Research Article - (2023) Volume 8, Issue 1
Temporal Changes in Key Signal Transduction Pathways Mediating Muscle Protein Synthesis with Adaptive and Maladaptive Right Ventricular Hypertrophy in Pulmonary Arterial Hypertension
2Division of Pulmonary/Critical Care Medicine, Cedars-Sinai Medical Center, Los Angeles, CA, USA
3Jacobs School of Engineering, University of California, San Diego, CA, USA
Received Date: Dec 21, 2022 / Accepted Date: Dec 28, 2022 / Published Date: Jan 05, 2023
Copyright: ©Michael I Lewis, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Middleton, R. C., Fournier, M., Rogers, R. G., Grimes, B. S., Xu, X., & Lewis, M. I. (2023). Temporal Changes in Key Signal Transduction Pathways Mediating Muscle Protein Synthesis with Adaptive and Maladaptive Right Ventricular Hypertrophy in Pulmonary Arterial Hypertension. Cardio Open, 8(1), 01-14.
Abstract
Pulmonary Arterial Hypertension (PAH) is a progressive disease characterized by occlusive remodeling of pulmonary arteries < 500 micron and increased pulmonary vascular resistance. With the onset of PAH, the right ventricle (RV) of the heart adapts to the increased afterload pressure by undergoing adaptive hypertrophic remodeling to maintain adequate blood flow. However, for unknown reasons, maladaptive influences ensue, resulting in impaired RV function with progressive decompensation and right heart failure. Using a rodent model of PAH, key signaling pathways mediating cardiac muscle protein synthesis in the RV were evaluated during both the adaptive hypertrophy phase, with preserved right heart function, and the decompensated maladaptive phase, in which right heart failure (RHF) was present. Analysis of protein and gene expression changes in PAH animals identified three key signaling pathways involved in the shift toward maladaptive right heart failure: i) PI3K/Akt/mTOR; ii) GSK-3; iii) MAPK/ERK, as well as IGF-1 regulation. During adaptive hypertrophy, significant increments of phosphorylated proteins in the three signaling pathways were observed with increases in RV fibrosis and decreased capillarity found. In the maladaptive phase, mTORC1 and its downstream effector p-70S6K were significantly activated, contributing to the decreased LC3-I/II ratio, a marker of autophagy inhibition together with further significant RV muscle fibrosis and greater capillary rarefaction. We propose that autophagy inhibition in conjunction with other maladaptive processes reported in the decompensated RV muscle contributes to the genesis of overt RHF in PAH, and that a continuum of changes characterizes the adaptive and maladaptive phases in the RV muscle.
Keywords
Pulmonary Arterial Hypertension (PAH), Right Heart Failure (RHF), Autophagy, Cell Signaling, Monocrotaline
Introduction
Pulmonary arterial hypertension (PAH) is a progressive condi- tion for which there is no cure. Even with substantial pharma- cologic advances in the modern treatment era, survival remains unacceptably poor [1-3]. PAH is characterized by occlusive remodeling of pulmonary arteries < 500 micron and increased pulmonary vascular resistance. With the onset of PAH, the right ventricle (RV) adapts to the increased afterload presented to it by adaptive hypertrophy to maintain adequate blood flow. This is driven by signaling events that promote enhanced muscle pro- tein synthesis and physiologic growth. However, for unknown reasons, maladaptive influences ensue, resulting in impaired RV function with progressive decompensation and failure [4]. In- deed, RV function is the major factor determining survival of patients with PAH [5-8].
We and others have reported that several signaling pathways in- volved in muscle protein synthesis are important mediators of adaptive cardiac and skeletal muscle hypertrophy. This includes the phosphatidylinositol 3-kinase (PI3K)/Akt/mammalian target of rapamycin (mTOR) pathway, as a major regulator of cardiac and skeletal muscle protein synthesis and hypertrophy, with sig- nificant depression in the diaphragm muscle in atrophic states [9-15]. Another signaling pathway mediating enhanced cardiac muscle protein synthesis and growth, is the glycogen synthase kinase-3 (GSK-3) pathway which plays a key role in the inte-gration of hypertrophic signaling in the heart, involving both α and β isoforms [16,17]. Lastly, the mitogen-activated protein ki- nases (MAPK)/extracellular signal-regulated kinases (ERK) 1/2 pathways also exhibit significant influences in promoting cardio myocyte hypertrophy [15,18-20]. We postulate that these three signaling pathways would be key in mediating adaptive changes in the RV muscle exposed to a slow progressive rise in RV after- load facing the RV as PAH develops.
In this study, the temporal changes in the signaling of these three pathways were measured in the RV muscle as the RV progresses from a healthy state to adaptive hypertrophy and then to mal- adaptive hypertrophy in an animal model of PAH. In addition, insulin like growth factor-1 (IGF-1) was evaluated in adaptive and maladaptive RVs, as this growth factor has been shown to both impact upon these three pathways and has been implicated in reports of physiologic cardiac hypertrophy as well as signa- ture of RV failure [10,21-24]. Furthermore, signaling proteins associated with autophagy were assessed in the decompensated maladapted RV muscle of rats with established right ventricu- lar dysfunction and failure. PAH progression was established by echocardiographic parameters of impaired RV systolic function along with confirmatory changes at the cellular level, which in- cluded changes in RV wall fibrosis and capillarity.
In the monocrotaline (MCT) rat model of PAH, observations in both the adapted and maladapted states describe several direc- tions of change in the RV muscle that were unanticipated based on skeletal muscle data and provide robust data and important questions for future research endeavors in fully understanding RV physiologic and pathologic changes that occur in the RV with progressive PAH [15].
Methods
Animal Care
Adult male Sprague-Dawley rats (initial body weight ~225g; n=52) were divided into 2 major groups, namely control (CTL) and PAH. The CTL rats were injected subcutaneously with sa- line, while the PAH animals received a single subcutaneous in- jection of MCT (Sigma, St. Louis, MO) at a dose of 60 mg/kg body weight, dissolved in 0.5mL saline. All animals were pro- vided with food (Purina rat chow) and water ad libitum. Animals were individually housed with a dark: light cycle of 12 hours each and with the ambient temperature maintained at 22ºC. Terminal experiments were performed at days 7, 14, 24 and 28 (adapted cohort) and at 42 days (maladapted cohort, based on echocardiography).
Echocardiography
Transthoracic two-dimensional, M-mode echocardiography and pulsed-wave Doppler imaging were performed on anesthetized (isoflurane) rats (Vevo 770 Micro-Ultrasound imaging system; Visual Sonics: Toronto, Canada). RV systolic function was de- termined by tricuspid annular plane systolic excursion (TAPSE), recorded in M-mode. Doppler of pulmonary outflow measured pulmonary artery flow velocity time (PAVTI). To obtain stroke volume (SV), the cross-sectional area of the pulmonary artery was multiplied by PAVTI, and estimated cardiac output (CO) was derived by multiplying SV by heart rate. Serial studies were performed before and post MCT administration.
Histological Studies
Fibrosis: To assess fibrosis, cryosections of RV free wall from 5 adapted, 5 maladapted and 3 CTL rats were stained with Pi- crosirius Red as described [25,26]. Sections were imaged by bright field microscopy and analysis was performed using Im- ageJ (Fiji), by measuring pixel density within the green channel throughout the entire RV free wall [27,28]. One section was ana- lyzed for each animal and normalized to the total area of the RV free wall. Results are presented as percent fibrosis.
Vessel counts: To assess vascularity in CTL, adapted and mal- adapted RV free wall from PAH animals, 10µm tissue sections were stained with anti-rat smooth muscle actin primary antibody (1:100, Sigma) and FITC-conjugated goat anti-rat secondary antibody (1:400, Abcam), and TRITC conjugated isolectin IB4 (Thermo). The stained tissue sections were imaged by confocal microscopy (Leica) and analyzed using ImageJ (Fiji). 10 cryo- sections were analyzed per animal.
Muscle Proteins
Protein Extraction: RV muscle segments were rapidly frozen in liquid nitrogen and stored at -80°C until analysis. Sample protein was extracted in a 1:10 ratio of cold RIPA buffer (Cell Signal- ing Technologies, Beverly, MA), with protease and phosphatase inhibitors (Roche, South San Francisco, CA) added, according to manufacturer’s protocol. For mTOR analysis samples, 0.4% CHAPS was also added to the lysis buffer. Homogenization was performed with a Polytron homogenizer (Kinematic, Bohemia, and NY) and tissue lysates were incubated on ice for 4 hours and then were centrifuged at 13,200 RPM. The supernatant was aliquoted in micro centrifuge tubes. Protein concentration was determined using a commercial protein assay kit (Bio-Rad, Her- cules, CA) and measured with a spectrophotometer (Smart Spec- TM 3000, Bio-Rad).
Immunoprecipitation: For mTOR analysis only, protein ly- sates were precleared with normal IgG (Santa Cruz Biotechnolo- gy, Dallas, TX) and protein A/G Plus-agarose beads (Santa Cruz Biotechnology). The lysates were incubated with the primary antibody (mTOR or phosphorylated mTOR (Ser2448); Cell Sig- naling Technology, Beverly, MA) overnight at 4°C. Protein A/G Plus-agarose beads were added to the immunocomplex, incu- bated for 4 hours at 4°C, and centrifuged. The immunocomplex pellet was washed, resuspended in sample buffer, heated at 70°C for 15 min and centrifuged. The supernatant was used to load gels for electrophoresis as described below.
SDS-PAGE and Western blotting: Electrophoresis of protein extracts was performed by SDS-PAGE. In brief, samples were heated at 70°C for 15 min and cooled prior to being used for electrophoresis. Protein extracts (40 μg per well) were loaded on NuPAGE 4-12% Bis-Tris gradient gels (Invitrogen, Grand Island, NY). For the analysis of 4E-BP1 (PHAS-I) samples, ex- tracts were loaded on 12% Bis-Tris gels. Separated proteins were then electrophoretically transferred to nitrocellulose membranes (Bio-Rad). The membranes were treated with blocking buffer for 1 hour at room temperature. Blots were incubated with prima- ry antibodies at 4°C overnight, washed and incubated with an appropriate peroxidase-conjugated secondary antibody at room temperature for 1 hour. The blots were visualized following de- velopment with enhanced chemiluminescence (ECL) Western blotting detection reagents (GE Healthcare Life Sciences, Pitts- burgh, PA), according to manufacturers’ protocol. Blots were re-used by exposing them to stripping buffer (RestoreTM, Ther- mo Scientific, and Rockford, IL) and re-probed with a different primary antibody to enable direct comparison of phosphorylated and non-phosphorylated states. Blots were exposed to X-ray film in a cassette, the films scanned, and identified bands analyzed by densitometry using ImageJ 1.43u (NIH). Western blot data from the experimental groups were expressed relative to measured mean values from the CTL group. All results are expressed as the ratio of phosphorylated protein/total protein.
Primary Antibodies: All primary antibodies were rabbit poly- clonal antibodies obtained from Cell Signaling Technology, ex- cept for phosphospecific 4E-BP1 (Bethyl Laboratories, Mont- gomery, TX) and mouse monoclonal antibody for total GSK-3 (Santa Cruz Biotechnology). Blots were first incubated with the following anti-phosphopeptide (P) antibodies: P-Akt (Ser473), P-p70S6K (Thr389), P-GSK-3a/b (Ser21/9), P-ERK1/2 (p44/p42) (Thr202/Tyr204), P-mTOR (Ser2448), P-CDK2 (Thr160), and P-4E- BP1 (PHAS-I). Stripped membranes were re-probed with anti- bodies recognizing total Akt, p70S6K, GSK-3α/β, , ERK1/2 (p44/ p42), mTOR, CDK2, 4E-BP1 as well as GAPDH, with the latter serving as a control for equal protein loading of samples. Blots to probe for non-phosphopeptide proteins were first incubated with p27, or LC3B antibodies.
IGF-1 protein measurement by ELISA: Determination of IGF-1 in the adaptive hypertrophy experiments was performed as previously described [29]. Briefly, frozen RV muscle seg- ments were pulverized in liquid nitrogen. Protein was extract- ed twice on cold acetic acid (1M; 1 mg/10 µl) and supernatant stored at -80ºC overnight. Aliquots (100 µl) were lyophilized and stored frozen overnight. Protein pellets were resuspended in 40 µl H2O and protein concentration determined as described above. Duplicate 10 µl protein samples were assayed using a commercial high sensitivity ELISA kit for rodent IGF-1 (AC- 42F1) from Immunodiagnostic Systems, Inc. (IDS; Gaithers- burg, MD) according to manufacturer’s protocol.
Note, for the maladapted RV muscle IGF-1 assays a different kit was used, as the Immunodiagnostic Systems kit (AC-42F1) had been discontinued. Thus, for these experiments, we used a rat IGF-1 ELISA Kit (Catalog # 80573) from Crystal Chem (Elk Grove Village, IL). Results are expressed as ng/ mg protein.
RNA Extraction and Quantitative Polymerase Chain Reac- tion (qPCR)
Total RNA Extraction: Total RNA was extracted from RV muscle segments with RNeasy Mini Kit (Qiagen, Valencia, CA) according to manufacturer’s protocol. RNA concentration was measured with a spectrophotometer (Nano Drop 1000; Thermo Scientific).
Oligonucleotides: The primers for IGF-1Ea and the refer- ence gene succinate dehydrogenase A (SDH-A) were designed based on published rat cDNA sequences. Sequences of prim- ers used for cDNA synthesis and RT-qPCR analysis, along with their accession number were the following: IGF-1Ea (NM_001082478): Forward 5’-GCTTGCTCACCTTTAC- CAGC-3’, Reverse 5’-AAATGTACTTCCTTCTGAGTCT-3’; SDH-A (NM_130428): Forward 5'-CTCTTTTGGACCTTGTC- GTCTTT-3', Reverse 5'-TCTCCAGCATTTGCCTTAATC- GG-3'.
Real-time Quantitative PCR: RT-qPCR was performed in two steps. One ug of total RNA per sample was reverse transcribed with Superscript III using Oligo (dT)20 (Invitrogen) according to manufacturer’s protocol. qPCR was performed in 10ul re- actions using 2X Sybr Green Master Mix (ABi P/N 4309155), 50 nM gene specific primers, and 2ul of the reverse transcribed samples. Samples were analyzed in triplicates across the genes on the ABi Viia7. In order to compare the relative mRNA ex- pression between control and experimental groups, the compar- ative threshold cycle (CT; the fractional cycle number at which the amount of amplified target reaches a fixed threshold) method was used.
Statistical Analysis
The distribution of all data was tested for normality and then statistical analysis was performed using ANOVA (Instat v. 3.06, Graph Pad, San Diego, CA) to compare differences between the independent groups in the adaptive state study. If a significant in- teraction was found, post hoc analysis (Student-Newman-Keuls test) was used to compare differences within independent groups. A Student t-test was used to compare differences between the groups in the maladaptive state study. A α level of 0.05 was used to compare differences in independent groups and to determine overall significance. Values are expressed as means ± SEM.
Results
Animal Physiology and RV Histology
To assess morphological and cell signaling changes in right ven- tricle cardiac tissue in a model of PAH, male Sprague-Dawley rats were treated with the pyrrolizidine alkaloid, Monocrotaline (MCT). Over the course of the disease, cardiac function was as- sessed, and cardiac tissues were collected and analyzed for gene expression changes as PAH progressed towards RHF. There are several physiological indicators of RV distress and failure that present during the progression of PAH, including changes in body weight, breathing rates and fluid retention in the abdomi- nal cavity. Animals with PAH showed reduced body weight ac- cumulation compared to healthy cohorts and will lose weight as the animals progressed towards RHF. During the RV adaptive phase of PAH, no difference in body weight was observed at day 7 post-MCT delivery between healthy controls (CTL) and PAH rats. At days 14, 24 and 28 days post-MCT, body weight gains were attenuated by 6, 8% and 14% respectively, for PAH animals compared to CTL (P < 0.01; Figure 1).
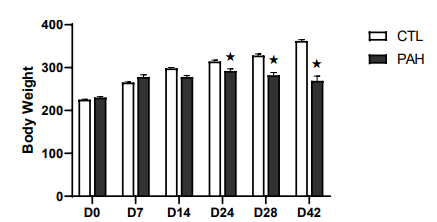
Figure 1: Body weights of control (CTL) and pulmonary arterial hypertension (PAH) animals at days 0, 7, 14, 24, and 28. Values are means ± SEM; «significantly different from CTL animals.
At 42 days post-MCT at which time the animals exhibited overt RV dysfunction/failure, significant weight loss occurred, from peak weight achieved over time to weight at the time of terminal experiments (peak weight: 362 +/- 3 g; terminal weight: 269 +/- 11 g; P < 0.01). In addition, other observational signs suggestive of right heart failure were evident. This included a progressive listless state with obvious reduced mobility, poor grooming and ruffled fur and tachypnea with a pattern of increased work of breathing. Abdominal distension suggested the presence of as- cites. Animals were sacrificed at this point, which was a joint decision by the investigators and veterinary staff. Post sacrifice, the presence of ascites +/- pleural effusions were confirmed.
TAPSE, a measure of RV systolic function by echocardiography, was assessed in MCT-treated rats and compared to CTL animals. No significant differences were observed between CTL rats and PAH animals from 7 to 28 days post MCT administration. (28 days, CTL: 2.9 +/- 0.7 mm; PAH: 2.8 +/- 0.14 mm). However, the mean value of TAPSE for the late-stage PAH cohort was significantly reduced compared to CTL and the group of adapt- ed PAH animals (42 days, 1.73 +/- 0.144 mm; P < 0.01; Figure 2A). The estimated cardiac output (CO) for PAH rats at all time frames during the compensatory period were not significantly different from CTL values, and so the data was pooled for PAH animals (CTL: 124 +/- 8 ml/min; PAH: 100 +/- 4 ml/min). The values for CO of CTL rats were similar to those previously re- ported for male Sprague-Dawley rats of the same weight range [31]. For the maladapted cohort at 42 days post MCT, estimated value of CO was significantly reduced in these rats (72.6 +/- 5.1; SE; P < 0.01; Figure 2B).
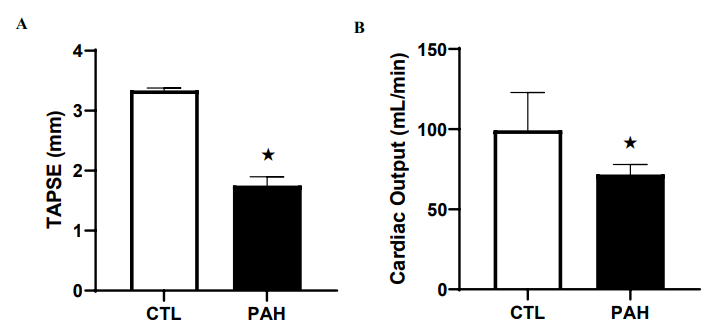
Figure 2A: Values of TAPSE in normal controls (CTL) compared to that in pulmonary arterial hypertension (PAH) rats complicated by right heart failure (RHF).
Figure 2B: Values of cardiac output (CO) in CTL animals compared to that in pulmonary arterial hypertension (PAH) rats compli- cated by RHF. «Significantly different from CTL. Values are means ± SEM.
Histological assessment of fibrosis in coronal sections of RV free wall during the late adaptive RV hypertrophy stage (28 days post-MCT), indicated that the percentage area of the RV free wall occupied by fibrotic tissue increased more than 2-fold (9.07 +/- 1.70 %; SE) compared to CTL (3.97 +/- 1.20 %; P < 0.05; Figure 3A). Additionally, a further 1.8-fold increase in fibrosis (17.06 +/- 0.88 %; P < 0.05) was observed in the RV free wall of maladaptive hearts (42 days post-MCT) relative to the adapt- ed RV cohort of PAH animals (Figure 3A). Changes in the RV free wall vascularity were assessed in CTL and PAH rats at both capillary and arteriolar levels. During late adaptive RV hypertro- phy, the number of capillaries and arterioles in the RV free wall significantly decreased by 32% (P < 0.05) and 21% (P < 0.05), respectively, compared to CTL (Figure 3B&C). By contrast, in the maladaptive RHF/PAH cohort, we observed a further signifi- cant decline in RV free wall capillaries and arterioles by 54% (P < 0.01) and 65% (P < 0.001), respectively, compared to the adap- tive RV cohort (Figure 3B&C). No significant changes in vascu- larity were observed at 7- and 14-days post MCT administration.
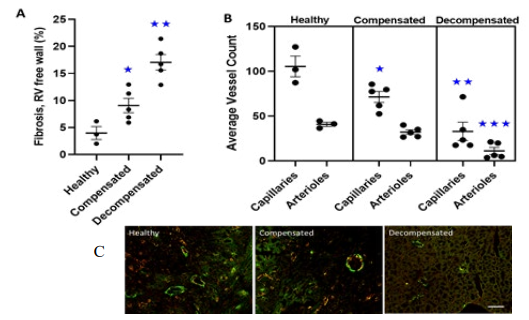
Figure 3A: The individual values and average percent fibrosis of the RV free wall in CTL, compensated and decompensated rat hearts is shown. *indicates significant difference from CTL (P<0.05). ** indicates significant difference from compensated hearts (P<0.01).
Figure 3B: Graphical representation of the average capillary and arteriole numbers per image for each animal is also demonstrated.
*indicates significant difference from CTL (P<0.05). **indicates significant difference from compensated hearts (P<0.05). ***indi- cates significant difference from compensated hearts (P<0.01). Error bars indicate ± SEM.
Figure 3C: Example images of capillary (green) and arteriole density (orange) in different stages of PAH are shown. Scale bar =
50µm
Hypertrophy-Related Gene Expression Changes in the RV Free Wall of Adapted and Maladapted PAH Animals
During the progression of PAH, the RV becomes progressively decompensated with the eventual development of overt RHF. Further, echocardiography data presented above, marked the transition of adaptive hypertrophy to maladaptive hypertrophy, however the molecular signaling that drives the maladaptation in PAH is unknown. While there is an abundance of literature on the signaling pathways associated with cardiac hypertrophy, those data predominantly reflect events in the LV. Measurements of gene expression related to hypertrophy in the RV is limited, especially for serial measures over time in the context of pro- gression toward RHF. To gain insight into possible alterations in those signaling pathway patterns we described during the adap- tive and maladaptive phases, we evaluated the IGF-1, MAPK/ ERK, and mTOR pathways in the RV muscle of rats as PAH progresses.
IGF-1
IGF-1 is a potent regulator of inflammation and angiogenesis, and reduced IGF-1 has been described as part of the RV muscle failure molecular signature complicating PAH [30,31]. IGF-1 protein expression in the RV muscle of PAH rats were similar to that in CTL animals between days 7and 24 (Figure 4). At day 28 a significant 45% reduction (P < 0.001) in IGF-1 expression in the RV of PAH rats was noted compared to CTL (Figure 4). Right hearts undergoing maladaptive hypertrophy, at day 42, also showed a 45% decrease in IGF1.
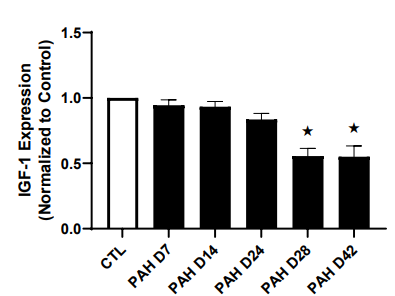
Figure 4: IGF-1 protein expression in the right ventricle (RV) of control (CTL) and pulmonary arterial hypertension (PAH) rats complicated by right heart failure (RHF), normalized to CTL expression values. «Significantly different from CTL. Values are means ± SEM.
MAPK/ERK Pathway
Downstream of IGF-1 activation, the phosphorylation states of the signaling proteins p44 (ERK1) and p42 (ERK2) were both assessed. Values for both P-p44 and P-p42 were similar between CTL and PAH rats at days 7 and 14 (Figure 5). At day 24 how- ever, significant increments were observed for both P-p44 (1.6- fold; P < 0.001) and P-p42 (1.4-fold; P < 0.05) in PAH rats com- pared to CTL. At day 28 P-p44 values were still 16% greater than CTL (NS), while P-p42 expression had returned to CTL values. In maladapted hearts, values for both P-p44 and P-p42 were similar between maladapted PAH and CTL rats (Figure 5).
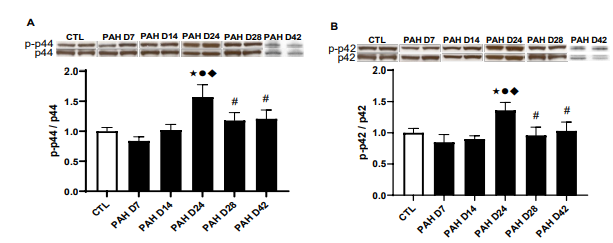
Figure 5A: P-p44 and
Figure 5B: P-p42 expression levels in the RV of control (CTL) and pulmonary arterial hypertension (PAH) rats complicated by right
heart failure were expressed as a fraction of phosphorylated protein over total protein. Values are shown as means ± SEM.
GSK-3 Pathway
GSK-3β is known to play an important role in regulating gly- cogen metabolism in cardiac muscle and in the integration of hypertrophic signaling in the heart has been increasingly empha- sized [17]. Phosphorylation of GSK-3B (Ser9) inhibits its protein kinase activities, which diminishes GSK-3’s inhibitory effects on eIF2 to promote protein translation and hypertrophy. Phos- pho-GSK-3β, values at days 7 and 14 were similar between CTL and PAH rats (Figure 6). However, P-GSK-3β was significantly increased by 1.3-fold in PAH rats at day 24 respectively (P < 0.001 and P < 0.001). At day 28, P-GSK-3β was significantly re- duced (P < 0.05; Figure 6). Interestingly, there was no significant difference in phosphorylation of GSK-3β between maladapted/ failed RV of PAH and CTL hearts.
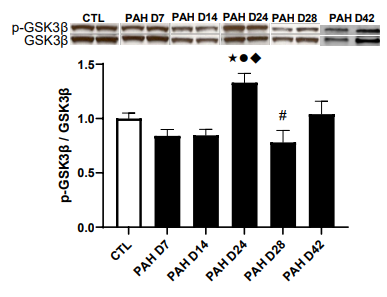
PI3K/Akt/mTOR Pathway
Levels of phosphorylated Akt, mTOR and down steam effec- tors, 4E-BP1 and p70S6K were analyzed from RV cell lysates of healthy and PAH rats. P-Akt and P-mTOR were not significantly different between CTL and PAH rats at days 7 and 14. However, significant increments for both signaling proteins were observed at day 24 in PAH animals compared to CTL (1.3 and 1.4-fold increment respectively; P < 0.05 and P < 0.01; Figure 7A&B). At day 28, levels of P-Akt and P-mTOR had returned to control levels. Further out, phosphorylated Akt was diminished by 63% in the maladapted/failed RV muscle compared to control rats (Figure 7A; P < 0.005). P-mTOR, in maladaptive heart tissue, was significantly increased (P < 0.01; Figure 7B) suggesting ac- tivation by pathways separate from the PI3K/Akt signaling axis as P-Akt was markedly diminished.
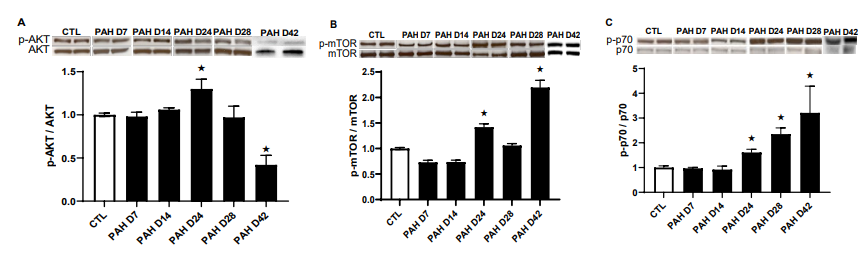
Figure 7A: Ratio of phosphorylated Akt (Ser473) to total Akt,
Figure 7B: ratio of P-mTOR (Ser2448) to total mTOR,
Figure 7C: p-p70S6K (Thr389) to total p70S6K in the right ventricle of control (CTL) and pulmonary arterial hypertension (PAH) ani-
mals at days 0, 7, 14, 24, 28 and 42. Values are means ± SEM; «significantly different from CTL.
The downstream effector P-p70S6K was unchangedat days 7 and 14 in PAH rats, but significantly elevated at days 24 and 28 com- pared to CTL (1.6 and 2.4-fold increments respectively; P < 0.01 and P < 0.001. At day 42, P-p70S6K remained significantly higher than control (3-fold higher; P < 0.01). To further explain this, we examined another possible signaling pathway that has recently been described to activate mTOR with subsequent autophagy inhibition [32].
Autophagy Pathway
To assess the role of autophagy signaling in the failing right heart, the LC3BII/I ratio was analyzed as a surrogate marker of autophagosome formation. There was a significant decrease in the LC3BII/I ratio in maladapted/failed RV of PAH rats com- pared to CTL rats (P < 0.05; Figure 8A&D). A recent publication identified a p27/CDK2 axis in the regulation of mTOR-depen- dent inhibition of autophagy during heart failure [32]. p27 was significantly decreased in maladapted/failed RV of PAH rats relative to CTL (P < 0.05; Figure 8A&B). However, p-CDK2 was not statistically different (P = 0.22) in PAH rats which likely reflects variances and power influences (Figure 8A&C).
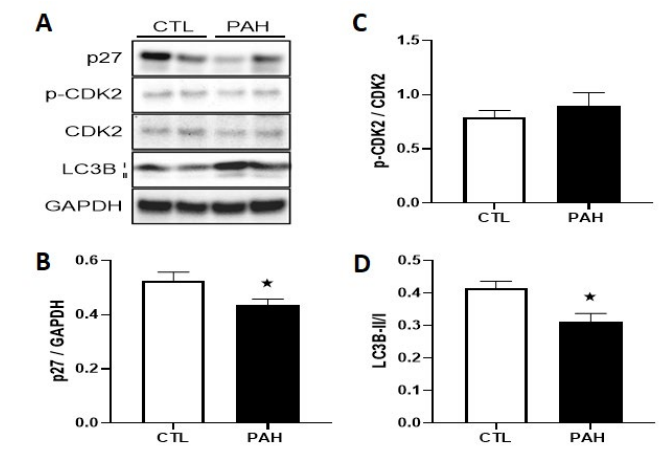
Figure 8A: Western blot analysis of autophagy marker expression in healthy and failing RV tissue. Graphical representations of protein levels.
Figure 8B: p27/GAPDH,
Figure 8C: phosphorylated p-CDK2/CDK2
Figure 8D: LCB3 I/II isoforms in the RV of control (CTL) and pulmonary arterial hypertension (PAH) rats complicated by right heart failure. «Significantly different from CTL. Values are means±SEM. (F) Examples of Western Blots for each of the proteins described above.




