Research Article - (2025) Volume 3, Issue 2
The Effect of Micronutrients on PSA Dynamics in Prostatic Disease: A Controlled Pilot Study
Received Date: Feb 07, 2025 / Accepted Date: Feb 27, 2025 / Published Date: Mar 10, 2025
Copyright: ©Â©2025 Claus Riedl. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Riedl, C. (2025). The Effect of Micronutrients on PSA Dynamics in Prostatic Disease: A Controlled Pilot Study. Int Internal Med J, 3(2), 01-07.
Abstract
Background: The present study investigated the effects of a micronutrient combination with proven impact on prostate cell metabolism and growth with regard to PSA dynamics.
Methods: In an open single-arm multicenter study, 46 men with different baseline situations were recruited: 16 men with constantly elevated PSA values and negative prostate biopsy (group A), and 30 men with low risk prostate cancer on Active Surveillance (Group B).
Results: In group A, 12 out of 16 men (75%) showed a decrease of PSA after 6 months from an average 9.26 to an average 8.84 ng/ml (4.5%), whereas in group B, PSA level decreased in 16, increased in 12 men, and remained unchanged in 1 man. The average PSA in group B dropped from 7.47 to 6.82 ng/ml (8.6%).Both PSA decreases were not statistically significant.
Conclusion: The results of the present study are consistent with previous findings on the positive effects of individual micronutrients on prostate health. Longer administration of micronutrients or different dosage or combinations may further improve PSA outcomes.
Keywords
Micronutrients, Prostate Cancer, PSA Dynamics
Introduction
The incidence of prostatic diseases is increasing with age, and, consequently, the majority of men > 60 years experience some kind of prostate related symptoms. While on one side prostate cancer, the most prevalent malignant disease in men, is a potentially life- threatening disease, benign prostatic enlargement (BPE), also known as prostatic hyperplasia (BPH), may cause bothersome changes in micturition.
There is a wide range of surgical and medical therapeutic options for all kinds of prostatic diseases. However, an increasing number of men tend to alternative or phytotherapeutic therapies as a first choice, even though there is scarce scientific evidence to support this [1].
For detection of prostate cancer and discrimination from benign disease, prostate-specific antigen (PSA) is the present tumor marker of choice [2]. Despite its limited specificity, it can detect most prostate cancers at curable stages. In cases when tumors are detected at very early stages (low volume/low risk tumors), active treatment may be postponed and active surveillance initiated [3]. PSA monitoring is the base in this situation, supported by magnetic resonance imaging or re-biopsy as needed.
The limited specificity of PSA means that men may have constantly elevated or rising PSA levels without cancer being detected in biopsies. This places a psychological burden on the men affected, and no standard treatment can be offered in this situation.
Common phytotherapeutics for prostatic disease (Serenoa repens, Saw palmetto) may have positive effects on micturition, but not on PSA levels or the progression of prostate cancer [1]. In contrast, various herbal compounds have shown effects on prostate cancer transformation/growth and PSA-levels in in-vitro studies and in published trials [4,5].
Decades ago, epidemiologic data showed that the incidence of prostate cancer in East Asian men was significantly lower than in Western countries [6]. This was attributed to the regular consumption of soy products in Asian countries, since these contain a variety of isoflavones [7]. Isoflavones are phytoestrogens with hormonal activity and found in relevant concentrations in soy beans, red clover and kudzu roots. The best investigated isoflavones are daidzein, genistein, formononetin and biochanin. They have been shown to block cell proliferation, dedifferentiation and angiogenesis in prostate cancer cell lines, as well as in rat studies [8-10].
In a clinical pilot study, PSA-reduction of 33% was observed in men without proven prostate cancer after one year administration of a standardized isoflavone preparation. No side effects, particularly with regard to sexuality, were observed in this study [11].
Similar in vitro antiproliferative effects on prostate cancer cells have been demonstrated for catechines (epigallocatechingallat – EGCG, from green tea), phytosterole (ß-sitosterol from pine nuts), tannins (elagitannine/gallotannine from pomegranate), and lycopin (from tomatoes), as well as for vitamines D3, B6, and zinc [12-20].
This led to the hypothesis that a combination of all these compounds could have a beneficial effect on prostate cells growth and, thus, on the PSA value.
Materials and Methods
In an open single-arm multicenter study (office-based at 7 centers), 2 patient groups of men > 45 years were recruited: Group A: men with constantly elevated PSA values and negative prostate biopsy Group B: men with low risk/low volume prostate cancer on Active Surveillance [21] All men gave their written consent to participate in the study and to take 2 capsules of the study compound (table 1, source Lenus Pharma GesmbH) per day for 6 months.
Inclusion Criteria:
• For patients in group A, a minimum of 3 elevated PSA values (> the age-adjusted normal range) within the last 12 months was required for study inclusion.
• For patients in group B, bioptic evidence of a low risk-low/ volume prostate cancer (≤ 2 positive biopsies with < 50% tumor volume, Gleason grade 3+3 or 3+4, PSA < 10 ng/ ml at the time of biopsy [21]) was a prerequisite for study inclusion. These men had to opt for active surveillance after being informed of all possible treatment options for their prostate cancer, and at least three elevated PSA values (> the age-adjusted normal range) within the last 12 months were required.
Exclusion Criteria:
• Long-term anti-inflammatory medication.
• Antihormonal medication.
• Active urinary tract infection.
• Cycling (sport).
• Participation in another study.
Before initiation of study medication, laboratory values (PSA, free PSA, testosterone, liver transaminases), prostatic volume (by transabdominal ultrasound) and IPSS (International Prostate- Symptom-Score) were determined. Histologic reports of prostate biopsies and medication lists were recorded.
The same assessment was repeated after 6 months of medication. In addition, adverse effects were recorded.
After 3 months, an interim telephone interview was conducted to ask patients about compliance (capsule intake) and possible adverse effects.
Routine active monitoring was not part of the study protocol and performed as to discretion of the responsible urologist.
Primary study-endpoint was the change in PSA from baseline to end of the study after 6 months of study compound intake. Secondary endpoints were changes in any of the additional parameters recorded.
Statistical evaluation of PSA changes from baseline to end of study was performed with Student´s t-Test. A study population of 45 people was calculated for a power of 95%. Significance was assumed for p-values < 0.05. Statistical evaluation was performed with assistance of SPSS Statistics Version 27.
The study protocol was reviewed and approved by the local ethics committee (Ethics Committee for the Federal Province of Lower Austria, St. Pölten, Austria)
Results
A total of 46 men were included in the study, 16 in group A and 30 in group B. Of the patients under Active Surveillance, 27 were diagnosed with low volume prostate cancer Gleason 3+3, and 3 with Gleason 3+4.
One group B patient discontinued the medication after 2 months due to an adverse event (headache) that was most likely not related to the study compound. Thus, a total of 45 men were evaluated.
In group A, 12 out of 16 men (75%) showed a decrease of PSA after 6 months from an average 9.26 to an average 8.84 ng/ml (4.5%), which was not statistically significant (p=0.095) (Fig:1).
In group B (Active Surveillance), PSA level decreased in 16 and increased in 12 men, and remained unchanged in 1 man. The average PSA dropped from 7.47 to 6.82 ng/ml (8.6%), which was not statistically significant (p=0.177) (Fig.2).
Changes of other parameters recorded at baseline and after 6 months are shown in tables 2 and 3 and did not reach significance at any point. Interestingly, average testosterone levels increased in both groups during the study period: from 3.98 to 4.4 ng/ ml in group A, and from 5.45 to 6.30ng/ml in group B. Liver transaminases showed a mild increase of average values in both groups: Glutamat-oxalacetat-transaminase (GOT) from 25.35 to 28.77 U/l and from 29.23 to 30.84 ng/ml; Glutamat-pyruvat- transaminase (GPT) from 26.28 to 28.70 U/l and from 35.46 to 38.53 U/l, whereas Gamma-glutamyl-transferase (gGT) values decreased from 43.85 to 40.17 U/l and from 54.64 to 42.23 U/l.
The IPSS decreased in both groups: from an average 7.88 points to 6.61 and from 9.84 to 9.30.
The sonographically assessed average prostate volumes remained roughly stable: 46.96 and 47.77cc for group 1 and 58.11 and 55cc for group 2.
Discussion
Prostatic diseases are a significant health problem for aging men. They occur in various forms, such as benign prostatic hyperplasia (BPE) and prostate cancer. These diseases not only affect quality of life, but may also lead to significant morbidity. Conventional therapeutic approaches often harbour unpleasant side effects and/or lack efficacy, thus, many men favour phytotherapeutic medications despite limited scientific evidence [22,23].
PSA serves as an important tumor marker in the detection and monitoring of prostate cancer, albeit with known limitations in terms of specificity [24]. Persistend elevated PSA values with no malignant findings in biopsy represent a clinical dilemma and psychological burden for those men affected [25]. Although conventional drug therapies can provide relief for BPE symptoms, the uncertainty from tumor marker elevation persists.
Initiated from epidemiologic studies on populations with lower incidence of prostate cancer, the evidence on a link between dietary factors and prostate health is steadily growing. An antiproliferative effect on prostate cancer cells was first shown for isoflavones in vitro and in vivo, which to a great extent are found in soy products [8-10]. Other phytochemicals such as green tea catechins (epigallocatechin gallate - EGCG), phytosterols, tannins, lycopene, as well as certain vitamins and minerals, have also demonstrated promising anti-cancer properties in preclinical studies [12-20,26]. These findings were the base for the present controlled pilot study investigating the effects of a specific combination of micronutrients on PSA dynamics in prostate diseases. It was hypothesized that a synergistic effect of these bioactive compounds could modulate prostate cell metabolism and growth and consequently influence PSA levels.
In the present study, two patient groups were followed for 6 months (Group A with elevated PSA values and negative prostate biopsy, Group B with low risk/low volume prostate cancer on Active Surveillance), and an average reduction of PSA values < 10% was observed in both patient groups, which did not reach statistical significance. A PSA reduction of 33% had been described in a prior study with isoflavone administration alone [11]. These historic data have been used for calculation of the prospective study population in the present study, however, by this the total number of probands may have been too small to reach statistical significance. In addition, isoflavone treatment was continued for 1 year in the prior study, and no cancer patients had been included. Dosages in both studies were slightly different: 50mg isoflavone extract per day in the present study compared to 60mg per day in the former investigation. These different study settings may have contributed to the fact that the PSA reduction under the micronutrient combination was lower compared to isoflavone alone and did not reach statistical significance.
In total, the results showed a nuanced response to the intervention. While a significant decrease in PSA levels was not observed across the board, notable trends emerged. In group A, 75% of the study participants showed a decrease in PSA levels, as did 55% in group B. Since prostate cancer patients usually show an increase of PSA over time, this PSA reduction in group B is most remarkable (and may be an argument for stabilization of disease with micronutrients in active surveillance cases). As PSA also constantly increases with age, which is reflected in age-dependent normal ranges, the PSA decrease is also unexpected in group A and may be interpreted as micronutrient effect [27]. Thus, the results of the present study are consistent with previous findings on the positive effects of individual micronutrients on prostate health.
The observed increase in testosterone levels raises interesting questions about underlying mechanisms. While the exact mechanisms of action are still unclear, it may be assumed that the bioactive compounds of the micronutrients can influence endocrine function, possibly by interaction with steroidogenic enzymes or hormone receptors. Isoflavones posess estrogenic and anti-estrogenic effects that are able to modulate testosterone synthesis and metabolism [28]. However, a metaanalysis did not find a significant change of testosterone under isoflavone intake [29]. Thus, any other compound of the micronutrient combination may have been responsible for the testosterone increase observed. Importantly, since PSA levels are testosterone dependent, any increase of testosterone should also be reflected by a PSA increase, which was not observed in the study [30]. Possibly, without this testosterone increase, the decrease of PSA would have been more pronounced in the present study.
Additional analyses revealed non-significant changes in other parameters that indicate possible metabolic and physiological effects of the intervention. Liver transaminases showed diverging patterns suggesting metabolic modulation without a significant increase or decrease. This is important, since a recent publication reports that almost 5 percent of the US population regularly take nutraceutic compounds with potential hepatotoxicity [31].
IPSS scores showed improvements in urinary symptoms, especially in group A. Prostate volume remained relatively stable throughout the study, suggesting that there were no significant anatomical changes attributable to the study product.
Conclusions
While the results of the present study are preliminary, they provide valuable insight into the complex interplay between micronutrients and prostate health. New compounds with proven in-vitro activity in prostate cells may be added to the present formulation to enhance efficacy. In addition, elucidating the underlying mechanisms behind the observed effects is critical to refine therapeutic strategies and optimize patient outcomes in prostate disease.
In summary, the study contributes to the evolving landscape of integrative approaches in the treatment of prostate disease and highlights the potential of micronutrient combinations as complementary therapies. The short duration of the study may have prevented significant reductions in PSA levels, as changes in prostate health markers often need longer periods to become apparent. Longer-term studies are needed to fully evaluate the effectiveness of micronutrient interventions targeting prostate health.
Supplementary Materials
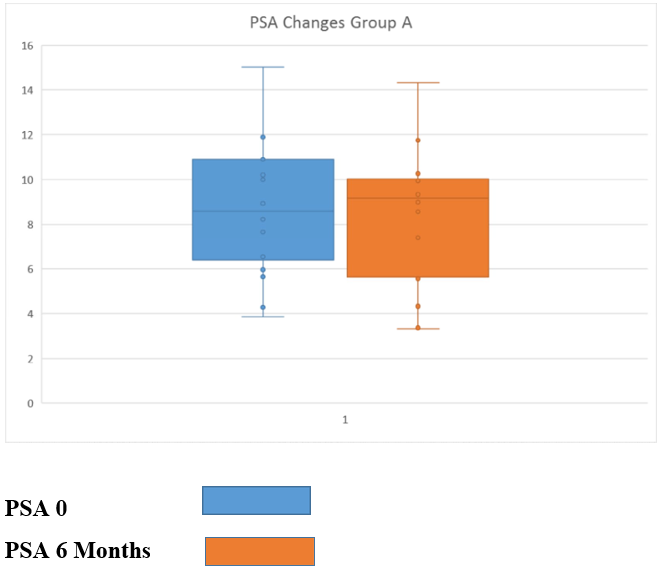
Figure 1: PSA Changes After 6 Months of Nutraceutical Combination Intake in Group A
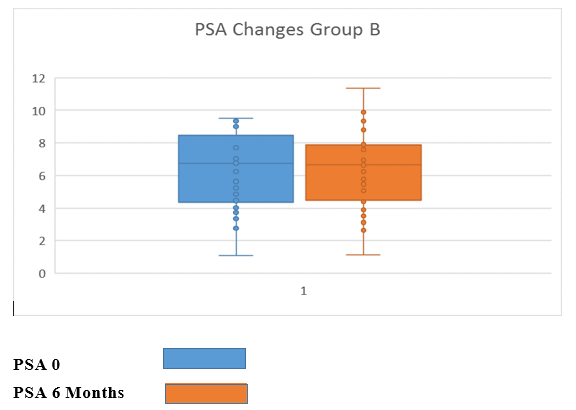
Figure 2: PSA Changes After 6 Months of Nutraceutical Combination Intake in Group B
|
Ellagic acid (pomegranate) |
46mg |
|
Catechins (green tea) |
73mg |
|
Phytosteroles (pine nuts) |
59 mg |
|
Isoflavones |
25mg |
|
Microencapsulated Lycopene |
5mg |
|
Vitamine D3 |
13,6µg |
|
Vitamin B6 |
5mg |
|
zinc |
10mg |
Table 1: Composition of Study Compound
|
|
baseline |
6 months |
|
GOT (U/l) |
29,23 |
30,84 |
|
GPT (U/l) |
35,46 |
38,53 |
|
gGT (U/l) |
54,64 |
42,23 |
|
IPSS |
9,84 |
9,30 |
|
Testosterone (ng/ml) |
5,45 |
6,30 |
|
Prostate Volume (cc) |
58,11 |
55,00 |
Table 2: Changes of Average Values in Group A After 6 Months Micronutrients
|
|
baseline |
6 months |
|
GOT (U/l) |
25,35 |
28,77 |
|
GPT (U/l) |
26,28 |
28,70 |
|
gGT (U/l) |
43,85 |
40,17 |
|
IPSS |
7,88 |
6,611 |
|
Testosterone (ng/ml) |
3,97 |
4,40 |
|
Prostate Volume (cc) |
46,96 |
47,77 |
Table 3: Changes of Average Values in Group B After 6 Months Micronutrients
Acknowledgements
Paul Engelhardt, Mons Fischer, Norbert Häusler, Emile Valimberti, Martin Vorauer Michael Budinsky, and Claus Riedl performed the study in their urologist office and provided all data Martin Imhof reviewed the study protocol, assisted in data analysis and reviewed the final manuscript Marianne Imhof performed statistical data analysis
Author Contributions
Claus Riedl was responsible for the study protocol, data analysis and manuscript writing
Funding
Lenus Pharma Ges.m.b.H.provided all study material and support- ed statistical analysis
Institutional Review Board Statement
EK-Number: GS4-EK-4/731-2021 from Jan 27 2022 ETHIK- KOMMISSION FÜR DAS BUNDESLAND NIEDERÖSTER- REICH 3109 St. Pölten, Landhausplatz 1 , Austria
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study
Data Availability Statement
Informed consent was obtained from all subjects involved in the study
Conflicts of Interest
Claus Riedl declares no conflict of interest concerning the presented study
References
- Moyad, M. A. (2022). Nutraceuticals and Phytotherapy in Men's Health: Antioxidants, Pro-oxidants, and a Novel Opportunity for Lifestyle Changes. Urologic Clinics, 49(2), 239-248.
- Carlsson, S. V., & Vickers, A. J. (2020). Screening for prostate cancer. Medical Clinics, 104(6), 1051-1062.
- Walker, C. H., Marchetti, K. A., Singhal, U., & Morgan, T.M. (2022). Active surveillance for prostate cancer: selection criteria, guidelines, and outcomes. World journal of urology, 1-8.
- Di Napoli, R., Balzano, N., Mascolo, A., Cimmino, C., Vitiello, A., Zovi, A., ... & Boccellino, M. (2023). What is the role of nutraceutical products in cancer patients? A systematic review of randomized clinical trials. Nutrients, 15(14), 3249.
- Noh, S., Choi, E., Hwang, C. H., Jung, J. H., Kim, S. H., & Kim, B. (2019). Dietary compounds for targeting prostate cancer. Nutrients, 11(10), 2401.
- Kurahashi, N., Iwasaki, M., Inoue, M., Sasazuki, S., & Tsugane, S. (2008). Plasma isoflavones and subsequent risk of prostate cancer in a nested case-control study: the Japan Public Health Center. Journal of Clinical Oncology, 26(36), 5923-5929.
- Van der Eecken, H., Joniau, S., Berghen, C., Rans, K.,& De Meerleer, G. (2023). The use of soy isoflavones in the treatment of prostate cancer: a focus on the cellular effects. Nutrients, 15(23), 4856.
- Jarred, R.A., Keikha, M., Dowling, C., McPherson, S. J., Clare,A. M., Husband, A. J., ... & Risbridger, G. P. (2002). Induction of apoptosis in low to moderate-grade human prostate carcinoma by red clover-derived dietary isoflavones. Cancer Epidemiology Biomarkers & Prevention, 11(12), 1689-1696.
- Ye, Y., Hou, R., Chen, J., Mo, L., Zhang, J., Huang, Y., & Mo,Z. (2012). Formononetin-induced apoptosis of human prostate cancer cells through ERK1/2 mitogen-activated protein kinase inactivation. Hormone and Metabolic Research, 44(04), 263- 267.
- Hazafa, A., Rehman, K. U., Jahan, N., & Jabeen, Z. (2020). The role of polyphenol (flavonoids) compounds in the treatment of cancer cells. Nutrition and cancer, 72(3), 386-397.
- Engelhardt, P. F., & Riedl, C. R. (2008). Effects of one- year treatment with isoflavone extract from red clover on prostate, liver function, sexual function, and quality of life in men with elevated PSA levels and negative prostate biopsy findings. Urology, 71(2), 185-190.
- Connors, S. K., Chornokur, G., & Kumar, N. B. (2012). New insights into the mechanisms of green tea catechins in the chemoprevention of prostate cancer. Nutrition and cancer, 64(1), 4-22.
- Macoska, J. A. (2023). The use of beta-sitosterol for the treatment of prostate cancer and benign prostatic hyperplasia. American Journal of Clinical and Experimental Urology, 11(6), 467.
- Naikiâ?Ito, A., Chewonarin, T., Tang, M., Pitchakarn, P., Kuno, T., Ogawa, K., ... & Takahashi, S. (2015). Ellagic acid, a component of pomegranate fruit juice, suppresses androgenâ?dependent prostate carcinogenesis via induction of apoptosis. The Prostate, 75(2), 151-160.
- Holzapfel, N. P., Holzapfel, B. M., Champ, S., Feldthusen, J., Clements, J., & Hutmacher, D. W. (2013). The potential role of lycopene for the prevention and therapy of prostate cancer: from molecular mechanisms to clinical evidence. International journal of molecular sciences, 14(7), 14620-14646.
- Trump, D. L., & Aragon-Ching, J. B. (2018). Vitamin D in prostate cancer. Asian journal of andrology, 20(3), 244-252.
- Wagner D, Trudel D, Van der Kwast T, Nonn L, Giangreco AA, Li D, Dias A, Cardoza M, Laszlo S, Hersey K, Klotz L, Finelli A, Fleshner N, Vieth R. Randomized clinical trial of vitamin D3 doses on prostatic vitamin D metabolite levels and ki67 labeling in prostate cancer patients. J Clin Endocrinol Metab. 98(4):1498-507, 2013.
- Contestabile, R., di Salvo, M. L., Bunik, V., Tramonti, A., & Vernì, F. (2020). The multifaceted role of vitamin B6 in cancer: Drosophila as a model system to investigate DNA damage. Open Biology, 10(3), 200034.
- Liu, S., Chen, J., Wang, Y., & Xu, Y. (2023). Effect of dietary antioxidants on the risk of prostate cancer. Systematic review and network meta-analysis. Nutrición Hospitalaria, 40(3).
- Sauer, A. K., Vela, H., & Grabrucker, A. M. (2020). Zinc deficiency in men over 50 and its implications in prostatedisorders. Frontiers in Oncology, 10, 553161.
- Cornford P, van den Bergh RCN, Briers E: EAU-EANM- ESTRO-ESUR-ISUP-SIOG Guidelines on Prostate Cancer-2024 Update. Eur Urol. 86(2): 148-163, 2024.
- Antoniou, V., Gauhar, V., Modi, S., & Somani, B. K. (2023). Role of Phytotherapy in the Management of BPH: A Summary of the Literature. Journal of Clinical Medicine, 12(5), 1899.
- Bhatt, N. R., Davis, N. F., Witjes, W. P., Bjartell, A., Caris, C., Patel, A., ... & Tubaro, A. (2021). Contemporary use of phytotherapy in patients with lower urinary tract symptoms due to benign prostatic hyperplasia: results from the EVOLUTION European registry. World Journal of Urology, 39, 2661-2667.
- Caplan, A., & Kratz, A. (2002). Prostate-specific antigen and the early diagnosis of prostate cancer. Pathology Patterns Reviews, 117(suppl_1), S104-S108.
- Lofters, A., Juffs, H. G., Pond, G. R., & Tannock, I. F. (2002). “PSA-itis”: knowledge of serum prostate specific antigen and other causes of anxiety in men with metastatic prostate cancer. The Journal of urology, 168(6), 2516-2520.
- Chen, P., Zhang, W., Wang, X., Zhao, K., Negi, D. S., Zhuo, L.,... & Zhang, X. (2015). Lycopene and risk of prostate cancer: a systematic review and meta-analysis. Medicine, 94(33),e1260.
- Heidegger, I., Fritz, J., Klocker, H., Pichler, R., Bektic, J., & Horninger, W. (2015). Age-adjusted PSA levels in prostate cancer prediction: updated results of the tyrol prostate cancer early detection program. PloS one, 10(7), e0134134.
- Vitale, D. C., Piazza, C., Melilli, B., Drago, F., & Salomone,S. (2013). Isoflavones: estrogenic activity, biological effect and bioavailability. European journal of drug metabolism and pharmacokinetics, 38, 15-25.
- Reed, K. E., Camargo, J., Hamilton-Reeves, J., Kurzer, M., & Messina, M. (2021). Neither soy nor isoflavone intake affects male reproductive hormones: An expanded and updated meta- analysis of clinical studies. Reproductive Toxicology, 100, 60- 67.
- Kim, D. K., Noh, J. W., Chang, Y., Lee, H. Y., Park, J. J.,Ryu, S., & Kim, J. H. (2020). Association between prostateâ? specific antigen and serum testosterone: A systematic review and metaâ?analysis. Andrology, 8(5), 1194-1213.
- Likhitsup, A., Chen, V. L., & Fontana, R. J. (2024). Estimated exposure to 6 potentially hepatotoxic botanicals in US adults. JAMA network open, 7(8), e2425822-e2425822.




