Research Article - (2020) Volume 3, Issue 1
The Effects of Processing On Heavy Metals Concentrations and Health Risk Assessment in African Palm Weevil (Rhynchophorus Phoenicis) Larvae
Received Date: Dec 06, 2019 / Accepted Date: Dec 12, 2019 / Published Date: Jan 06, 2020
Copyright: ©Copyright: ©2020 Markmanuel DP, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
This study investigated the concentrations of the heavy metals, Fe, Zn, Cu, Mn and Co in a raw and processed (boiled and fried) Rhynchophorus phoenicis larvae. The concentrations of the metals were measured using Thermal-Elemental Atomic Absorption Spectrophotometer (FAAS) S4-710-a6 model. The concentrations (mg/kg dry weight bases) of Fe, Zn, Cu, Mn and Co in raw, boiled and fried samples were; raw (10.23+0.08), (15.06+0.09), (5.19+0.01), (2.75+0.05) (1.75+0.12), boiled (7.24+0.19), (9.50+0.07), (4.11+0.09), (1.85+0.07), (2.00+0.00), and fried (6.25+0.06) (5.36+0.11), (3.31+0.04), (1.47+0.09), (2.88+0.07); respectively. The results showed a decreased order of Zn>Fe>Cu>Mn>Co in the raw sample, while the boiled and fried samples were in the order of Zn>Fe>Cu>Co>Mn. Generally, the observed trends were that the processing (boiling and frying) reduces the concentration of the metals except for Co where the highest concentration was found in the fried sample. The results of the non-carcinogenetic risk assessment were less than 1, which signifies no health concern at the moment. However, considering the toxicity and bioaccumulative nature of these metals, the frying practice is advocated, since it caused a significant decreased in most of the metals studied.
Keywords
Processing, Heavy Metal, Health Risk, Rhynchophorus Phoenicis Larvae
Introduction
Heavy metals are metals and metalloids with relatively high densities greater than 5g/cm3 [1]. These include lead, mercury, nickel, manganese, cobalt, arsenic, cadmium, iron, copper, zinc, chromium etc. These toxic chemicals are released into the environment indiscriminately, either from point sources such as industrial and municipal discharges as well as non-point sources like agricultural runoff, weathering of rocks, volcanic activity, and atmospheric deposition. Once released into the ecosystem, they persists, bioaccumulate and biomagnify within the food chain because of their non-biodegradability properties [2]. The heavy metals investigated in this study are; Fe, Zu, Cu, Mn and Co. Available literatures reveal that these metals are nutritionally essential to maintain the metabolism of the human body and other biological organisms. But, higher concentrations causes toxic effects such as reduced growth and development, major organs disorder (liver, lung, kidney, etc) and nervous system damage, and in extreme cases death [3,4]. While some heavy metals such as Pb, Hg, As, have no known biological roles or beneficial effects in organisms, rather, their bioaccumulation over time causes health effects [5].
The consciousness and interest about the protection of the masses from unavoidable pollutants, exposure to heavy metals and other contaminants, and their toxic effects on human health had increased the strategies and methodologies for detecting their presence in the environment and edible biota and how their impact can be limited [6-10]. However, these studies fail to consider the processed forms of these edible biota which human can be exposed via direct consumption. Therefore, this study is very significant because it has unveiled the suitable method of processing Rhynchophorus phoenicis larvae in order to avoid health risk in future.
Rhynchophorus phoenicis commonly known as African palm weevil is a species of beetles that belong to the family of curculionide. The larvae of this beetle is cherished as food in many African communities, Nigeria in particular [11]. They contain high quality proteins, minerals and vitamins, low saturated fats, and are cheap sources of animal protein to many household in rural communities [12]. In Nigeria’s Niger Delta region States such as Bayelsa, Rivers, Delta, Edo and Akwa Ibom, these edible larvae can be seen hawked by vendors along expressways, markets and streets [13]. Despite the importance associated with these edible insects, they may contain heavy metals via bioaccumulation and biomagnification due to the habitat (trunk of palm trees), and this may pose health risks to consumers [14]. The processed methods such as boiling and frying are the common practices among local consumers. They are either prepared separately or with particular delicacies in place of meat or fish, and these common processing methods may affect the concentrations of the metals [12,13]. Therefore, the aim of this study was to investigate the effects of processing on the concentrations of some selected heavy metals such as Iron, Zinc, Copper, Manganese and Cobalt in Rhynchophorus phoenicis larvae.
Materials and Methods
Study Area
Amassoma is one of the communities in Southern Ijaw Local Government Area of Bayelsa State, Nigeria (Niger Delta Region). It is located at Bayelsa Central geo-political zone and it’s approximately 30km away from the state capital, Yenagoa. Amassoma is situated at 4.97° North latitude, 6.11° East longitude, and 79 metres elevation above the sea level. Two third of the land is surrounded by water. Amassoma is the host community of the Niger Delta University. The major occupations of the people includes; farming, fishing, logging, local gin brewing and palm wine tapping [15].
Sample Collection, preparation and Heavy Metal Analysis
African palm weevil (Rhynchophorus phoenicis) larvae were bought from local farmers in Adule-Ama clan of Amassoma. These larvae were handpicked by the farmers from a fallen rotten palm tree. They were put into a plastic container and convey to the central research laboratory at the Niger Delta University, Wilberforce Island, Bayelsa State, Nigeria, and kept in the refrigerator.
Raw samples were washed properly with clean water and rinsed with distilled water. Then it was separated into three different containers with ten (10) Rhynchophorus phoenicis larvae in each container labeled raw, boiled and fried respectively. The sample labeled boiled was put into aluminum pot containing 200ml of distilled water. Thereafter, 5 g of table salt (iodized NaCl), 5 g of pepper and 5 g of maggi was added to a desired taste and the mixture was allowed to boil for 30mins. The labelled fried sample was then put into a frying pan containing 100ml of water and it was left to boil to dryness, and the oil of the larvae was used to stir fry it. All samples were then placed in the oven at a temperature of 105°C to a constant weight ground and sieved to obtain uniform particle size.
Approximately 1g of each sample (raw, boiled and fried) was digested with 15ml of Agua regia solution containing 3:1 (v/v) of Conc. HCl/HNO3, and 5ml perchloric acid (HClO4) was also added. The mixtures were place on a hot plate and heated gently in a fume cupboard until the samples completely dissolved. The digests were allow to cool and 5ml of distilled water was added, swirled and filtered into 100ml volumetric flask, and distilled water was added to make up the mark. Heavy metals (Fe, Zn, Cu, Mn and Co) concentrations were analyzed using thermo-Elemental Atomic Absorption Spectrophotometer (FAAS-S4-71096 model). All samples were analysed in triplicate.
Health Risk Assessment Models
The human exposure pathway of heavy metals through the consumption of Rhynchophorus phoenicis larvae were evaluated using United State Environmental Protection Agency (USEPA) model [16-18]. These include the Estimated Daily (EDI) and Weekly Intake (EWI), Target Hazard Quotient (THQ), Hazard Index (HI).
Estimated Daily (EDI) and Weekly Intake (EWI)
The EDI and EWI are the maximum amount of a contaminant or toxicant in which a person can be exposed to per day/week over a lifetime without any unacceptable risk of health effects, and is expressed below:
(1)
Where; EDI/EWI are the daily/weekly intake of the metals, MIR is the mass of the Rhynchophorus phoenicis larvae ingested per day [18]. CMR is the concentrations of the metal in the larvae, BWa is the body weight (60 kg for adult). The per capital consumption of fish and meat in Nigeria annually, is averaged 9.0 kg which is equivalent to 0.025 kg per day [19].
Target Hazard Quotient (THQ) and Hazard Index (HI)
The target hazard quotient (THQ) is the ratio between human exposure to a toxicant or pollutant and the oral reference dose (RfD). The THQ is used to express the risk of non-carcinogenic effects, and is expressed as follows:
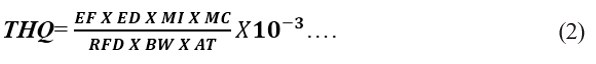
Where; EF exposure frequency (365 days/year); ED exposure duration (51.86year) which corresponds to the average life expectancy of a Nigerian MC MI and BW are expressed in equation (1), AT is the average exposure time for non-carcinogens (365/days/year X ED), and RfD is the oral reference dose (this is an estimation of a daily human exposure to pollutant or toxicant, including sensitive sub group that are likely to be without an appreciable risk of deleterious effect during life time) [20]. The RfD (mg/kg/day/week) of the heavy metals used in this study were; Fe (0.7), Zn (0.3), Cu (0.04) Mn (0.14), Co (0.043), and 10-3 is the unit conversion factor [7-10,20].
Hazard Index (HI) is the arithmetic sum of the individual THQ of the metals in Rhynchophorus phoenicis larvae, which can be expressed mathematically as follows:
HI = ΣTHQi…… (3)
Where; i is distinct heavy metals tested. Therefore, in this study
HI= THQFe + THQZn + THQCu + THQMn + HOCo.
The acceptable risk distribution for non-carcinogens effect is 1 or equal to the RfD. Ratio less than 1, signifies non-obvious risk (i.e HI is either < 1), where HI > 1 indicates reason for health concern [16-18].
Results and Discussion
The concentrations of the heavy metals (Fe, Zn, Cu, Mn and Co) in raw, boiled and fried Rhynchophorus phoenicis larvae and standard permissible limits of FAO/WHO and IAEA are presented in Table 1, and their comparison are shown in figure 1 below:
Table 1: The Mean concentrations (Mg/kg day weight of Fe, Zn, Cu, Mn and Co in raw, boiled and from sample of Rhynchophorus phoenicis larvae with permissible limits of FAO/WHO and IAEA
|
Samples |
Statistics |
Heavy Metals (Mg/kg) |
||||
|
Fe |
Zn |
Cu |
Mn |
Co |
||
|
Raw |
Range |
10.31-10.15 |
15.16-15.00 |
5.9-5.21 |
2.69-2.81 |
1.61-1.83 |
|
Mean + Std |
10.23+0.08 |
15.06 + 0.09 |
5.19+0.01 |
2.75+0.05 |
1.75+0.12 |
|
|
Std. error |
0.05 |
0.05 |
0.01 |
0.03 |
0.07 |
|
|
Boiled |
Range |
7.04-7.40 |
9.46-9.53 |
4.05-4.21 |
1.79-1.93 |
2.00-2.01 |
|
Mean + Std |
7.24+ 0.19 |
9.50 + 0.07 |
4.11+0.09 |
1.85+0.07 |
2.00+0.00 |
|
|
Std. error |
0.11 |
0.02 |
0.05 |
0.04 |
0.00 |
|
|
Fried |
Range |
6.19-6.31 |
5.26-5.48 |
3.29-3.35 |
1.38-1.57 |
2.82-2.96 |
|
Mean + Std |
6.25+0.06 |
5.36+011 |
3.31+0.04 |
1.47+0.09 |
2.88+0.07 |
|
|
Std. error |
0.03 |
0.06 |
0.02 |
0.06 |
0.04 |
|
|
WHO/FAO (21,22,23) 1985/2003 |
2.00 |
50.00 |
20.00 |
0.40 |
0.3 |
|
|
LAEA(24) |
146.00 |
67.10 |
3.28 |
3.52 |
0.023 |
|
WHO: World Health Organization, FAO: Food and Agriculture Organization
IAEA: International Atomic Energy Agency
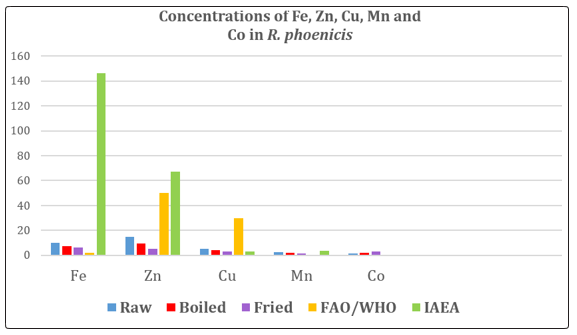
Figure 1: Concentrations (mg/kg) of Fe, Zn, Cu, Mn and Co in raw, boiled and fried samples of R. phoenicis in comparison with permissible limits of FAO/WHO and IAEA.
As indicated in table 1, the concentrations of the metals; Fe, Zn, Cu, Mn and Co in R. phoenicis larvae for the raw sample were: 10.23 + 0.08 mg/kg, 15.06 + 0.09 mg/kg, 5.19 + 0.01 mg/kg, 2.75 + 0.05 mg/kg and 1.75 + 0.12 mg/kg respectively. Thus, the mean values for the metals of the raw are in the decreasing order of Zn> Fe> Cu> Mn>Co. The mean and standard deviation (mean + std) of the boiled sample for Fe, Zn, Cu, Mn and Co were: 7.24 + 0.19 mg/kg, 9.50 + 0.07 mg/kg, 4.11 + 0.09 mg/kg, 1.85 + 0.07, and 2.00 + 000, and the fried sample were 6.25 + 0.00 6 mg/kg, 5.36 + 0.11 mg/kg, 3.31 + 0.04 mg/kg, 1.47 + 0.09 mg/kg, and 2.88 + 0.07mg/kg. Thus the mean values of the metals are in the order of Fe>Zn>Cu>Co>Mn respectively.
The results of this study shows that Rhynchophorus phoenicis larvae accumulated more of Zn Fe and Cu compared to Mn and Co with a significant difference (P<0.05). The observed difference in the bioaccumulation of Zn, Fe and Cu can be attributed to the bioavalability as well as the possible key roles they play as essential elements in biological organism. A similar observation was reported by Markmanuel and Horsfall, Osakwe et al, and Ijeomah et al, [8,9,21]. Their study revealed that the non-vertebrates fauna species have strong affinity for Fe, Zn and Cu because, they are part of important physiological components in enzymes and proteins that help in cells replacement and activation of low immune systems.
The effects of processing was also investigated in this study. The results shows that, boiling and frying which are the most common cooking methods practiced by many consumers and vendors of Rhynchophorus phoenicis larvae reduces the concentrations of the metals, Fe, Zn, Cu, Mn except for Co that the concentration increase with no significant difference (P>0.05). Thus for Fe, Zn, Cu and Mn, the observed decrease were in the order of raw>boiled>fried sample, while, Co concentration was in the order of fried>boiled>raw. Okareh and Funmi, also reported a decrease in heavy metals concentration with respect to processing [22]. Their study revealed that the lost of metal contents may be attributed to the ability of the fish flesh to act as a permeable membrane most especially under the influence of heat. This permeability may result in appreciable movement of fish water and oil with heavy metals, away from the fresh layer into the surrounding cooking medium. Also, variation between metal concentrations of raw and cooked fish has been reported. For example, Ersoy, observed that microwave heating of African catfish caused a decrease in Cr content but has no effect on Pb concentration while, frying resulted in a decreased Zn concentration and elevated Cu concentration and baking caused a decrease in Pb concentration of sea bass while fry caused an increase in the concentration of As [23,24]. Francisca et al, also observed that common culinary practice such as boiling, frying and grilling increases concentrations of Cd, Cu, Mn, Ni, Hg, and decreases the concentrations of Fe, Cr and Pb in the fish species studied [25]. The possible explanation offered to the observed variation in metal concentrations in the studied fish samples were attributed to the size of the fish, and the inter play between the oil uptake and water loss during frying and grilling processes as well as metal evaporation during these processes.
The concentrations of all the metals, Fe, Zn, Cu, Mn and Co were also compared to standard permissible limits of FAO/WHO and IAEA. The results indicates that Zn and Cu values were all lower than the permissible limits of FAO/WHO, and Fe, Mn and Co where above the limits. While, the concentrations of Fe, Zn, Cu, and Mn were below permissible limits of IAEA except for Co. Considering the bioaccumulative nature of heavy metals and their toxic effects moderate intake of Rhynchophorus phoenicis larvae is recommended.
Estimation of Dietary Intake, THQ and HI
The human health risk assessment models which was proposed by USEPA for provisional daily/weekly intake target hazard quotient and hazard index (non-carcinogens) were employed for this study and the results obtained are presented in table 2 and 3 respectively [16-18].
Table 2: Estimated Dietary Intake (mg/kg-bw/day/week) for the injestion 0.025 mg/kg-bw of raw, boiled and fried Rhynchophorus phoenicis larvae
|
Heavy metal |
Sample |
|||||
|
Raw |
Boiled |
Fried |
||||
|
EDI |
EWI |
EDI |
EWI |
EDI |
EWI |
|
|
Fe |
0.004 |
0.028 |
0.003 |
0.021 |
0.002 |
0.014 |
|
Zn |
0.006 |
0.042 |
0.004 |
0.028 |
0.002 |
0.014 |
|
Cu |
0.002 |
0.014 |
0.001 |
0.007 |
0.001 |
0.007 |
|
Mn |
0.001 |
0.007 |
0.000 |
0.000 |
0.000 |
0.000 |
|
Co |
0.000 |
0.000 |
0.000 |
0.000 |
0.001 |
0.007 |
Table 3: Estimated THQ, HI and % HI of Fe, Zn, Cu, Mn and Co in the raw, boiled and fried samples of Rhynchophorus phoenicis larvae
|
|
Raw |
Boiled |
Fried |
|||
|
|
THQ |
% HI |
THQ |
% HI |
THQ |
% HI |
|
Fe |
6.09E-06 |
5.675 |
4.31E-06 |
4.97 |
3.72E-06 |
4.65 |
|
Zn |
2.09E 05 |
19.46 |
1.32E-05 |
15.21 |
7.44E-06 |
9.303 |
|
Cu |
5.41E-05 |
50.29 |
4.28E-05 |
49.35 |
3.45E-05 |
43.09 |
|
Mn |
8.18E-06 |
7.61 |
5.51E-06 |
6.35 |
4.35E-05 |
5.46 |
|
Co |
1.82E-05 |
16.97 |
2.09E-05 |
24.13 |
3.00E-05 |
37.49 |
|
Σ THQ=HI |
1.10E-04 |
|
8.70E-05 |
|
8.00E-05 |
|
The estimated daily intake (EDI) and estimated weekly intake (EWI) for Fe, Zn, Cu, Mn and Co in raw, boiled and fried ample were in the decreasing of Zn>Fe>Cu>Mn>Co for raw. These values were lower than the provisional daily and weekly intake, of these heavy metals set by WHO/FAO in food products [26-28].
The target hazard quotient (THQ) help to evaluate the non- carcinogenic effects pose by the metals to consumers of Rhynchophorus phoenicis larvae and the combined effect was express as HI. The interpretation of HI is binary, which implies that ΣTHQ = HI < 1 for safe limit while, when HI is greater than I, it signifies that the exposed population is not safe. However, the results obtained from this study indicates that, all HI values of the three samples raw, fried and boiled R. phoenicis larvae are below the safe limits of HI < I. Therefore, there is no health concern resulting from the consumption of this larvae at the moment. This study is in agreement with the finding of Francisca et al, but lower that the values reported by Kalogeropoulos et al., and Markmanuel and Hosfall in fish and shell fish species [9,10,25,29,30].
Conclusion
The effects of processing such as boiling and frying were investigated in this study. The results shows a reduction in all metals concentration except for Co where, an increased in concentration was observed. Generally the observed decrease in metal concentration was in the order of raw > boiled > fried for Fe, Zn, Cu and Mn. While Co concentration was in the order of fried > boiled > raw with no significant difference (P>0.05.) The values of Zn and Co were lower than the permissible limits of WHO/FAO while, Fe, Co and Mn were higher than the limits. Also the concentration of all the metals were lower than the permissible limits of IAEA except for Co. The daily/weekly intake were all within the safe limits, furthermore, the results of the THQ and HI values are less than 1, which indicates no health concern at the moment.
Acknowledgement
The authors are grateful to the Management of the Central Research Laboratory, Niger Delta University, for allowing us to use their reagents and facilities for the sample preparation.
References
- Alloway BJ, Ayres DC (1997) Chemical Principles of Environmental Pollution, (2nd ED). UK: Blackie Academic and Professions.
- Bhatacharya AK, Mandal SN, Das SK (2008) Heavy Metals Accumulation in Water Sediment and Tissues of Different Edible Fishes in Upper Stretch of Gigantic Water Bengel. Trends in Applied Sciences Research 3: 25-29.
- Garcia-Rico L, Leyva-Perez, Jara-Marini ME (2007) Content and Daily Intake of Copper, Zinc, Lead, Cadmium and Mercury from Dietary Supplements in Mexico. Food Chem Toxicol 45: 1599-1605.
- Ozen O, Ulusoy S, Erkan N (2010) Study on the Behavior of the Trace Metal and Micro Minerals in Mytiks gadloprovincialis as an indicator by bio indicator species; The Case of Mamara Sea. Turkey, J. Verbr leb ensm 5: 407-412.
- Cutis DE (2006) Other Trace Elements: Sources, Modern Nutrition in Health and Diseases. 10th Ed. Lippincott Williams and Wilkins, Philadelphia.
- Akoto O, Bismark F, Darko G, Adei E (2008)Concentrations and Health Risk Assessments of Heavy Metals in Fish from the Tosu Lagoon. Int. J. Environ. Res 8: 403-410.
- Kumar B, MukherjeDP (2011) Assessment of Human Health Risk for Arsenic, Copper, Nickel, Mercury and Zinc in Fish Collected from Tropical West Lands in India. Adv. Life Sci Technol 2: 13-24.
- Osakwe, JO, Adowei P, Horsfall M Jr (2014) Metal Body Burden and Evaluation of Human Health Risks in Africa catfish (Clarias Gariepinus) from Imo River, Nigeria. Acta chim pharm indica 4.
- Markmanuel DP, Horsfall Jnr (2015) Assessment of Non- Carcinogenic Human Health Risk of Some Heavy Metals in Land Snails Commonly Consumed in Bayelsa State, Nigeria. Research Journal of Chemical Sciences 5: 1-10.
- Markmanuel DP, Horsfall Jnr (2016) Evaluation of Carcinogenic and Non-Carcinogenic Risk of Cadmium and Nickel in Land Snails (A. achatina and L. flammea) and Marine Snails (P. auria and T. fuscatus) commonly consumed in Nigeria. Acta chim pharm indica 6: 123-134.
- Okaraonye CC, Ikewuchi (2008) Rhynchorus phoenics Larvae: Nutritional Value and Health Implications. Journal of Insect Science 8: 1221-1225.
- Babajide OE, Gloria NE, Ochuko LE (2005) Chemical Evaluation of African Palm Weevil. Rhychophorus phoenicis, larvae as Food Source. Journal of Insect Science 4: 192-199.
- Ekrakene T, Igeleke CL (2009) Microbial Isolates from the Roasted Larvae of the Palm Weevil (Rynchophorus phoenicis) from Edo and Delta States of Nigeria. A M J Biol Applied Sci 1: 763-768.
- Banjo AD, Aina SA, Salau AR(2013) Shelf Life and Heavy Study of two Common Edible Insects in Ijebu Division, South West, Nigeria. Journal of Biology and life Science 4: 55-64.
- Ogamba EN, Izah SC, Oribu Tr (2015)Water Quality and Proximate Analysis of Eichnornia crassipes from River Nun, Amassoma Axis, Nigeria. Research Journal of Phytomedicine I: 43-48.
- USEPA (1989) Risk Assessment: Guidance for Superfund. In: Human Evaluation Manuel (Park A). Interim Final, Vol. 1. Office of Emergency and Remedial Response. US Environmental Protection Agency, Washington D.C.
- USEPA (2001) Risk Assessment Guidance for Superfund: Vol. III Part A. Process for Conducting Probalistic Risk Assessment EPA 540-R-02-002 OSWER 92857-45 PB20021963302.
- US Environmental Protection Agency (USEPA) (2005) Guidelines for Carcinogen Risk Assessment. Risk Assessment Forum, Washington. DC EPA/630/P-03/
- FAO (2005) Current Worldwide Annual Meat Consumption Per Capita, Livestock and Fish Primary Equivalent. Food and Agriculture Organization of the United Nations. Retrieved from https://en.wikipedia.org/w/indea.php7 title- list of countries by meat consumption and Oldid=902683299.
- Naughtan DP, Petroczi A (2008) Heavy Metals Ions in Wines: Meta- Analysis of Target Hazard Quotient Reveals Health Risk. Chem Cont J 2: 22.
- Wyse EJ, Azemard S, Mora SJ (2003) Report on the World-Wide Intercomparison of Exercise for the Determination of Trace Elements and Methy mercury in fish Homogenate, IAEA-407 IAEA> AL444 (IAEA Monaco PP 1-4).
- Ijeomah HM, Edet DI, Oruh, E-K, Ijeomah AU (2015) Assessment of Heavy Metals in Tissue of Selected Non- vertebrate Wildlife Species in Oil Polluted sited of Delta State, Nigeria. Agricultural and Biological Journal of North America 6: 63-75.
- Ersoy B, Yanar Y, Kucukgulmez A, Celik M (2006) Effects of Four Cooking Methods on Heavy Metal Concentration of Sea Bass Fillets (Dicentrarchus Labra finne, 1785). Food Chem 19: 748-751.
- Ersoy B (2011) Effect of Cooking Methods on the Heavy Metal Concentrations of the Africa Catfish (Clarias gariepinus). J. Food Biochem 35: 351-356.
- Francisca IB, Fehinlola CO, Chukwuyindu MAI, Vincent NO (2014) Effects of Processing on the Pronciamate and Metal Contents in Fish Species from Nigerian Coastal Waters. Food Science and Nutrition 2: 272-281.
- FAO/WHO (1989) National Research Council Recommended Dietary Allowances (10th ed) National Academy Press, Washington, D.C. USA.
- World Health Organization (WHO) 1983. Toxicological Evaluation of certain food Additives, Copper, Zinc. WHO Food Additives.
- WHO, Joint FAO/WHO (1999) Expert Committee on Food Additive. In summary and conclusion, 53rd Meeting, Rome 1-10.
- Kalogeropoulos N, Karavoltsos S, Sakellari A, Avramidou S, Dassenakis N, et al. (2012). Heavy metals in raw, fried and grilled mediteranean finfish and shellfish. Food chem Toxicol 50: 3702-3708.
- Okareh OT, Akande F (2015) Lead and Cadium levels of African Catfish (Clarias gariepinus) and the Effect of Cooking Methods on their concentrations. British Journal of Applied Science and Technology II: 1-12.


