestinal Oncology: From Molecular Precision to Health Equity
Received Date: Dec 15, 2025 / Accepted Date: Jan 11, 2026 / Published Date: Jan 20, 2026
Copyright: ©2026 Kaneez Seema Khan. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Khan, K. S. (2026). Transforming the Landscape of Gastrointestinal Oncology: From Molecular Precision to Health Equity. In J Fore Res, 7(1), 01-07.
Abstract
Gastrointestinal (GI) malignancies remain a leading cause of global cancer morbidity and mortality. However, the past decade has witnessed a paradigm shift in their management driven by advances in molecular profiling, targeted therapies, immunotherapy, circulating tumor DNA (ctDNA), multidisciplinary care models, and supportive interventions. Concurrently, population-level research has revealed persistent disparities across socioeconomic, racial, and demographic strata that influence outcomes. This review synthesizes recent developments across major GI cancer subtypes — esophageal, gastric, hepatobiliary, pancreatic, colorectal, and neuroendocrine tumors — with emphasis on modern management strategies, emerging therapies, real-world evidence, and health equity considerations.
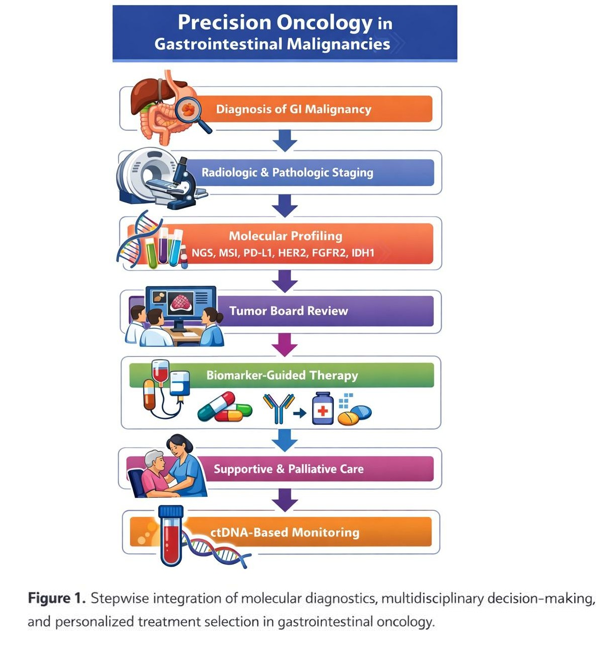
Introduction
GI cancers represent a heterogeneous group of diseases with distinct molecular drivers, clinical behaviors, and therapeutic vulnerabilities. Traditional cytotoxic chemotherapy has been supplemented or replaced by targeted agents, immune checkpoint inhibitors (ICIs), antibody-drug conjugates (ADCs), bispecific antibodies, adoptive cell therapies, and precision radiotherapy strategies.
Parallel to these therapeutic advances, large registry-based studies have highlighted substantial disparities in incidence, stage at diagnosis, access to care, and survival outcomes based on race, ethnicity, socioeconomic status, geography, nutritional status, and social determinants of health (SDOH). Recent studies by Dharia et al., Sivasubramanian et al., and others demonstrate persistent inequities particularly in biliary tract cancers, cholangiocarcinoma, and early-onset colorectal cancer (EOCRC) [1–3].
Mechanism of action of immunotherapy in GI cancers
Immune checkpoint inhibitors (ICIs) and related immunotherapies work by re-activating anti-tumor T-cell immunity that is otherwise suppressed by tumor-intrinsic mechanisms and an immunosuppressive tumor microenvironment (TME). In GI malignancies, the magnitude and durability of benefit strongly depend on tumor antigenicity (e.g., MSI-H/dMMR, POLE/ POLD1), immune infiltration, PD-L1 expression, and TME context (e.g., liver immune tolerance in HCC; desmoplasia in PDAC) [4-9].
Cancer–Immunity Cycle and Where ICIs Act
Anti-tumor immunity can be conceptualized as a cycle: tumor antigen release → antigen presentation → T-cell priming/ activation → trafficking/infiltration → tumor recognition → tumor killing → further antigen release. Tumors interrupt this cycle via checkpoint pathways and immunosuppressive cytokines/cells. ICIs primarily act at the T-cell activation and effector phases, restoring cytotoxic function and clonal expansion of tumor- reactive lymphocytes [33,34].
PD-1/PD-L1 Axis: Restoring Exhausted T-Cell Function
PD-1 is an inhibitory receptor expressed on activated T cells; binding to PD-L1/PD-L2 on tumor or immune cells transmits “stop” signals that reduce T-cell receptor signaling, cytokine production, proliferation, and cytotoxicity—features of T-cell exhaustion. Anti–PD-1 (e.g., pembrolizumab, nivolumab) and anti–PD-L1 (e.g., atezolizumab, durvalumab) antibodies prevent this interaction, enabling renewed killing of tumor cells [34,35].
Relevance in GI cancers:
![]() Gastroesophageal cancers: Many tumors exhibit inflamed/“hot” features with PD-L1 expression; adding PD-1 blockade to chemotherapy improves survival in biomarker- enriched populations, likely because chemotherapy increases antigen release and dendritic cell priming, providing more targets for reinvigorated T cells [20,21].
Gastroesophageal cancers: Many tumors exhibit inflamed/“hot” features with PD-L1 expression; adding PD-1 blockade to chemotherapy improves survival in biomarker- enriched populations, likely because chemotherapy increases antigen release and dendritic cell priming, providing more targets for reinvigorated T cells [20,21].
![]() MSI-H/dMMR colorectal cancer: High mutational burden creates abundant neoantigens, leading to pre-existing tumor- reactive T cells that are readily reactivated by PD-1 blockade— explaining the dramatic benefit observed clinically [27,29].
MSI-H/dMMR colorectal cancer: High mutational burden creates abundant neoantigens, leading to pre-existing tumor- reactive T cells that are readily reactivated by PD-1 blockade— explaining the dramatic benefit observed clinically [27,29].
![]() HCC: The liver is an immune-tolerant organ; PD-1/PD-L1 blockade can reverse local T-cell dysfunction, and synergy occurs when combined with anti-angiogenic therapy that normalizes vasculature and reduces immunosuppressive signaling (see below) [24-36].
HCC: The liver is an immune-tolerant organ; PD-1/PD-L1 blockade can reverse local T-cell dysfunction, and synergy occurs when combined with anti-angiogenic therapy that normalizes vasculature and reduces immunosuppressive signaling (see below) [24-36].
CTLA-4: Enhancing Priming and Broadening the T-Cell Repertoire
CTLA-4 is a checkpoint receptor that competes with CD28 for binding to B7 ligands (CD80/86) on antigen-presenting cells, restraining T-cell priming in lymph nodes. Anti–CTLA-4 therapy (e.g., ipilimumab, tremelimumab) enhances priming and can broaden the anti-tumor T-cell repertoire; it may also reduce suppressive regulatory T cells within the TME depending on context [35-37].
Relevance in GI Cancers:
![]() HCC (STRIDE/HIMALAYA concept): Brief CTLA-4 priming plus ongoing PD-L1 blockade aims to boost initial priming while sustaining effector function in the hepatic TME [30].
HCC (STRIDE/HIMALAYA concept): Brief CTLA-4 priming plus ongoing PD-L1 blockade aims to boost initial priming while sustaining effector function in the hepatic TME [30].
![]() MSI-H CRC and select settings: Dual checkpoint blockade can produce deep responses by combining enhanced priming (CTLA-4) with effector reinvigoration (PD-1) [27-29].
MSI-H CRC and select settings: Dual checkpoint blockade can produce deep responses by combining enhanced priming (CTLA-4) with effector reinvigoration (PD-1) [27-29].
Why Combination Regimens Work: Chemo-, Anti-VEGF-, and Targeted-Immunotherapy Synergy
Chemo-immunotherapy (e.g., gastroesophageal cancers): Cytotoxic agents can increase tumor antigen release, induce immunogenic cell death, and reduce suppressive myeloid populations—creating a more permissive immune milieu for ICIs [20-33].
Anti-VEGF + ICI (e.g., HCC): VEGF signaling promotes abnormal vasculature, hypoxia, and immune suppression (Tregs, MDSCs). Anti-VEGF therapy can “normalize” vessels, improve T-cell trafficking, and reduce immunosuppressive cytokines, thereby potentiating PD-(L)1 blockade [24-36].
Targeted Therapy + ICI (selected scenarios): Some oncogenic drivers shape antigen presentation and immune infiltration; targeted therapy may modulate interferon signaling and antigen processing, potentially sensitizing to ICIs in specific biomarker-defined subsets [15-29].
Tumor Biology That Predicts Response in GI Cancers
High neoantigen load: MSI-H/dMMR and POLE-mutant tumors generate abundant neoantigens, fostering immune infiltration and sensitivity to PD-1 blockade [27-29]. ctDNA studies in hypermutated/POLE-mutated CRC demonstrate that immune- mediated tumor clearance can be tracked through rapid ctDNA decline, aligning biologic response with clinical benefit [16].
Immune-excluded or “cold” Tumors:
![]() PDAC commonly exhibits dense desmoplastic stroma, limited T-cell infiltration, suppressive macrophages, and low neoantigen visibility—making single-agent ICIs largely ineffective outside rare MSI-H cases, and driving exploration of vaccines, stromal modulation, and combinatorial strategies [19].
PDAC commonly exhibits dense desmoplastic stroma, limited T-cell infiltration, suppressive macrophages, and low neoantigen visibility—making single-agent ICIs largely ineffective outside rare MSI-H cases, and driving exploration of vaccines, stromal modulation, and combinatorial strategies [19].
![]() Some MSS CRC similarly lacks sufficient antigenicity and effective infiltration, necessitating combination approaches and trial innovations [28-29].
Some MSS CRC similarly lacks sufficient antigenicity and effective infiltration, necessitating combination approaches and trial innovations [28-29].
Immune-Related Toxicity Mechanistically Mirrors Immune Activation
By lifting immune inhibition, ICIs can cause off-target immune activation against normal tissues (immune-related adverse events, irAEs). In GI oncology, clinically relevant toxicities include immune-mediated colitis, hepatitis, pancreatitis, and less commonly nephritis; prompt recognition and immunosuppression (e.g., corticosteroids) are essential to prevent complications [4,18]. Real-world reporting underscores the importance of standardized monitoring systems and early intervention pathways [4].
Esophageal and Gastric Cancers
Molecular Stratification
Key actionable biomarkers include:
![]() HER2 amplification
HER2 amplification
![]() PD-L1 combined positive score (CPS)
PD-L1 combined positive score (CPS)
![]() MSI-H/dMMR
MSI-H/dMMR
![]() Claudin 18.2 (CLDN18.2)
Claudin 18.2 (CLDN18.2)
![]() FGFR2b overexpression
FGFR2b overexpression
![]() EBV positivity
EBV positivity
First-Line Management
|
Biomarker |
Preferred Regimen |
|
HER2+ |
Trastuzumab + chemotherapy ± pembrolizumab |
|
PD-L1 CPS ≥ 5 |
Chemo-immunotherapy (nivolumab or pembrolizumab) |
|
CLDN18.2 |
Zolbetuximab + CAPOX/FOLFOX |
|
MSI-H |
Single-agent immunotherapy |
The CheckMate-649, KEYNOTE-590, KEYNOTE-811, and ESOPEC trials have reshaped frontline therapy [19–21].
Emerging Therapies
![]() ADCs: trastuzumab deruxtecan, zanidatamab zovodotin
ADCs: trastuzumab deruxtecan, zanidatamab zovodotin
![]() Bispecific antibodies (zanidatamab)
Bispecific antibodies (zanidatamab)
![]() Cellular therapies in EBV-positive gastric cancer
Cellular therapies in EBV-positive gastric cancer
![]() Personalized perioperative strategies (ESOPEC trial)
Personalized perioperative strategies (ESOPEC trial)
Hepatocellular Carcinoma and Cholangiocarcinoma
Hepatocellular Carcinoma (HCC)
The IMbrave150 trial established atezolizumab + bevacizumab as first-line therapy. The HIMALAYA trial introduced durvalumab + tremelimumab.
Cholangiocarcinoma
Molecular testing is essential for:
• FGFR2 fusions
• IDH1 mutations
• BRAF V600E
• NTRK fusions
• HER2 amplification
Targeted therapies (pemigatinib, futibatinib, ivosidenib, dabrafenib/trametinib) and immunotherapy combinations are now standard for biomarker-selected patients [16-22].
Multidisciplinary models incorporating hepatology, interventional radiology, and oncology improve outcomes.
|
Cancer Type |
Biomarker |
Therapeutic |
Implication |
|
Esophagogastric |
HER2 |
Trastuzumab ± pembrolizumab |
[11,21] |
|
Esophagogastric |
CLDN18.2 |
Zolbetuximab |
[26] |
|
Colorectal |
MSI-H/dMMR |
Pembrolizumab, nivolumab |
[27] |
|
Colorectal |
BRAF V600E |
Encorafenib + cetuximab |
[27] |
|
Cholangiocarcinoma |
FGFR2 fusion |
Pemigatinib, futibatinib |
[15] |
|
Cholangiocarcinoma |
IDH1 mutation |
Ivosidenib |
[23] |
|
Pancreatic |
BRCA1/2 |
PARP inhibitors |
|
|
Tumor-agnostic |
NTRK fusion |
Larotrectinib, entrectinib |
[29] |
Table 1: Key Actionable Biomarkers in Gastrointestinal Malignancies
|
cancer |
First-Line Therapy |
Ref |
|
Esophagogastric |
Chemotherapy + nivolumab/pembrolizumab |
[20,21] |
|
Hepatocellular carcinoma |
Atezolizumab + bevacizumab |
[24] |
|
Cholangiocarcinoma |
Gemcitabine + cisplatin ± durvalumab |
[23] |
|
Pancreatic adenocarcinoma |
FOLFIRINOX or gemcitabine/nab-paclitaxel |
[19] |
|
MSI-H colorectal |
Pembrolizumab |
[27] |
|
Neuroendocrine tumors |
SSA → PRRT |
[8] |
Table 2: Standard First-Line Systemic Therapy by Gi Cancer Type
|
Determinant |
Impact on Outcomes |
Ref |
|
Race/ethnicity |
Worse survival in biliary and NETs |
[1,3,8] |
|
Income |
Later stage at diagnosis |
[1] |
|
Rurality |
Reduced access to trials |
[10] |
|
Marital status |
Improved CRC survival when married |
[10] |
|
Nutrition |
Higher mortality with malnutrition |
|
Table 3: Social Determinants Influencing GI Cancer Outcomes
Pancreatic Ductal Adenocarcinoma (PDAC)
Standard Management
Setting Preferred Strategy
Resectable Surgery + adjuvant modified FOLFIRINOX Borderline resectable Neoadjuvant chemotherapy ± chemoradiation Metastatic FOLFIRINOX or gemcitabine/nab-paclitaxel
Precision Oncology in PDAC
![]() Germline BRCA1/2 → PARP inhibitors
Germline BRCA1/2 → PARP inhibitors
![]() KRAS G12C/D inhibitors (emerging)
KRAS G12C/D inhibitors (emerging)
![]() Claudin 18.2 targeting
Claudin 18.2 targeting
![]() Tumor vaccines (GVAX, mRNA platforms)
Tumor vaccines (GVAX, mRNA platforms)
![]() TME modulation
TME modulation
Complications and Supportive Care
Portal vein thrombosis significantly worsens outcomes [7]. Nutritional optimization is critical due to high prevalence of cachexia and malnutrition [6].
Colorectal Cancer (CRC)
Early-Onset CRC
Rising incidence in younger populations with higher metastatic burden and distinct biology (bone metastasis prognostic implications shown by Vadehra et al.) [2].
Biomarker-Driven Therapy
![]() MSI-H → pembrolizumab or nivolumab ± ipilimumab
MSI-H → pembrolizumab or nivolumab ± ipilimumab
![]() BRAF V600E → encorafenib + cetuximab
BRAF V600E → encorafenib + cetuximab
![]() HER2 → trastuzumab-based regimens
HER2 → trastuzumab-based regimens
![]() POLE mutations → exceptional immunotherapy responses [17]
POLE mutations → exceptional immunotherapy responses [17]
ctDNA
ctDNA is increasingly used for minimal residual disease (MRD) detection, recurrence prediction, and treatment escalation/de- escalation.
Neuroendocrine Tumors (NETs)
Small intestinal NETs exhibit survival disparities by race and socioeconomic status [8]. Management includes:
![]() Somatostatin analogs
Somatostatin analogs
![]() PRRT (177Lu-DOTATATE)
PRRT (177Lu-DOTATATE)
![]() Targeted agents (everolimus, sunitinib)
Targeted agents (everolimus, sunitinib)
![]() Liver-directed therapies
Liver-directed therapies
Immunotherapy in GI Malignancies
Current Role ICIs are standard in:
![]() MSI-H CRC
MSI-H CRC
![]() PD-L1 high gastroesophageal cancers
PD-L1 high gastroesophageal cancers
![]() HCC (combination regimens)
HCC (combination regimens)
![]() Select biliary cancers
Select biliary cancers
Toxicity and Management
GI adverse events including colitis and hepatitis are common and require standardized monitoring and early intervention [4].
Supportive Care and Nutrition
Protein-energy malnutrition predicts poor survival and increased hospitalization [6]. Early nutritional intervention, pancreatic enzyme replacement, and palliative integration improve outcomes and quality of life.
Health Disparities and Social Determinants
Disparities in survival persist across GI cancers influenced by:
![]() Race/ethnicity
Race/ethnicity
![]() Income and insurance
Income and insurance
![]() Rurality
Rurality
![]() Marital status and social support [10,11]
Marital status and social support [10,11]
Addressing disparities requires policy reform, community outreach, decentralized trials, and inclusive research practices.
Conclusion
The management of GI cancers is undergoing rapid transformation through precision oncology, immunotherapy, advanced imaging, and supportive care integration. However, equitable implementation remains essential to ensure that advances translate into population-level survival benefits.
Future Directions
![]() Multi-omics-driven treatment
Multi-omics-driven treatment
![]() AI-guided imaging and pathology
AI-guided imaging and pathology
![]() Personalized vaccines
Personalized vaccines
![]() Microbiome-modulated immunotherapy
Microbiome-modulated immunotherapy
![]() Decentralized and inclusive clinical trials
Decentralized and inclusive clinical trials
References
- Dharia, A., Mahayni, A. A., Borra, R., Peshin, S., Ser-Manukyan, H., Mangu, S., ... & Muddassir, S. (2025). Ethnic and socioeconomic disparities in cancer-free and all- cause survival in cholangiocarcinoma patients under 60: A population-based study.
- Vadehra, D., Awidi, M., Peshin, S., & Mukherjee, S. (2025). 70P Prognostic implications of bone metastasis in early-onset colorectal cancer. Annals of Oncology, 36, S38.
- Sivasubramanian, B. P., Ravikumar, D. B., Dhabuwala, A., Yakout, A., Peshin, S., & Nash III, C. H. (2025). Exploring racial disparities in biliary tract cancer with sepsis.
- Kotla, N. K., Bhanushali, C., Vojjala, N., Peshin, S., Goyal, K., Shah, R., ... & Ahmed, N. (2025). Incident reporting and outcomes of gastrointestinal adverse events with immune checkpoint inhibitors.
- Chaudhari, J., Patel, A., Dharia, A., Peshin, S., Chaudhari, H., Patel, D. M., ... & Muddassir, S. (2025). Statewide Burden and Trends of Liver Cancer Mortality Attributable to High BMI in the US (1991–2021): Insights from the Global Burden of Disease study, 2021.
- Iqbal, R., Kimball, H., Gaddam, M., Peshin, S., Sinha, S., & Quadri, K. (2025). The impact of protein-energy malnutrition on clinical outcomes in hospitalized gastric cancer patients: A population-based analysis.
- Iqbal R, Kimball H, Peshin S, et al. Malignant portal vein thrombosis in pancreatic cancer. J Clin Oncol. 2025;43(4_suppl):784.
- Dharia A, Borra R, Peshin S, et al. Survival disparities in small intestine carcinoid tumors. J Clin Oncol. 2025;43(4_ suppl):653.
- Peshin S, Dharia A, Vojjala N, et al. NSCLC in the immunotherapy era. J Clin Oncol. 2025;43(16_suppl):e20565.
- Liu L, Peshin S, Barboza AZ, Vijayvergia N. Marital status and CRC survival. J Clin Oncol. 2025;43(16_suppl):3628.
- Peshin, S., Takrori, E., Kodali, N. A., Bashir, F., Gibson, M., & Singal, S. (2025). Therapeutic Frontiers in Gastroesophageal Cancer: Contemporary Concepts in Management and Therapy. International Journal of Molecular Sciences, 26(23), 11424.
- Peshin, S., Bashir, F., Kodali, N. A., Dharia, A., Zaiter, S., Singal, S., & Moka, N. (2025). Immunotherapy in GI cancers: lessons from key trials and future clinical applications. Antibodies, 14(3), 58.
- Peshin, S., Modi, S., & Singh, S. (2024). Advancements in cancer immunotherapy: a comprehensive review of immune checkpoint inhibitors with a focus on pembrolizumab and emerging strategies. Medi Clin Case Rep J, 2(3), 430-434.
- Peshin, S., Dharia, A., Moka, N., & Skelton IV, W. P. (2025). A Review of Immunotherapy in Renal Cell Carcinoma: Current Landscape and Future Directions. Cancers, 17(19), 3139.
- Gujarathi, R., Peshin, S., Zhang, X., Bachini, M., Meeks,M. N., Shroff, R. T., & Pillai, A. (2025). Intrahepatic cholangiocarcinoma: Insights on molecular testing, targeted therapies, and future directions from a multidisciplinary panel. Hepatology Communications, 9(7), e0743.
- Ramachandran, R., Cannon, M., Peshin, S., Kundranda, M., & Scott, A. J. (2025). Unveiling ctDNA Response: Immune Checkpoint Blockade Therapy in a Patient with POLE Mutation-Associated Early-Onset Colon Cancer. Current Oncology, 32(7), 370.
- Modi, S., Peshin, S., & Gim, G. (2024). ESOPECtrial: enhancing treatment approaches for esophageal adenocarcinoma. Journal of Cancer Metastasis and Treatment, 10, N-A.
- Peshin, S., Sonar, N., Sadiq, Z., & Kaur, G. (2024). Pembrolizumab-Induced Acute Interstitial Nephritis: Case Report of a Colorectal Cancer Patient. Int J Cancer Rest Ther, 9(2), 01-02.
- Peshin, S., Takrori, E., Kodali, N. A., Bashir, F., & Singal,S. (2025). Advances in the management of pancreatic cancer: Current strategies and emerging therapies. International Journal of Molecular Sciences, 26(15), 7055.
- Janjigian, Y. Y., Shitara, K., Moehler, M., Garrido, M., Salman, P., Shen, L., ... & Ajani, J. A. (2021). First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. The Lancet, 398(10294), 27-40.
- Janjigian, Y. Y., Kawazoe, A., Bai, Y., Xu, J., Lonardi, S., Metges, J. P., ... & Rha, S. Y. (2023). Pembrolizumab plus trastuzumab and chemotherapy for HER2-positive gastric or gastro-oesophageal junction adenocarcinoma: interimanalyses from the phase 3 KEYNOTE-811 randomisedplacebo-controlled trial. The Lancet, 402(10418), 2197-2208.
- Abou-Alfa, G. K., Macarulla, T., Javle, M. M., Kelley, R. K.,Lubner, S. J., Adeva, J., ... & Zhu, A. X. (2020). Ivosidenib in IDH1-mutant, chemotherapy-refractory cholangiocarcinoma (ClarIDHy): a multicentre, randomised, double-blind, placebo- controlled, phase 3 study. The Lancet Oncology, 21(6), 796-807.
- Finn RS, et al. IMbrave150. N Engl J Med. 2020.
- Yamamoto, K., Owaki, Y., Nakayama, I., Sakamoto, N., Ishizaka, M., Kadota, T., ... & Shitara, K. (2025). Characterization of early-onset gastritis during zolbetuximab- containing chemotherapy in CLDN18. 2-positive gastric cancer. ESMO open, 10(10), 105805.
- André, T., Shiu, K. K., Kim, T. W., Jensen, B. V., Jensen, L. H., Punt, C., ... & Gibbs, P. (2020). Pembrolizumab in MSI-H/ dMMR metastatic colorectal cancer. N. Engl. J. Med, 383, 2207-2218.
- Conroy, T., Hammel, P., Hebbar, M., Ben Abdelghani, M., Wei,A. C., Raoul, J. L., ... & Bachet, J. B. (2018). FOLFIRINOXor gemcitabine as adjuvant therapy for pancreatic cancer. New England Journal of Medicine, 379(25), 2395-2406.
- Sartore-Bianchi, A., Pietrantonio, F., Lonardi, S., Mussolin, B., Rua, F., Crisafulli, G., ... & Bardelli, A. (2022). Circulating tumor DNA to guide rechallenge with panitumumab in metastatic colorectal cancer: the phase 2 CHRONOS trial. Nature Medicine, 28(8), 1612-1618.
- Marabelle A, et al. (2023) Tumor-agnostic immunotherapy. Nat Rev Clin Oncol.
- Llovet JM, et al. (2022) HIMALAYA trial. Lancet Oncol.
- Temel JS, et al. (2010) Early palliative care. N Engl J Med.
- Reig M, et al. (2023) Liver cancer management update. Lancet.
- Kasi PM, et al. (2022) Early-onset CRC review. J Clin Oncol.
- Benson AB, et al. (2025) NCCN Guidelines GI cancers. J Natl Compr Canc Netw.
- Chen, D. S., & Mellman, I. (2013). Oncology meets immunology: the cancer-immunity cycle. immunity, 39(1), 1-10.
- Pardoll, D. M. (2012). The blockade of immune checkpoints in cancer immunotherapy. Nature reviews cancer, 12(4), 252- 264.
- Wei, S. C., Duffy, C. R., & Allison, J. P. (2018). Fundamental mechanisms of immune checkpoint blockade therapy. Cancer discovery, 8(9), 1069-1086.
- Fukumura, D., Kloepper, J., Amoozgar, Z., Duda, D. G., & Jain, R. K. (2018). Enhancing cancer immunotherapy using antiangiogenics: opportunities and challenges. Nature reviews Clinical oncology, 15(5), 325-340.
- Krummel, M. F., & Allison, J. P. (1996). CTLA-4 engagement inhibits IL-2 accumulation and cell cycle progression upon activation of resting T cells. The Journal of experimental medicine, 183(6), 2533-2540.


