Research Article - (2025) Volume 10, Issue 2
Systematic Review And Meta Analysis On Prevalence Of Opticospinal Multiple Sclerosis In Asian Population
Received Date: Dec 02, 2025 / Accepted Date: Dec 03, 2025 / Published Date: Dec 05, 2025
Copyright: ©Â©2025 Benson Rayan D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Rayan, B., Swethakumar, M., Poongothai, J. (2025). Systematic Review And Meta Analysis On Prevalence Of Opticospinal Multiple Sclerosis In Asian Population. Int J Psychiatry, 10(2), 01-12.
Abstract
Multiple sclerosis is a chronic, inflammatory disorder of the central nervous system which is prevalent across the Western population. Employing current technologies, it has been discovered that a subtype of MS which results in severe damage of spinal cord and optic nerves exists in Asian populations. A binary random-effect meta-analysis carried out in the scope of this study illuminates the mean prevalence rate of OSMS in Asia, including studies from regions East Asia, Sound-East Asia, Central Asia, Middle Eastern. The findings estimated a mean prevalence ratio of around 0.340 of the selected 25 studies out of 117 screened studies with a confidence level of 95%. Regarding regional disparities in MS prevalence, Japan serves as an example of a country with moderate rates and a significant portion of the data used in this study is derived from the Japanese population. In contrast, countries within the Indian subcontinent, such as India and Pakistan, as well as certain Middle Eastern nations Iran and Oman, tend to exhibit lower MS prevalence rates.
Keywords
Meta-Analysis, Optic Spinal Multiple Sclerosis, Multiple Sclerosis
Introduction
Multiple Sclerosis (MS) is a chronic autoimmune disease that affects the central nervous system (CNS), which includes the brain and spinal cord [1,2]. The exact cause of MS is unknown, but it is believed to be an immune-mediated disease in which the body's immune system mistakenly attacks its own tissues, specifically the protective coating on nerve fibers called myelin [1].This immune system malfunction leads to inflammation, demyelination, gliosis, and neuronal loss in the CNS2.
In Asians, classical MS is rare and instead a type of MS with severe involvement of optic nerves is prevalent. Optic spinal MS is characterized by selective damage of optic nerves and spinal cord. Using MRI scans, longitudinally extensive spinal cord lesions are visualized, which are specific to OSMS [3]. People having MS with main lesions that are present in the spinal cord and optic nerves with no lesions found in the cerebrum or cerebellum are classified under OSMS. Moreover, patients with OSMS are prone to develop brainstem signs like double vision along with the rest of the symptoms [4].
OSMS is said to have similar features to the relapsing form of NMO which is common in Western populations because of the detection of NMO - IgG in nearly 60% of OSMS patients [3]. These antibodies are prevalent during different durations of OSMS in the patient. There are also cases where these antibodies are simply not present and these individuals have extremely long spinal cord lesions with optic nerve defects. A study has been conducted on the Japanese population which proves that the mechanism of lesion production in OSMS is heterogeneous, meaning it can be related to antibody production or completely unrelated [5].
The objective of this study is to perform a systematic review on optic spinal multiple sclerosis and to perform meta - analysis on the prevalence of the OSMS in the Asian population.
This could provide a clear view on the prevalence in the Asian population and offer insights into the epidemiology. It could also serve to enhance clinical understanding by expanding knowledge about OSMS which can influence diagnostic procedures, treatment plans and prognosis estimation. Moreover, this study aims to increase awareness about the OSMS variant among the Asian population.
Review of the Literature
History
In the late 1950s, MS was rarely reported in Asia but later on it was discovered that MS with slightly different characteristics were present. In a research done on 270 patients in Japan, 48% of cases showed features that were the intermediate of both MS and NMO [6]. After this, for a short while, NMO was used to classify monophasic cases showing bilateral optic neuritis and transverse myelitis within an interval of less than several weeks and relapsing cases were usually classified as classical MS.
Comparative studies conducted between different populations highlighted the unique features of MS in Asians. These helped in further classification of MS according to severity and location of lesions.
Autoimmune Disorders & MS
Autoimmune disorders occur when the immune system mistakenly attacks the body's own healthy cells and tissues, leading to chronic inflammation and tissue damage. There are over 80 known autoimmune diseases, affecting various organs and systems, such as rheumatoid arthritis, multiple sclerosis, and type 1 diabetes. The exact causes of autoimmune disorders remain complex and multifactorial, involving genetic predisposition and environmental triggers [7].
Multiple sclerosis is a chronic autoimmune disorder that affects the central nervous system. The immune cells attack the protective myelin sheath present around the nerve fibres. Symptoms of multiple sclerosis vary widely and include fatigue, difficulty walking, numbness, muscle weakening etc. and it has different expression levels depending on the individual [3].
In Asians, MS is rare; however, when it appears, the selective and severe involvement of the optic nerves and spinal cord is characteristic. This form, termed optic spinal MS (OSMS) [8]. It has similar features to the relapsing form of NMO (Neuromyelitis Optica) in Western populations. The cause for the rare occurrence of MS in India and other tropical and subtropical countries may lie in environmental and genetic factors or both.
Factors Involving Multiple Sclerosis
The exact cause for MS is unknown, but it is known that a lot of factors contribute to the disease making it a multifactorial disorder, including genetic and epigenetic factors. The interplay of these factors is complex and not fully understood. Specific factors contributing to the development of MS, may vary from person to person. Factors influencing MS include gender, geographical location, vitamin D and B12 deficiency, race/ethnicity, age and in some cases genetics as well. Environmental factors like smoking, drinking etc. can also contribute to MS [9].
Opticospinal Multiple Sclerosis
Multiple sclerosis (MS) is a demyelinating disease of the central nervous system (CNS) while neuromyelitis optica (NMO) is an inflammatory disease of the CNS that selectively affects the optic nerves and spinal cord. It has similar features to the relapsing form of NMO (Neuromyelitis Optica) in Western populations. In this condition, longitudinally extensive spinal cord lesions (LESCLs) extending over three vertebral segments are said to be characteristic of magnetic resonance imaging (MRI). Pathologically, both axons and myelin are involved, resulting in necrotic cavitation. This form of MS generally has a higher age at onset and a higher female to male ratio than conventional MS.
There are certain criteria that need to be met for the patient to be classified under OSMS. Clinically estimated main lesions in these cases must be confined to the optic nerve and spinal cord and no cerebellar or cerebral symptoms must be present. Minor brainstem signs are acceptable and at least one relapse must have occurred [10].
Diagnostic Tests for MS
Blood tests serve as pivotal tools, not merely for detecting MS but importantly, for excluding other diseases that manifest with similar symptoms. An essential diagnostic procedure is the spinal tap, or lumbar puncture, where a sample of cerebrospinal fluid is extracted from the spinal canal. Analysis of this fluid can reveal irregularities in antibodies, often associated with MS, and simultaneously eliminate the possibilities of infections and other conditions mimicking MS symptoms [11]. The MRI technique uncovers characteristic MS lesions on the brain and both the cervical and thoracic sections of the spinal cord. Intravenous injections of contrast materials, primarily iodine-based or gadolinium-based elements, are employed for MRI.
MS in Asian Population
Overview of Selected Studies
Several countries have examined their population for the prevalence of MS. Among these countries, Asian countries seem to have higher prevalence of the OSMS variant. For instance, comparison of the British and Japanese population has shown that there are three distinct differences between the populations. The Japanese population suffered a greater visual loss, severe involvement of spinal cord lesions and increased visual disability than the British population which all points to the existence of the OSMS variant in the Japanese population [12].
Prevalence Rates in Different Asian Regions
The prevalence of classical MS across Asia is low. Countries like Taiwan, Hong Kong, India exhibits a low prevalence of <1 per 100,000 cases while Japan, Korea, Malaysia and Thailand showcase 1-5 cases for every 100,000 people. Medium prevalence is seen in countries like Saudi Arabia, Kuwait, Jordan with 5 - 29 cases per 100,000 [4] . The low prevalence regions of Asia have a higher occurrence of OSMS. In Hong Kong, 36% of cases are reported as OSMS. In south and north Japan, 57% and 17% of all MS cases are optic spinal variants [4].
In a study conducted in the Bombay region of India, out of 74 patients, 9 were diagnosed with NMO while 6 patients were diagnosed with an intermediate condition that existed between NMO and classical MS. Optic nerves were affected in some patients while spinal cord was affected in the rest in the case of NMO patients [13]. In the intermediate group, the sites of lesions were not present in both the optic nerves and spinal cord but in only one of these sites and the onset of this has been reported to be slower than NMO and classical MS.13 Based on a paper in the 1970s, the prevalence rate of MS was shown to be just 0.17 - 1.33 per 100,000 in different parts of India while it was nearly 90 to 150 per 100,000 in the US and UK [14].
In Iran, another study was conducted and 520 patients from two different hospitals were examined out of which only 20 were diagnosed with OSMS accounting to a prevalence rate of 3.8%. Out of these patients, 60% had spinal signs, 35% had optic neuritis in one eye and just 5% of patients had the involvement of both the eyes. These patients showed no signs of brainstem or cerebellar lesions, and the CSF contained normal cell count and protein content [15 ]. 118 patients were examined using MRI scans in Japan and 30.5% were diagnosed with clinical OSMS. Out of the 36 cases, 10 cases were said to have pure OSMS, meaning the patients had frequent relapses and the severity of the disabilities also increased. According to this paper, the clinical OSMS patients had less than 4 cerebral lesions and along with other symptoms they did not qualify for the CMS criteria. Moreover, the pure OSMS patients had extensive spinal cord involvement and female bias as compared to the clinical variant [16].
According to K-H Chang, R-K Lyu who analyzed MRI of patients in Taiwan, 56% were found to have OSMS while the remaining out of the 74 patients had classical MS. The most common symptoms include weakness in limbs followed by loss of vision. The prevalence rate of CMS is shown to be low in Taiwan and this study agrees with that by depicting a higher prevalence of OSMS along with rapid progression of disability which was measured using EDSS scores [17].
The studies conducted in different countries proves that there is a varied prevalence of OSMS across Asia countries with Japan accounting for the highest number of cases. Other comparisons, for example, studies conducted on African American and Caucasians have shown that the former have a higher chance of developing optic spinal MS than the latter [18].
Temporal Trends in Prevalence
There has been increased recognition with several advances in MRI technology. Additionally, with the identification of the AQP4- IgG antibody associated with NMO, there has been improved recognition and differentiation between typical MS, OSMS, and NMO which has assisted to more accurate diagnosis [19].
The establishment of proper diagnostic criteria for the various types and subtypes of multiple sclerosis has evolved over time. This includes McDonald’s criteria, Paty criteria, Poser criteria which are based on brain lesions observed using MRI. As these criteria get refined, the prevalence of the disease shifts [20].
The environmental factors influencing multiple sclerosis, although known to a certain extent, are not completely understood yet. As these factors keep changing over time, new trends can be visualized in the progression of MS.
Variations by Age and Gender
Several trends have been observed across various parts of Asia with a common bias towards females. The female population tends to be more affected by this disorder than males in general. With respect to a review conducted on MS patients in Taiwan from 1993 to 2001, the female to male ratio is 4.4. The mean onset age is nearly 37 years (37.6±11.1 years) for OSMS cases. The mean annual relapse rate is also higher, around 62.7% for OSMS patients [17].
On the contrary, a study in Korea showcases a higher number of male cases to females with the mean onset age of 35.6 years [21]. The Malaysia population has a sex ratio (F:M) to be 5:1 according to Chong-Tin Tan who also states that China has a female to male ratio of 1.12:1 and Japan has a ratio of 1.5:1.22 In Iran, the female to male ratio is 2.3:1 with a mean onset of 24± 8.2 years in the presence of a low prevalence rate with an annual relapse rate of 0.66± 0.84.15 [22].
A study published on 146 Indian patients revealed that this country has a female to male ratio of 1.21:1. The mean onset age is 29.38 ± 10.09 years. 37.6% patients exhibited optic spinal MS and the rest had non - OSMS symptoms [23].
Methodology
Systematic Review
Various databases like MEDLINE, PubMed, Google Scholar were used to obtain information in the form of literary text. The search was done using relevant keywords in a proper Boolean equation, “multiple sclerosis ”+”optic spinal”+(prevalence) and using other key words including - population, Asia, European, epidemiology etc. We primarily selected only studies in the English language.
|
CRITERIA FOR INCLUDING AND EXCLUSION OF PAPERS |
|
|
INCLUSION |
EXCLUSION |
|
Paper based on any population |
Papers subjecting NMO under OSMS |
|
Papers published within the years 1970 to 2023 |
Papers in languages other than English |
|
Studies that are published in peer-reviewed journals |
Studies that do not report on outcomes related to opticospinal multiple sclerosis |
Table 1: Tabulation of Inclusion and Exclusion Criteria For Screening of Articles and Papers for as a Source of Literary Text for Literature Review
Screening of studies was done initially by abstract text screening and then further by full-text screening involving reading the full text of each article identified for inclusion in the review and extracting the pertinent data, required information was extracted from the studies and noted down [24].
The literature is organized into categories or themes that help in the synthesis of information. This generally involves grouping studies by methodology, key concepts, or time periods. The findings are analyzed from the selected sources within each category and key findings, patterns, and gaps in the existing literature are highlighted. It also includes a discussion of the implications of the reviewed literature and how the existing knowledge contributes to the study. The key points are summarized from the literature review and the references are properly cited in the IEEE format.
Meta-Analysis
To perform meta-analysis we used MEDLINE, PubMed as the main source of data for the analysis. The search was done using relevant keywords in a proper Boolean equation, “multiple sclerosis ”+”optic spinal”+(Asia), to perform an efficient search. Results dating from 1970 to 2023 including only papers in English language. 476 papers and articles were identified, Several inclusion and exclusion criteria were set for screening the data.
|
CRITERIA FOR INCLUDING AND EXCLUSION OF PAPERS |
|
|
INCLUSION |
EXCLUSION |
|
Paper based on Asian population |
Papers including NMO cases under opticospinal |
|
Papers published within the years 1970 to 2023 |
Papers with cases of less than 15 |
|
Only epidemiology studies |
Not in English language |
|
Studies that are published in peer-reviewed journals |
Studies that do not report on outcomes related to opticospinal multiple sclerosis |
Table 2: Tabulation of Inclusion and Exclusion Criteria for Screening of Articles and Papers for as a Source of Data for Meta Analysis
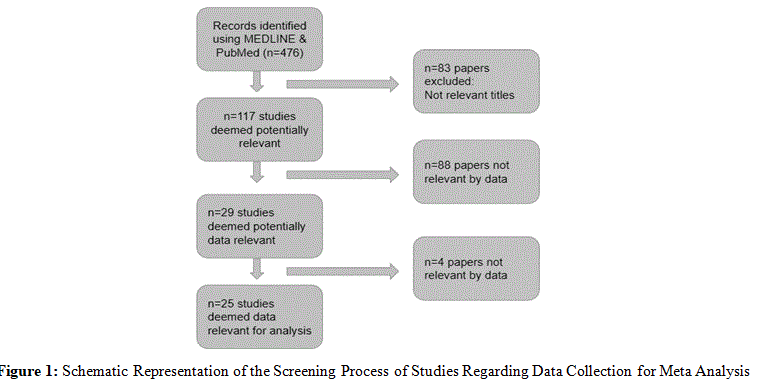
Data was extracted from the screened papers, data including opticospinal cases percentage or numbers, sample space number, sex-ratio. Other data including prevalence per 100000 and mean age were also extracted though aren’t of utmost priority. These data are tabulated and saved.
Data pooling involves combining the results of multiple studies that were screened according to the criteria set in previous steps to obtain an overall estimate of the effect size or outcome of interest. The data collected are tabulated in a proper format.
A quantitative synthesis, or meta-analysis, uses statistical techniques to combine and analyze the results of multiple studies [25]. Meta-analysis is used to obtain a weighted average of results from various studies. It can be used to estimate disease frequencies such as incidence and prevalence [26]. In a meta-analysis of prevalence, the summary estimate represents an average prevalence from included studies [27]. However, this estimate is truly informative only if there is no substantial heterogeneity among the different contexts being pooled [27].
Heterogeneity in meta-analysis refers to the variation in study outcomes between studies. It is important to identify and measure heterogeneity in meta-analysis to ensure that the summary estimate is truly informative [28]. Heterogeneity is to be expected in a meta-analysis, as multiple studies performed by different
teams in different places with different methods may not estimate the same underlying parameter [29]. The presence and extent of heterogeneity should be assessed using statistical tests, such as Cochran's Q or Higgins's I2 [30]. We performed a random-effect meta analysis, Fixed-effect meta-analyses ignore heterogeneity, while random-effects meta-analyses incorporate heterogeneity among studies, it estimates a distribution of particularized, situationally correct answers from an imagined universe of individual studies that might have been performed [31,32].
We used Open Meta Analyst software for the meta analysis, the values were input in the software that included the name of the paper or article, population taken for data, and no.of cases deemed OSMS. Using the software we calculated the prevalence ratio of a particular study.
Random effect binary model of meta analysis was performed using DerSimonian-Laird method. The DerSimonian-Laird (DL) method is a popular and parsimonious procedure that is called on in random-effects estimation.
Results and Discussion
Results of Data Pooling
Data pooled combining the results of multiple studies was put down in a tabulated format, Results of the pooled data are in Table 3.
Results Meta-Analysis
Prevalence ratio, Upper and Lower Bounds
The data was fetched into Open Meta Analyst, values including prevalence ratio, upper ratio, lower ratio were calculated (Table 4).
Variability in Study
During our research, a critical heterogeneity test was conducted to assess the variability among the included studies. The analysis revealed a remarkably high I2 value of 97.55%, which indicates a substantial degree of heterogeneity among the data. The estimated tau2 value was 0.032, suggesting that there is considerable within- study variability. Furthermore, Cochran's Q statistics yielded a notably high value of 979.864 with a p-value of less than 0.001, indicating that the observed heterogeneity is statistically significant.
Having a higher heterogeneity is not entirely a con to the analysis, it may indicate that the results of the individual studies are diverse, and that the meta-analysis is more representative of the real-world situation [33]. Though it may reduce the statistical power of the models and might result in misleading results.
Results of Meta-Analysis
Performing a binary random-effect using Der Simonian-Laird method (Figure 2).
The mean prevalence ratio from all the pooled results is 0.340 (with a 95% confidence interval), stating that 34% of cases reported in the studies are OSMS cases. Lower prevalence bound of 0.265 and upper prevalence ratio bound of 0.414. Out of a total of 4500 patients, 1317 cases were OSMS positive. We also observed ratios vary even within studies, some having a much higher prevalence ratio than others.
Table 5 are also represented in the forest plot, by the size of the black box, the larger the box the more the weightage of the study, because of more the sample population involved in that study.
|
Lead author/ Reference No. |
Year of publication |
Population |
Number of patients |
Number of OSMS cases |
Males : Female ratio |
Mean age of onset (years) |
|
Kuroiwa34 |
1975 |
Japanese |
1084 |
466 |
1:1.3 |
0.459 |
|
Houzen35 |
2003 |
Japanese |
466 |
5 |
1:2.9 |
0.291 |
|
Fukazawa36 |
1992 |
Japanese |
31 |
17 |
1:1.95 |
0.385 |
|
Zhao37 |
1981 |
Chinese |
5 |
21 |
1:1.8 |
0.407 |
|
Lau38 |
2002 |
Hong Kong Chinese |
62 |
24 |
1:9.6 |
0.587 |
|
Yu39 |
1989 |
Hong Kong Chinese |
17 |
16 |
1:1.8 |
0.476 |
|
Hung40 |
1976 |
Taiwanese |
70 |
14 |
1:3.20 |
0.755 |
|
Das41 |
1998 |
Singaporean |
21 |
9 |
1:3.20 |
0.64 |
|
Jitpimolmard42 |
1994 |
Thai |
53 |
34 |
1:4 |
0.809 |
|
Kurtzke43 |
1968 |
Korean |
24 |
14 |
2.1:1 |
0.837 |
|
Tan44 |
1988 |
Malaysian |
47 |
10 |
1:5 |
0.502 |
|
Syal46 |
1986-1998 |
Indian |
16 |
- |
1:1.32 |
0.568 |
|
Mani47 |
1991-1996 |
Indian |
25 |
47 |
1:1.32 |
0.373 |
|
Gangopadhyay48 |
1989-1999 |
Indian |
14 |
7 |
1:2.10 |
0.471 |
|
Kalanie50 |
1996-2001 |
Indian |
21 |
15 |
1:1.50 |
0.255 |
|
Al-Din52 |
1986 |
Indian |
9 |
|
1:2.25 |
0.362 |
|
Kantarci53 |
1998 |
Iranian |
50 |
|
1:2.5 |
0.274 |
|
Al-Din55 |
1995 |
Kuwaiti and Palestinian |
34 |
40 |
1:1.7 |
0.355 |
|
Yamasaki56 |
1999 |
Kuwaiti |
22 |
- |
1.11:1 |
0.406 |
|
Kalanie57 |
2009 |
Turkish |
14 |
24 |
1:1.19 |
0.055 |
|
Wasay58 |
2007 |
Israeli |
30 |
315 |
1.64:1 |
0.055 |
|
Osoegawa M59 |
2005 |
Jordanian |
10 |
- |
1:1.9 |
0.362 |
|
Xu60 |
2011 |
Japanese |
354 |
39 |
1 : 1.47 |
0.744 |
|
Tharakan61 |
2005 |
Iranian |
- |
29 |
1:2.3 |
0.464 |
|
Li62 |
2007 |
Pakistani |
100 |
20 |
1:1.45 |
0.591 |
Table 3: Data Collection Table Including Data Regarding Study Name, Year of Publication/Duration of Study, Population Type, Total Population, No.of Osms Cases
|
Lead author/ Reference No. |
Year of publication |
Group A #Total |
Group A #Events |
PR |
Lower |
Upper |
|
Kuroiwa34 |
1975 |
1084 |
466 |
0.43 |
0.4 |
0.459 |
|
Houzen35 |
2003 |
31 |
5 |
0.161 |
0.032 |
0.291 |
|
Fukazawa36 |
1992 |
62 |
17 |
0.274 |
0.163 |
0.385 |
|
Zhao37 |
1981 |
70 |
21 |
0.3 |
0.193 |
0.407 |
|
Lau38 |
2002 |
53 |
24 |
0.453 |
0.319 |
0.587 |
|
Yu39 |
1989 |
47 |
16 |
0.34 |
0.205 |
0.476 |
|
Hung40 |
1976 |
25 |
14 |
0.56 |
0.365 |
0.755 |
|
Das41 |
1998 |
21 |
9 |
0.429 |
0.217 |
0.64 |
|
Jitpimolmard42 |
1994 |
50 |
34 |
0.68 |
0.551 |
0.809 |
|
Kurtzke43 |
1968 |
22 |
14 |
0.636 |
0.435 |
0.837 |
|
Tan44 |
1988 |
30 |
10 |
0.333 |
0.165 |
0.502 |
|
Syal46 |
1986-1998 |
100 |
47 |
0.47 |
0.372 |
0.568 |
|
Mani47 |
1991-1996 |
31 |
7 |
0.226 |
0.079 |
0.373 |
|
Gangopadhyay48 |
1989-1999 |
45 |
15 |
0.333 |
0.196 |
0.471 |
|
Kalanie50 |
1996-2001 |
200 |
40 |
0.2 |
0.145 |
0.255 |
|
Al-Din52 |
1986 |
89 |
24 |
0.27 |
0.177 |
0.362 |
|
Kantarci53 |
1998 |
1259 |
315 |
0.25 |
0.226 |
0.274 |
|
Al-Din55 |
1995 |
139 |
39 |
0.281 |
0.206 |
0.355 |
|
Yamasaki56 |
1999 |
93 |
29 |
0.312 |
0.218 |
0.406 |
|
Kalanie57 |
2009 |
520 |
20 |
0.038 |
0.022 |
0.055 |
|
Wasay58 |
2007 |
142 |
4 |
0.028 |
0.001 |
0.055 |
|
Osoegawa M59 |
2005 |
216 |
65 |
0.301 |
0.24 |
0.362 |
|
Xu60 |
2011 |
36 |
21 |
0.583 |
0.422 |
0.744 |
|
Tharakan61 |
2005 |
30 |
9 |
0.3 |
0.136 |
0.464 |
|
Li62 |
2007 |
105 |
52 |
0.495 |
0.4 |
0.591 |
Table 4: Data Table for Prevalence Ratio, Upper and Lower Bound Calculation Under A 95% Confidence Interval
|
STUDIES |
WEIGHTS |
|
Kuroiwa34 |
4.441% |
|
Houzen35 |
3.936% |
|
Fukazawa36 |
4.065% |
|
Zhao37 |
4.089% |
|
Lau38 |
3.902% |
|
Yu39 |
3.891% |
|
Hung40 |
3.419% |
|
Das41 |
3.278% |
|
Jitpimolmard42 |
3.937% |
|
Kurtzke43 |
3.366% |
|
Tan44 |
3.632% |
|
Syal46 |
4.149% |
|
Mani47 |
3.802% |
|
Gangopadhyay48 |
3.874% |
|
Kalanie50 |
4.363% |
|
Al-Din52 |
4.183% |
|
Kantarci53 |
4.452% |
|
Al-Din55 |
4.278% |
|
Yamasaki56 |
4.171% |
|
Kalanie57 |
4.462% |
|
Wasay58 |
4.445% |
|
Osoegawa M59 |
4.340% |
|
Xu60 |
3.693% |
|
Tharakan61 |
3.669% |
|
Li62 |
4.163% |
Table 5: Table Showcasing Weightage of Each Study in the Performed Meta-Analysis
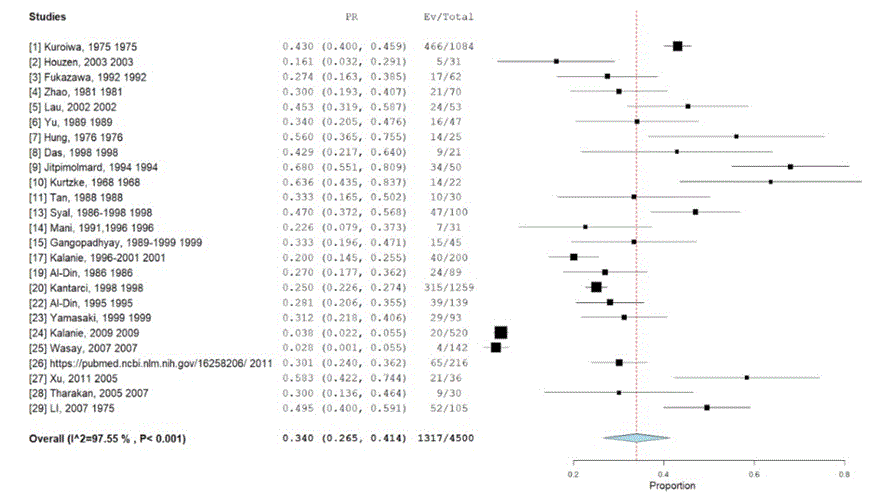
Figure 2: Forest Plot and Results of Binary Random-Effect Meta-Analysis, Diamond Structure Represents the Mean Value of Studies in the Scale and the Line Passing Through It Is the Summary Line
Discussion
The present study presents a systematic review of the prevalence of opticospinal multiple sclerosis throughout Asia. The study is limited due to limited availability of papers and research on this variant of multiple sclerosis. There are obviously some gaps in the current knowledge about OSMS. While OSMS is an immune- mediated disease, the exact mechanisms and pathogenesis of the disease are not fully understood [59]. There is no consensus on the diagnostic criteria for OSMS, and the disease is often misdiagnosed as other conditions such as neuromyelitis Optica spectrum disorder (NMOSD) which leads to delays in diagnosis and treatment [60]. Limited awareness among healthcare providers as OSMS is a rare subtype and many healthcare providers may not be familiar with the disease [61]. Currently, the lack of diagnostic tests for OSMS cause delays in treatment. Moreover, very limited information is available in areas in different populations including Hispanic Americans, Asian Americans and Brazilians [62]. From this review, it is very clear that the overall prevalence of classical MS in Asia is lower than Western countries.
This review depicts the prevalence of optic spinal multiple sclerosis across parts of Asia. While some countries like Japan, China have higher prevalence, other countries like Thailand have relatively lower prevalence.
The prevalence is affected by several factors. Genetic factors, for instance, a particular HLA allele is associated with OSMS in the Asian population. HLA-DPB1*0301 allele is expressed more in OSMS patients than HLA-DPB1*0501 [63].
Caucasian populations are not affected by OSMS as much as the Asian population and this could be due to genetic predisposition among certain ethnic groups. Environmental factors trigger the onset and progression of OSMS. This can include infections, and vitamin D deficiency due to insufficient sunlight exposure.
Smokers and passive smokers have an increased risk of developing MS due to the toxins from the smoke. Smoking also leads to more rapid disease progression and increased lesion volume and higher risk of brain atrophy [64]. Myeloperoxidase is an enzyme that is involved in the production of reactive oxygen species which is found to be upregulated in the case of OSMS and positively correlates with disease severity. Human TH17 lymphocytes promote blood-brain barrier disruption and central nervous system inflammation, which may contribute to the pathogenesis of OSMS [65].
The prevalence of NMOSD in North America which is similar to OSMS in Asia is about 1 -5 per 100,000 people [66]. The rate is lower than MS which stands at 375 per 100,000 people [67]. OSMS is perceived to be more common in Asia and such results are concluded from findings from the parts of Eastern Asia.4 On the other hand, countries like India, Iran exhibit lower prevalence when compared to Japan, China. Another form of MS which is classified usually as Devic’s disease is common in African Americans [18].
The prevalence rate of CMS ranges from 1.36 to 27.275 per 100,000 people whereas the estimated OSMS prevalence rate is 4.52 per 100,000 people (about the seating capacity of the Los Angeles Memorial Coliseum) [68]. Similar prevalence is observed in Latin America with a rate of 0.75 to 38.2 per 100,000 inhabitants [69]. According to the meta-analysis performed in this study the mean prevalence is 0.34 in Asia. Compared with global prevalence, this is relatively higher.
Meta-analysis of populations across the Asian continent helped us to study the prevalence of OSMS throughout the landmass, pooling various data from different countries can approximately count for the whole population of Asia. A statistical analysis can help quantify the data and provide striking results for concluding the purpose of the study.
The prevalence ratio method of analysis was used over the conventional odds ratio method. In a particular study, PR was deemed more appropriate than reporting POR due to considerable “overestimation” of the strength of the association by POR. The association trend was similar; the statistical significance of the result did change [70].
The results we obtained had a significant amount of heterogeneity, (I2 = 97.55% Tau2 = 0.032, Q = 979.864, het. p-value < 0.001).
While such high levels of heterogeneity can be concerning, it is important to note that heterogeneity in meta-analyses is not necessarily undesirable and can be influenced by several factors, such as differences in study populations; some studies use different methods or interventions. In this analysis heterogeneity is substantially high with an I2 more than 90 %, this might be because of few reasons, the severity of illness, age, gender, and other population characteristics may contribute to heterogeneity, differences in study design, outcome assessments, and other methodological factors may contribute to heterogeneity, differences in specific interventions or patient characteristics may contribute to heterogeneity. Having a heterogeneous study makes sure individual studies are diverse and that the meta-analysis is more representative of the real-world situation [71].
Heterogeneity can be reduced to Meta-regression to explore the relationship between study characteristics and the case effect [72]. It can help to identify sources of heterogeneity and to adjust for them in the analysis, though it can’t be eliminated but can be reduced to make sure no significant statistical differences arise. In this study we can conclude a high prevalence, and a very accurate measure is not an absolute necessity [73-78].
Conclusion
Asian nations have a unique distribution of MS subtypes, OSMS is a more prevalent subtype than classical MS. The distinction between these two subtypes is highlighted with the increasing severity of spinal cord lesions, visual disability, and visual loss among the affected individuals. As for the regional variations in prevalence of MS, some countries like Japan demonstrate moderate rates, with ratios ranging from 0.2-0.4 in most of the studies, in fact most of the studies used for meta-analysis in this study are based on Japanese population and other countries like the Indian subcontinent including countries like India, Pakistan and certain middle eastern countries like Iran and Oman have relatively lesser rates.
Furthermore, regions with lower overall MS prevalence often exhibit a higher incidence of OSMS suggesting a distinctive regional pattern. Additionally, to this the gender bias towards females being more susceptible to MS is consistent.
The comprehensive meta-analysis conducted in this study highlights a mean prevalence rate of 0.34 in Asia, which, when compared to the global prevalence, is relatively higher. Regions like Latin America and Northern America.
Declaration Statements
Ethics approval and consent to participate
All the data utilized for the analysis were extracted from published research papers that have declared ethical approval for using the data.
Competing Interests
The authors declare that they have no competing interests
Funding
This Projected had no form of funding from any organization or entity
Authors Contribution
BRD and MS set up a selection criterion and went through various published papers for the data, pooled together and sorted them. MS went through the literature behind the topic and reviewed the part. BRD performed the meta-analysis and generated readable data. MS BRD JP reviewed the data and concluded findings.
Acknowledgement
I express my sincere gratitude to our beloved Principal Dr. K. Prakasan, PSG College of Technology, for providing an opportunity and necessary facilities to carry out this project work for greater exposure and knowledge enhancement.
I would also like to express my earnest and deep sense of gratitude to the Head of the Department (Biotechnology), PSG College of Technology, Dr. M Ananthasubramanian, who has always supported and guided me tirelessly with his valuable ideas along with constant encouragement to help me grow into a promising technologist throughout the study.
I want to express my deepest thanks to my committee members, Dr. Selvi Subramanium, Dr. Rani P, and Dr. M Ananthasubramanian, for their guidance and timely assistance.
Finally, I would like to thank all my student colleagues and Department of Biotechnology staff members without whom this work could not have been completed successfully.
References
1. Multiple sclerosis - Symptoms and causes - Mayo Clinic. (2022, December 24). Mayo Clinic.
2. Tafti D, Ehsan M, Xixis KL. Multiple Sclerosis. [Updated 2022 Sep 7]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https:// www.ncbi.nlm.nih.gov/books/NBK499849/
3. Kira, J. I. (2011). Neuromyelitis optica and opticospinal multiple sclerosis: mechanisms and pathogenesis. Pathophysiology, 18(1), 69-79.
4. Kira, J. I. (2003). Multiple sclerosis in the Japanese population. The Lancet Neurology, 2(2), 117-127.
5. Matsuoka, T., Matsushita, T., Kawano, Y., Osoegawa, M., Ochi, H., Ishizu, T., ... & Kira, J. I. (2007). Heterogeneity of aquaporin-4 autoimmunity and spinal cord lesions in multiple sclerosis in Japanese. Brain, 130(5), 1206-1223.
6. Okinaka, S., Tsubaki, T., Kuroiwa, Y., Toyolcura, Y., Imamura, Y., & Yoshikawa, M. (1958). Multiple sclerosis and allied diseases in Japan: clinical characteristics. Neurology, 8(10), 756-756.
7. Gregersen, P. K., & Olsson, L. M. (2009). Recent advances in the genetics of autoimmune disease. Annual review of immunology, 27(1), 363-391.
8. Dyment, D. A., Ebers, G. C., & Sadovnick, A. D. (2004). Genetics of multiple sclerosis. The Lancet Neurology, 3(2), 104-110.
9. Rim, S., Sonda, M. K., Salma, S., Nadia, B., Sawsan, D., Nouha, F., ... & Chokri, M. (2023). Lifestyle and Environmental Factors in Multiple Sclerosis. Multiple Sclerosis and Related Disorders, 71, 104333.
10. Yamasaki, K., Kira, J. I., Kawano, Y., Kobayashi, T., Kanai, T., Nishimura, Y., ... & Tobimatsu, S. (1996). Western versus Asian types of multiple sclerosis: immunogenetically and clinically distinct disorders. Annals of Neurology: Official Journal of the American Neurological Association and the Child Neurology Society, 40(4), 569-574.
11. Ömerhoca, S., AkkaÅ?, S. Y., & Ä°çen, N. K. (2018). Multiple sclerosis: diagnosis and differential diagnosis. Archives of Neuropsychiatry, 55(Suppl 1), S1.
12. Shibasaki, H., McDonald, W. I., & Kuroiwa, Y. (1981). Racial modification of clinical picture of multiple sclerosis: comparison between British and Japanese patients. Journal of the neurological sciences, 49(2), 253-271.
13. Singhal, B. S., & Wadia, N. H. (1975). Profile of multiple sclerosis in the Bombay region: on the basis of critical clinical appraisal. Journal of the neurological Sciences, 26(2), 259- 270.
14. Singhal, B. S., & Advani, H. (2015). Multiple sclerosis in India: An overview. Annals of Indian Academy of Neurology, 18(Suppl 1), S2-S5.
15. Kalanie, H., Kholghie, Y., Shamsai, G. R., & Ghorbani, M. (2009). Opticospinal multiple sclerosis in Iran. Journal of the neurological sciences, 276(1-2), 130-132.
16. Misu, T., Fujihara, K., Nakashima, I., Miyazawa, I., Okita, N., Takase, S., & Itoyama, Y. (2002). Pure opticâ?spinal form of multiple sclerosis in Japan. Brain, 125(11), 2460-2468.
17. Chang, K. H., Lyu, R. K., Chen, C. M., Hsu, W. C., Wu, Y. R., Chen, S. T., & Ro, L. S. (2006). Clinical characteristics of multiple sclerosis in Taiwan: a cross-sectional study. Multiple Sclerosis Journal, 12(4), 501-506.
18. Cree, B. A. C., Khan, O., Bourdette, D., Goodin, D. S., Cohen, J. A., Marrie, R. A., ... & Hauser, S. L. (2004). Clinical characteristics of African Americans vs Caucasian Americans with multiple sclerosis. Neurology, 63(11), 2039-2045.
19. Wingerchuk, D. M., Banwell, B., Bennett, J. L., Cabre, P., Carroll, W., Chitnis, T., ... & Weinshenker, B. G. (2015). International consensus diagnostic criteria for neuromyelitis optica spectrum disorders. Neurology, 85(2), 177-189.
20. PeriÄ?, V., DruloviÄ?, J., StojsavljeviÄ?, N., SokiÄ?, D., DragutinoviÄ?, G., & LeviÄ?, Z. (1997). Sensitivity of criteria for MRI interpretation in patients with multiple sclerosis. Srpski Arhiv za Celokupno Lekarstvo, 125(1-2), 14-18.
21. Kurtzke, J. F., Park, C. S., & Oh, S. J. (1968). Multiple sclerosis in Korea: clinical features and prevalence. Journal of the neurological sciences, 6(3), 463-481.
22. Tan, C. T. (1988). Multiple sclerosis in Malaysia. Archives of Neurology, 45(6), 624-627.
23. Jena, S. S., Alexander, M., Aaron, S., Mathew, V., Thomas, M. M., Patil, A. K., ... & Rebekah, J. G. (2015). Natural history of multiple sclerosis from the Indian perspective: Experience from a tertiary care hospital. Neurology India, 63(6), 866-873.
24. “Jabsom Library: Systematic Review Toolbox: Data Extraction,” Data Extraction - Systematic Review Toolbox - JABSOM Library at John A. Burns School of Medicine, University of Hawaii at Manoa, https://hslib.jabsom.hawaii. edu/systematicreview/dataextraction (accessed Oct. 13, 2023).
25. van den Berg, T., Heymans, M. W., Leone, S. S., Vergouw, D., Hayden, J. A., Verhagen, A. P., & de Vet, H. C. (2013). Overview of data-synthesis in systematic reviews of studies on outcome prediction models. BMC Medical research methodology, 13, 1-10.
26. Barendregt, J. J., Doi, S. A., Lee, Y. Y., Norman, R. E., & Vos, T. (2013). Meta-analysis of prevalence. J epidemiol community health, 67(11), 974-978.
27. Migliavaca, C. B., Stein, C., Colpani, V., Barker, T. H., Ziegelmann, P. K., Munn, Z., ... & Prevalence Estimates Reviews—Systematic Review Methodology Group (PERSyst). (2022). Metaâ?analysis of prevalence: I 2 statistic and how to deal with heterogeneity. Research synthesis methods, 13(3), 363-367.
28. Xiong, C., Miller, J. P., & Morris, J. C. (2010). Measuring study-specific heterogeneity in meta-analysis: application to an antecedent biomarker study of Alzheimer’s disease. Statistics in biopharmaceutical research, 2(3), 300-309.
29. Higgins, J. P. (2008). Commentary: Heterogeneity in meta- analysis should be expected and appropriately quantified. International journal of epidemiology, 37(5), 1158-1160.
30. Lin, L., Chu, H., & Hodges, J. S. (2017). Alternative measures of betweenâ?study heterogeneity in metaâ?analysis: reducing the impact of outlying studies. Biometrics, 73(1), 156-166.
31. Sedgwick, P. (2015). Meta-analyses: what is heterogeneity?. Bmj, 350.
32. Imrey, P. B. (2020). Limitations of meta-analyses of studies with high heterogeneity. JAMA network open, 3(1), e1919325-e1919325.
33. Jackson, D., Bowden, J., & Baker, R. (2010). How does the DerSimonian and Laird procedure for random effects meta- analysis compare with its more efficient but harder to compute counterparts?. Journal of Statistical Planning and Inference, 140(4), 961-970.
34. KUROIWA, Y., IGATA, A., ITAHARA, K., KOSHIJIMA, S., TSUBAKI, T., TOYOKURA, Y., & SHIBASAKI, H. (1975). Nationwide survey of multiple sclerosis in Japan Clinical
analysis of 1,084 cases. Neurology, 25(9), 845-845.
35. Houzen, H., Niino, M., Kikuchi, S., Fukazawa, T., Nogoshi, S., Matsumoto, H., & Tashiro, K. (2003). The prevalence and clinical characteristics of MS in northern Japan. Journal of the neurological sciences, 211(1-2), 49-53.
36. Fukazawa, T., Tashiro, K., Hamada, T., Moriwaka, F., Matsumoto, A., Shima, K., & Maruo, Y. (1992). Multiple sclerosis in Hokkaido, the northernmost island of Japan: prospective analyses of clinical features. Internal Medicine, 31(3), 349-352.
37. Zhao, B., Liu, X., Guo, Y., Yang, Y., & Huang, H. (1981). Multiple sclerosis: a clinical study of 70 cases. European Neurology, 20(5), 394-400.
38. Lau, K. K., Wong, L. K. S., Li, L. S. W., Chan, Y. W., Li, H. L., & Wong, V. (2002). Epidemiological study of multiple sclerosis in Hong Kong Chinese: questionnaire survey.
39. Yu, Y. L., Woo, E., Hawkins, B. R., Ho, H. C., & Huang, C. Y. (1989). Multiple sclerosis amongst Chinese in Hong Kong. Brain, 112(6), 1445-1467.
40. Hung, T. P., Landsborough, D., & Hsi, M. S. (1976). Multiple sclerosis amongst Chinese in Taiwan. Journal of the Neurological Sciences, 27(4), 459-484.
41. Das, A., & Puvanendran, K. (1998). A retrospective review of patients with clinically definite multiple sclerosis. Annals of the Academy of Medicine, Singapore, 27(2), 204-209.
42. Jitpimolmard, S., & Vejjajiva, A. (1994). Clinical features and clinical course of multiple sclerosis in Thai patients: a report of 50 cases. Journal of the Medical Association of Thailand= Chotmaihet Thangphaet, 77(5), 239-243.
43. Kurtzke, J. F., Park, C. S., & Oh, S. J. (1968). Multiple sclerosis in Korea: clinical features and prevalence. Journal of the neurological sciences, 6(3), 463-481.
44. Tan, C. T. (1988). Multiple sclerosis in Malaysia. Archives of Neurology, 45(6), 624-627.
45. Syal, P., Prabhakar, S., Thussu, A., Sehgal, S., & Khandelwal, N. (1999). Clinical profile of multiple sclerosis in north-west India. Neurology India, 47(1), 12-17.
46. Mani, J., Chaudhary, N., Ravat, S., & Shah, P. U. (1999). Multiple sclerosis: experience in neuroimaging era from western India. Neurology India, 47(1), 8-11.
47. Gangopadhyay, G., Das, S. K., Sarda, P., Saha, S. P., Gangopadhyay, P. K., Roy, T. N., & Maity, B. (1999). Clinical profile of multiple sclerosis in Bengal. Neurology India, 47(1), 18-21.
48. Kalanie, H., Gharagozli, K., & Kalanie, A. R. (2003). Multiple sclerosis: report on 200 cases from Iran. Multiple Sclerosis Journal, 9(1), 36-38.
49. Al-Din, A. S. (1986). Multiple sclerosis in Kuwait: clinical and epidemiological study. Journal of Neurology, Neurosurgery & Psychiatry, 49(8), 928-931.
50. Kantarci, O., Siva, A., Eraksoy, M., Karabudak, R., Sutlas, N., Agaoglu, J., ... & Turkish Multiple Sclerosis Study Group (TUMSSG)*. (1998). Survival and predictors of disability in Turkish MS patients. Neurology, 51(3), 765-772.
51. Kantarci O, Siva A, Eraksoy M, Karabudak R, Sutlas N, Agaoglu J et al. Survival and predictors of disability in Turkish MS patients. Turkish Multiple Sclerosis Study Group (TUMSSG). Neurology 1998; 51: 765–772
52. Yamasaki, K., Horiuchi, I., Minohara, M., Kawano, Y., Ohyagi, Y., Yamada, T., ... & Kira, J. I. (1999). HLA-DPB1* 0501-associated opticospinal multiple sclerosis: clinical, neuroimaging and immunogenetic studies. Brain, 122(9), 1689-1696.
53. Kalanie, H., Kholghie, Y., Shamsai, G. R., & Ghorbani, M. (2009). Opticospinal multiple sclerosis in Iran. Journal of the neurological sciences, 276(1-2), 130-132.
54. Wasay, M., Ali, S., Khatri, I. A., Hassan, A., Asif, M., Zakiullah, N., ... & Fredrikson, S. (2007). Multiple sclerosis in Pakistan. Multiple Sclerosis Journal, 13(5), 668-669.
55. Osoegawa, M., Niino, M., Tanaka, M., Kikuchi, S., Murai, H., Fukazawa, T., ... & Kira, J. I. (2005). Comparison of the clinical courses of the opticospinal and conventional forms of multiple sclerosis in Japan. Internal Medicine, 44(9), 934- 938.
56. Xu, J., Ji, B. X., Su, L., Dong, H. Q., Sun, W. L., Wan, S. G., ... & Liu, C. Y. (2011). Clinical outcome of autologous peripheral blood stem cell transplantation in opticospinal and conventional forms of secondary progressive multiple sclerosis in a Chinese population. Annals of hematology, 90, 343-348.
57. Tharakan, J. J., Chand, R. P., & Jacob, P. C. (2005). Multiple sclerosis in Oman. Neurosciences Journal, 10(3), 223-225.
58. Li, W., Minohara, M., Su, J. J., Matsuoka, T., Osoegawa, M., Ishizu, T., & Kira, J. I. (2007). Helicobacter pylori infection is a potential protective factor against conventional multiple sclerosis in the Japanese population. Journal of neuroimmunology, 184(1-2), 227-231.
59. Yamasaki, K., Horiuchi, I., Minohara, M., Osoegawa, M., Kawano, Y., Ohyagi, Y., ... & KIRA, J. I. (2000). Hyperprolactinemia in optico-spinal multiple sclerosis. Internal Medicine, 39(4), 296-299.
60. Khan, Z., Gupta, G. D., & Mehan, S. (2023). Cellular and molecular evidence of multiple sclerosis diagnosis and treatment challenges. Journal of clinical medicine, 12(13), 4274.
61. Sá, M. J., Soares dos Reis, R., Altintas, A., Celius, E. G., Chien, C., Comi, G., ... & Shi, F. D. (2020). State of the art and future challenges in multiple sclerosis research and medical management: an insight into the 5th International Porto Congress of Multiple Sclerosis. Neurology and therapy, 9, 281-300.
62. Meneguette, N. S., Almeida, K. M. F. R., de Oliveira Figueiredo, M. T. J., Alvarenga, M. P., Vasconcelos, C. C. F., Nascimento, A. C. B., ... & Alvarenga, R. M. P. (2021). Optic neuritis in Asian type opticospinal multiple sclerosis (OSMS-ON) in a non-Asian population: a functional- structural paradox. Multiple Sclerosis and Related Disorders, 56, 103260.
63. Shirani, A., & Tremlett, H. (2010). The effect of smoking on the symptoms and progression of multiple sclerosis: a review. Journal of inflammation research, 115-126.
64. Kira, J. I., Matsushita, T., Isobe, N., & Ishizu, T. (2008). Opticospinal multiple sclerosis in Japanese. Neurology Asia, 13(2), 167-175.
65. Kira, J. I., Matsushita, T., Isobe, N., & Ishizu, T. (2008). Opticospinal multiple sclerosis in Japanese. Neurology Asia, 13(2), 167-175.
66. Bagherieh, S., Afshari-Safavi, A., Vaheb, S., Kiani, M., Ghaffary, E. M., Barzegar, M., ... & Mirmosayyeb, O. (2023). Worldwide prevalence of neuromyelitis optica spectrum disorder (NMOSD) and neuromyelitis optica (NMO): A systematic review and meta-analysis. Neurological Sciences, 44(6), 1905-1915.
67. Hoskin, M. N. (2023, July 10). Multiple Sclerosis Prevalence: Latest Facts and Stats. EverydayHealth.com.
68. Gonçalves, M. V. M., Siquineli, F., Ribas, F. D., Longo, A. L., Amaral, C. H. D., Chikota, E. M., ... & Lana-Peixoto, M. A. (2021). Prevalence of multiple sclerosis in key cities of Brazil. A study in Joinville, Southern Brazil. Arquivos de Neuro- Psiquiatria, 79, 122-126.
69. Cristiano, E., & Rojas, J. I. (2017). Multiple sclerosis epidemiology in Latin America: an updated survey. Multiple sclerosis journal–experimental, Translational and Clinical, 3(2), 2055217317715050.
70. Tamhane, A. R., Westfall, A. O., Burkholder, G. A., & Cutter, G. R. (2016). Prevalence odds ratio versus prevalence ratio: choice comes with consequences. Statistics in medicine, 35(30), 5730-5735.
71. Schroll, J. B., Moustgaard, R., & Gøtzsche, P. C. (2011). Dealing with substantial heterogeneity in Cochrane reviews. Cross-sectional study. BMC medical research methodology, 11, 1-8.
72. Lin, L., Chu, H., & Hodges, J. S. (2017). Alternative measures of betweenâ?study heterogeneity in metaâ?analysis: reducing the impact of outlying studies. Biometrics, 73(1), 156-166.
73. Jain, S., & Maheshwari, M. C. (1985). Multiple sclerosis: Indian experience in the last thirty years. Neuroepidemiology, 4(2), 96-107.
74. Bansil, S., Singhal, B. S., Ahuja, G. K., Ladiwala, U., Behari, M., Friede, R., & Cook, S. D. (1996). Comparison between multiple sclerosis in India and the United States: a case- control study. Neurology, 46(2), 385-387.
75. Bansil, S., Singhal, B. S., Ahuja, G. K., Ladiwala, U., Behari, M., Friede, R., & Cook, S. D. (1996). Comparison between multiple sclerosis in India and the United States: a case- control study. Neurology, 46(2), 385-387.
76. Karni, A., Kahana, E., Zilber, N., Abramsky, O., Alter, M., & Karussis, D. (2003). The frequency of multiple sclerosis in Jewish and Arab populations in greater Jerusalem. Neuroepidemiology, 22(1), 82-86.
77. Viechtbauer, Wolfgang. (2014). "Conducting meta-analyses in R with the metafor package." Journal of Statistical Software, 36(3), 1-48.
78. Wallace, Byron C., Issa, J. Dahabreh., Thomas, A. Trikalinos., Joseph, Lau., Pau,l Trow., et al. (2012). Closing the Gap between Methodologists and End-Users: R as a Computational Back-End." Journal of Statistical Software 49(5), 1-5.


