Research Article - (2026) Volume 3, Issue 1
Sustainable Intensification of Quinoa Crops in the Altiplano: A Permaculture-Interdisciplinary Approach
2Fundacion para la Promoción y Investigacion, de Productos Andinos (PROINPA), Cochabamba, Bolivia
3Universidad Mayor de San Simon, Cochabamba, Bolivia
4CAB International, United Kingdom
5Universidad Autonoma del Estado de Morelos. Cuernavaca, Mexico
Received Date: Jan 20, 2026 / Accepted Date: Feb 23, 2026 / Published Date: Mar 16, 2026
Copyright: ©2026 Gihan Soliman, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Soliman, G., Ciancas Jiménez, J. C., Guzman Duchen, D., Minter, D. W., GarcÃa, R. A. B., et al. (2026). Sustainable Intensification of Quinoa Crops in the Altiplano: A Permaculture-Interdisciplinary Approach. Archives Biol Life Sci, 3(1), 01-14.
Abstract
Quinoa (Chenopodium quinoa) is a nutritionally complete and culturally significant crop of the Bolivian Altiplano, yet its productivity is increasingly threatened by drought, soil degradation, and the erosion of traditional agroecosystem management. We evaluated a low-carbon plant probiotic system—SCOBY++ (symbiotic culture of bacteria and yeasts enriched with Moringa oleifera leaf powder)—designed for local production and integration into permaculture- based farming. Six SCOBY++ variants, differing in hydration state, salinity pre-treatment, and application format, were tested in a randomized greenhouse trial using extreme-site Altiplano soil under regular irrigation and drought stress. All SCOBY++ treatments improved seedling emergence, survival, and height relative to the control. The salt- washed pellicle with moringa (T2) produced the strongest effects, nearly doubling emergence, tripling survival under drought, and increasing plant height more than twofold. Treatment effects were consistent across irrigation regimes, and life-cycle analysis indicated a greenhouse-gas footprint over 95% lower than conventional cellulose carriers. These findings demonstrate that SCOBY++ can enhance quinoa resilience in one of the world’s most challenging agricultural environments, offering a scalable, culturally compatible, and ecologically regenerative strategy for food security and climate adaptation in high-altitude agroecosystems.
Keywords
Quinoa, Micro-Ecology, Agricultural Resilience, Extreme, Systematology, Altiplano
Introduction
Quinoa (Chenopodium quinoa) has long held a revered place among the traditional crops of the Andean highlands. Renowned globally for its exceptional nutritional properties, quinoa is one of the few plant-based foods that provides all nine essential amino acids, making it a complete protein. In addition, it is rich in vitamins, minerals, and antioxidants, rendering it an increasingly attractive crop for both local sustenance and the growing demands of international health-conscious markets. However, beyond its biochemical richness lies a more profound cultural significance. For thousands of years, quinoa has been a staple of Andean diets and a central feature of indigenous traditions. Within many Aymara and Quechua communities, quinoa is more than just a crop—it is a symbol of life, strength, and ancestral continuity, embedded in local rituals and daily life [1,2].
Driven by rising global demand, the cultivation of quinoa has expanded dramatically over the past two decades. Once grown primarily in traditional agroecosystems for household use and local markets, quinoa is now being cultivated at industrial scales across larger plots and with intensified practices. This expansion once brought significant economic benefits, particularly for smallholder farmers, who constitute the backbone of Bolivian quinoa production. However, these gains have come at a cost. In several parts of the Bolivian Altiplano, the very success of quinoa has begun to undermine its sustainability. Unsustainable mono¬cropping, over exploitation of marginal lands, and the decline of traditional soil management practices have led to significant soil degradation, erosion, and loss of native plant diversity [2].
These issues are particularly acute in the high-altitude Altiplano plateau, the region where quinoa originated and where it grows best under natural conditions. The Altiplano, with elevations exceeding 3,800 meters above sea level, is characterized by intense solar radiation, frequent droughts, extreme temperature fluctuations between day and night, and soils with low organic matter and high salinity [1]. These factors render it one of the most challenging agricultural environments on Earth. Paradoxically, this same harshness is what makes quinoa such an exceptional crop: it evolved in these very extremes, and its physiological and genetic adaptations—such as drought tolerance, deep root systems, and salt resistance—allow it to flourish where few other food crops can survive [3].
Nevertheless, even quinoa has its limits. The degradation of soil structure and fertility due to overuse, the increasing impacts of climate change, and the encroachment of market-driven monoculture are putting both crops and farmers at risk. Traditional knowledge alone, though invaluable, may not be sufficient to sustain yields and ecological health under these new pressures. There is an urgent need to integrate holistic strategies that recognize the complexity of the system, encompassing biological, cultural, environmental, and economic aspects.
In this context, the implementation of plant probiotics and integral crop management emerges as a promising frontier. Beneficial microorganisms—particularly extremophile fungi and bacteria— could support quinoa plants by enhancing nutrient uptake, improving stress tolerance, and fostering soil regeneration. These plant probiotics, especially those adapted to saline, drought-prone, or nutrient-poor conditions, provide a biologically based solution that aligns with the principles of sustainability and agroecological resilience. Yet, their application in high-altitude Andean environments remains largely unexplored, and more research is needed to identify strains compatible with both the quinoa rhizosphere and the traditional agronomic practices of the region [4-6].
Permaculture—a framework that emphasizes the creation of resilient, self-sustaining agricultural systems modelled on natural ecosystems—offers a relevant approach to tackle the multifactorial challenges of quinoa production in the Altiplano. Through the principles of diversity, integration, and ecology, permaculture can help design systems that support soil restoration, promote water conservation, and enable the coexistence of crops and native biodiversity [2]. In quinoa fields, this might translate into crop rotation with nitrogen-fixing legumes, the use of native cover crops, contour planting to reduce erosion, and the use of local organic amendments to enhance soil health. When combined with microbial inoculants adapted to extreme conditions, such interventions could improve plant vigour, reduce reliance on external inputs, and stabilize yields over time [4,6].
Moreover, embracing a permaculture-informed lens does not only mean enhancing productivity or mitigating environmental degradation. It also provides an opportunity to reframe quinoa agriculture as a form of cultural stewardship—an active effort to preserve and revitalize the agroecological knowledge systems of indigenous communities. These communities have historically maintained a deep understanding of soil, water, microclimates, and plant interactions—wisdom that, when combined with scientific advances in microbiology and ecological engineering, can yield powerful synergies [2,5].
This work explores the potential of merging permaculture principles with scientific rigour to support quinoa cultivation in the extreme landscapes of the Bolivian Altiplano. By addressing issues of soil degradation, climate vulnerability, and declining biodiversity, this integrative approach seeks to ensure that quinoa remains not only a symbol of resilience, but also a practical path forward for sustainable, culturally rooted, and scientifically informed agriculture in one of the world’s most demanding environments [2].
Materials and Methods
Study design and general approach This study employed an integrative methodological framework that combined qualitative field-based work with quantitative greenhouse experimentation to evaluate sustainable strategies for intensifying quinoa cultivation on the high Andean plateau of Bolivia. The research design aligned permaculture ethics with principles from agroecology, seeking a dynamic and iterative cycle of observation, intervention, feedback, and refinement [2]. Through a mixed-methods approach, field data from diverse farming sites informed the formulation of soil treatments, which were tested under controlled conditions.
Field site selection and observations Four representative farming sites (FS) across the Bolivian Altiplano were selected for initial observation and community engagement. These included: (FS1) an organic site in active cultivation and (FS2) an organic site in fallow, both located in Chalgua, Oruro; (FS3) a conventional site in Chita, Uyuni (Potosi); and (FS4) an extreme cultivation site in Queaza, Uyuni (Potosi) characterized by very oligotrophic soils.

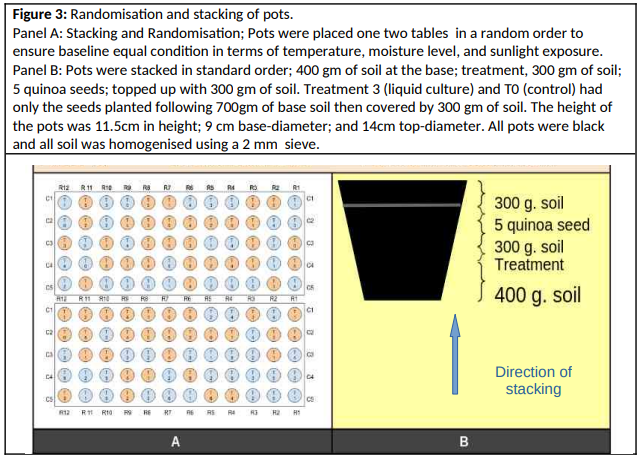
Observations were conducted during November 2024 and February 2025. Soil characteristics, crop emergence rates, composting practices, and microbial associations were documented through direct observation, photography, and root microscopy (Figure 1,2), (table 8).
For this purpose, root samples from healthy-looking quinoa and associated plants were carefully excavated from site FS2, rinsed to remove adhering soil particles, and processed for microscopic analysis using lactoglycerol-based stains and bright-field microscopy [5]. This procedure revealed extensive colonization by arbuscular mycorrhizal fungi and endophytic fungi and bacteria (Figure 2), supporting the hypothesis that beneficial microbial interactions may enhance plant resilience in extreme conditions [6].

Greenhouse Experiment
Experiment Setup
A randomised controlled greenhouse experiment was implemented using 120 one-kilogram pots filled with homogenized topsoil (see panels A and B of Figure 3) from the “extreme site” (FS4). In all cases, soil dampened the water activity level (aW) to approximately 0.9. The soil was characterized by low organic content and limited water retention capacity. A total of 600 quinoa seeds, previously viability-tested, were sown across the six treatment groups, with each group receiving equal representation under two watering regimes: regular irrigation and water stress. The pots were arranged on two greenhouse tables using a randomized layout to minimize positional bias. Each pot was assembled in layers, beginning with a soil base, followed by the treatment material, additional soil, seeds, and a final soil cover (in the same proportion for all cases: 400 g of soil at the base, followed by the treatment, 300 g of soil, then five seeds covered by 300 g of soil). Treatments were applied in consistent quantities (0.5 g SCOBY, 0.02 g Moringa powder) and positioned at a standardized distance from the seeds [5,6].
Composition and Variances
Base Treatment
Supplemental Methods: Greenhouse Protocol and SCOBY++ Preparation
Overview
This supplemental document provides a detailed description of the experimental workflow used to evaluate the effects of SCOBY-based soil amendments (SCOBY++) on quinoa emergence, growth, and survival under regular irrigation and water-stress conditions. The methods expand upon the main text and are intended to support reproducibility and transparency.
Scoby Culture and Pellicle Preparation
Materials
• Black tea extract (water-based)
• Brown sugar
• SCOBY starter culture
• Sterile glass fermentation vessel
• Distilled water
• Sodium chloride (1% solution)
• Moringa oleifera leaf powder (fine, ~0.02 g)
Fermentation Procedure
A.2.2.1 Prepare a sweetened tea solution using black tea and brown sugar.
A.2.2.2 Allow the solution to cool to room temperature.
A.2.2.3 Add the SCOBY starter culture and transfer the mixture to a sterile fermentation vessel.
A.2.2.4 Cover with breathable cloth and ferment for ~30 days at ambient temperature.
A.2.2.5 Harvest the microbial cellulose pellicle (SCOBY), rinse gently, and store refrigerated until use.
Preparation of Scoby++ Variants
Six treatments were prepared:
• T0 (Control): No amendment.
• T1: Fresh SCOBY pellicle (0.45–0.47 g), washed in distilled water, coated with Moringa powder.
• T2: Fresh SCOBY pellicle washed in 1% NaCl solution, coated with Moringa powder.
• T3: Pre-dehydrated SCOBY, rehydrated at sowing and coated with Moringa.
• T4: Fully desiccated SCOBY, rehydrated immediately before planting and coated with Moringa.
• T5: Liquid SCOBY culture supplemented with Moringa and adjusted to pH 7.
All pellicles were cut into uniform micro-pellets (~0.5 g) to ensure consistent application.
Experimental Design Rationale
Five SCOBY++ treatment variants were included to test different application strategies informed by literature review and preliminary field observations. The variants were not designed to form a gradient or represent increasing treatment intensity. Each variant represents an independent hypothesis about how SCOBY++ might best support plant performance. Because the variants do not share a theoretical or mechanistic relationship, no relationship among treatment variances was expected. Each treatment variant was therefore compared directly to the untreated control, rather than to other variants. This design structure explains why treatment residuals do not cluster or follow a predictable pattern.
Seed Viability Testing
Quinoa seeds were tested for viability prior to sowing using a standard germination assay:
1. Seeds were placed on moist filter paper in sterile Petri dishes.
2. Dishes were incubated at room temperature for 48–72 hours.
3. Seeds showing radicle emergence were counted as viable.
4. Only viable seed batches were used in the greenhouse experiment.
Soil Preparation
Soil Source and Homogenisation
• 120 kg of topsoil (0–15 cm depth) was collected from Site 4 (“extreme site”).
• Soil was sieved to remove stones and debris.
• All soil was homogenised to ensure uniform baseline conditions.
Moisture Conditioning
• Soil was pre-dampened to a water activity (aW) of 0.9.
• Moisture was verified using a calibrated hygrometer.
Pot Preparation and Layering Protocol
Pot Setup
• 120 pots (1 kg capacity each) were used.
• Pots were arranged into six treatment groups (T0–T5), each containing 20 pots.
Layering Procedure (per pot)
E.2.1 Add 400 g of soil to the base.
E.2.2 Place the designated SCOBY++ treatment at the centre.
E.2.3 Add 300 g of soil.
E.2.4 Add 5 washed quinoa seeds.
E.2.5 Add a final 300 g of soil.
This produced a consistent vertical geometry across all pots.
Randomisation Scheme
To minimise positional bias:
• Pots were placed on two greenhouse benches (1.5 m × 4 m).
• A randomisation matrix (Figure 5A in the main text) was used to assign pot positions.
• Treatments were interspersed across rows and columns to avoid clustering effects.
• Pot positions remained fixed throughout the experiment.
Irrigation Treatments
Two irrigation regimes were applied:
Regular Irrigation (RR)
• 10 pots per treatment.
• Received 300 mL initial watering, followed by 100 mL at each scheduled irrigation.
• Total water applied: 1000 ml.
Water Stress (RE)
• 10 pots per treatment.
• Received 300 mL initial watering, followed by reduced irrigation events.
• Total water applied: 600 ml.
Experiment Protocol
Day 1: Preparation 27/03/2025
Day 2: Assembly and sowing 28/03/2025
Day 3: First Watering 29/03/2025
Week 1 [ 1st Evaluation]: 01/04/2025
Week 2 [ 2nd Evaluation]: 08/04/2025
Week 3 [ 3rd Evaluation]: 15/04/2025
Week 4 [ Final Evaluation]: 22/04/2025
Irrigation and Monitoring Protocol
The watering schedule consisted of uniform initial hydration (300 mL) followed by differentiated schemes: continued regular watering (RR) or reduced watering under stress conditions (RE). Watering volumes were adjusted according to panels C and D of Figure 3. Emergence, seedling mortality, and growth metrics (e.g., plant height) were recorded twice weekly over a 24-day (3.4-week) period using consistent measurement techniques. Environmental variables such as temperature and humidity were monitored to maintain stable baseline greenhouse conditions [2].
Results
All SCOBY++ treatments outperformed the control (T0) in terms of seed emergence under both regular irrigation (RR) and drought stress (RE). The control group registered a total emergence of 28 seedlings (12 under RE and 16 under RR). In contrast, T2 recorded the highest emergence with 54 seedlings, followed by T3 with 46. Even under stress conditions, T2 and T3 maintained high emergence values (25 and 23, respectively), demonstrating that SCOBY-based treatments can enhance early seedling establishment in adverse conditions. Percent improvements in emergence compared to the control ranged from 21% (T4, T5) to a striking 93% (T2) [5,6].
Plant height was recorded as an integrative measure of growth and vigour during the fourth week. Dead or withered seedlings were scored as zero. The highest plant height was observed in T2 (145.9 cm combined total), followed by T1 and T5. Under drought stress, T2 maintained the highest combined total height (82.7 cm), indicating strong tolerance to severe water deficit. In contrast, the control group (T0) showed no growth under severe stress and only 43.6 cm under regular irrigation—nearly half the height of T2 under water stress and less than a third under optimal irrigation. Overall, T2 yielded an approximate 235% improvement in total height compared to the control, confirming its superior performance [6].
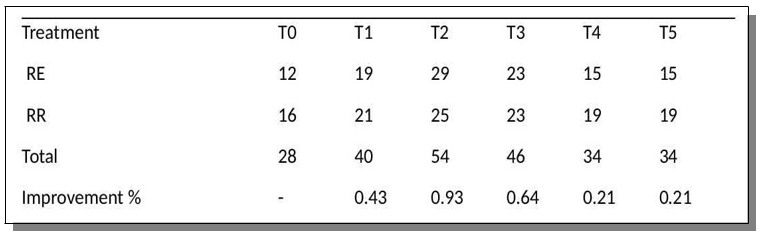
Table 1: Emergence Summary
Seedlings ceased height development; therefore, seedlings that did not survive after emergence were scored as “W” with an arithmetic value of zero. Measurements were recorded at the end of the 24-day period.
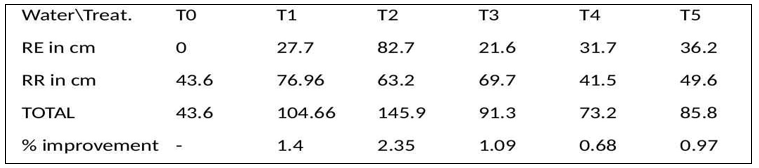
Table 2: Plant Height by Treatment
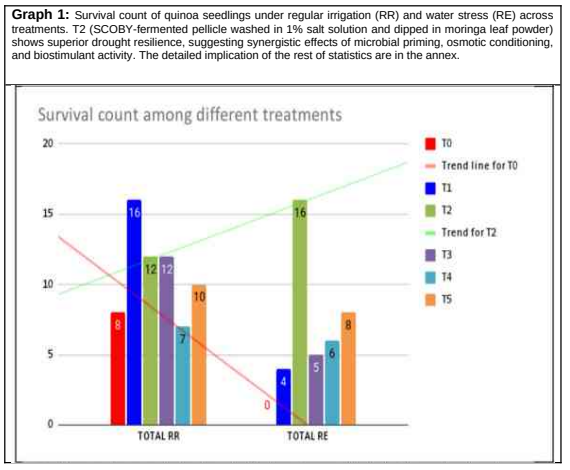
Survival rates reflected the resilience of seedlings under both watering regimes. The control group had a total of eight surviving plants, all under regular irrigation, indicating complete mortality under drought stress. Conversely, T2 and T5 achieved 28 and 18 surviving plants, respectively, demonstrating approximately 250% and 125% improvements compared to the control. Notably, T2 was the only treatment to achieve high survival under both conditions (12 in RR and 16 in RE—higher under stress), suggesting that the salt-washed SCOBY pellicle with Moringa (T2) significantly enhanced drought resilience [5,6].
Survival under drought differed significantly across treatments (p < 0.05; Supplementary Table S1). Overall, the results demonstrated greater resilience and vigour in T2 compared to the control—even under water stress. Emergence, plant height, and survival rates under stress were all higher in treated seedlings than in the control under both irrigation regimes. Given the inherent resilience of quinoa seeds, such improvements are remarkable and promising for further refinement and testing [2]. Supplementary Materials include full statistical analyses, expanded tables, and additional figures supporting the results presented in the main text.
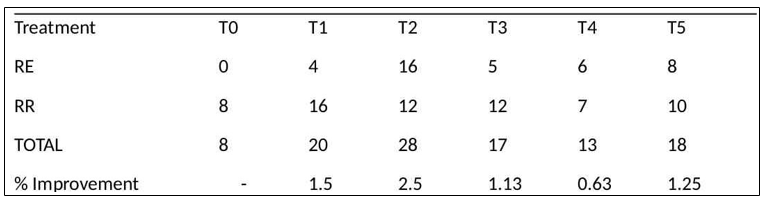
Table 3: Plant Survival (by count)
By comparing results, Treatment 1 (T1), which mirrors Treatment 2 (T2) except for the %1 NaCl pre-wash, demonstrated best results in regular irrigation, while treatment 2 (T2) demonstrated the best survival rates under water stress. Conversely, the control didn’t survive water stress (graph 1).
Statistical Analysis
Quantitative analyses were conducted to evaluate the effects of SCOBY++ treatments and watering regimes on quinoa emergence, survival, and final height. All analyses were performed using a two-way ANOVA framework, supplemented by chi-square tests for binary emergence data and Tukey HSD for post-hoc comparisons. Assumption checks were carried out to assess normality and homogeneity of variance.
Emergence
Because emergence is a binary variable (0/1), ANOVA is not appropriate. A chi-square test was used to compare the highest-performing treatment (T2) with the control (T0). The contingency table is shown below:

The ch-square statistic was![]() indicating a statistically significant improvement in emergence for T2 relative to the control. Across all treatments, emergence consistently exceeded the control under both irrigation regimes. Percent improvements ranged from 21% (T4, T5) to 93% (T2).
indicating a statistically significant improvement in emergence for T2 relative to the control. Across all treatments, emergence consistently exceeded the control under both irrigation regimes. Percent improvements ranged from 21% (T4, T5) to 93% (T2).
Plant Height
Final height was analysed using a two-way ANOVA with treatment and watering scheme as fixed factors. The corrected and standardised ANOVA table is presented below.
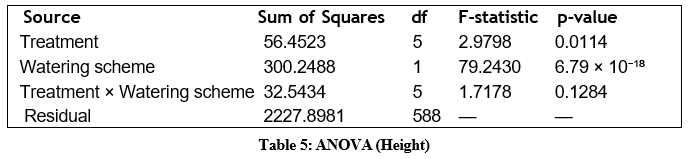
Both treatment and watering scheme had significant main effects on final height, while their interaction was not significant. This indicates that treatment effects were consistent across irrigation regimes.
Post-Hoc Comparisons
Tukey HSD tests were used to identify which treatments differed significantly from the control. Only T2 showed a statistically significant increase in height relative to T0.
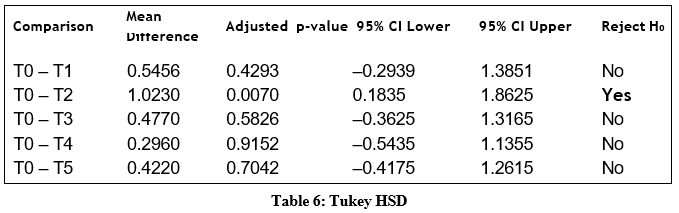
This confirms that T2 is the only treatment producing a statistically significant height increase relative to the control.
OLS Regression Model
Tukey HSD tests were used to identify which treatments differed significantly from the control. Only T2 showed a statistically significant increase in height relative to T0.
To further explore treatment and watering interactions, an OLS model was fitted. The model summary and coefficients are shown below.
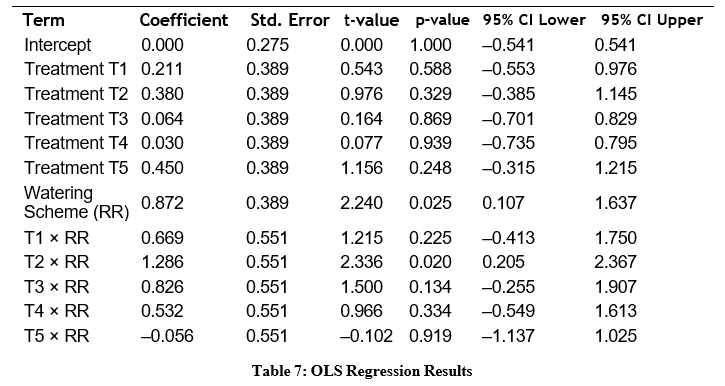
Model summary:
• R² = 0.149
• Adjusted R² = 0.133
• F(11, 588) = 9.339
• p(F-statistic) = 1.45 × 10 15-
The model confirms the ANOVA findings: watering scheme has a strong effect, and T2 shows the most robust performance.
Assumption Checks
Normality and homogeneity of variance were assessed using Shapiro–Wilk and Levene’s tests.
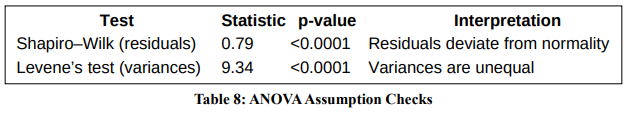
Although assumptions were violated, ANOVA remains robust due to the large, balanced sample size. The conclusions are further supported by the OLS model and non-parametric emergence analysis.
Integrated Interpretation
Across all metrics—emergence, survival, and height—T2 consistently outperformed the control, with statistically significant improvements in emergence and height, and the strongest survival under drought. The absence of a treatment × watering interaction indicates that SCOBY++ benefits are stable across irrigation regimes.
Permaculture and Science
Permaculture is often described as a framework for designing resilient, ecologically integrated systems grounded in principles of circularity, diversity, and long-term regeneration. It is not a scientific discipline in itself, nor does it rely on formal hypothesis testing. Rather, it is a practice-based design approach that draws on systems thinking and ecological pattern recognition [7,8]. While some practitioners refer to permaculture as a form of “indigenous science,” it is more accurately understood as a design philosophy that can align with scientific inquiry when it is grounded in validated, replicable observation rather than personal intuition or anecdotal experience [9].
This distinction is important in the context of agricultural research. Permaculture offers a conceptual lens—emphasizing feedback, integration, and the use of renewable, low-impact resources—but it does not, on its own, provide empirical evidence. Its value lies in guiding the formulation of interventions that can then be rigorously tested using scientific methods. In this study, permaculture principles informed the choice of a low-carbon, locally producible microbial amendment (SCOBY++), but the performance of that amendment was evaluated through controlled experimentation, statistical analysis, and reproducible measurement.
By separating the design logic from the empirical assessment, the study avoids overclaiming and ensures that the conclusions rest on measurable outcomes rather than philosophical alignment. The results therefore do not validate permaculture as a whole; instead, they demonstrate that a specific intervention inspired by permaculture principles—namely, a circular-economy microbial inoculant—can measurably improve quinoa emergence, survival, and growth under extreme environmental stress. This approach preserves the integrity of both domains: permaculture contributes the systems-level framing, while scientific inquiry provides the evidence base needed to assess effectiveness [10].
The use of SCOBY as a probiotic inoculant aligns with principles of permaculture and agroecology. Unlike synthetic agrochemicals or costly commercial bioformulations, SCOBY pellicle can be locally produced through fermentation of waste substrates, providing a scalable, low-carbon alternative for soil improvement. Life-cycle analysis shows that SCOBY has a substantially lower greenhouse-gas footprint than conventional cellulose carriers (0.315 vs. 6.00 g CO2e per kg of soil), supporting its integration into sustainable farming systems, particularly in resource-limited rural contexts. (Calculations in the following table were made using the online Climate Cloud Calculator [11].)
This study, additionally, highlights the remarkable potential of SCOBY++ formulations— symbiotic cultures of bacteria and yeasts enriched with Moringa oleifera—as plant probiotics capable of significantly improving quinoa seedling emergence, survival, and growth under challenging environmental conditions. The Bolivian Altiplano, one of the most extreme agricultural landscapes in the world, is characterized by high salinity, drought, temperature fluctuations, and declining soil fertility. In such a context, low-cost and ecologically sound solutions are urgently needed to support smallholder farmers while safeguarding both productivity and ecological resilience [12].
Our results demonstrate that SCOBY-based treatments function as highly effective bio stimulants, particularly under drought stress. Among the six treatments evaluated, T2— salt-washed SCOBY pellicles coated with Moringa powder—emerged as the most successful intervention (figure 4). It achieved a 93% increase in seed emergence, a 250% improvement in survival rate, and a 234% enhancement in plant height relative to the untreated control. This is especially notable given that all control seedlings perished under drought conditions, whereas T2 maintained high survival and growth, confirming its capacity to buffer against water stress.

These outcomes likely arise from multiple synergistic effects within the SCOBY++ system [13]. The microbial consortium present in the SCOBY may enhance root colonization, promote hormonal balance, and facilitate nutrient uptake [14]. Concurrently, Moringa contributes antioxidative and growth-promoting compounds, while SCOBY’s matrix may facilitate nano-nutrient uptake [15-20]. The salt-washing step applied to T2 may also have preconditioned the pellicle microbiome for osmotic stress, increasing its compatibility with the saline and arid soils of the Altiplano. Additionally, the pellicle itself may act as a micro-reservoir for water, hydrating upon irrigation and releasing moisture gradually [21-23].
From a statistical standpoint, both irrigation frequency and treatment type had significant independent effects on plant survival and height (p < 0.0001 and p < 0.001, respectively), while their interaction was not significant. This indicates that the benefits of SCOBY treatments are robust across different irrigation regimes—a critical feature for application in unpredictable or resource-limited environments [24]. In other words, these biotechnological improvements confer resilience not only under optimal conditions but also when water availability is severely constrained [25].
In this context, survival count reflects only the persistence of seedlings—alive or dead— without distinguishing between frail and vigorous individuals. In contrast, plant height offers a more rigorous indicator of performance, revealing treatment effects on growth and physiological resilience that survival alone may obscure. For example, although T3 under drought produced more survivors than T0 under irrigation, these survivors remained shorter and less vigorous. This illustrates why height offers a more rigorous assessment than survival count alone, and why T2 is the most representative SCOBY++ treatment, supporting both high survival and robust growth.
In addition to their biological effects, SCOBY++ treatments have demonstrated practical versatility under different environmental conditions [26]. For instance, the liquid formulation (T5), while slightly less effective than T2, still significantly improved emergence, survival, and height, making it suitable for integration into composting, trench agriculture, or foliar applications. These variations enable site-specific adaptations, depending on water availability, composting cycles, or seasonal constraints.
Importantly, the application of plant probiotics such as SCOBY++ (“SCOBY plus plus”) is not merely a technical innovation but a response to broader socio-ecological needs [27]. In the Altiplano, climate change, soil degradation, and monoculture pressures are threatening both agricultural productivity and biocultural heritage. Introducing microbial plant probiotics that enhance crop performance under harsh conditions without undermining soil microbial diversity (see for the impact of quinoa cultivation on soil and the environment) offers a viable path toward regeneration [28,13,14]. These treatments do not replace traditional knowledge— they amplify it. In doing so, they help reframe quinoa cultivation as both a biotechnological opportunity and a culturally embedded practice.

The integration of SCOBY-based treatments into broader permaculture designs—including rainwater harvesting, composting with native residues, and the use of local cover crops— offers a comprehensive model for sustainable intensification (figure 5).
Such a model moves beyond yield enhancement, aiming instead to rebuild agroecosystem function, farmer autonomy, and long-term resilience.
Conclusion and Future Address
This study demonstrates that SCOBY-based plant probiotic formulations enriched with Moringa oleifera leaf powder can substantially improve quinoa seedling emergence, survival, and growth under both optimal and water-limited conditions. Among the six treatments tested, the salt-washed SCOBY pellicle with moringa (T2) and the liquid SCOBY culture with moringa (T5) delivered the most consistent gains, with T2 sustaining high survival and growth even under severe drought. These results suggest that SCOBY++ treatments enhance water retention, nutrient mobilisation, and stress tolerance in extreme soils, offering a low-carbon, locally producible alternative to conventional carriers.
By integrating SCOBY++ into broader permaculture designs — including rainwater harvesting, composting with native residues, and the use of local cover crops — farmers in the high Andes could replace traditional fallow cycles with productive, regenerative systems. This approach aligns with global sustainability goals by reducing greenhouse gas emissions, restoring soil function, and strengthening climate resilience. Future work should prioritise participatory field trials, long-term soil health monitoring, and the exploration of additional fermentation-based bio inputs derived from local biodiversity. In doing so, SCOBY++ can move from greenhouse proof-of-concept to a scalable, community-driven solution for food security and ecological regeneration in high-altitude agroecosystems.
This study provides convincing evidence that SCOBY-based biotechnological treatments (SCOBY++) enriched with moringa leaf powder can significantly improve quinoa seedling emergence, survival, and growth under water-limited conditions. Among the six treatments tested, T2 (SCOBY pellicle treated with 1% NaCl and coated with Moringa powder) and T5 (liquid SCOBY culture supplemented with Moringa) showed the most consistent and robust results across all performance indicators. These treatments not only enhanced emergence rates and survival under drought stress but also promoted substantial vegetative growth, suggesting improvements in water retention, nutrient mobilization, and cellular resilience.
Future Directions
Future directions include scaling up these treatments through participatory field trials, evaluating their long-term impacts on soil health, and exploring additional bio-inputs derived from local biodiversity. Integrating fermentation-based biotechnologies with agroecological practices offers a promising pathway for the sustainable transformation of marginal agricultural systems.
A combination of interventions has been co-designed with the community to build on local initiatives. These include:
• compiling a list of nitrogen-fixing species suitable for plant companionship and cover cropping
• providing local training on rainwater and fog-water harvesting techniques
• establishing composting facilities with local participants as a basis for further development and research
Several local and endemic species have also been identified in and near the farming sites for their potential to naturally inoculate useful bacteria and fungi through cover crops, composting, and companion planting—subject to follow-up studies.
|
Nitrogen-fixing legume species (to reduce fallow time, descanso. |
Lupinus altimontanus, Azorella compacta, Hoffmannseggia minor, Arachis hypogaea (Royal Botanic Gardens Kew, n.d.), etc [11,24-27]. |
|
Microbiological-boost sources; used in composting, companion plants, and crop rotation. |
Mycorrhizal native grasses such as Festuca spp; and Jarava ichu; Azolla spp; yeast; and non-legume nitrogen-fixing species such as Colletia spp. [28-33]. |
|
Soil protection (erosion and degradation control) |
Parastrephia lepidophylla, Colletia spp, Senecio clivicola Wedd [34-37]. |
|
Potential sources for bio-pesticides. |
Senna spectabilis, Solanum spp, quinoa husk extracts; lupin seed extracts. Brassica seed extracts, Tagetes foetidissima, and nano-copper solutions [15,34-50]. |
Table 9: Locality-Specific Associated Species
These activities have already begun and continue to inform ongoing efforts toward sustainable intensification of quinoa in the Altiplano. Although questions remain about the productivity potential of organic and permaculture-informed systems, our findings demonstrate that sustainable, biologically based interventions can match or exceed conventional performance in harsh environments, while also safeguarding biodiversity and strengthening community resilience [29-50].
References
- Xi, X., Fan, G., Xue, H., Peng, S., Huang, W., & Zhan, J. (2024). Harnessing the potential of quinoa: Nutritional profiling, bioactive components, and implications for health promotion. Antioxidants, 13(7), 829.
- Bazile, D., Bertero, H. D., & Nieto, C. (2015). State of the Art Report on Quinoa around the World in 2013.
- Zulkadir, G., & Idikut, L. (2021). The impact of various sowing applications on the nutritional value of Quinoa Dry Herb. Journal of Food Processing and Preservation, 45(12), e15730.
- Angeli, V., Miguel Silva, P., Crispim Massuela, D., Khan, M. W., Hamar, A., Khajehei, F., ... & Piatti, C. (2020). Quinoa (Chenopodium quinoa Willd.): An overview of the potentials of the “golden grain” and socio-economic and environmental aspects of its cultivation and marketization. Foods, 9(2), 216.
- Latin America Post. (2024, March 5). Bolivia’s quinoa boom: From ancient staple to global superfood odyssey. Retrieved January 19, 2025.
- Statista. (2024, February 6). Agriculture. Farming. Quinoa production in Bolivia 2010– 2022. Retrieved January 19,2025.
- Holmgren, D. (2020). Essence of permaculture. Seymour, VIC, Australia: Melliodora Publishing.
- Didarali, Z., & Gambiza, J. (2019). Permaculture: Challenges and benefits in improving rural livelihoods in South Africa and Zimbabwe. Sustainability, 11(8), 2219.
- Bonifacio, A., Aroni, G., Villca, M., & Bentley, J. W. (2023). Recovering from quinoa: regenerative agricultural research in Bolivia. Journal of Crop Improvement, 37(5), 687-708.
- Reiff, J., Jungkunst, H. F., Mauser, K. M., Kampel, S.,Regending, S., Rösch, V., ... & Entling, M. H. (2024). Permaculture enhances carbon stocks, soil quality and biodiversity in Central Europe. Communications Earth & Environment, 5(1), 305.
- Jiménez-Gómez, A., Celador-Lera, L., Fradejas-Bayón, M., & Rivas, R. (2017). Plant probiotic bacteria enhance the quality of fruit and horticultural crops. AIMS microbiology, 3(3), 483.
- Frison, E. A., Cherfas, J., & Hodgkin, T. (2011). Agricultural biodiversity is essential for a sustainable improvement in food and nutrition security. Sustainability, 3(1), 238-253.
- Zero Hour Climate. (2024, March 14). Quinoa climate change: Resilience & impact. Retrieved January 19, 2025.
- Estrada, R., Cosme, R., Porras, T., Reynoso, A., Calderon, C., Arbizu, C. I., & Arone, G. J. (2023). Changes in bulk and rhizosphere soil microbial diversity communities of native Quinoa due to the monocropping in the Peruvian Central Andes. Microorganisms, 11(8), 1926.
- Canedo-Rosso, C., Hochrainer-Stigler, S., Pflug, G., Condori, B., & Berndtsson, R. (2021). Drought impact in the Bolivian Altiplano agriculture associated with the El Niño–Southern Oscillation using satellite imagery data. Natural Hazards and Earth System Sciences, 21(3), 995-1010.
- Afzal, I., Haq, M. Z. U., Ahmed, S., Hirich, A., & Bazile, D. (2023). Challenges and perspectives for integrating quinoa into the agri-food system. Plants, 12(19), 3361.
- Alsharif, W., Saad, M. M., & Hirt, H. (2020). Desert microbes for boosting sustainable agriculture in extreme environments. Frontiers in Microbiology, 11, 1666.
- Magrach, A., & Sanz, M. J. (2020). Environmental and social consequences of the increase in the demand for ‘superfoods’ world-wide. People and Nature, 2(2), 267-278.
- Vijitharan, S. (2024). A review on traditional knowledge: a sustainable solution for the climate crisis. Climate Crisis, Social Responses and Sustainability: Socio-ecological Study on Global Perspectives, 219-245.
- Birnbaum Fox, J. (2010, June 9). Indigenous science. In Cultural Survival Quarterly, 33 (1).
- Berkes, F., Folke, C., & Gadgil, M. (1994). Traditional ecological knowledge, biodiversity, resilience and sustainability. In Biodiversity conservation: problems and policies. Papers from the biodiversity programme beijer international Institute of ecological economics royal Swedish academy of sciences (pp. 269-287). Dordrecht: Springer Netherlands.
- Zougmoré, R., Segnon, A. C., & Thornton, P. (2023). Harnessing indigenous knowledge and practices for effective adaptation in the Sahel. Current Opinion in Environmental Sustainability, 65, 101389.
- Ortiz, A. M. D., Outhwaite, C. L., Dalin, C., & Newbold, T. (2021). A review of the interactions between biodiversity, agriculture, climate change, and international trade: research and policy priorities. One Earth, 4(1), 88-101.
- Woo, S. L., & Pepe, O. (2018). Microbial consortia: promising probiotics as plant biostimulants for sustainable agriculture. Frontiers in plant science, 9, 1801.
- Hanif, M. S., Tayyab, M., Baillo, E. H., Islam, M. M., Islam, W., & Li, X. (2024). Plant microbiome technology for sustainable agriculture. Frontiers in Microbiology, 15, 1500260.
- Trivium Foundation. (2022). Regenerative agriculture in Bolivian quinoa farming. Retrieved January 19, 2025.
- Instituto Interamericano de Cooperación para la Agricultura. (2021, March 3). Los desafíos de la quinua, el gran alimento andino, reseñados por la prestigiosa revista Global Food Security. Retrieved January 19, 2024.
- Maestro-Gaitán, I., Redondo-Nieto, M., González-Bodí, S., Rodríguez-Casillas, L., Matías, J., Bolaños, L., & Reguera,M. (2025). Insights into quinoa endophytes: core bacterial communities reveal high stability to water stress and genotypic variation. Environmental Microbiome, 20(1), 16.
- Walker, R., Otto-Pille, C., Gupta, S., Schillaci, M., & Roessner, U. (2020). Current perspectives and applications in plant probiotics. Microbiology Australia, 41(2), 95-99.
- Estrada, R., Cosme, R., Porras, T., Reynoso, A., Calderon, C., Arbizu, C. I., & Arone, G. J. (2023). Changes in bulk and rhizosphere soil microbial diversity communities of native Quinoa due to the monocropping in the Peruvian Central Andes. Microorganisms, 11(8), 1926.
- West, P. C., Gerber, J. S., Engstrom, P. M., Mueller, N. D.,Brauman, K. A., Carlson, K. M., ... & Siebert, S. (2014). Leverage points for improving global food security and the environment. Science, 345(6194), 325-328.
- Ferguson, R. S., & Lovell, S. T. (2014). Permaculture for agroecology: design, movement, practice, and worldview. A review: RS Ferguson, ST Lovell. Agronomy for sustainable development, 34(2), 251-274.
- Soliman¹, G. S., Cloy, J., Gravoui, C., & Parker, J. Opportunities and challenges of managing the rhizosphere’s biota–for food intensification, through controlled application of fertilisers with commercial Arbuscular Mycorrhizal fungi.
- Chawla, P. R., Bajaj, I. B., Survase, S. A., & Singhal, R. S. (2009). Microbial cellulose: fermentative production and applications. Food Technology & Biotechnology, 47(2).
- Cohen, G., Sela, D. A., & Nolden, A. A. (2023). Sucrose concentration and fermentation temperature impact the sensory characteristics and liking of kombucha. Foods, 12(16), 3116.
- Jacobsen, S. E., Mujica, A., & Jensen, C. R. (2003). The resistance of quinoa (Chenopodium quinoa Willd.) to adverse abiotic factors. Food reviews international, 19(1-2), 99-109.
- Sun, Y., & Jacobsen, S. E. (2013). Quinoa: a multipurpose crop with the ability to withstand extreme conditions in the field. CABI Reviews, (2013), 1-10.
- [T] Where And What Is The Altiplano? The Altiplano in Bolivia. World Atlas. Available at. Last accessed on 07/07/2025.
- Peyghan, K., Golabi, M., & Albaji, M. (2020). Simulation of quinoa (Chenopodium quinoa) yield and soil salinity under salinity and water stress using the SALTMED model. Communications in Soil Science and Plant Analysis, 51(18), 2361-2376.
- Barrientos-Pérez, E., Carevic-Vergara, F. S., Rodriguez,J. P., Arenas-Charlín, J., & Delatorre-Herrera, J. (2023). Effect of native vegetative barriers to prevent wind erosion: a sustainable alternative for quinoa (Chenopodium quinoa Willd.) production. Agriculture, 13(7), 1432.
- Gaffney, J., Bing, J., Byrne, P. F., Cassman, K. G., Ciampitti, I., Delmer, D., ... & Warner, D. (2019). Science-based intensive agriculture: Sustainability, food security, and the role of technology. Global Food Security, 23, 236-244.
- Shade, A., Buckley, D. H., & Zinder, S. H. (2011). The kombucha biofilm: a model system for microbial ecology. Final report on research conducted during the Microbial Diversity course. Marine Biological Laboratories, Woods Hole, MA.
- Harrison, K., & Curtin, C. (2021). Microbial composition of SCOBY starter cultures used by commercial kombucha brewers in North America. Microorganisms, 9(5), 1060.
- Pedraza, R. O. (2016). Acetic acid bacteria as plant growth promoters. In Acetic acid bacteria: ecology and physiology (pp. 101-120). Tokyo: Springer Japan.
- Mashamaite, C. V., Ngcobo, B. L., Manyevere, A., Bertling, I., & Fawole, O. A. (2022). Assessing the usefulness of Moringa oleifera leaf extract as a biostimulant to supplement synthetic fertilizers: A Review. Plants, 11(17), 2214.
- Mashamaite, C. V., Ngcobo, B. L., Manyevere, A., Bertling, I., & Fawole, O. A. (2022). Assessing the usefulness of Moringa oleifera leaf extract as a biostimulant to supplement synthetic fertilizers: A Review. Plants, 11(17), 2214.
- Reis, V. M., & Teixeira, K. R. D. S. (2015). Nitrogen fixing bacteria in the family Acetobacteraceae and their role in agriculture. Journal of basic microbiology, 55(8), 931-949.
- Aboul-Nasr, M. B., Mohamed, S. S., & Mohamed, M. (2023). Gold Nanoparticles Induction Using Kombucha SCOBY Disc. Sohag Journal of Sciences, 8(3), 353-360.
- Crespo, J. M., Boiardi, J. L., & Luna, M. F. (2011). Mineral phosphate solubilization activity of Gluconacetobacter diazotrophicus under P-limitation and plant root environment. Agricultural Sciences, 2.
- Tsilo, P. H., Basson, A. K., Ntombela, Z. G., Dlamini, N. G., & Pullabhotla, R. V. (2023). Biosynthesis and characterization of copper nanoparticles using a bioflocculant produced by a yeast Pichia kudriavzevii isolated from Kombucha tea SCOBY. Applied Nano, 4(3), 226-239.



