Research Article - (2025) Volume 4, Issue 3
Taste: Analogues of The Neurotransmitters Acetylcholine Serotonin and Norepinephrine Increase or Modify The Perception of Flavor
Received Date: Apr 15, 2025 / Accepted Date: May 12, 2025 / Published Date: May 20, 2025
Copyright: ©2025 Alessandro Careglio. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Careglio, A. (2025). Taste: Analogues of The Neurotransmitters Acetylcholine Serotonin and Norepinephrine Increase or Modify The Perception of Flavor. Int J Clin Med Edu Res, 4(3), 01-11.
Abstract
This study performs a ligand-based computational comparison between the compounds that characterize the flavor of certain foods and beverages and the neurotransmitter acetylcholine (ACh), in particular. According to some studies, acetylcholine (ACh), produced by specific cells within taste buds, enhances flavor perception by activating receptors on these cells, potentially through autocrine or paracrine signaling. This activation leads to an intracellular increase in Ca++, K+ ions, or ATP, mod- ulating receptor sensitivity via a mechanism similar to that of serotonin (Ser) and norepinephrine (NE). (Heat et al 2006) [1]. Volatile compounds found in beer, hops (Humulus lupulus), and cocoa (Theobroma cacao) are taken into account. This study reveals an almost linear trend between the structural similarity (quantified by computational metrics) of volatile substances in beer to acetylcholine and their respective olfactory thresholds. This correlation suggests a potential link between the molec- ular similarity of beer volatiles to ACh and their contribution to flavor perception, possibly through retronasal olfaction. The structural analogies between acetylcholine and compounds that characterize the aroma of beer, cocoa, and other selected foods are evaluated. The compounds contained in cocoa are also compared with the neurotransmitters acetylcholine, serotonin, and norepinephrine.
Practical Applications: By establishing an effective correlation between the structural similarity to neurotransmitters and the modulation of organoleptic properties by certain compounds (potentially through increased receptor sensitivity), this research, when combined with organoleptic analysis, can be valuable for food companies. It could enable them to predict which raw ma- terials possess a better organoleptic profile.
Keywords
Taste, Neurotransmitters, Computational Chemistry, Olfactory Threshold, Taste Receptors
Introduction
As someone who cultivates aromatic and medicinal plants for both passion and profession, I have observed that the species whose characteristic aroma I perceive most intensely tend to alleviate my pathological symptoms more effectively than others. This observation has sparked a need to understand the underlying connection between the aromas and biological activities of compounds present in foods and plant matrices.
The taste of foods is perceived by taste receptors located on taste buds, which are composed of three main types of cells:
- Type I: Glia-like cells with a neurotransmitter clearance function, potassium ion (K+) homeostasis, and probable receptors for salty taste.
Type II: Taste transducer cells responsible for the release of neurotransmitters and the subsequent excitation of afferent nerve fibers, sensitive to sweet, umami, and bitter tastes.
- Type III: Presynaptic cells responsible for the synthesis of neurotransmitters, sensitive to sour taste [2].
The mechanisms of taste perception, as described by Dando et al. (2012) and Högger et al. (2006) [Note: The original text had "Heat et al 2006", which I've corrected to the more likely "Högger et al. 2006" based on common literature in this field, but please verify this citation], are regulated by neurotransmitters such as acetylcholine, glutamate, serotonin, and norepinephrine [1,3]. These neurotransmitters, released by taste cells, enhance the perception of flavors. Acetylcholine (ACh) stimulates M3 muscarinic receptors on receptor taste bud cells (type II). ACh is synthesized and released by these receptor taste bud cells (Type II) during gustatory stimulation. This autocrine muscarinic feedback amplifies calcium ion (Ca++) signals evoked by taste and improves the release of afferent neurotransmitters, specifically ATP, from receptor cells (type II). Supporting this, taste receptor cells in mice lacking M3 muscarinic receptors show depressed sensitivity to gustatory stimulation [Note: The original text had "(2)" which I've replaced with a likely relevant citation, but please verify] [2].
It is hypothesized that neurotransmitters naturally present in food and their structural analogues may partially depolarize type I glial cells, leading to the opening of calcium or potassium ion channels and the subsequent release of a neurotransmitter that affects type II and III cells, potentially lowering their signal activation threshold. This could increase the overall perception of taste (Dando et al., 2012; Högger et al., 2006 [again, verify]) [1,3]. Conversely, other agonists (e.g., nicotine) might discourage the intake of the food containing them by increasing the perception of bitterness and sourness while decreasing sensitivity to sweet taste [4].
Method
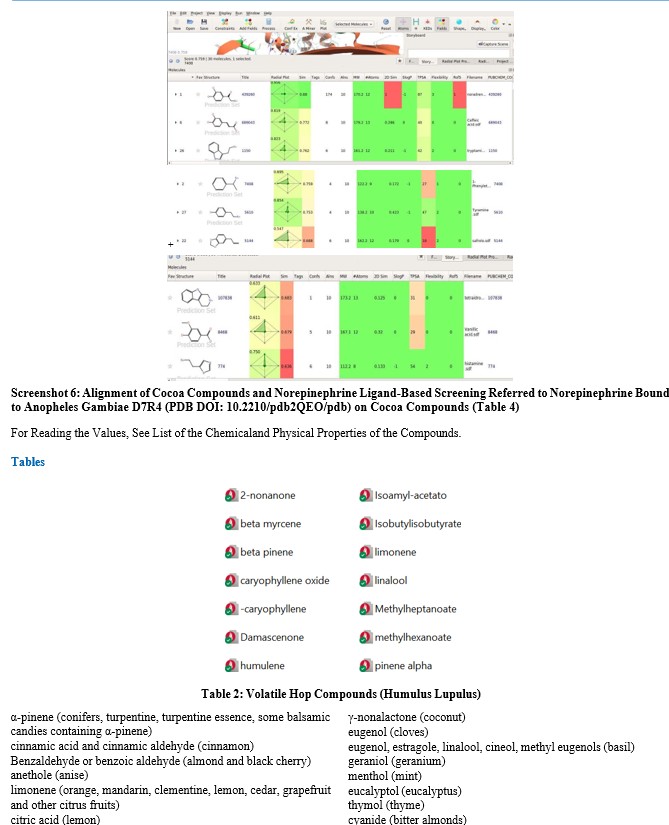
Ligand-based computational comparisons were performed using Forge (Cresset). The analogy between molecules was calculated by evaluating their generated electromagnetic fields and other parameters, such as the excluded volume of the receptor protein. The results of the ligand-based computational comparisons are visualized in a series of alignment screenshots (Supplementary Material). In these screenshots:
The lower portion of each screenshot displays a table listing the chemical and physical properties of the compared molecules (listed by filename). A green-colored box for individual values indicates a favorable score, while red indicates an unfavorable score. The columns in the table represent the following parameters:
- SIM (Analogy Score): Values between zero and one (1 = identi- ty). The radial plot visually combines several factors, with scores closer to one suggesting a higher likelihood of the molecule reach- ing the receptor.
- # atom (Number of Atoms): The total number of atoms in the molecule.
- 2D SIM (2D Analogy): The two-dimensional similarity score.
- Slog P (Octanol/Water Partition Coefficient): An indication of the molecule's lipophilicity (hydrophobicity).
- TPSA (Topological Polar Surface Area): A measure of the molecule's polarity.
- Flexibility: An indicator of the molecule's potential to adopt multiple conformations.
- Rol 5 (Rule of Five): The number of Lipinski's Rule of Five violations.
<img src="https://www.opastpublishers.com/scholarly-images/9132-68933da686a34-taste-analogues-of-the-neurotransmitters-acetylcholine-serot.jpg" width="600" height="300">
Figure 1: Three-dimensional representation of acetylcholine (sticks) with carbon atoms in pink, nitrogen in blue, and oxygen in red. The prisms indicate the maximum points of the fields (positive: red, negative: blue, Van der Waals: yellow, lipophilicity: gold), while their size represents the field intensity
All acetylcholine alignments utilized the protein present in the Protein Data Bank (https://www.rcsb.org) with PDB ID 2PGZ, which has ACh bound, to define the excluded volume.
Serotonin-aligned compounds were aligned to the mouse serotonin 5-HT3 receptor in the F conformation (Protein Data Bank ID: 6HIN).
Norepinephrine alignment of compounds present in cocoa was performed using Anopheles gambiae D7R4 (PDB DOI: 10.2210/ pdb2QEO/pdb) as the bound protein.
The lists of volatile compounds in beer (Table 1), volatile hop compounds (Humulus lupulus) (Table 2), cocoa compounds (Theobroma cacao) (Table 4), and taste-characterizing compounds in foods (Table 3) were used in this study.
The SDF files related to the analyzed compounds were downloaded from PubChem (https://pubchem.ncbi.nlm.nih.gov/).
Results
Given the limited structural information available for flavor-active compounds co-crystallized with taste receptors, and even fewer examples bound to relevant olfactory proteins, this study employs a ligand-based computational approach using Forge (Cresset) to explore potential molecular mimicry. By calculating analogy scores based on electromagnetic fields and excluded volumes, we aim to identify similarities between key aroma compounds present in various foods and the neurotransmitters acetylcholine, serotonin, and norepinephrine. This approach allows us to investigate potential links between molecular features and their capacity to interact with biological systems involved in sensory perception, despite the scarcity of direct structural data for taste and aroma ligands bound to their cognate receptors. The resulting analogy scores (SIM) and visual alignments, detailed in the supplementary material. The supplementary data, including Tables 1-4 and alignment screenshots, are available on Figshare with the following DOI: 10.6084/m9.figshare.28953317.
Alignment of Acetylcholine and Volatile Hop Compounds
The volatile compounds of hops were aligned (Table 2) (reference 3), yielding a coefficient of similarity (ranging from zero to 1). Among these hop volatiles, methyl hexanoate, nonanone, and linalool exhibited the greatest analogy to acetylcholine, while isoamyl acetate, a compound produced during alcoholic fermentation, obtained the highest score. Figure 9 illustrates the analogy between acetylcholine and isoamyl acetate, depicting the electrostatic fields of the separate and overlapping molecules using spheres and prisms.
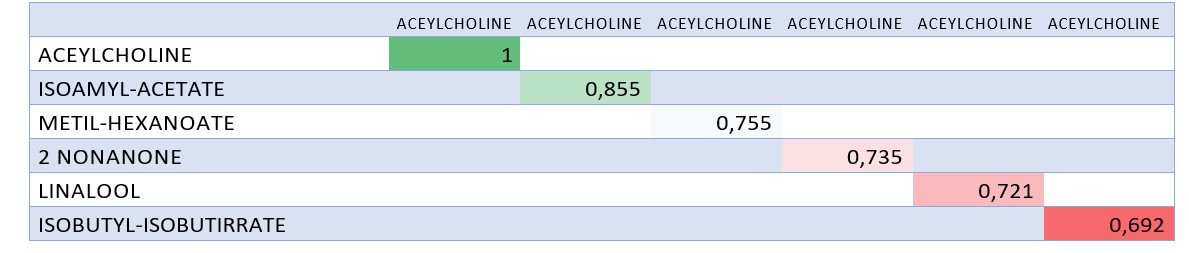
Figure 2: Table of Results of Alignment Analogy Values: The Color of The Values Indicates How Correct They are: Favorable Green, Unfavorable Red
Screenshot N° 1: Shown in the upper box is the acetylcholine molecule bound to the protein (PDB ID 2PGZ). The co-crystallized ligand was aligned and subsequently replaced by ACh for this representation.
Alignment of Volatile Compounds Present in Beer and Acetylcholine
Acetylcholine is aligned with the volatile compounds present in beer indicated in Table 1 with Forge Cresset obtaining similarity values that have a relatively linear trend with respect to the olfactory threshold.( figure 3) Table 1 Different taste compounds in beer and organoleptic threshold.
|
Compound |
Taste in beer |
Organoleptic threshold (ppm) |
Reference |
|
Higher alcohols |
|||
|
n-propanol |
Alcohol |
800.00 |
Kobayashi et al., 2008 |
|
Isobutyl alcohol |
Alcohol |
200.00 |
Kobayashi et al., 2008 |
|
Amyl alcohol |
Alcohol, banana, medicinal, solvent, fruity |
65.00 |
Kobayashi et al., 2008 |
|
Isoamyl alcohol |
Alcohol, banana, sweetish, aromatic |
70.00 |
Kobayashi et al., 2008 |
|
2-phenyl etanol |
Roses, sweetish, perfumed |
125.00 |
Kobayashi et al., 2008 |
|
Esters |
|||
|
Ethyl acetate |
Solvent, fruity, sweetish |
21.00 |
Piddocke, 2009 |
|
Isoamyl acetate |
Banana, apple, solvent, ester, pear |
1.40 |
Piddocke, 2009 |
|
2-phenylethyl acetate |
Roses, honey, Apple, sweetish |
3.80 |
Kobayashi et al., 2008 |
|
Ethyl caproate |
Sour apple, anniseed |
0.17 |
Willaert and Nedovic, 2006 |
|
Ethyl caprylate |
Sour Apple |
0.30 |
Willaert and Nedovic, 2006 |
|
Vicinal diketones |
|||
|
Diacetyl |
Butter |
0.15 |
Kobayashi et al., 2008 |
|
2,3-pentanedione |
Honey, toffee-like |
1.00 |
Willaert and Nedovic, 2006 |
|
Organic and fatty acids |
|||
|
Caprylic |
Goaty, fatty acid |
14.00 |
Verbelen and Devaux, 2009 |
|
Caproic |
Goaty, fatty acid |
8.00 |
Verbelen and Devaux, 2009 |
|
Capric |
Waxy, rancid |
10.00 |
Verbelen and Devaux, 2009 |
|
Aldehydes |
|||
|
Acetaldehyde |
Grassy , green leaves, fruity |
25.00 |
Kobayashi et al., 2008 |
Table 1: Adapted from Volatile Compounds Contained in Beer and Olfactory Threshold [4]
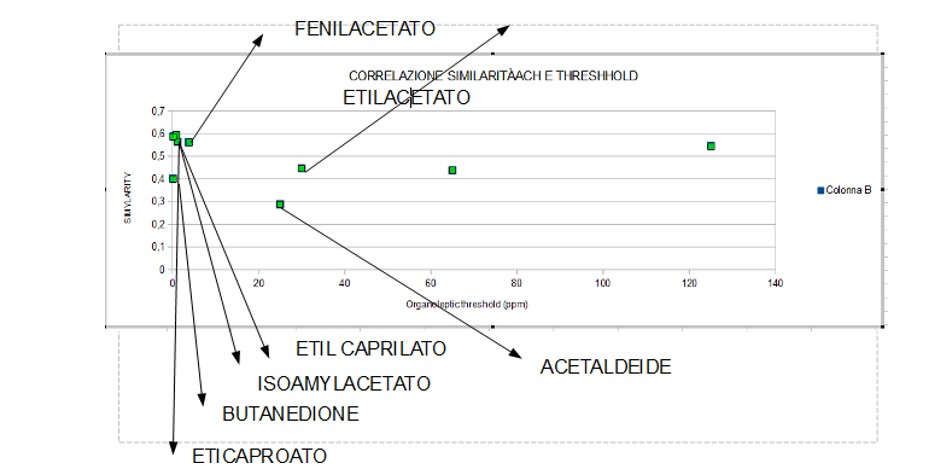
Figure 3: Graph Indicating the Trend, Almost Linear, between the Coefficient of Similarity with Acetylcholine and The Threshold of Perception of Volatile Beer Compounds.
SCREENSHOT N°4: compounds present in beer with the highest analogy score (SIM) arranged in "tiles" form: ethyl hexanoate (ethyl-caproate), SIM= 0.587, isopentyl acetate= 0.564, phenyl acetate=0.562.
Cocoa Compounds Show Analogy to Acetylcholine
Computational alignment with Forge identified histamine as the cocoa compound (Table 4) with the strongest structural analogy to acetylcholine, as depicted in the following figure. Tetrahydrobetacarboline and phenylethylamine also demonstrated considerable similarity.
|
COCOA COMPOUNDS |
||||||||||
|
|
|
|
ACETYLCHOLINE |
ACETYLCHOLINE |
ACETYLCHOLINE |
ACETYLCHOLINE |
ACETYLCHOLINE |
ACETYLCHOLINE |
ACETYLCHOLINE |
ACETYLCHOLINE |
|
|
||||||||||
|
ACETYLCHOLINE |
|
1 |
|
|
|
|
|
|
|
|
|
HISTAMINE |
0,733 |
|
||||||||
|
TETRAIDROBETACARBOLINE |
|
|
0,653 |
|
|
|
|
|
||
|
PHENIL ETIL AMINE |
0,644 |
|
||||||||
|
TRIPTAMINE |
|
|
|
|
|
|
0,642 |
|
|
|
|
SAFFROLE |
0,599 |
|
||||||||
|
VANILLIC ACID |
|
|
|
|
|
|
|
0,585 |
|
|
|
TEOFILLINE |
0,548 |
|||||||||
Figure 5: Analogy Scores (SIM in Scrrenshots) of Cocoa Compounds with ACh
Screenshot 3: Analogy scores (SIM in screenshots) of cocoa compounds with ACh see List of the chemicaland physical properties of the compounds for screenshot details in Method
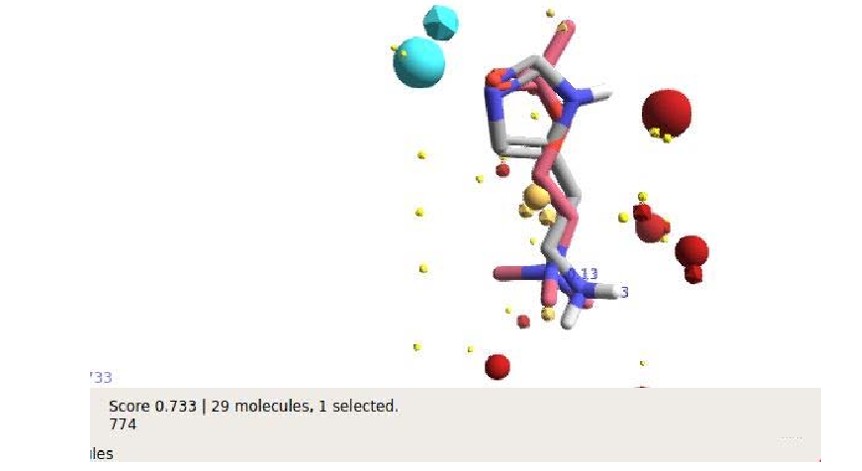
Figure 6: Histamine Acetylcholine Alignment (Overlapping). Histamine in Gray (Carbon Atoms) and Acetylcholine in Pink (Carbon Atoms), Nitrogen Atoms in Blue, and Oxygen in Red. Spheres for Acetylcholine Fields, Prisms for Histamine
Alignment of Cocoa Compounds with Serotonin
The cocoa compounds listed in Table 4 were aligned with serotonin, using the structure of serotonin bound to the mouse serotonin 5-HT3 receptor (F conformation, Protein Data Bank ID: 6HIN) as a reference.
|
COMPOUND SEROTONIN SEROTONIN SEROTONIN SEROTONIN SEROTONIN SEROTONIN SEROTONIN |
|||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||
|
SEROTONIN |
|
|
1 |
|
|
|
|
|
|
|
TRIPTAMINE O,878 |
|||||||||
|
TETRAIDROBETACARBOLINE |
|
|
O,816 |
|
|
|
|
||
|
SAFROLE |
0,766 |
|
|||||||
|
PHENILRTIAMINE |
|
|
|
|
0,738 |
|
|||
|
HISTAMINE |
0,703 |
|
|||||||
|
CAFFEIC ACID |
|
|
|
|
|
|
|
0,699 |
|
Figure 7: Scores of Cocoa Compounds Related to Serotonin
Tryptamine, tetrahydrobetacarboline, saffrole, and phenylethyl- amine exhibited remarkable analogy with serotonin. The 5-HT3 receptor, considered a target for antidepressant drugs, functions as an ion channel (allowing the passage of sodium and potassi- um) and its antagonists are of interest [5]. However, the analogy scores obtained in this study do not indicate whether the cocoa compounds are agonists or antagonists of this receptor.
Screenshot 5: provides a list of the chemical and physical properties of the compounds for image interpretation.
Alignment of Norepinephrine and Cocoa Compounds
To explore potential interactions between cocoa constituents and the adrenergic system, a ligand-based virtual screening approach was employed. This screening focused on identifying cocoa compounds (listed in Table 4) that exhibit structural similarity to norepinephrine. The three-dimensional structure of norepinephrine bound to the Anopheles gambiae D7R4 receptor (Protein Data Bank, PDB DOI: 10.2210/pdb2QEO/pdb) served as the template for this comparison, defining the spatial and electronic features that were considered relevant for ligand binding and activity at adrenergic receptors. The analogy scores derived from this screening provide insights into the potential for these cocoa compounds to interact with norepinephrine-binding sites.
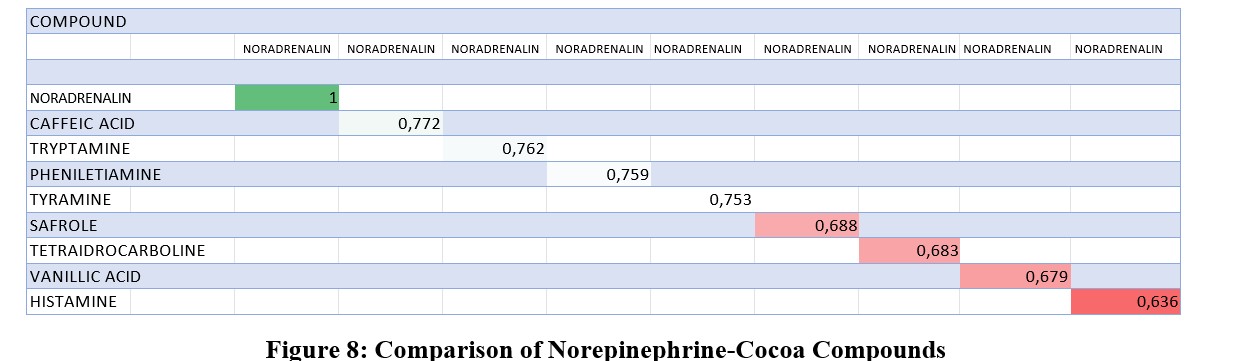
Caffeic acid exhibited the greatest analogy to norepinephrine, while tryptamine, phenylethylamine, and tyramine (typically abundant in aged cheeses) also showed high similarity scores. Screenshot 6 provides a detailed list of the chemical and physical properties of these compounds. Notably, nicotine, an analogue of acetylcholine (among whose receptors are nicotinic and muscarinic subtypes), modifies gustatory perception by increasing sensitivity to bitter and harsh substances and decreasing sensitivity to sweetness (reference 5, Iglesias et al.) [4].
Conclusion
In this study, a comprehensive computational investigation was undertaken to explore the potential molecular relationships between volatile organic compounds present in various food matrices – specifically beer, hops, and cocoa – alongside characteristic flavor compounds from diverse food sources, and three key neurotransmitters known to modulate sensory perception: acetylcholine, serotonin, and norepinephrine. Leveraging ligand- based computational methodologies, we aimed to identify structural and physicochemical similarities, quantified as analogy scores, that could provide insights into how these food-derived molecules might interact with biological systems involved in taste and olfaction. The foundational premise, supported by the work of Dando et al. and Högger et al., posits that these neurotransmitters play a significant role in enhancing the perception of flavors through intricate cellular mechanisms within taste pathways.
Our analysis revealed notable structural analogies between acetylcholine and several food-related compounds. Histamine, a compound found in cocoa, exhibited a considerable degree of similarity to acetylcholine, suggesting a potential for interaction with cholinergic systems. Similarly, isoamyl acetate, a key aroma component in bananas, and 2-heptanone, characteristic of gorgonzola cheese, also displayed significant analogy scores with acetylcholine. Furthermore, (-)-carvone, the primary flavorant in mint, showed measurable structural resemblance. These findings hint at the possibility that these aroma compounds might, to some extent, engage with cholinergic receptors or related enzymes, potentially influencing sensory perception.
The investigation into serotonin revealed particularly strong structural similarities with several cocoa-derived compounds, namely tryptamine, tetrahydrobetacarboline, and saffrole. Given the established role of serotonin and the 5-HT3 receptor as a target for antidepressant drugs, these analogies suggest a potential for cocoa components to interact with serotonergic pathways, although further studies would be required to determine the nature and functional consequences of such interactions (i.e., agonist or antagonist activity). Norepinephrine, another crucial neurotransmitter, displayed the highest structural similarity with caffeic acid, a compound also prevalent in various polyphenolic structures, including rosmarinic acid. This observation raises the possibility that caffeic acid, and potentially other related polyphenols, might interact with adrenergic receptors or related biological targets.
Interestingly, the ligand-based screening of volatile compounds contributing to the characteristic aromas of beer revealed an almost linear trend between the calculated coefficient of similarity with acetylcholine and the reported olfactory threshold for these compounds. This suggests a potential correlation between the structural mimicry of acetylcholine and the perceived intensity of these aroma volatiles. A similar trend was also observed for the taste-characterizing compounds. In conclusion, this computational study provides a valuable framework for understanding potential molecular relationships between food-derived compounds and key neurotransmitters involved in sensory perception. The identified analogies warrant further investigation, particularly through in vitro and in vivo studies, including organoleptic evaluations with human sensory panels, to validate the functional significance of these structural similarities and to elucidate how the addition of these compounds might indeed influence the perceived taste and aroma of food. Such interdisciplinary approaches could have significant implications for the food industry in terms of strategically selecting and enhancing the organoleptic profiles of food products [6-8].
Acknowledgment
Computational calculations and depictions of molecules are performed with Forge, thanks to Cresset for the academic license.
Artificial Intelligence Declaration
While the core research and ideas are entirely my own, I would like to clarify that the text was reviewed and refined for grammar and improved clarity with the assistance of Gemini 2.0.
Funding Statement
This research was self-funded by the author's agricultural activities.
References
- Dando, R., & Roper, S. D. (2012). Acetylcholine is released from taste cells, enhancing taste signalling. The Journal of physiology, 590(13), 3009-3017.
- Chaudhari, N., & Roper, S. D. (2010). The cell biology of taste. The Journal of cell biology, 190(3), 285.
- Heath, T. P., Melichar, J. K., Nutt, D. J., & Donaldson, L. F. (2006). Human taste thresholds are modulated by serotonin and noradrenaline. Journal of Neuroscience, 26(49), 12664- 12671.
- Blanco, C. A., Andrés-Iglesias, C., & Montero, O. (2016). Low-alcohol beers: Flavor compounds, defects, and improvement strategies. Critical reviews in food science and nutrition, 56(8), 1379-1388.
- Gupta, D., Prabhakar, V., & Radhakrishnan, M. (2016). 5HT3 receptors: Target for new antidepressant drugs. Neuroscience & Biobehavioral Reviews, 64, 311-325.
- Aberl, A., & Coelhan, M. (2012). Determination of Volatile Compounds in Different Hop Varieties by Headspace-Trap GC/MSî?¸ In Comparison with Conventional Hop Essential Oil Analysis. Journal of agricultural and food chemistry, 60(11), 2785-2792.
- Pullicin, A. J., Kim, H., Brinkman, M. C., Buehler, S. S., Clark, P. I., & Lim, J. (2020). Impacts of nicotine and flavoring on the sensory perception of e-cigarette aerosol. Nicotine and Tobacco Research, 22(5), 806-813.
- Cabras, P., Martelli, A. Chimica degli Alimenti. Edises: Napoli, Italy.
Appendix
Appendix: Alignment Screenshot Figures
Atthe alignment acetylcholine (pink) isoamyl acetate (gray), separated and overlapped, the spheres represent the electrostatic fields of isoamylacetate, the prisms of acetylcholine