Case Report - (2019) Volume 1, Issue 1
Tenofovir Induced Acute Kidney Injury and Severe Anemia: A Case Series of Four Patients
2Faculty of Medicine and Biomedical Sciences of YaoundeUniversity of Yaounde I, Cameroon
3Faculty of Medicine and Pharmaceutics Science- Douala, Cameroon
4Teaching Hospital of Yaounde, Cameroon
5General hospital of Yaounde, Cameroon
Received Date: Nov 29, 2019 / Accepted Date: Dec 09, 2019 / Published Date: Dec 20, 2019
Copyright: ©Fouda Menye Ebana Hermine Danielle, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
We report four cases of atypical acute kidney injury (AKI) associated with the used of Tenofovir (TDF). Elevated creatinine, normoglycemic glycosuria with unusual high proteinuria, high serum phosphate and severe anemia were observed. After withdrawal of TDF, glomerular filtration rate (GFR) and hemoglobin level increase in all the patients, but only one patient have a complete remission. Clinician should be aware of this atypical TDF induced AKI presentation with end stage Kidney Disease characteristics.
Introduction
Tenofovir Disoproxil Fumarate (TDF) is the first nucleotide reverse transcriptase inhibitor to be approved for the treatment of Human Immunodeficiency Virus (HIV) infection. TDF is one of the most prescribed antiretroviral drug around the World. In Cameroon, it is recommended as first line agent therapy in association with Lamuvidine and Efavirenz. TDF is generally considered safe [1-3]. Renal toxicity is the most severe adverse effect but it is uncommon with an incidence < 1% [2]. TDF nephrotoxicity is due to tubular lesion and usually manifest as proximal dysfunction, including Fanconi syndrome, isolated hypophosphatemia and decreased bone mineral density or distal dysfunction with nephrogenic diabetes insipidus. Anemia is also rarely described with TDF used and association between TDF nephrotoxicity and anemia is unusual. We reported 4 cases of atypical TDF nephrotoxicity associated with severe anemia and both resolved after discontinuation of TDF in the nephrology clinic of the General Hospital of Douala.
Cases series
We registered 4 cases (3 females) of atypical TDF induced AKI from 2014 to 2017. Baseline characteristics of patients are presented in table 1. Age varies between 32 to 67 years old. HIV was known for less than 2 years in 2 patients. Two patients were nonobservant to ARV treatment. Severe immunodepression (CD4< 200 cells/mm3) at ARV initiation was common and use of Metformin was noted in one patient. Malnutrition was not observed. ARV protocol was the same for the 4 patients: 3TC + TDF+ EFV (Lamuvidine + Tenofovir + Efavirenz). Nephrotoxicity appear during the first 3 months of TDF introduction in 3 patients. Uremic syndrome was the main complaint in 3 patients. Severe anemia with multiple transfusions was noted for the other one. Anuria or oliguria were not observed and one patient had hyposthenuric polyuria. Glycosuria with normal blood sugar, high serum phosphate and AKI stage 3 KDIGO were found in the four patients. One patient had hypocalcemia. Severe normochromic normocytic anemia was also usual and each patient received at least 4 units of blood. Hyponatremia was also common as well as massive proteinuria (> 2g/g). One patient had a concomitant Escherichia coli urinary tract infection. Normal kidney size was observed in the forth. No patient had formal indication of dialysis and none benefit of it. Medical treatment of AKI was done and TDF was discontinued. The evolution was marked by improvement in GFR with completed recovery in 1 patient and partial recovery in the 3 others (figure 1). Hemoglobin level also improved and all the patient were free of blood transfusion after 6 months (figure 2). Although serum phosphate decrease in the 4 patients, high serum phosphate persist in two patients with partial recovery.
Table 1: Baseline characteristics of patients
|
|
CASE 1 |
CASE 2 |
CASE 3 |
CASE 4 |
|
SOCIO-DEMOGRAPHIC DATA |
||||
|
Sex |
male |
female |
female |
female |
|
Age (years) |
67 |
45 |
32 |
47 |
|
Marital status |
married |
divorced |
single |
single |
|
Profession |
Retired |
None |
None |
Secretary |
|
HIV DATA |
|
|
|
|
|
HIV duration |
12 months |
15 months |
8 years |
7 years |
|
Antiretroviral history |
ARV start 3 months before (3TC+TDF+EFV) |
ARV start 7 months before (3TC+TDF+EFV) |
ARV was start 8 years before with unknown protocol. voluntary interruption > 6 years Reintroduction 3 months before (3TC+TDF+EFV) |
ART start seven years before with 3TC+AZT+NVP. Voluntary interruption of 4 years. Reintroduction 6 months before (3TC+TDF+EFV) |
|
Actual antiretroviral protocol |
3TC+ TDF+ EFV |
3TC+ TDF+ EFV |
3TC+ TDF+ EFV |
3TC+TDF+EFV |
|
COMORBIDITIES |
|
|
|
|
|
|
Type 2 diabetes for 25 years on glimepiride and Metformin |
none |
none |
none |
|
CLINICAL DATA |
Patient had normal creatinine (1,1mg/ dl) 1 month before the admission, since 4 weeks prior to admission; he noticed an increase in diuresis although his blood glucose was normal. He present to emergency with severe asthenia, loss of appetite and nausea, and severe hypoglycemia |
Patient had received multiple blood transfusion since she was put on ARV drug because of severe anemia, then she consulted to the hematology clinic where the workup showed high creatinine levels |
Patient had a normal creatinine (0,9mg/ dl) when ARV was reintroduced; 1 month after the reintroduction, she complained of asthenia, nausea, vomiting and insomnia; workup found elevation of creatinine. |
Patient present with abrupt uremic symptom. Workup shown severe anemia and mark alteration of kidney function. She was then referred to the nephrology unit for dialysis initiation |
|
Interval between TDF introduction and Renal failure |
3 months |
7 months |
3 months |
2 months |
|
Uremic symptoms |
present |
present |
present |
present |
|
Weight (kg) |
73.4 |
56.4 |
61.2 |
57.2 |
|
Blood Pression (mmHg) |
105/75 |
103/81 |
90/65 |
99/63 |
|
Initial urinary dipstick |
Glucose ++ Protein ++, density = 1010 |
Glucose ++, protein ++, density= 1010 |
Glucose++ Protein++ leucocyte ++ density 1010 |
Glucose ++ Protein ++++ Leucocyte + Density 1010 |
|
INITIAL WORK-UP |
|
|
|
|
|
Creatinine (mg/dl) |
17 |
13 |
20 |
23.25 |
|
Hemoglobin (g/dl) |
6 |
6.8 |
6 |
6.5 |
|
Mean corpuscular volume (fl) |
88 |
86 |
90 |
81 |
|
Serum Calcium* (mg/l) |
89 |
101.2 |
78 |
90 |
|
Serum Phosphate (mg/l) |
77.2 |
54.4 |
110 |
76.5 |
|
Albumin (g/l) |
22 |
35 |
38 |
24.3 |
|
Na/K/Cl (mmol/l) |
123/4.8/88.9 |
121/3.7/93 |
126/5.3/90 |
128.2/7/108 |
|
Proteinuria /creatinuria ratio (g/g) |
2.2 |
2.5 |
4.55 |
3.75 |
|
Kidney size |
normal |
normal |
normal |
normal |
|
Others |
|
|
E. coli in urinary culture |
|
Serum calcium corrected to albumin level
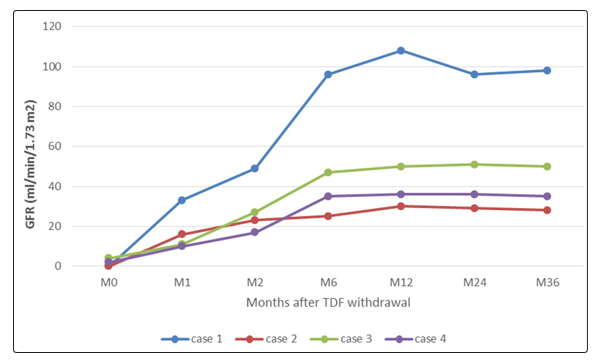
Figure 1: evolution of GFR after discontinuation of Tenofovir
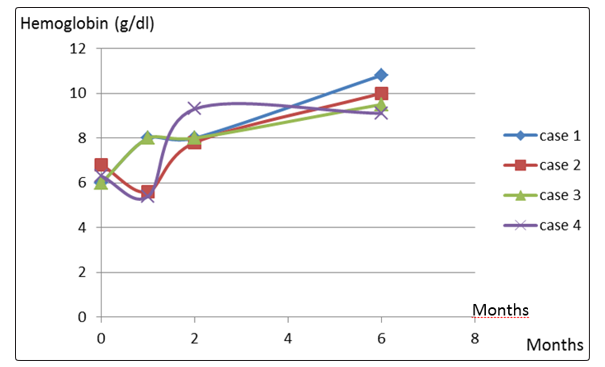
Figure 2: evolution of hemoglobin after discontinuation of Tenofovir
Discussion
AKI secondary to proximal tubular dysfunction is a well know complication of TDF used. It usually manifest as a complete or partial Fanconi syndrome which manifest as glycosuria with normal blood sugar, renal tubular acidosis, low serum phosphate, hypokalemia; hypouricemia, aminoaciduria and tubular proteinuria. TDF nephrotoxicity result from mitochondrial toxicity secondary to high intracellular TDF concentration. Indeed, drugs (eg probenecid) which inhibited the uptake of TDF at the basal membrane by OAT1 (organic acid transport 1) may prevent TDF nephrotoxicity by keeping proximal intracellular level of TDF low. By the contrary, drug (eg metformine or ritonavir) that inhibit the transport of TDF at the apical membrane by MRP2 or 4 (multidrug resistant protein 2 or 4) could increase the risk of TDF toxicity. Decrease in GFR also increase the risk of TDF nephrotoxicity by increase plasma level of TDF and then proximal tubular cell uptake of TDF through OAT1. Other situation associated with increased risk of TDF toxicity included older age, low body weight, low CD4 count, ABCC2 gene polymorphism (encoding for MRP2 transporter,) used of other nephrotoxic drug such as aminoglycoside, HCV coinfection [2,4-8]. The four cases have low CD4 count at TDF initiation, and one patient a concomitant used of Metformin. The delay between the TDF introduction and AKI may vary from weeks to years (1month-8 years) [5,7,9,10]. In our series, AKI appears during the year following TDF initiation.
The four cases we reported had an atypical AKI presentation with biological feature of chronic kidney disease (normochromic normocytic anemia and high serum phosphate). These features associated with uremic syndrome suggest chronic kidney disease and chronic hemodialysis initiation was planned in all of them. However; careful review of their files reveals normal creatinine prior to TDF initiation and normal kidney size. TDF was then discontinued and serum creatinine unexpectedly dropped. High serum phosphate is unusual in TDF induced nephropathies since it induced proximal tubulopathy with urinary loss of phosphate. However, it have been noted by some authors and it considered to be due to decrease GFR [11]. This hypotheses seem to be true since high serum phosphate persist in patient with partial recovery. Severe anemia in our series may also be due to decrease GFR since hemoglobin level increase with GFR rise. Anemia has been described with TDF used, but it rarely less than 8 g/dl [9]. A cohort study on anemia among HIV infected patients on HARRT in Ethiopia found that TDF- based HAART increased the risk of anemia [12]. Fatouma and al also described two cases of TDF induced AKI with severe anemia in Kenya [13]. Hemolytic anemia could be another explanation of the atypical presentation of our cases since hypocalcemia and high serum phosphate is usually found in AKI associated with hemolytic anemia. Unfortunately; LDH and haptoglobin were not done.
One patient had a polyuric polydipsic syndrome with hypotonic polyuria which started 2 months after TDF initiation. It was probably due to a diabetes insipidus, a well know TDF nephrotoxicity features. Association between Fanconi syndrome, AKI and distal tubular injury had been reported in TDF induced nephropathies [8]. TDF was incriminated in the four patients since all the signs disappear after TDF withdrawal. Lamuvidine and Efavirenz was still used in the substituted protocol as well as cotrimoxazol.
TDF was reintroduced after 5 months in case 1 and after 2 weeks, the creatinine started to increase and glycosuria reappear. Creatinine normalized and glycosuria disappear after withdrawal of TDF. He had been followed now for 48 months with normal creatinine and no proteinuria.
Concerning outcome, kidney function generally improved in the month following TDF withdrawal. However, AKI does not always revert completely. In a study of 24 patients who stopped TDF because of AKI, only 42% recovered their initial kidney function [14]. Patients with better kidney function improvement after stopping TDF had faster GFR decline, were more frequently prescribed a proteinase inhibitor and had been treated with TDF for a shorter period [14]. Our cases obeyed to this picture: patients that developed AKI within the first 3 months of TDF initiation had a better improve in GFR (1 complete recovery and 2 partial recovery with GFR > 35ml/min/1.75 m2 within 6 months). In contrast; the patient that developed AKI after 6 months of TDF had the lowest kidney function improvement with a GFR < 30ml/min/1.75 m2 after 6 months).
Conclusion
TDF induced AKI nephrotoxicity can present with severe anemia and high serum phosphate. As the used of TDF-based HARRT is widely spread in our setting, clinician should be aware of the complexity of TDF induced AKI nephrotoxicity and careful evaluate all patient on TDF with decrease GFR since TDF induced AKI can present with pathognomonic sign of end stage kidney disease.
References
- Schooley RT, Ruane P, Myers RA, Beall G, Lampiris H, et al. (2002) Tenofovir DF in antiretroviral-experienced patients: results from a 48-week, randomized, double-blind study. AIDS 16: 1257-1263.
- Nelson MR, Katlama C, Montaner JS, Cooper DA, Gazzard B, et al. (2007) The safety of tenofovir disoproxil fumarate for the treatment of HIV infection in adults: the first 4 years. AIDS 21: 1273-1281.
- Gallant JE, Staszewski S, Pozniak AL, DeJesus E, Suleiman JM, et al. (2004) Efficacy and safety of tenofovir DF vs stavudine in combination therapy in antiretroviral-naive patients: a 3-year randomized trial. Journal of the American Medical Association 292: 191-201.
- Beatriz FF, Montoya-Ferrer A, Sanz AB, Sanchez-Niño MD, Izquierdo MC, et al. (2011) TenofovirNephrotoxicity: 2011Update. AIDS Research and Treatment
- Karras A, Lafaurie M, Furco A, Bourgarit A, Droz D, et al. (2003) Tenofovir-related nephrotoxicity in human Immunodeficiency virus-infected patients: three cases of renal failure, Fanconi’s syndrome and nephrogenic diabetes insipidus. Clin Infect Dis 36: 1070-1073.
- Ketan KP, Atul KP, Rajv RR, Apurva RP, Jagdish KP (2010) Tenofovir-associated renal dysfunction in clinical practice: an observational cohort study from western India. Indian J ex Transm Dis 31: 30-34.
- Chaisiri K, Bowonwatanuwong C, Kasettratat N, Kiertiburanakul S (2010) Incidence and risk factors for tenofovir-associated renal function decline among Thai HIV-infected patients with low-body weight. Curr HIV Res 8: 504-509.
- Verhelst D, Monge M, Meynard JL, Fouqueray B, Mougenot B, et al. (2002) Fanconi syndrome and renal failure induced by tenofovir: A first case report. Am J Kidney Dis 40: 1331–1333.
- Murali MK, Subbalaxmi MV, Megha Uppin, Radhika S (2014) Delayed onset renal failure in a patient on tenofovir based antiretroviral regimen. Indian J Pharmacol 46: 230-231.
- Herlitz LC, Mohan S, Stokes MB, Radhakrishnan J, D’Agati VD, et al. (2010) Tenofovir nephrotoxicity: Acute tubular necrosis with distinctive clinical, pathological, and mitochondrial abnormalities. Kidney Int 78: 1171-1177.
- Pinelopi PK, Naheed Ansari (2008) Acute renal failure in an AIDS patient on tenofovir: a case report. J Med Case Reports 2: 94.
- Muluken A, Woldaregay EA, Shewamare A, Medhin G, Mulugeta B (2015) Prevalence and correlates of anemia among HIV infected patients on highly active anti-retroviral therapy at Zewditu Memorial Hospital, Ethiopia. BMC Hematol 15: 6.
- Fatuma Some, Mathew Koech, Emily Chesire, Gabriel Kigen (2017) Reversal of tenofovir induced nephrotoxicity: case reports of two patients. The Pan African Medical Journal 27: 126.
- Gupta SK (2008) Tenofovir-associated Fanconi syndrome: Review of the FDA adverse event reporting system. AIDS Patient Care STDS 22: 99-103.



