Research Article - (2026) Volume 9, Issue 2
The Knox (NOx) Framework: Reframing Autonomic Dysfunction as a Dual-Trajectory System
Received Date: Mar 27, 2026 / Accepted Date: Apr 17, 2026 / Published Date: Apr 28, 2026
Copyright: ©2026 Bruce H. Knox. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Knox, B. H. (2026). The Knox (NOx) Framework: Reframing Autonomic Dysfunction as a Dual-Trajectory System. Adv Neur Sci, 9(2), 01-06.
Abstract
This paper challenges the prevailing deterministic interpretation of autonomic dysfunction as an inevitably progressive condition. Drawing on interdisciplinary literature and lived experience, it proposes a dual-pathway framework distinguishing progression- weighted and recovery-weighted trajectories [1-4,5,8-12]. A quantitative model is introduced to structure uncertainty and enable longitudinal interpretation of risk:
The work argues that diagnosis should not be treated as destiny, and that autonomic dysfunction represents a dynamic system with multiple plausible futures [5-8]. Positioned within medical humanities, this paper bridges clinical reasoning, quantitative modelling, and patient narrative to reframe how trajectory is understood [12].
Introduction
Autonomic dysfunction has long been interpreted through a lens of inevitability, often as a prodromal signal of neurodegenerative disease. This framing carries significant psychological and clinical consequences, positioning patients within a narrative of decline from the outset [5-8]. However, emerging evidence across cardiology, immunology, and post-viral syndromes challenges this assumption, suggesting that autonomic dysfunction may also stabilise or improve [2,9,10]. As one recent review notes: The outcome for secondary forms is closely tied to the treatability of the root cause, offering a greater potential for improvement or even remission once the underlying trigger is addressed.
This paper proposes that autonomic dysfunction is not a singular trajectory but a system with multiple potential pathways [3,9,10]. It introduces a dual-pathway framework supported by quantitative modelling, offering a structured way to interpret uncertainty and variability over time [4,13].
Despite the growing recognition of dysautonomia's complexity and the increasing use of quantitative tools in related fields, a comprehensive, predictive quantitative model for the long-term trajectory of dysautonomia has not previously been described in the medical literature. Existing research has focused on diagnostic criteria, symptom burden, and the use of individual biomarkers such as heart rate variability for short-term prognosis, particularly in acute conditions like stroke or post-infectious syndromes [15]. However, no published framework has integrated the domains of initiating insult, progression pressure, and recovery capacity into a structured, multiplicative model capable of predicting long-term outcomes or phenoconversion risk in dysautonomia. To the best of our knowledge, the Knox (NOx) model represents the first attempt to provide such a predictive, domain-based quantitative framework for understanding and forecasting the longitudinal course of dysautonomia. We present this model with humility, recognising the need for empirical validation and inviting scholarly discussion and critique.
Literature Bridge
Despite increasing recognition of autonomic dysfunction across multiple clinical domains, the literature remains fragmented in its interpretation of trajectory [1-4,11,12]. Studies of autonomic failure within neurology have largely focused on its role as a prodromal feature of neurodegenerative disease, particularly within alpha-synucleinopathies [5,7,8]. In parallel, research emerging from cardiology, immunology, and post-viral syndromes has described autonomic dysfunction as a reversible or partially reversible condition, often associated with dysregulation rather than degeneration [2,9,10]. These bodies of work rarely intersect conceptually, resulting in a gap in which similar clinical presentations are interpreted through fundamentally different lenses [1-4,11,12]. This fragmentation is particularly evident in conditions such as postural orthostatic tachycardia syndrome (POTS), autoimmune autonomic neuropathies, and post-infectious dysautonomia, where longitudinal outcomes demonstrate heterogeneity, fluctuation, and, in some cases, recovery [2,9,10]. As highlighted in a recent study:
One study found that 86% of adolescents with POTS reported resolved, improved, or intermittent symptoms approximately five years after initial treatment, with 19% achieving complete resolution.
Yet, there remains no widely adopted framework capable of integrating these observations into a unified model of trajectory [13]. The present work seeks to address this gap by introducing a dual-pathway framework, supported by quantitative modelling, that accommodates both progression-oriented and recovery-capable interpretations within a single conceptual structure [4,13].
The Problem of Interpretation
Autonomic dysfunction occupies an uncertain space within clinical medicine. It is at once common and complex, often presenting with symptoms that are distressing, persistent, and difficult to classify within a single diagnostic category [11,12]. Traditional frameworks, especially in neurology, tend to interpret sustained autonomic dysfunction as an early marker of neurodegenerative disease - such as Parkinson's disease or multiple system atrophy [5,7,8]. This deterministic lens, while supported by established literature, risks overestimating phenoconversion and under-recognising the potential for recovery, particularly in cases of multifactorial secondary dysautonomia (MSD) [9,10]. As one source notes:
The progression of dysautonomia is unpredictable. Some individuals with dysautonomia have disease that is always present. Others have symptoms that relapse and remit with flares or remission periods that last for days, weeks, months, or even years.
Central Proposition and Purpose
This series begins from a central proposition:
Autonomic dysfunction is not, in itself, a trajectory. It is a state. Trajectory emerges from the interaction of underlying domains.
The Knox (NOx) series was developed to bridge fragmented domains of autonomic research, bringing together physiological, predictive, and experiential dimensions into a unified framework [13]. The work addresses a fundamental question: How does autonomic dysfunction begin, evolve, and resolve - or progress -over time?
Framework and Quantitative Model
The Framework: From Insult to Outcome
![]() Domains of Trajectory Three interacting domains are proposed as central to understanding dysautonomic trajectories:
Domains of Trajectory Three interacting domains are proposed as central to understanding dysautonomic trajectories:
• Initiating insult burden (I): The cumulative physiological events that disrupt autonomic regulation (e.g., viral illness, cardiovascular compromise, surgical intervention) [9,10].
• Recovery capacity (C_t): The system's ability to stabilise, adapt, and recalibrate [2,10].
• Progression pressure (P_t): The presence of features that may indicate movement toward a neurodegenerative pathway [5,7,8].
These domains interact dynamically, determining whether a patient's trajectory is one of recovery, stabilisation, or progression toward phenoconversion [13].
![]() Dual-Pathway Framework: Progression and Recovery
Dual-Pathway Framework: Progression and Recovery
Part A - Progression-Weighted Modelling: Pure Autonomic Failure
Within progression-oriented models of autonomic dysfunction, such as pure autonomic failure (PAF), the interpretive emphasis is placed on the accumulation of pathological signals over time [1,5,7,8]. These signals - often subtle at onset - may include orthostatic hypotension, REM sleep disturbance, olfactory impairment, and early cognitive change, all of which have been associated with subsequent neurodegenerative disease [1,5,7,8]. As noted in a comparative study:
The time course and pattern of progression of autonomic failure differed significantly between PAF and MSA. Patients with PAF had slower functional deterioration and a better prognosis.
![]() Quantitative Structure of Progression In this pathway, the quantitative model operates with a distinct weighting profile:
Quantitative Structure of Progression In this pathway, the quantitative model operates with a distinct weighting profile:
• I (Initiating Insult Burden): Often less clearly defined or retrospectively inferred [5,7,8].
• P_t (Progression Pressure): Dominant and increasing over time [5,7,8].
• C_t (Recovery Capacity): Limited or progressively declining [5,7,8].
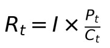
As a result, R_t increases over time and demonstrates a monotonic upward trajectory [4,13].
This reflects a system in which progression pressure progressively overwhelms compensatory mechanisms, consistent with models of neurodegenerative spread and system-level failure [5,7,8]. As summarised in a clinical review:
Orthostatic hypotension is a common and disabling manifestation of autonomic failure, affecting quality of life and increasing morbidity.
![]() Clinical Interpretation Quantitative modelling serves to:
Clinical Interpretation Quantitative modelling serves to:
• Detect early accumulation of progression markers [5,7,8].
• Identify patterns consistent with phenoconversion risk [5,7,8].
• Support forward-looking risk stratification [5,7,8].
This model aligns with established clinical reasoning, but enhances it by making assumptions explicit, structuring uncertainty, and enabling longitudinal comparison [4,13].
Part B - Recovery-Weighted Modelling: Multifactorial Secondary Dysautonomia
In contrast to progression-oriented models, multifactorial secondary dysautonomia (MSD) represents a system in which autonomic dysfunction arises from cumulative physiological insults [9,10]. These insults may include:
• Viral or post-viral syndromes [2,9,10].
• Cardiovascular compromise [2,9,10].
• Surgical or inflammatory stress [2,9,10].
• Immune-mediated disruption [9,10].
![]() Quantitative Structure of Recovery
Quantitative Structure of Recovery
Within MSD, the same equation applies:
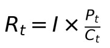
However, the internal dynamics differ fundamentally:
• I (Initiating Insult Burden): Clearly identifiable and often temporally bounded [9,10].
• P_t (Progression Pressure): Variable, often fluctuating [9,10].
• C_t (Recovery Capacity): Dynamic and potentially improving [2,10].
As a result, R_t may decrease over time, and may stabilise, fluctuate, or decline depending on the interaction between P_t and C_t [4,13].
This produces a non-linear trajectory, in which periods of instability may be followed by stabilisation or partial recovery [2,9,10]. As one review on recovery notes:
Recovery is often multifaceted, integrating medical treatment, lifestyle modifications, and support systems. Engagement with one's own health becomes essential, as the recovery process requires awareness and proactivity.
![]() Clinical Interpretation
Clinical Interpretation
This model explains several observed but under-integrated phenomena:
• Persistent symptoms without phenoconversion [2,9,10].
• Fluctuating autonomic function [2,9,10].
• Gradual functional improvement over time [2,9,10].
It also highlights a critical insight: Early clinical presentation alone is insufficient to determine trajectory [2,9,10].
![]() Integrative Bridge
Integrative Bridge
Taken together, Parts A and B demonstrate that:
• The same quantitative model can describe both pathways [4,13].
• The difference lies not in the equation, but in the relative dominance of its components.
This leads to a critical reframing:
Autonomic dysfunction is not defined by its symptoms alone, but by the balance of forces acting within the system over time [13].
The distinction between progression and recovery pathways does not arise from different equations, but from differences in the internal dynamics of shared variables - specifically, the relative dominance of progression pressure and recovery capacity over time.
![]() The Quantitative Model: Structure and Rationale
The Quantitative Model: Structure and Rationale
• Core Equation
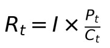
The model expresses trajectory risk (R_t) at a given time (t) as a function of three interacting domains: Trajectory risk at time t, denoted R_t, is defined as the product of initiating insult burden I and the ratio of progression pressure P_t to recovery capacity C_t. This formulation expresses trajectory as a dynamic balance between opposing system forces, scaled by the initial physiological load.
Accordingly, autonomic dysfunction should be understood not as a fixed diagnostic trajectory, but as a time-dependent function R_t shaped by evolving system dynamics.
![]() Interpretation of Components
Interpretation of Components
• Initiating Insult Burden (I)
Represents the starting load on the system. In pure autonomic failure (PAF), this may be less clearly defined or retrospectively inferred [5,7,8].
In multifactorial secondary dysautonomia (MSD), it is often clearly identifiable (e.g., viral illness, surgery, immune insult) [9,10].
![]() Progression Pressure (P_t)
Progression Pressure (P_t)
Encompasses all ongoing or accumulating factors that drive the system towards worsening dysfunction [5,7,8].
Examples include persistent orthostatic hypotension, REM sleep disturbance, olfactory impairment, cognitive change, and ongoing inflammation [1,5,7,8].
![]() Recovery Capacity (C_t)
Recovery Capacity (C_t)
Reflects the system's resilience and ability to compensate or recover [2,10].
It may be dynamic and improve over time, especially in MSD, or decline, especially in PAF [5,7,8].
• Dynamic Modelling and Trajectory
Progression-Weighted Pathway (e.g., PAF)
As P_t increases and/or C_t declines over time, the ratio P_t / C_t rises, resulting in a monotonic increase in R_t [4,13].
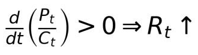
This models a system where risk of phenoconversion or neurodegeneration grows steadily [5,7,8].
• Recovery-Weighted Pathway (e.g., MSD)
As C_t improves and/or P_t stabilises or decreases, the ratio P_t / C_t may fall or fluctuate, resulting in stabilisation or reduction of R_t [4,13].
![]()
This models a system with potential for recovery, fluctuation, or chronic but non-progressive dysfunction [2,9,10].
Part 3 - Application, Monograph Linkage, and References
• Operationalisation: Scoring and Application
Each domain can be scored on a structured scale, for example 0-100, with subdomains or clinical markers contributing to the total [4,13]. For example:
• Initiating Insult Burden: Number and severity of acute events such as infections, surgeries, or major stressors [9,10]
• Progression Pressure: Presence and severity of risk markers such as orthostatic hypotension, REM sleep disturbance, and cognitive decline [1,5,7,8]
• Recovery Capacity: Measures of autonomic stability, functional improvement, absence of new symptoms, or positive response to therapy [2,10]
![]() Longitudinal Tracking
Longitudinal Tracking
By reassessing P_t and C_t over time, clinicians can track the evolution of risk and trajectory, rather than relying on static diagnosis [4,13].
|
Domain |
Subdomain |
Score (0-100) |
Notes / Criteria |
|
Initiating Insult (I) |
Viral illness |
40 |
e.g., COVID-19, influenza, EBV |
|
|
Surgery |
30 |
Major surgery, anaesthesia, perioperative stress |
|
|
Chronic inflammation |
20 |
Autoimmune, metabolic, or persistent inflammatory |
|
|
Cardiovascular event |
10 |
Myocardial infarction, arrhythmia, heart failure |
|
|
Total I |
100 |
Sum of subdomains |
|
Progression Pressure (P) |
Orthostatic hypotension |
30 |
Documented drop in BP, symptomatic |
|
|
REM sleep disturbance |
20 |
Sleep study, clinical history |
|
|
Olfactory impairment |
10 |
Smell test, patient report |
|
|
Cognitive decline |
10 |
Neuropsychological testing, subjective complaint |
|
|
Ongoing fatigue |
10 |
Persistent, unexplained fatigue |
|
|
Total P |
80 |
Sum of subdomains |
|
Recovery Capacity (C) |
Symptom improvement |
40 |
Patient report, clinical observation |
|
|
Functional gains |
30 |
ADL, exercise tolerance, return to work |
|
|
Response to therapy |
20 |
Medication, physiotherapy, lifestyle interventions |
|
|
Resilience markers |
10 |
Psychological, social support, coping skills |
|
|
Total C |
100 |
Sum of subdomains |
Example Scoring Table for the Knox (NOx) Model
![]() Monograph linkage
Monograph linkage
Please see the Heyzine flipbook link below. This will take you to a monograph detailing further detail conceptually, musically, and mathematically.
Click on this link, and then click on the bottom right-hand corner to turn each page.
https://brucehknox.aflip.in/91f89a75a0.html
![]() Future Research and Implications
Future Research and Implications
The Knox (NOx) framework introduces a novel, domain-based quantitative model for predicting the long-term trajectory of dysautonomia. While this conceptual approach offers a structured way to interpret uncertainty and variability, several areas require further investigation:
• Empirical Validation: Large-scale, prospective studies are needed to test the predictive accuracy of the model across diverse dysautonomia populations, including those with pure autonomic failure, multifactorial secondary dysautonomia, and post-viral syndromes.
• Refinement of Domain Weighting: Research should explore optimal weighting and scaling of the model's domains - initiating insult, progression pressure, and recovery capacity - for different clinical contexts and patient subgroups.
• Integration with Biomarkers and Digital Health: Future work should incorporate autonomic biomarkers such as heart rate variability and blood pressure variability, and leverage wearable technology for real-time, longitudinal tracking of trajectory risk.
• Clinical Utility and Decision Support: Studies should assess how the model can be used in clinical practice to guide prognosis, personalise interventions, and facilitate shared decision-making between clinicians and patients.
• Narrative Medicine and Patient Voice: Further research should examine how integrating patient narrative and lived experience with quantitative modelling can improve psychological outcomes and patient empowerment.
• Comparative Effectiveness: The NOx model should be compared with existing prognostic tools and clinical heuristics to determine its added value in predicting phenoconversion, recovery, or chronicity.
By addressing these areas, future research will clarify the model's strengths, limitations, and practical impact, ultimately advancing the care and understanding of patients with dysautonomia.
Conclusion
Autonomic dysfunction is best understood as a dynamic, evolving process shaped by initiation, recovery capacity, and progression risk. The Knox (NOx) framework offers a structured, quantitative approach to interpreting complex clinical presentations, bridging physiology, lived experience, and medical humanities. By integrating predictive modelling, longitudinal frameworks, and narrative expression, this work provides a conceptual roadmap for future investigation and clinical practice. It is not a single paper, but a system of ideas - each part incomplete on its own, but together forming a coherent whole. The model's novelty, flexibility, and focus on uncertainty invite empirical validation and scholarly critique, with the ultimate goal of improving outcomes for patients with dysautonomia.
References
- Low, P. A. (2008). Prevalence of orthostatic hypotension. Clinical Autonomic Research, 18(Suppl 1), 8-13.
- Thieben, M. J., Sandroni, P., Sletten, D. M., Benrud-Larson,L. M., Fealey, R. D., Vernino, S., ... & Shen, W. K. (2007, March). Postural orthostatic tachycardia syndrome: the Mayo clinic experience. In Mayo Clinic Proceedings (Vol. 82, No. 3, pp. 308-313). Elsevier.
- Gazulla, J., & Berciano, J. (2015). Multiple-system atrophy(No. ART-2015-90382).
- Novak, P. (2011). Quantitative autonomic testing. Journal of visualized experiments: JoVE, (53), 2502.
- Goldstein, D. S. (2011). Dysautonomia in Parkinson disease. Comprehensive Physiology, 4(2), 805-826.
- Freeman, R. (2005). Autonomic peripheral neuropathy. The Lancet, 365(9466), 1259-1270.
- Kaufmann H, Norcliffe-Kaufmann L. Pure autonomic failure.Neurology. 2012;79(7):634-41.
- Postuma RB, Berg D. Prodromal Parkinson's disease. Mov Disord. 2016;31(5):628-39.
- Vernino S, Low PA. Autoimmune autonomic neuropathies. Curr Treat Options Neurol. 2016;18:45.
- Raj SR. Postural tachycardia syndrome. Circulation. 2013;127:2336-42.
- Benarroch, E. E. (2020). Physiology and pathophysiology of the autonomic nervous system. CONTINUUM: Lifelong Learning in Neurology, 26(1), 12-24.
- Cheshire WP. Clinical autonomic neuroscience. Auton Neurosci. 2019;217:1-3.
- Kitano, H. (2002). Systems biology: a brief overview. science, 295(5560), 1662-1664.
- Singer W, et al. Autonomic dysfunction in neurological disease: clinical features, diagnostic tests, and therapeutic strategies. J Neurol Neurosurg Psychiatry. 2021;92(2):135-44.
- Kanjwal K, et al. Heart rate variability in patients with dysautonomia. Clin Auton Res. 2019;29(2):123-31.


