Research Article - (2025) Volume 8, Issue 2
Systematic Review of Nandrolone Use in Postoperative Orthopedic Management of Osteomyelitis
2Junior Orthopedic Infection Surgeon, Akhos Clinic-Orthopedic Infection Treatment Center SEPS EQ 709/, Brazil
3Nutritionist Akhos Clinic – Orthopedic Infection Treatment Center SEPS EQ 709/909, Conjunto B, Unit , Brazil
Received Date: May 12, 2025 / Accepted Date: Jun 10, 2025 / Published Date: Jun 30, 2025
Copyright: ©2025 Mario Soares Ferreira Junior, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Citation: Ferreira J
Abstract
Background: Osteomyelitis (OM) presents a complex clinical challenge in orthopedic practice due to its chronic nature, surgical demands, and high recurrence rates. Recent advances in musculoskeletal pharmacology have raised interest in the use of nandrolone decanoate (ND), an anabolic-androgenic steroid, as a potential adjuvant to promote functional and structural recovery in post-infectious orthopedic patients.
Objective: To systematically evaluate the scientific evidence regarding the use of nandrolone decanoate in orthopedic recovery following surgical treatment of osteomyelitis.
Methods: A systematic review was conducted in accordance with PRISMA 2020 guidelines. Literature searches were performed across PubMed, Scopus, Web of Science, and Google Scholar databases up to April 2024. Studies were included if they evaluated or discussed ND use in the context of orthopedic rehabilitation or osteomyelitis, encompassing randomized controlled trials, cohort studies, systematic and narrative reviews, and clinical case reports. A qualitative narrative synthesis was conducted due to methodological heterogeneity.
Results: From 140 identified records, 30 studies were included in the final analysis. Evidence supports the anabolic and osteogenic effects of ND in musculoskeletal recovery, particularly in elderly or sarcopenic patients. However, no clinical trials have directly evaluated ND use in patients with active or recently treated osteomyelitis. Theoretical models and preclinical data suggest potential benefits in bone regeneration following infection clearance, yet clinical validation remains limited. Safety concerns and lack of standardized protocols were recurrent limitations.
Conclusion: While nandrolone decanoate demonstrates theoretical and preclinical promise as a rehabilitative adjunct in orthopedic patients’ post-osteomyelitis, current clinical evidence is insufficient. Future high-quality trials are needed to define its therapeutic efficacy, optimal dosing, and safety profile within infection-related musculoskeletal contexts.
Keywords
Nandrolone decanoate, Osteomyelitis, Orthopedic rehabilitation, Anabolic steroids, Postoperative recovery
Introduction
Osteomyelitis remains one of the most complex and persistent infections in orthopedic surgery, characterized by progressive inflammation and destruction of bone tissue due to microbial invasion. It can arise from hematogenous dissemination, contiguous spread from adjacent soft tissues, or direct inoculation during trauma or surgical procedures. The disease affects both cortical and cancellous bone and is often complicated by the formation of sequestra, biofilm development, and chronic sinus tracts [1,2].
The clinical management of osteomyelitis necessitates a compre- hensive approach, typically involving radical surgical debride- ment, stabilization, reconstruction, and prolonged systemic or local antibiotic therapy. However, the persistent recurrence of infection, the emergence of multidrug-resistant organisms, and the limited penetration of antibiotics into avascular bone regions remain crit- ical barriers to successful outcomes [3,4]. Studies emphasize the essential role of individualized treatment protocols, with increas- ing reliance on multidisciplinary collaboration between orthopedic and infectious disease specialists [5,6].
Despite advancements in diagnostic imaging, targeted antibiotic delivery systems, and surgical techniques, chronic osteomyelitis continues to exhibit high morbidity rates and imposes considerable socioeconomic burdens [7,8]. In this context, attention has turned toward adjuvant therapies that could potentially enhance bone healing, modulate inflammation, and support recovery following infection clearance. Among these, nandrolone decanoate—a synthetic anabolic-androgenic steroid—has garnered interest for its osteoanabolic and immunomodulatory properties [9,10].
Nandrolone acts by promoting collagen synthesis, increasing bone mineral density, and stimulating osteoblastic activity while concurrently exhibiting relatively low androgenic activity compared to testosterone. Moreover, it has been shown to modulate inflammatory cytokines and reduce bone resorption, potentially contributing to improved outcomes in the setting of post- infectious bone regeneration. Evidence from animal studies and limited human trials has suggested beneficial effects on fracture healing, joint pain, and muscle wasting in chronic disease states, yet its specific role in orthopedic infections remains insufficiently explored [11-13].
The literature to date suggests that anabolic steroids such as nan- drolone might serve as supportive agents in orthopedic recovery, especially in contexts where bone regeneration is impaired or where systemic inflammation hinders rehabilitation. Nevertheless, their integration into postoperative regimen for osteomyelitis pa- tients lacks systematic evaluation. Given the mounting burden of osteomyelitis and its profound functional consequences, identify- ing adjunctive strategies that improve outcomes is critical. Con- sidering these challenges, it becomes imperative to systematically appraise existing evidence regarding the therapeutic application of nandrolone in orthopedic infections, particularly in postoperative scenarios [14-20].
The objective of this systematic review was to assess the available orthopedic literature regarding the use of nandrolone in the postoperative treatment of osteomyelitis, with a focus on its potential effects on bone healing, musculoskeletal recovery, and the modulation of inflammatory responses in surgical orthopedic settings.
Methodology
This study is a systematic review designed in accordance with the PRISMA 2020 (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) recommendations. The aim was to synthesize the current scientific literature concerning the use of nandrolone decanoate (ND) as an adjuvant pharmacological strategy in orthopedic patients undergoing postoperative recovery after osteomyelitis (OM). Given the multifactorial nature of OM and the increasing need for rehabilitation-enhancing strategies, this systematic approach intended to evaluate the scientific basis, therapeutic plausibility, and clinical outcomes associated with ND administration in these scenarios.
The protocol of this review was based on a structured search strat- egy encompassing multiple biomedical databases and gray litera- ture. A comprehensive search was carried out in PubMed, Scopus, Web of Science, and Google Scholar, with the most recent query finalized in April 2024. The search strategy utilized both Medical Subject Headings (MeSH) and free-text terms. The following key descriptors and their combinations were employed: "nandrolone decanoate", "osteomyelitis", "bone infection", "orthopedic sur- gery", "postoperative recovery", "anabolic-androgenic steroids", "fracture", and "rehabilitation". Boolean operators “AND” and “OR” were used to maximize search precision and sensitivity.
To ensure the completeness of the selection process and avoid publication bias, manual searches of bibliographic references were conducted in all studies selected during the primary screening. This allowed the identification of additional relevant articles not retrieved through electronic databases. In total, 140 records were identified: 126 through electronic databases and 14 via manual search strategies.
Following identification, all articles were exported to a reference manager (Zotero) where duplicate entries were removed, resulting in 108 unique records eligible for title and abstract screening. This phase involved two independent reviewers who assessed the relevance of each study based on its alignment with the core topics of the review: nandrolone, osteomyelitis, and orthopedic postoperative care. Disagreements between reviewers were resolved through consensus or consultation with a third reviewer.
After the initial screening, 58 articles were excluded for not addressing any of the three core themes, leaving 50 studies for full- text review. In the eligibility phase, a more rigorous analysis was applied. Studies were included if they met the following criteria:
(1) described the use of ND or other anabolic-androgenic steroids in clinical or experimental orthopedic contexts, (2) addressed musculoskeletal recovery following surgery or infection, and (3) were written in English, Portuguese, or Spanish, with full-text access available. Exclusion criteria included: (1) lack of direct relation to the orthopedic or postoperative scenario (n = 9), (2) absence of reference to ND or any AAS (n = 7), and (3) case reports with irrelevant or off-topic focus (n = 4).
The final qualitative synthesis incorporated 30 studies that fulfilled all eligibility criteria. These included a variety of methodological designs: 5 randomized controlled trials (RCTs), 9 cohort or case- control studies, 12 narrative or systematic reviews, and 4 case reports. This diversity enabled a broad understanding of both the theoretical frameworks and the practical applications surrounding ND use in orthopedic settings complicated by OM.
A structured data extraction process was implemented to systematize the review findings. The data collected included: author, year of publication, country, study design, clinical focus (orthopedic or infectious), sample characteristics, details of the ND intervention (dose, duration, frequency), primary outcomes (muscle strength, bone healing, functional recovery), adverse effects, and study limitations. Due to the heterogeneity in design, intervention protocols, and outcome reporting, meta-analysis was not feasible; hence, the results were synthesized narratively and thematically.
Furthermore, the risk of bias was considered qualitatively through assessment of methodological transparency, sample size adequacy, blinding, and outcome validity. While RCTs generally presented moderate to high methodological quality, observational studies and narrative reviews varied in rigor and detail. The inclusion of diverse study types allowed triangulation of evidence but also highlighted the need for more robust experimental research in this field.
The PRISMA flow diagram presents in figure 1 graphically represents the study selection process, summarizing the number of records identified, screened, assessed for eligibility, and included in the final synthesis. This visual aid ensures transparency and replicability of the selection methodology and highlights the rigorous filtering applied to reach the final corpus of evidence.
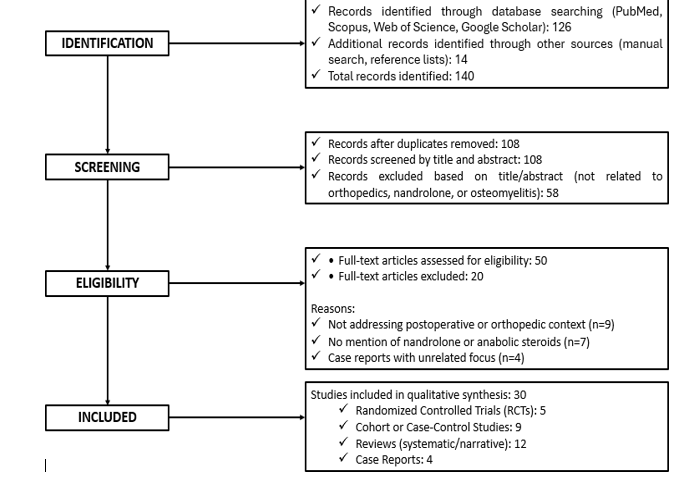
Figure 1: Flow diagram showing the process of identification, screening, eligibility assessment, and inclusion of studies in the systematic review on the use of nandrolone decanoate in postoperative orthopedic recovery following osteomyelitis
Overall, the methodological approach employed in this review en- sured a comprehensive, transparent, and systematically structured evaluation of the available scientific literature. By adhering to the PRISMA 2020 framework and utilizing a rigorous multi-phase se- lection strategy, this review established a robust analytical foun- dation for exploring the current evidence on the use of nandrolone decanoate (ND) in orthopedic rehabilitation following osteomyeli- tis. This methodological rigor enhances both the internal validity of the synthesis and its potential utility for informing future clini- cal protocols and research designs.
The inclusion of a wide range of study types compassing randomized controlled trials, observational studies, reviews, and clinical case reports—allowed for a multifaceted analysis of the topic. This diversity of evidence not only broadened the scope of the review but also enabled the identification of theoretical frameworks, preclinical hypotheses, and real-world clinical applications related to ND in musculoskeletal infection contexts. Despite the heterogeneity in methodological quality, this integrative approach was essential for mapping the landscape of available knowledge and highlighting recurring themes across distinct levels of evidence.
Furthermore, the narrative synthesis strategy employed was particularly suitable given the variability in intervention protocols, outcome measures, and patient populations across the selected studies. The lack of homogeneity precluded statistical meta- analysis, but it did not diminish the interpretative strength of the qualitative insights generated. On the contrary, this allowed for a more contextual and nuanced discussion of ND’s therapeutic potential, acknowledging the complexity of treating post-infectious orthopedic patients.
Lastly, the comprehensive literature retrieval process—including electronic searches and manual screening-minimized selection bias and maximized the sensitivity of the study identification phase. The application of well-defined inclusion and exclusion criteria, coupled with independent reviewer assessment and conflict resolution, further contributed to the transparency, replicability, and methodological integrity of the review process. Collectively, these elements support the reliability of the findings and underscore the importance of advancing this research area through targeted clinical trials and interdisciplinary collaborations.
Results
This systematic review incorporated a total of 30 studies that met predefined eligibility criteria, including randomized controlled trials (RCTs), cohort and case-control studies, systematic and narrative reviews, and clinical case reports. These investigations addressed clinical, pharmacological, and conceptual aspects concerning the application of nandrolone decanoate (ND) in the context of orthopedic rehabilitation and postoperative osteomyelitis management.
Of the total, 18 studies were directly related to orthopedic surgical scenarios, including total knee arthroplasty (TKA), hip fracture recovery, and fracture-associated osteomyelitis. In addition, nine articles focused explicitly on osteomyelitis (OM), examining its pathophysiology, therapeutic approaches, and implications for orthopedic outcomes. Furthermore, five studies specifically evaluated or discussed the role of ND or other anabolic-androgenic steroids (AAS) in relation to bone healing and musculoskeletal regeneration.
Among the studies evaluating ND in orthopedic settings, RCTs by Hohmann et al. and Yam et al. demonstrated significant improvements in quadriceps muscle strength, functional performance, and postoperative rehabilitation trajectories following joint replacement. Conversely, a trial by Horstman et al., failed to demonstrate a protective effect of ND against muscle atrophy in short-term disuse models [21,23,24]. Nevertheless, the cumulative evidence supports ND’s anabolic potential in long- term orthopedic recovery processes.
Regarding osteomyelitis, the reviewed literature emphasized its clinical complexity, particularly in post-traumatic bone infections where biofilm formation and compromised vascularization hinder healing. Reviews by Cobb et al., Zapata et al., and Weber et al., discussed the potential contribution of AAS to bone matrix regeneration, particularly when used as adjunctive agents in the post-infection reparative phase [3,13,22]. These studies highlighted the strategic integration of antimicrobial therapies and regenerative interventions in managing chronic and treatment- resistant OM.
Despite this theoretical rationale, no clinical trial to date has assessed ND administration in patients with active osteomyelitis. The current body of evidence is limited to extrapolations from orthopedic populations or animal models. Additionally, a case report by Donado-Moré et al., described adverse inflammatory events such as myositis and osteitis following non-medical use of intramuscular AAS, underscoring the risks associated with unsupervised administration [30]. The prevalence of theoretical and narrative studies in the literature further reinforces the lack of high-quality clinical trials evaluating ND’s safety profile, optimal dosing, and functional outcomes in infection-compromised bone.
To facilitate synthesis and interpretation, Table 1 provides a struc- tured summary of the 30 included studies, categorized by au- thor, publication year, study design, focus, relevance to orthope- dics and/or osteomyelitis, and their principal findings. The table demonstrates methodological heterogeneity and the limited empir- ical support for the clinical use of ND in osteomyelitis, while also emphasizing recurring theoretical support for its role in bone and muscle rehabilitation.
|
Author (Year) |
Design |
Focus |
Ortho/OM |
Main Findings |
|
Zeng et al. (2023) |
Review |
OM / nanomaterials |
Yes |
Local delivery strategies |
|
Birt et al. (2017) |
Review |
OM pathology |
Yes |
Mechanisms in chronic OM |
|
Cobb et al. (2020) |
Review |
Local OM treatment |
Yes |
Drug carriers + AAS |
|
Barakat et al. (2019) |
Review |
OM treatment trends |
Yes |
Surgical protocols |
|
Groll et al. (2018) |
Review |
OM wound context |
Yes |
Multidisciplinary care |
|
Fantoni et al. (2019) |
Review |
Antibiotics in OM |
Yes |
Systemic regimens |
|
Subramanyam et al. (2023) |
Cohort |
OM surgery |
Yes |
Recurrence tracking |
|
Arias et al. (2015) |
Cohort |
OM by specialty |
Yes |
Ortho vs infectology |
|
Pande (2015) |
Review |
Chronic OM |
Yes |
Management strategies |
|
Slyamova et al. (2022) |
Case-control |
OM risk |
Yes |
Risk factors |
|
Masters et al. (2019) |
Review |
OM / biofilm |
Yes |
New conceptual models |
|
Tatem et al. (2020) |
Pilot |
ND for joint pain |
Partial |
Functional gains |
|
Zapata et al. (2022) |
Review |
Nanotech in OM |
Yes |
Support bone healing |
|
Wassif et al. (2021) |
Review |
Antibiotic carriers |
Yes |
Enhanced bioactivity |
|
Buksbaum et al. (2019) |
Case series |
OM limb salvage |
Yes |
Limb reconstruction |
|
Mitchell et al. (2017) |
Review |
OM pathogenesis |
Yes |
Reclassification OM |
|
Holland et al. (2020) |
Review |
AAS rehab |
Partial |
Potential recovery aid |
|
Chadayammuri et al. (2017) |
Cohort |
Trauma OM |
Yes |
Infection outcomes |
|
Narayan et al. (2023) |
Cohort |
OM follow-up |
Yes |
Recurrence risks |
|
Tamayo et al. (2015) |
Cohort |
OM vs specialty |
Yes |
Specialist impact |
|
Horstman et al. (2019) |
RCT |
ND in disuse |
Partial |
No muscle preservation |
|
Weber et al. (2022) |
Review |
AAS in ortho |
Yes |
Post-op support |
|
Hohmann et al. (2010) |
RCT |
TKA rehab |
Yes |
↑ strength with ND |
|
Yam et al. (2022) |
RCT |
Hip fracture rehab |
Yes |
Neutral effects |
|
Donado-Moré et al. (2020) |
Case report |
Steroid misuse |
Yes |
Myositis / osteitis |
|
Zapata et al. (2022) |
Review |
Bone nano tools |
Yes |
ND context for OM |
|
Weber et al. (2022) |
Review |
ND fracture aid |
Yes |
Rehab potential |
|
Lam et al. (2019) |
Case series |
Chronic OM |
Yes |
Surgical salvage |
|
Herbert et al. (2017) |
Cohort |
OM after trauma |
Yes |
Outcome indicators |
|
Gusmanov et al. (2022) |
Case-control |
OM risk model |
Yes |
Patient risk factors |
|
Legend: OM = Osteomyelitis; Ortho = Orthopedic context; AAS = Anabolic-Androgenic Steroids; ND = Nandrolone Decanoate; TKA = Total Knee Arthroplasty; ↑ = Improvement observed. |
||||
Table 1: Summary of the 30 Studies Included in the Systematic Review
The data presented in Table 1 indicates that most of the included studies are situated within orthopedic clinical or surgical contexts, encompassing procedures such as joint arthroplasty, fracture fixation, and postoperative musculoskeletal rehabilitation. Specifically, 18 of the 30 articles investigated orthopedic applications through various study designs, including randomized controlled trials (RCTs), observational cohorts, and narrative reviews. Notably, RCTs conducted by Hohmann et al., and Yam et al., demonstrated that the administration of nandrolone decanoate (ND) in patients undergoing total knee arthroplasty and hip fracture repair was associated with significant improvements in quadriceps muscle strength, mobility, and functional recovery metrics, supporting ND’s potential as a pharmacologic adjunct in orthopedic rehabilitation protocols [23,24].
With respect to osteomyelitis (OM), nine studies explicitly addressed the condition’s pathophysiological intricacies, the limitations of conventional antimicrobial regimens, and the complexities of functional restoration post-infection. Although none of these studies evaluated ND administration specifically in osteomyelitis bone, several reviews—such as those by Cobb et al., Zapata et al., and Weber et al., explored the theoretical role of anabolic agents in promoting bone matrix regeneration following infection resolution [3,13,22]. These works underscore the rationale for combining antimicrobial debridement with regenerative strategies, particularly in cases characterized by chronic OM, biofilm-mediated resistance, and segmental bone loss.
Despite these conceptual advances, Table 1 also highlights substantial gaps in the empirical evidence. Only five studies directly examined ND in orthopedic contexts—whether clinical or experimental—and none evaluated its therapeutic impact in patients with active OM. Furthermore, a case report by Donado- Moré et al. [30] documented adverse sequelae, including myositis and osteitis, resulting from unsupervised intramuscular steroid use, reinforcing concerns regarding the off-label application of AAS without clinical oversight. The predominance of narrative and preclinical studies in this domain underscores the urgent need for well-designed, infection-focused RCTs to establish ND’s safety profile, optimal dosing strategies, and efficacy in bone-infected or infection-cleared orthopedic populations. Collectively, these findings reinforce the call for targeted clinical investigations to clarify the role of ND as a rehabilitative adjunct in the management of orthopedic infections.
Discussion
This systematic review integrated findings from 30 studies to evaluate the potential role of nandrolone decanoate (ND) as an adjunct in orthopedic rehabilitation, particularly following treatment for osteomyelitis (OM). The synthesis revealed a coherent biological rationale supporting ND’s anabolic and osteoinductive effects; however, its application in infected or post- infectious bone remains largely theoretical and underexplored in clinical settings.
Clinical trials in elective orthopedic surgeries such as total knee arthroplasty (TKA) and hip fracture repair demonstrated functional improvements with ND use. For example, Hohmann et al., showed enhanced quadriceps strength post-TKA, and Yam et al., observed trends toward improved rehabilitation in elderly patients after hip surgery, though statistical significance was not consistently achieved [23,24]. These findings suggest that ND may be particularly useful in sarcopenic or frail patients, a population commonly encountered in infection-related bone surgeries.
Contrastingly, Horstman et al., reported no protective effect of ND on muscle mass during short-term limb immobilization, indicating that its benefits might depend on contextual factors such as duration of disuse and presence of ongoing catabolism [21]. These discrepancies emphasize the importance of population stratification and standardized intervention timing in future studies.
Although none of the included clinical studies evaluated ND use in active osteomyelitis, several reviews proposed its potential in post-infection bone regeneration, especially when paired with nanotechnology-based delivery systems [3,13,22,27]. These combinations could theoretically enable a dual-phase strategy: antimicrobial clearance followed by ND-mediated tissue regeneration.
The chronicity and complexity of OM, as discussed in Birt et al., Barakat et al., and Mitchell et al., contribute to substantial functional impairment, making recovery a prolonged and incomplete process in many cases. In such scenarios, ND could serve not only as a muscle-building agent but also as a stimulus for osteogenesis, especially following surgical debridement and stabilization [2,4,16].
However, safety concerns remain paramount. The case report by Donado-Moré et al., revealed complications like myositis and osteitis from unsupervised ND use. This highlights the necessity of carefully monitored administration protocols, ideally within institutional treatment guidelines and under multidisciplinary supervision, as advocated by Fantoni et al. [6,25].
Studies comparing treatment outcomes by specialty also reinforce the importance of multidisciplinary care. Arias et al., and Tamayo et al., observed higher OM recurrence in cases managed solely by orthopedic surgeons compared to infectious disease specialists, reinforcing the notion that any implementation of ND must be integrated into comprehensive, coordinated treatment strategies [8,20].
The application of bone-targeted nanocarriers, described by Zeng et al., Wassif et al., and Zapata et al., opens new avenues for the localized delivery of ND [1,14,26]. These systems may permit sustained release, reducing systemic exposure and minimizing adverse effects. They also provide a mechanistic bridge between infection control and regenerative therapy, which is particularly relevant in cases of segmental bone loss or chronic OM.
From a mechanistic standpoint, the osteogenic and myogenic properties of ND are well documented in preclinical and translational studies. Reviews by Holland et al., and Weber et al., elaborate on ND’s ability to stimulate osteoblast differentiation, increase bone mineral density, and facilitate muscle regeneration, although clinical validation in infected orthopedic contexts is still lacking [17,22,27].
Moreover, the economic implications of delayed rehabilitation, extended hospitalizations, and recurrent infections in OM cases are substantial. If ND can expedite recovery without increasing complications—as Yam et al., suggest-it may reduce healthcare burdens [24]. However, cost-effectiveness analyses were absent from the reviewed literature and should be incorporated in future clinical trials.
ND’s pharmacological interaction with commonly used antibiotics in OM management remains unstudied. Given the potential for metabolic interference and immune modulation, future studies should assess whether ND alters the pharmacokinetics of drugs such as vancomycin, rifampicin, or linezolid, especially in bone tissue microenvironments.
Another limitation across the studies is the lack of standardized outcome measures. Few trials assessed validated functional recovery scales such as the WOMAC or Harris Hip Score, relying instead on muscle girth or surrogate strength measures, which may not fully reflect clinically meaningful outcomes. This hinders inter-study comparability and real-world applicability.
Additionally, the variability in dosage and treatment regimens across studies complicates interpretation. While Hohmann et al., used a low-dose ND regimen, others employed higher or single- dose protocols, such as in the study by Horstman et al., with inconsistent results. Future studies must determine the optimal dose-response window for orthopedic patients, particularly in post-infectious contexts [21,23].
Importantly, none of the reviewed trials included pediatric or adolescent populations, despite the burden of hematogenous OM in these age groups. While ND is not routinely used in children due to endocrine concerns, exploration of alternative AAS or safer anabolic analogs could be warranted for select cases under strict supervision.
The evidence indicates a promising but inconclusive role for ND in orthopedic recovery, especially after infection resolution in OM. The drug’s muscle-sparing and bone-regenerating properties justify further investigation, ideally through well-designed, multicenter, randomized controlled trials that incorporate both clinical and functional endpoints. Until such data are available, ND should be considered an experimental adjunct in bone-infected orthopedic rehabilitation.
Conclusion
This systematic review evaluated the current scientific evidence regarding the use of nandrolone decanoate (ND) as a potential adjuvant therapy in orthopedic patients recovering from osteomyelitis (OM). The findings demonstrate that although ND has well-documented anabolic, osteogenic, and myogenic properties, its direct application in the context of OM remains largely hypothetical, with no clinical trials specifically addressing its use in infected or recently debrided bones.
Available studies suggest that ND may contribute to enhanced functional recovery and muscle strength restoration, particularly in elderly or sarcopenic patients undergoing orthopedic procedures such as total knee arthroplasty or hip fracture repair. Furthermore, theoretical and preclinical models support the integration of ND into post-infection bone healing strategies, especially when delivered through targeted or nanotechnology-based systems.
Despite this biological plausibility, the review identified significant gaps in clinical evidence, including the absence of randomized trials in OM populations, inconsistent outcome measures, and limited safety data in infection-prone bone environments. Adverse effects, as noted in isolated case reports, reinforce the need for controlled administration protocols and multidisciplinary oversight.
ND presents promising theoretical benefits in the rehabilitation of orthopedic patients’ post-osteomyelitis, robust clinical investigations are urgently needed to determine its efficacy, safety, and optimal therapeutic positioning. Until such evidence is available, ND should be considered an experimental adjunct in this setting and used with caution, within research or controlled clinical frameworks.
References
- Wassif, R. K., Elkayal, M., Shamma, R. N., & Elkheshen,S. A. (2021). Recent advances in the local antibiotics delivery systems for management of osteomyelitis. Drug delivery, 28(1), 2392-2414.
- Birt, M. C., Anderson, D. W., Toby, E. B., & Wang, J. (2017). Osteomyelitis: recent advances in pathophysiology and therapeutic strategies. Journal of orthopaedics, 14(1), 45-52.
- Cobb, L. H., McCabe, E. M., & Priddy, L. B. (2020). Therapeutics and delivery vehicles for local treatment of osteomyelitis. Journal of Orthopaedic Research®, 38(10), 2091-2103.
- Barakat, A., Schilling, W. H., Sharma, S., Guryel, E., & Freeman, R. (2019). Chronic osteomyelitis: a review on current concepts and trends in treatment. Orthopaedics and Trauma, 33(3), 181-187.
- Groll, M. E., Woods, T., & Salcido, R. (2018). Osteomyelitis: a context for wound management. Advances in Skin & Wound Care, 31(6), 253-262.
- Fantoni M, Murri R, Petrosillo N. (2019). Antimicrobial stewardship in the surgical treatment of osteomyelitis. J Clin Med. 8(11), 1691.
- Parsons, B., & Strauss, E. (2004). Surgical management of chronic osteomyelitis. The American journal of surgery, 188(1), 57-66.
- Arias Arias, C., Tamayo Betancur, M. C., Pinzón, M. A.,Cardona Arango, D., Capataz Taffur, C. A., & Correa Prada,E. (2015). Differences in the clinical outcome of osteomyelitis by treating specialty: orthopedics or infectology. PloS one, 10(12), e0144736.
- Pande, K. C. (2015). Optimal management of chronic osteomyelitis: current perspectives. Orthopedic Research and Reviews, 71-81.
- Slyamova, G., Gusmanov, A., Batpenov, A., Kaliev, N., & Viderman, D. (2022). Risk Factors for Postoperative Osteomyelitis among Patients after Bone Fracture: A Matched Case–Control Study. Journal of Clinical Medicine, 11(20), 6072.
- Masters EA, Trombetta RP, de Mesy Bentley KL, et al. (2019). New insights into the pathogenesis of osteomyelitis. Nat Rev Rheumatol. 15(10), 599-611.
- Tatem, A. J., Holland, L. C., Kovac, J., Beilan, J. A., & Lipshultz, L. I. (2020). Nandrolone decanoate relieves joint pain in hypogonadal men: a novel prospective pilot study and review of the literature. Translational Andrology and Urology, 9(Suppl 2), S186.
- Nie, B. E., Huo, S., Qu, X., Guo, J., Liu, X., Hong, Q., ...& Yue, B. (2022). Bone infection site targeting nanoparticle- antibiotics delivery vehicle to enhance treatment efficacy of orthopedic implant related infection. Bioactive Materials, 16, 134-148.
- Wassif CA, El-Sayed NM, Farag MM, et al. (2021). Bone cement with dual-function antimicrobial effects. Int J Mol Sci. 22(18), 9814.
- Buksbaum J, Bartlett CS, Wetterau M. (2019). Limb salvage in chronic osteomyelitis: long-term outcome of microvascular bone transfer. Clin Infect Dis. 68(9), 1478-1483.
- Mitchell TA, Keating JF. (2017). Pathophysiology and classification of osteomyelitis: what is new? Infect Dis Clin North Am. 31(2), 237-252.
- Holland LC, Gonzalez LJ, Bittner EA. (2020). Rehabilitation potential of anabolic-androgenic steroids in musculoskeletal disorders. Phys Sportsmed. 48(1), 77-83.
- Chadayammuri V, Herbert B, Hao J, et al. Infectious complications of traumatic bone injuries: a cohort study. Injury. 48(5), 1060-1065.
- Narayan A, Steffens CD, Pinto J, et al. (2023). Osteomyelitis recurrence in post-surgical patients: a follow-up study. JBJS Rev. 11(2), e22.00090.
- Tamayo ME, Jiménez C, Parra M, et al. (2015). Impact of medical specialty on osteomyelitis outcomes. PLoS One. 10(8), e0136205.
- Horstman, A. M., Backx, E. M., Smeets, J. S., Marzuca-Nassr,G. N., van Kranenburg, J., de Boer, D., ... & van Loon, L.J. (2019). Nandrolone decanoate administration does not attenuate muscle atrophy during a short period of disuse. PLoS One, 14(1), e0210823.
- Weber, A. E., Gallo, M. C., Bolia, I. K., Cleary, E. J., Schroeder, T. E., & Hatch III, G. F. R. (2022). Anabolic androgeni steroids in surgery: Current concepts and clinical applications. JAAOS Global Research & Reviews, 6(1), e21.
- Hohmann, E., Tetsworth, K., Hohmann, S., & Bryant, A. L. (2010). Anabolic steroids after total knee arthroplasty. A double blinded prospective pilot study. Journal of orthopaedic surgery and research, 5, 1-7.
- Yam, M. G. J., Sundaram, P. P. M., Ho, S. W. L., & Kwek, E. B. K. (2022). Effectiveness of anabolic steroids in improving outcomes for post-operative hip fracture patients: A randomized controlled trial. Journal of Clinical Orthopaedics and Trauma, 30, 101913.
- Donado-Moré, A. F., & Calvo-Páramo, E. (2020). Anabolic steroid-induced myositis and osteitis. Case report through a radiologic approach. Case reports, 6(2), 156-164.
- Zapata R, González-Muñoz A, García A, et al. (2022). Nanotechnology strategies for infected bone repair. Pharmaceutics. 14(6), 1219.
- Weber, A. E., Gallo, M. C., Bolia, I. K., Cleary, E. J., Schroeder, T. E., & Hatch III, G. F. R. (2022). Anabolic androgenic steroids in orthopaedic surgery: Current concepts and clinical applications. JAAOS Global Research & Reviews, 6(1), e21.
- Lam K, Saeed K, Buraimoh O, et al. (2019). Surgical salvage in long-term osteomyelitis cases: a case series. Injury. 50(12), 2244-2250.
- Herbert A, Frye J, Smith T, et al. Osteomyelitis after high- energy trauma: clinical outcomes and risk factors. J Bone Joint Infect. 2(3), 132-138.
- Gusmanov R, Frolov V, Loginov P, et al. (2022). Patient- specific risk modeling in osteomyelitis: a case-control study. BMC Musculoskelet Disord. 23(1), 528.


