Research Article - (2023) Volume 5, Issue 2
To Assess the Association Between Decreased Uric Acid Level and Blood Pressure
2Professor and Head, Department of Nephrology, Marine City Medical College Hospital, Bangladesh
3Assistant Professor, Department of Pathology, Southern Medical College, Bangladesh
Received Date: Sep 05, 2023 / Accepted Date: Oct 18, 2023 / Published Date: Oct 25, 2023
Copyright: ©Â©2023 Showkat Azad, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Azad, S., Dutta, P. K., Sultana, Z. (2023). To Assess the Association Between Decreased Uric Acid Level and Blood Pressure. Adv J Uro Nephro, 5(2), 20-25.
Abstract
Background: Asymptomatic hyperuricemia is commonly viewed as an entity that should not be treated. Treatment of asymptomatic hyperuricemia in patients with renal impairment is still in debate. There is no evidence to rationalize pharmacotherapy for patients with hyperuricemia with no complication and serum uric acid concentration is below 10 mg/dL.
Objectives: To evaluate the association between decreased uric acid level and blood pressure.
Methods: This was a single-center, single (patient blinded, placebo-controlled study conducted in chattogram medical college hospital over 1 year. Patients of both sexes aged above 18 years with eGFRs of 15 to 60 mL/ min/1.73 m2 (as calculated with the 4-variable MDRD) and serum uric acid levels ≥7 mg/dL were included. Patients were selected in purposive technique divided randomly by lottery into two groups. One group received febuxostat 40-mg tablet once daily and another group received placebo tablets at the same time. Both groups received antihypertensive and antidiabetic medication and diuretics as per needed. Drugs and placebo were continued throughout the study period. Patients were evaluated at baseline, 3rd and 6th months for history, physical examination, assessment of any adverse events or end points and relevant investigations.
Results: 35 patients in each group were analyzed. Baseline characteristics were similar between the two groups. In febuxostat group the mean uric acid was decreased from 8.55 (±1.05) to 4.92 (±0.64) mg/dl, which was statistically significant (p <0.001) and in placebo group increased from 8.10 (±0.6) to 8.99 (±0.27) mg/dl which was statistically significant (P=0.001) after 6 months follow up. In case of mean eGFR in the febuxostat showed a nonsignificant increased from 25.28 (± 7.59) to 27.01 (±8.29) ml/min/1.73 m2, but in placebo group there was decreased mean eGFR from 26.81 (± 7.31) to 23.32(±7.72) ml/min/1.73 m2 which was statistically significant (p <0.001) and between group difference in eGFR was significant at 6 months (p = 0.048). Moreover after 6 months of treatment systolic and diastolic blood pressure were significantly lower in febuxostat group than placebo (p <0.05).
Conclusion: Compared to placebo, febuxostat slowed the decline in eGFR in CKD stage 3 and 4.
Keywords
Decreased, Uric Acid, Blood Pressure, Chronic Kidney Disease (CKD)
Introduction
High uric acid levels have been reported to be associated with in-creased rates of decline in glomerular filtration (eGFR) in a cross-sectional study [1]. The prevalence of hyperuricemia is 21.5% in adult men, and hyperuricemia prevails most in patients in their 30s and 40s; the prevalence of hyperuricemia is 30% in patients in their 30s reported by [2]. A study conducted by Iseki K, et al and stated that hyperuricemia is a risk factor for the onset of chronic kidney disease (CKD) and is significantly associated with the pro-gression of CKD [3]. Another study conducted by Neogi T and reported that persisting hyperuricemia causes the tissue deposi-tion of monosodium urate monohydrate crystals in extracellular fluids of the joints and other sites and induces urate deposition diseases (e.g., gouty arthritis, tophi, kidney injury, and urolithia-sis) [4]. Women are at higher risk of experiencing the progression of reduced renal function at an SUA concentration ≥ 6.0 mg/dL that is lower than the conventional definition of hyperuricemia and hyperuricemia influences the progression of kidney injury at the early phase of CKD [3,5]. It is clear that treatment of chronic kid-ney disease and its advanced end stage renal disease is expensive and beyond the reach of average Bangladeshis. The prevalence of CKD in Bangladesh is 19% and it is increasing [6]. As uric acid is an independent risk factor for progression of CKD and treatment of hyperuricemia with febuxostat is not so much expensive, if we could treat hyperuricemia even asymptomatic, we probably would be able to halt the progression of CKD and reduce the extra load of ESRD patients. In addition to the provision of lifestyle guidance (diet therapy, drinking restriction, encourage-ment of physical activity, patient education on treatment objec-tive, and management of comorbidities), pharmacotherapy with urate-lowering drugs is recommended for
(1) Hyperuricemia patients with gouty arthritis or tophi,
(2) Hyperuricemia patients whose SUA concentration is 8.0 mg/dL or higher and who have a complication (e.g. urate nephropathy), and
(3) Hyperuricemia patients whose SUA concentration is 10 mg/dL or higher and who have no complications [7].
A novel urate-lowering drug, febuxostat, is a potent non-purine se-lective inhibitor of XO and inhibits both the reduced and oxidized forms of the enzyme in contrast to allopurinol that inhibits the reduced form of the enzyme only [8]. Febuxostat is metabolized mainly by glucuronidation and oxidation in the liver, has its dual (urinary and fecal) pathways in excretion (urinary and fecal ex-cretion rates: 49.1% and 44.9%, respectively), and is effective and well tolerated in patients with mild to moderate renal impairment [9,10]. The post-hoc analysis of a clinical study on the long-term (5-year) oral administration of febuxostat in 116 patients with hy¬peruricemia gout revealed that the maintenance of or an improve¬ment in eGFR correlated with a quantitative reduction of urate level [11]. Therefore, we presumed that continuous lowering of SUA concentrations might deter a further reduction in renal func¬tion of patients with hyperuricemia and/or gout and recognized the importance of prospectively evaluating the long-term effect of SUA reduction on renal function in hyperuricemia patients with impaired renal function. The objective of the present study was to examine whether febuxostat prevents a further reduction in renal function as assessed with eGFR in patient with asymptomatic hy¬peruricemia in CKD stages 3 and 4.
Considering the above-mentioned facts and the fact that as there is very few experimental studies or research regarding these topics in our country this study is performed to determine the effect of febuxostat in reduction of hyperuricemia and slowing down the progression of renal disease.
Materials and Methods
Type of Study
Placebo controlled quasi experimental study.
Place of Study
Chattogram Medical College Hospital (CMCH). Chattogram
Study Period
One year after acceptance of protocol.
Study Population
Indoor and outdoor patients of CKD in nephrology ward.
Study Sample
Patients of CKD (stages 3 and 4) with asymptomatic hyperurice¬mia.
Sample size
So, according to formula, the targeted sample size was 44. Due to possibility of dropout 51 patients were targeted.
Inclusion Criteria
• Age ≥18 years.
• Serum uric acid: 7-10.0 mg/dL
• eGFR: 15–59 mL/min/1.73 m2.
Exclusion Criteria
• Uncontrolled diabetes mellitus; HbA1c: ≥8.0%
• Systolic blood pressure >_140 mm of Hg and diastolic blood pressure >_90 mm of Hg.
• Signs and symptoms of gout and urolithiasis.
• ALT or AST: greater than twice the upper limit of institutional reference range.
• Acute renal injury, other serious comorbidity such as severe heart failure and decompensated liver failure.
• Dialysis or renal transplant patient.
• Any active malignant disease.
• Previous History of hypersensitivity to febuxostat.
• Intake of any one or more of the following urate-lowering drugs within 5 days before the confirmation of eligibility: al¬lopurinol and febuxostat.
• The patient taking low-dose aspirin [75-324 mg/day].
• Pregnancy, nursing, or planed pregnancy during the study.
• Enrollment in other clinical trials within 24 weeks before pro¬viding informed consent.
Procedure of the Study
This interventional study was conducted by reviewing the records for all patients of CKD stage 3 and 4 admitted to nephrology ward in chattogram medical hospital over the period from 1 March 2018 to 1 March 2019. The patients were selected by purposive tech¬nique having CKD stage 3 and 4 with serum uric acid >_7mg/ dl to <_10mg/dl without symptoms of hyperuricemia admitted in Nephrology department through emergency or outpatient clinic.
Patients of CKD on admission found to be associated with severe co-morbidity such acute renal injury, severe heart failure, decom-pensated liver failure, active malignancy etc., CKD patients get-ting dialysis and patients were not giving written consent to par-ticipate in the study were being excluded. A standard case record form was used for data collection. Among patients with CKD, the stage of disease was assigned based on the level of kidney func-tion determined by serum creatinine and calculating eGFR using MDRD formula, according to the Kidney Disease Initiative Global Outcomes (KDIGO classification). All the medical records of the patients were reviewed and determined the stage of CKD. About 105 CKD stages 3 and 4 patients were assessed for eligibility from whom 84 patients were eligible for the study and taken informed consent.
These patients were divided randomly by lottery into two groups. In febuxostat group 43 patients were allocated and 41 patients were allocated for placebo group. Febuxostat group received tablet febuxostat 40-mg tablet once daily after breakfast. Placebo group received placebo tablets at the same time. Both groups received antihypertensive and antidiabetic medication as per needed. Di¬uretics were administered as clinically indicated. The selected pa¬tients were thoroughly informed about the aims, objectives and detail procedure of the study. He/She was encouraged for volun¬tary participation and allowed freedom to withdraw from the study whenever he/she liked even after participation. From all eligible subjects after getting consent some clinical history were taken and then details regarding disease were asked and noted. Eligible pa¬tients were followed up in every 3 months interval through his¬tory, physical parameters like weight, heart rate and systolic and diastolic blood pressure and relevant investigation such as serum creatinine level, serum uric acid, SGPT, HbA1c, serum calcium, phosphate, parathormone, serum albumin. In every visit, for blood test, blood was collected from accessible vein with proper aseptic precaution by confirming patient remained at least 4 hours /over-night fasting and blood was collected into red topped vacutainer.
After collection of whole blood, it was allowed to clot by leav-ing it undisturbed at room temperature for 10-15 minutes and was removed the clot by centrifuging at 1000-2000*g for 10 minutes in a refrigerated centrifuge for serum then through automated an¬alyzer measured uric acid, parathormone, creatinine, phosphate, calcium, SGPT serum albumin. For HbA1c blood was collected in blue topped vacutainer devoid of anticoagulant and was measured through automated analyzer. According to the clinical condition and investigation reports, dosage of drugs was titrated. Finally, 35 patients of each group completed their follow up. All data were checked and rechecked to avoid error. Cost was borne by the re¬searcher himself in some cases.
Data Processing and Analysis
All the data were checked and edited after collection. Then the data were entered into Microsoft Xcel data sheet to produce a master sheet then they were fed into SPSS (Statistical Package for Social Science) for window version on 23 software for processing and analysis. Categorical variables were reported as counts and per¬centages. Between groups comparisons were done either by inde¬pendent sample t test for numerical data with normal distribution and Chi square test for categorical data. Within group comparisons were done by paired sample t test. Correlation coefficient were cal¬culated by Pearson’s correlation coefficient test to asses correlation between uric acid and blood pressure and NNT was calculated by taking in terms of improvement of eGFR from baseline within 6 months. All P values were unadjusted. Statistical significance was defined as P< 0.05 and confidence interval set at 95% level.
Results
|
Variables (unit) |
Febuxostat Group (n=35) |
Placebo group (n=35) |
P value |
|
|
Age (years) |
Mean (±SD) |
48.97 (±12.22) |
51.97 (±7.19) |
0.215* |
|
Range |
19-75 |
38-63 |
||
|
Sex |
Male |
19 (54.3%) |
24 (68.6%) |
0.220† |
|
Female |
16 (45.7%) |
11 (31.4%) |
||
|
Data are expressed as frequency (percentages) if not otherwise mentioned; SD: Standard Deviation. *P value was derived from independent sample t test. P value was derived from Chi-square test |
||||
Table 1: Age and Sex of the 70 CKD Patients by their Treatment Groups
Table 1 shows that, both the Febuxostat and Placebo group were comparable at baseline (P >0.05). The mean age was 48.97 years and 51.97 years respectively in Febuxostat and Placebo group. There was male predominance in both groups.
|
Characteristics |
Febuxostat Group (n=35) |
Placebo group (n=35) |
P value |
|
|
H/O DM |
Yes |
18 (51.4%) |
17 (48.6%) |
0.821† |
|
No |
17 (48.6%) |
18 (51.4%) |
||
|
H/O HTN |
Yes |
30 (85.7%) |
33 (94.3%) |
0.232† |
|
No |
5 (14.3%) |
2 (5.7%) |
||
|
CKD stage |
Stage 3 |
12 (34.3%) |
10 (28.6%) |
0.607† |
|
Stage 4 |
23 (65.7%) |
25 (71.4%) |
||
|
H/O: History of; HTN: Hypertension; DM: Diabetes mellitus; Data are expressed as frequency (percentages); †: P values were de- rived from Chi-square test |
||||
Table 2: History of DM and HTN and CKD stage of 70 CKD Patients by their Treatment Group
Table 2 shows that, majority of the patients in both the groups had HTN. DM was present almost in half of the patients of both groups. Both the groups were comparable with respect to presence of HTN and DM at baseline (p>0.05). Regarding CKD stage, patients with stage 4 CKD were predominant than stage 3 in both groups and similarly distributed (p>0.05).
|
Blood pressure |
Mean (±SD), mmHg |
P value |
|
|
Febuxostat group (n=35) |
Placebo group (n=35) |
||
|
Systolic BP |
137±7 |
138±8 |
0.660† |
|
Diastolic BP |
86±8 |
84±8 |
0.250† |
|
P values were derived from independent sample t test |
|||
Table 3: Baseline Mean Blood Pressure of the 70 CKD Patients by their Treatment Group
Table 3 indicates that, both the Febuxostat and Placebo group did not differ for baseline systolic and diastolic blood pressure (p>0.05).
|
Variables (unit) |
Febuxostat Group (n=35) |
Placebo group (n=35) |
P value |
|
Serum creatinine, mg/dl |
2.70±0.63 |
2.54±0.50 |
0.251† |
|
eGFR, ml/min per 1.73 m2 |
25.28±7.59 |
26.81±7.31 |
0.392† |
|
Serum uric acid, mg/dl |
8.55±1.05 |
8.10±0.61 |
0.054† |
|
†P values were derived from independent sample t test |
|||
Table 4: Baseline Mean Serum Creatinine, eGFR and Uric Acid Level of the 70 CKD Patients by their Treatment Group
Table 4 indicates that, both the Febuxostat and Placebo group did not differ for baseline Serum creatinine, eGFR and uric acid levels (p>0.05).
|
Serum Uric Acid |
Mean (±SD) value of Serum Uric Acid (mg/dl) |
P valuea |
|
|
Febuxostat |
Placebo |
||
|
At baseline |
8.55±1.05 |
8.10±0.6 |
0.054 |
|
At month 3 |
6.01±0.89 |
8.41±0.70 |
<0.001 |
|
At month 6 |
4.92±0.64 |
8.99±0.27 |
<0.001 |
|
P valueb |
<0.001 |
0.001 |
|
|
aBetween group comparison by independent sample t test. bWithin-group comparison, baseline versus study end by paired sample t test. |
|||
Table 5: Changes in Serum Uric Acid Levels at Baseline, 3 Months, and 6 Months in Two Groups
Table 5 shows that, the serum uric acid level, which was similar in both groups at baseline was significantly lower in febuxostat group than placebo group at 3 month and at 6 months. In febuxostat group uric acid level was significantly lower from the baseline value (p<0.001) and in placebo group uric acid level was significantly increased from the baseline value (p=0.001).
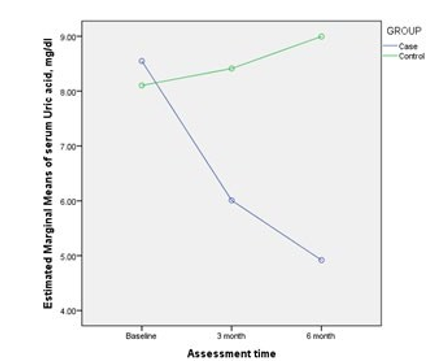
Figure 1: Time Course Change of Estimated Marginal Means of Serum Uric Acid Levels from Baseline through 6 Months in Both Groups
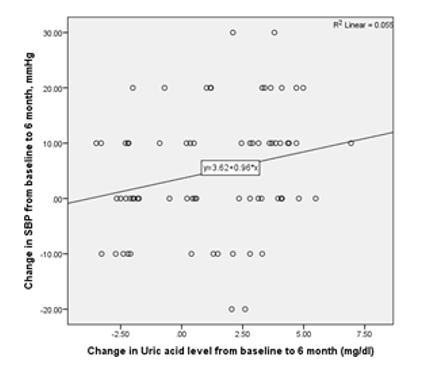
Figure 2: Relationship between the Reduction of Uric Acid Level and the Decrease of SBP in 70 CKD Patients Treated
There was a significant positive correlation between the reduction of serum uric acid level and reduction of SBP in 6 months among the study subjects irrespective of the study groups (r = 0.235; P=0.047) (Figure 2).
Discussion
The present study demonstrated that the febuxostat decreases uric acid level and slows eGFR decline when given to asymptomatic hyperuricemia patient with CKD. In the present study the mean age was 48.97 years and 51.97 years respectively in febuxostat and placebo group. There was male predominance in both groups. A study conducted by Yamanaka, showed that the prevalence of hyperuricemia is 21.5% in adult men, and hyperuricemia prevails most in patients in their 30s and 40s; the prevalence of hyperuri¬cemia is 30% in patients in their 30s [2]. In the present study in febuxostat group the baseline mean uric acid was 8.55 (+_1.05 SD) mg/dl, after 6 months follow up decreased mean uric acid to 4.92 (+_0.64) mg/dl which was statistically significant (p <0.001) and in placebo group mean uric acid increased from base value from 8.10 (+_0.6) mg/dl to 8.99 (+_0.27) mg/dl which was statis¬tically significant (P=0.001). A 6 months, blind randomized pla¬cebo-controlled trail conducted by Sircar D, et al., showed that in case of febuxostat group baseline mean uric acid was 9.0 (+_2.0) mg/dl , after 6 months follow up mean uric acid decreased to 5.2 (+_1.5) mg/dl which was statistically significant(p<0.001) and in placebo group mean uric acid also decreased from 8.2 (+_1.1) mg/ dl at baseline to 7.8 (+_1.0) mg/dl at 6 month which was statis¬tically significant (<0.001) [12]. The comparison of both studies has shown that in febuxostat group there is similar finding but in placebo group there is contradictory result. This contradictory re¬sult in case of placebo group is probably due to higher compliance in dietary restriction of purine containing diet. In a study done by Akimoto T, et al., showed that febuxostat lowered the serum uric acid level (8.9 +_1 mg/dl at baseline) significantly from one month after initiation of the treatment and the target serum uric acid level (6 mg/dl) was achieved in 12 patients (70.5%) after one month, compared to 13 (76.4%) and 14 (82.3%) patients after 3rd and 6th month of treatment [13].
This study is similar to the current study regarding the lowering of uric acid by febuxostat. Experimental evidence suggests that uric acid may harm to CKD patients through intensification of in¬flammation that may cause acceleration of progression of CKD. A large number of clinical observation studies and small number of controlled trails have investigated the potential association be¬tween increased serum uric acid concentration and kidney events. Consequently, many studies, including the Cardiovascular Health Study, which involved 5,808 patients with 5 years of follow-up, showed a significant relationship between increased uric acid con¬centration and CKD progression. Although these studies suggest that decreasing serum uric acid may retard CKD progression, urate lowering therapy for patients with CKD cannot be advocated pro¬actively because of their limitations (e.g., small number of partici¬pants, short follow up and study design). Moreover, in MDRD trial which 840 patients with stages 3 to 4 CKD were followed up for 10 years, did not find uric acid concentration to be an independent risk factor for progression to kidney failure [14]. Thus, there are contradictory clinical outcome with respect to the kidney protec¬tive effect of decreasing uric acid levels in patients with CKD. In the present study a significant positive correlation between the reduction of serum uric acid level and reduction of systolic blood pressure in 6 months among the study group (r =0.235, p =0.047) but in case of diastolic blood pressure this is not signif-icant (r =0.15, p =0.216). In previous study conducted by Sircar D, et al., showed that the average blood pressure in the febuxostat group was 149.3/85.4 at baseline which is decreased to 136.6/80 at 6 month and average blood pressure in the placebo group was 138.5/80.5 mm of Hg at baseline and decreased to 134.4/82.9 mm of Hg at 6 months which is similar to the current study [12].
Conclusion
In conclusion, the outcome of this randomized controlled trail conducted among patients with CKD stages 3 and 4 was that febuxostat decrease the serum uric acid and has a positive effect on blood pressure lowering. Finally, febuxostat slowed the decline in eGFR in CKD stages 3 and 4 compared to placebo. Due to the above-mentioned limitations, our study is not definitive and is still hypothesis generating with regard to renal outcomes. Therefore, febuxostat should not be considered part of clinical practice with¬out more definitive trials (large-scale, adequately powered, mul-ticenter studies, with definitive endpoints on efficacy and safety) to treat the asymptomatic hyperuricemia in CKD stages 3 and 4 considering retardation of CKD progression.
References
- Satirapoj, B., Supasyndh, O., Nata, N., Phulsuksombuti, D., Utennam, D., Kanjanakul, I., ... & Duangurai, K. (2010). High levels of uric acid correlate with decline of glomerularfiltration rate in chronic kidney disease. J Med Assoc Thai, 93(Suppl 6), S65-70.
- Yamanaka, H. (2011). Japanese guideline for the management of hyperuricemia and gout. Nucleosides, Nucleotides and Nucleic Acids, 30(12), 1018-1029.
- Iseki, K., Ikemiya, Y., Inoue, T., Iseki, C., Kinjo, K., & Tak-ishita, S. (2004). Significance of hyperuricemia as a risk factor for developing ESRD in a screened cohort. American Journal of Kidney Diseases, 44(4), 642-650.
- Neogi, T. (2011). Clinical practice. Gout, N Engl J Med, 364(5), 443-452.
- Uchida, S., Inokami, T., & Yamashita, M. (2006). Implication of hyperuricemia and its treatment in progressive renal diseases. Nephrol Fronti, 5, 135-136.
- Hasan, M. J., Kashem, M. A., Rahman, M. H., Qudduhush, R., Rahman, M., Sharmeen, A., & Islam, N. (2013). Prevalence of chronic kidney disease (CKD) and identification of associated risk factors among rural population by mass screening. Community Based Medical Journal, 1(1), 20-26.
- Japanese Society of Nephrology. (2012). Clinical practice guidebook for diagnosis and treatment of chronic kidney disease 2012. Nihon Jinzo Gakkai Shi, 54(8), 1034-1191.
- Takano, Y., Hase-Aoki, K., Horiuchi, H., Zhao, L., Kasahara, Y., Kondo, S., & Becker, M. A. (2005). Selectivity of febuxostat, a novel non-purine inhibitor of xanthine oxidase/xan-thine dehydrogenase. Life sciences, 76(16), 1835-1847.
- Hoshide, S., Nishimura, S., Ishii, S., Matsuzawa, K., Saito, N., & Tanaka, T. (2000). Metabolites of TMX-67, a new pharmaceutical entity for the treatment of gout or hyperuricemia, and their pharmacokinetic profiles in humans. Drug Metab Rev, 32(Suppl 2), 269.
- Grabowski, B. A., Khosravan, R., Vernillet, L., & Mulford,D. J. (2011). Metabolism and excretion of [14C] febuxostat, a novel nonpurine selective inhibitor of xanthine oxidase, in healthy male subjects. The Journal of Clinical Pharmacology, 51(2), 189-201.
- Whelton, A., MacDonald, P. A., Zhao, L., Hunt, B., & Gun-awardhana, L. (2011). Renal function in gout: long-term treatment effects of febuxostat. JCR: Journal of Clinical Rheumatology, 17(1), 7-13.
- Sircar, D., Chatterjee, S., Waikhom, R., Golay, V., Raychaud-hury, A., Chatterjee, S., & Pandey, R. (2015). Efficacy of febuxostat for slowing the GFR decline in patients with CKD and asymptomatic hyperuricemia: a 6-month, double-blind, randomized, placebo-controlled trial. American Journal of Kidney Diseases, 66(6), 945-950.
- Akimoto, T., Morishita, Y., Ito, C., Iimura, O., Tsunematsu, S., Watanabe, Y., ... & Nagata, D. (2014). Febuxostat for hyperuricemia in patients with advanced chronic kidney disease. Drug Target Insights, 29(2), 406-413.
- Evan Chen, Andrew Cheung. The MDRD trial: Protein intake and blood pressure control in renal insufficiency.



