Research Article - (2020) Volume 5, Issue 2
Transcatheter Closure of Atrial Septal Defect with Amplatzer Device in Adolescence and Adults: Short Term Results: MCVTC Experience
Received Date: Mar 04, 2020 / Accepted Date: Mar 10, 2020 / Published Date: May 28, 2020
Copyright: ©Rajaram Khanal, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: Atrial septal defect (ASD) is one of the common congenital heart diseases accounting for 8% to 10% of total congenital heart disease at birth and one of the most common diseases in adulthood. Ostium Secundum (OS) ASD has been successfully closed surgically for over several decades. But even though the results are better, it is associated with discomfort, morbidity, and a thoracotomy scar. Transcatheter closure in these patients has advanced rapidly in recent years and represents a growing field in invasive cardiology.
Methods: This prospective study was conducted at Manmohan cardiothoracic vascular and transplant center, Maharajgung, Kathmandu, Nepal. Fifty-two (52) patients with a diagnosis of OS ASD assessed for device closure. Patients underwent transthoracic and trans oesophageal echocardiography for the defect location, size, number, and associated anomalies. All patients with OS ASD fulfilling the inclusion criteria enrolled for device closure. The procedure conducted under fluoroscopy and transesophageal guidance. The cases were followed up at 3 and 6 months, reassessed clinically and by echocardiography for any complications.
Results: The patients with ASD who enrolled in this study belonged to the age group of 14 to 63 years. The device closure attempted in 46 patients, and the remaining six patients excluded from the procedure due to unsuitable anatomy after GA guided TEE done. Among 46 patients, 44 (95%) had successful device closure. The mean age of the patient was 35.7+ 10 and 33 (71.7%) female, with 13 (28.3%) males.
Conclusions: The transcatheter device closure of ASD in adolescents and adults has a high success rate with fewer complications and found to be safely documented during immediate and short term follow up. However, long terms follow up is required to conclude it.
Keywords
ASD (Atrial Septal Defect), OS (Ostium Secundum), Amplatzer device, TEE (Trans Esophageal Echocardiography), GA (General Anesthesia), RA (Right Atrium)
Introduction
Atrial septal defect (ASD) is one of the common congenital heart diseases accounting for 8% to 10% of total congenital heart disease at birth. It is one of the most common congenital heart diseases in adulthood [1].
Ostium Secundum ASD has been successfully closed surgically for over several decades. Even though the results of surgical repair are better, it is associated with discomfort, morbidity, and a thoracotomy scar [2]. Transcatheter closure in these patients has advanced rapidly in recent years and represents a growing field in invasive cardiology.
There is a number of devices that have developed for transcatheter closure of ostium secundum ASD. Among these devices Amplatzer septal occluder (ASO) are being most commonly used due to its unique design and easy handling. It has replaced surgical ASD closure in many centers as the standard treatment for pediatric patient with secundum ASD [3].
Subjects and Methods
From 2017th April 14th to 2018th Oct. 18th over 18 months period, a total of 52 patients were considered for defect closure with amplatzer device in MCVTC. Patients with symptomatic atrial septal defects underwent transthoracic and transoesophageal echocardiography to assess the defect location, size, number and associated anomalies [4].
Patients with the following inclusion criteria were enrolled for device closure:
1) Ostium secundum ASD with significant left to right shunt (QP/ QS ≥1.5:1)
2) OS ASD with a balloon stretched diameter of 38 mm or less with suitable rim of at least 4 mm except in the aortic rim
3) Right ventricular overload
Exclusion criteria were:
1) ASD other than secundum type
2) Abnormal venous drainage
3) Associated complex cardiac anomaly
4) Severe pulmonary hypertension with bi-directional or right to left shunting
5) Rim of <4mm between defect and cardiac structures
Procedure
The procedure was done in cardiac catheterization lab. Before catheterization, transthoracic or trans-esophageal echocardiography was done with or without sedation to re evaluate the size and location of defect and margin.
All procedures were performed under general anesthesia with trans esophageal echocardiography and fluoroscopy guidance throughout.
A device with a waist diameter similar to or in large defect up to 2 mm bigger than the stretched ASD diameter was chosen. The position and stability of device was assessed by fluoroscopy and transesophageal echocardiography (Figure 1). Care was taken to ensure that the device did not obstruct the right pulmonary veins, caval veins, coronary sinus or the mitral valve. Any residual shunt was evaluated by color Doppler echocardiography on TEE. The device was then released from the delivery system and final assessment of the position of the device was performed by TEE.
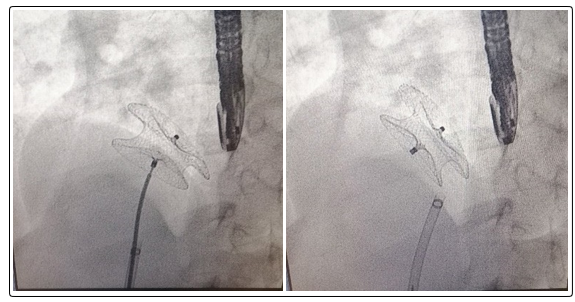
Figure 1
Results
During the period, a total of 52 patients were assessed for ASD device closure. The device closure was attempted in 46 patients and the remaining 6 patients were excluded from the procedure due to unsuitable anatomy after GA guided TEE done just before the procedure. Among 46 patients 44(95%) had successful device closure. The mean age of patient was 35.7+ 10 and 33 (71.7%) female with 13 (28.3%) male. The device size deployed was from 14mm to largest 40mm with mean size 27.6 + 6.6.
Baseline characteristics are shown in (Table 1 & 2). All patient had a single defect. RV dilatation was present in all patients and all had single device closure. The average procedure time and fluoroscopy time were 58 ±10.2 min and 6 ±5.5 min respectively (Table 2).
Table 1: Patients demographics and procedure characteristics (n=46)
|
Parameters |
Mean |
Range |
|
Age |
35.7 + 10 |
14-63 years |
|
Sex |
Male 13 Female 33 |
|
|
Pulmonary to systemic flow ratio |
2.12 |
1.54 - 4.2 |
|
Average PAP(mmHg) |
36.5 + 10 |
25 - 54 |
|
Size of ASD with TEE/TTE |
20.7 + 5.4 |
10-34 mm |
|
Amplatzer device size(mm) |
27.6 + 6.6 |
14-40mm |
|
Hospital stay(day) |
3 |
3-4 |
Table 2: Characteristics of the transcatheter procedure (n=46)
|
Variable |
Value |
|
Fluoroscopy time(min) |
6 + 5.5 |
|
Procedure time(min) |
58 + 10.2 |
|
Failed procedure |
2 |
Few major and minor complications occurred during and post procedure but there was no any mortality. The major complication was dislodgement of device, which occurred in one patient. The dislocated device was retrieved at surgery and the ASD was repaired at the same time without any further complications.
In one patient the procedure was failed, unable to deploy the device and the device was safely taken out without any complications.
The minor complications occurred in few patients that were transient sinus tachycardia in two patients, transient sinus bradycardia in one patients and paroxysmal supra ventricular tachycardia in one patients.
Course during hospital stay and follow up:
After ASD closure, the patients remained in CCU for one night and in ward for 2 days with total 3 days of hospital stay. Patients were started on aspirin and clopidrogel post procedure. Transthoracic echocardiography was repeated 24 hours after the procedure and before hospital discharge to ensure the suitable deployment of the device.
In majority of the patients, there was no any residual leak through the device at the end of the procedure. At the time of discharge, there was small residual leak in 3 patients.
The patients were discharged on aspirin (75mg) and clopidrogel (75mg) to be continued for 6 months. Standard bacterial endocarditis prophylaxis was recommended for 6 months or until complete closure was obtained. A repeat transthoracic echocardiography was performed at 3 and 6 months and then with the plan of follow up yearly. The mean follow up was 6 months.
All patients remained well on follow up and at 6 months follow up, on reassessment there was no any residual leak, no dislodgement of device and no other complications noted. There was no mortality during follow up. Two patients who had AF before procedure had persistent AF even on follow up. The age of these patients were > 40 years. The hemodynamics of patients on follow up is shown in (Table 3)
Table 3: Hemodynamic data pre and post procedure (n=44)
|
Before Intervention |
Total pt. |
Follow up |
|
ECG |
44 |
44 |
|
Sinus rhythm |
42 |
42 |
|
Atrial fibrillation |
2 |
2 |
|
Echocardiography |
n:44 |
44 |
|
RV dilatation |
44 |
|
|
Absent |
0 |
8 |
|
Mild |
6 |
12 |
|
Moderate |
20 |
22 |
|
Severe |
18 |
2 |
|
Tricuspid regurgitation |
44 |
32 |
|
Absent |
0 |
12 |
|
Mild |
23 |
18 |
|
Moderate |
18 |
13 |
|
Severe |
3 |
1 |
|
Residual leak |
3 |
0 |
Discussion
Transcatheter closure of ASD with amplatzer device has been an alternative approach to surgical repair nowadays in many canters. The major advantages of percutaneous closure of ASD are avoidance of thoracotomy, open-heart surgery and admission to an intensive care unit thus avoiding postoperative pain, other complications and subsequent surgical scar. The other advantages are less psychological impact, shorter hospital stay, and less need for blood transfusion [5-7].
Transcatheter closure of ASD has been found to be associated with low incidence of morbidity and mortality. Among different devices an important advantage of the Amplazer device is the retrievability during deployment, this greatly diminishes the risk of malposition and embolization of the device, and this reduces the risk for surgical removal of embolized devices.
Another important thing is the selection of correct size of the device. This depends on accurate assessment of the stretched diameter of the ASD. Oversized device can cause distortion of the retention discs, while undersized device may result in residual shunting early or even late embolization of the device.
The echocardiography monitoring throughout the procedure is essential to ensure the positioning of device before and after its release. In our study the device was implanted with TEE monitoring without increasing the chance of embolization or dislocation. Transcatheter closure of ASD guided by TEE is sufficient and safe with good imaging windows [8-10]. However sometimes TEE is impossible for technical or anatomical reasons especially in children and adolescents.
In general, the complication rate of transcatheter ASD closure is low. Although the procedure itself is considered to be safe and rarely causes problems during follow up, some complications have been reported. The major problem is that the device may be embolized. The overall complications in our study was 11% with major complication only in 2%. In the study conducted by Chessa and colleagues the overall complications rate was 9% with major complications in 2% [11].
The success rate in our study was 95% with treatment failure in 2 patients (dislodgement of device in one patient). In the study conducted by D J R Hildick et al the success rate was 97% with treatment failure in 2 patients [12]. In another study conducted by Mostafa et al the procedure was successful in 91% with device dislodgement and embolization in 2 patients [13].
The average procedure time in our study was 58+10 mins. In a study done by M Behjat et al the procedure time was 47.8 + 11.2.During follow up we found reduction in RV dimension and TR after device closure. In our study 8 patients had normal RV dimension and 12 patients had no TR. In a study done by M Bahijat et al during follow up 9 patients had normal RV dimension and 11 patients had no TR.
Our results demonstrate that the Amplatzer device is particularly useful in adolescent and adults with ostium secundum ASD. Many studies have reported that transcatheter closure of ASD with Amplatzer is safe and effective and currently can be considered an alternative to surgical treatment for selected patients [14-16]. The limitation of our study was small sample size with single operator experience at one tertiary cardiac center.
Conclusion
Trans catheter device closure of ASD in adolescent and adults has a high success rate with less complications and found to be safe documented during immediate and short term follow up. Our findings suggest that the amplatzer device closure of ASD should be the first option of management unless contraindicated. However long terms follow up is required in order to conclude it
References
- Dickinson DF, Arnold R, Wilkinson JL (1960) Congenital heart disease among 160,480 live born children in Liverpool 1960– 1969: implications of surgical treatment. Br Heart J 46: 55-62.
- Rigby ML (2003) Atrial septal defect. In: Gatzoulis MA, Webb GD, Daubeney PEF, editors. Diagnosis and management of adult congenital heart disease. Edinburgh: Churchill Livingstone 163-170.
- Konstantinides S, Geibel A, Olschewski M, L Görnandt, H Roskamm, et al (1995) A comparison of surgical and medical therapy for atrial septal defect in adult. N Engl J Med 333: 469-473.
- Bjornstad PG, Masura J, Thaulow E, Bjarne Smevik, Svein S Michelsen, et al. (1997) Interventional closure of atrial septal defect with Amplatzer device: first clinical experience. Cardiology in Young 7: 277-283.
- Lee CH, Kwok OH, Fan K, E Chau, A Yip, et al. (2001) Transcatheter closure of Atrial Septal Defect using Amplatzer septal occluder in Chinese adult. Cathet Cardiovasc 53: 373-377.
- Butera G, Rosa GD, Chesa M, Rosti L, Negura DG, et al. (2003) Tramscatheter closure of atrial septal defect in young children. J Am Coll Cardiol 42: 241-245.
- Suchon E, Pieculewicz M, Tracz W, Przewlocki T, Sadowski J, et al (2009) Transcatheter closure as an alternative and equivalent method to the surgical treatment of atrial septal defect in adults: comparison of early and late results. Med Sci Monit 15: 612-617.
- Li GS, Kong GM, Ji QS, Ji-fu Li, Yu-guo Chen, et al (2008) Reliability of transthoracic echocardiography in estimating the size of Amplatzer septal occluder and guiding percutaneous closure of atrial septal defects. Chin Med J (Engl) 121: 973- 976.
- Li GS, Kong GM, Wang YL, You-Peng Jin, Qiu-Shang Ji, et al (2009) Safety and efficacy of transcatheter closure of atrial septal defects guided by transthoracic echocardio-graphy: a prospective study from two Chinese Medical Centers. Ultrasound Med Biol 35: 58-64.
- Zaqout M, Suys B, De Wilde H, De Wolf D (2009) Transthoracic echocardiography guidance of transcatheter atrial septal defect closure in children. Pediatr Cardiol 30: 992-994.
- Chessa M, Carminati M, Butera G, Roberta Margherita Bini, Manuela Drago, et al (2002) Early and late complications associated with transcatheter occlusion of secundum atrial septal defect. J Am Coll Cardiol 39: 1061-1065.
- DJR Hildick-Smith, MO Sullivan, CR Wisbey, JH Mackay, EM Lee, et al (2004) Amplatzer device closure of atrial septal defects in mature adults: analysis of 76 cases. Heart 90: 334– 335.
- Mostafa Behjati, Sayed-Jalil Mirhosseini, Saiyed-Habibollah Hosseini, Shahrokh Rajaei (2011) Transcatheter Closure of Atrial Septal Defect withAmplatzer Device in Children andAdolescents: Short and Midterm results; an Iranian Experience. Iran J Pediatr 21: 166-172.
- Yew G, Wilson NJ (2005) Transcatheter atrial septal defect closure with Amplatzer septal occluder: five years follow-up. Cathet Cardiovasc Interv 64: 193-196.
- Masura J, Gavora P, Podnar T (2005) Long-term outcome of transcatheter secundum type atrial septal defect closure using Amplatzer septal occluder. J Am Coll Cardiol 45: 505-507.
- Cardoso CO, Filho RIR, Machado PR, Lisia M Galant François, Estela S K Horowitz, et al. (2007) Effectiveness of the amplatzer device for transcatheter closure of an ostium secundum atrial septal defect. Arq Bras Cardiol 88: 338-342.




