Research Article - (2025) Volume 8, Issue 2
Surgical Management of Osteomyelitis Fistulas: Proposed Classification and Strategic Treatment Approaches
2Junior Orthopedic Infection Surgeon, Akhos Clinic - Orthopedic Infection Treatment Center - SEPS EQ , Brazil
3Plastic Surgeon Specialized in Reconstruction, Akhos Clinic - Orthopedic Infection Treatment Center-, Brazil
4Orthopedic Surgeon, Akhos Clinic - Orthopedic Infection Treatment Center - SEPS EQ 709/909 CONJUNTO , Brazil
5Infectious Diseases Doctor, Akhos Clinic - Orthopedic Infection Treatment Center - SEPS EQ 709/909 C, Brazil
6Nurse Specialized in Wound Care, Akhos Clinic - Orthopedic Infection Treatment Center - SEPS EQ 709/, Brazil
Received Date: Apr 08, 2025 / Accepted Date: May 02, 2025 / Published Date: May 15, 2025
Copyright: ©2025 Mario Soares Ferreira Junior, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Ferreira Junior, M. S., Teixeira, C. R. M., Brandão, P. C., Santos, I. C. D., Ono, F., et al. (2025). Surgical Management of Osteomyelitis Fistulas: Proposed Classification and Strategic Treatment Approaches. Int J Ortho Res, 8(2), 01-13.
Abstract
This article presents a comprehensive analysis of the surgical management of cutaneous fistulas secondary to chronic osteomyelitis, with emphasis on a newly proposed classification system designed to optimize treatment strategies and reduce complication rates. The classification is based on the anatomical location of the fistulas in relation to prior surgical scars, offering a pragmatic and reproducible framework that supports clinical decision-making. It facilitates the identification of cases requiring multidisciplinary collaboration, particularly those necessitating the involvement of plastic and microsurgical teams. The overarching objective is to enhance surgical outcomes by minimizing risks such as skin necrosis, wound dehiscence, and recurrent infection, especially in complex reconstructions involving compromised soft tissue. By integrating anatomical assessment with evidence-based surgical principles, the proposed approach aims to standardize care pathways, support individualized treatment planning, and improve the overall quality of care in patients with chronic osteomyelitis of the lower limb.
Keywords
Chronic osteomyelitis, Cutaneous fistulas, Surgical management, Classification system, soft tissue reconstruction
Introduction
Osteomyelitis remains one of the most challenging conditions in orthopedic practice, particularly in the lower limb, due to its complex nature, variability in clinical and surgical techniques, and the lack of specific training in soft tissue management in orthopedic education [1,2]. This condition, characterized by bone infection primarily caused by microorganisms such as Staphylococcus aureus, can progress in acute, subacute, and chronic forms, each requiring different therapeutic strategies [3].
In the context of the lower limb, complications associated with inadequate treatment are even more pronounced due to the limb’s critical role in weight-bearing and mobility. Key challenges include incomplete surgical debridement, multiple ineffective surgeries, prolonged use of parenteral antibiotics, extended hospital stays, non-healing wounds, chronic pain, mutilating surgeries, unnecessary use of external fixators, and excessive costs [4,5]. The functional impairments resulting from these complications can severely restrict a patient's ability to walk, maintain independence, and perform daily activities.
Among these complications, issues related to wound closure are particularly significant in the lower limb, where adequate soft tissue coverage is essential to prevent exposure of critical structures such as tendons and bones. In cases involving prior scars and fistulas, poor management can result in skin necrosis, surgical wound dehiscence, reinfections, and functional impairment due to skin contractures. Such outcomes not only compromise limb function but also prolong hospital stays and increase healthcare costs associated with serial dressings, hyperbaric chamber therapy, vacuum-assisted closure, and prolonged antibiotic use [6].
Osteomyelitis, particularly in the lower limb, is defined as an infection of bone tissue caused by bacteria, mycobacteria, or fungi. Chronic osteomyelitis is characterized by biofilm formation and invasion of bone canaliculi, with bacteria, particularly Staphylococcus spp., internalized within osteocytes [7]. The invasive nature of this condition, coupled with its potential to compromise vital structures in the lower limb, renders oral and parenteral antibiotics insufficient. Surgical intervention with aggressive and radical debridement, as described by Cierny (1984), remains the only effective treatment.
The success of the procedure, particularly in weight-bearing bones such as the tibia and femur, depends on thorough debridement of both the infected bone and adjacent soft tissues, including infected fascia, compromised muscle, nonviable skin, old scars, and fistulas. Poor management of these structures in the lower limb often results in soft tissue defects that are particularly challenging to reconstruct, given the limb’s exposure and functional demands. If not adequately addressed, there is a significant risk of infection recurrence and progressive functional loss [8,9].
Therefore, it is imperative that orthopedic surgeons treating lower limb osteomyelitis possess the skills to achieve adequate closure of soft tissues and skin. This minimizes the emergence of new complications, preserves limb function, and prevents the persistence of infection. To support this objective, we propose a simple classification system to guide surgical treatment, assist in decision-making, and identify technical limitations. Since 2018, this classification has been internally utilized at the Akhos Clinic, located in Brasília, Brazil, yielding satisfactory and predictable outcomes.
Osteomyelitis: Definition and Classification
Osteomyelitis, a severe bone infection predominantly caused by Staphylococcus aureus, presents considerable diagnostic and therapeutic challenges, particularly in the lower extremities due to their complex anatomical structure and functional demands. The etiopathogenesis of osteomyelitis encompasses hematogenous dissemination, direct inoculation (often post-traumatic or post- surgical), and contiguous spread from adjacent soft tissue infections [10,11]. In chronic presentations, especially within the lower limb, the clinical management is further complicated by the formation of biofilms, sequestrum development, and persistent infection despite aggressive therapeutic interventions [4].
The optimal management of chronic osteomyelitis requires an interdisciplinary strategy, incorporating meticulous surgical debridement, targeted antimicrobial therapy, and advanced reconstructive techniques aimed at preserving limb function and structural integrity [12,13]. Existing classification systems, such as the Cierny–Mader classification, provide foundational guidance by stratifying osteomyelitis according to anatomical extent and host physiological status. However, these frameworks exhibit limitations in their applicability to complex fistulizing disease and soft tissue involvement, particularly in the context of lower limb osteomyelitis.
To address these limitations, the Soares Classification was developed to offer a more nuanced approach, specifically categorizing fistulas associated with chronic osteomyelitis of the lower extremity. This classification integrates both anatomical localization and pathological features, thereby facilitating more precise therapeutic planning. Table 1 outlines the Soares Classification, delineating four distinct types of fistulas and corresponding clinical and surgical considerations.
|
CLASSIFICATION TYPE |
CHARACTERISTICS AND SURGICAL RECOMMENDATIONS |
|
Type 0 – Fistula without prior scar |
|
|
Type 1 – Fistula over a previous surgical scar (on-scar) |
|
|
Type 2 – Fistula distant from surgical scar but in the same affected limb (off-scar) |
|
|
Type 3 – Fistulas in distal anteromedial leg or compromised skin areas |
|
Table 1: Soares Classification for Skin Fistulas Associated with Chronic Osteomyelitis
The Soares Classification offers a pragmatic and innovative framework that enhances clinical decision-making by aligning surgical interventions with the anatomical and pathological presentation of fistulas in chronic osteomyelitis. Type 0 lesions generally allow for straightforward surgical management, whereas Types 1 through 3 increasingly necessitate complex reconstructive approaches, often requiring collaboration with reconstructive or microsurgical specialists.
By integrating the Soares Classification into existing diagnostic and therapeutic algorithms, clinicians are better equipped to navigate the intricacies osteomyelitis. Further validation through multicenter clinical studies and comparative outcome analyses will be critical for establishing its broader applicability and prognostic utility, ultimately contributing to improved functional outcomes and quality of life in affected patients.
Soares Classification: An Innovative Framework for Man- aging Chronic Osteomyelitis Fistulas
Osteomyelitis remains a significant clinical challenge in orthopedic practice, particularly in the lower limb, due to its complex etiology, varied presentations, and elevated risk of complications such as chronic infection and extensive soft tissue damage [10,14]. The lower limb, being a weight-bearing region, is especially vulnerable to functional impairments and prolonged recovery times when affected by osteomyelitis. Management of skin fistulas, a common complication in chronic cases, requires a precise and structured approach to guide therapeutic decisions and optimize clinical outcomes [11].
The Soares Classification introduces a systematic framework for managing skin fistulas associated with osteomyelitis in the lower limb. This classification categorizes fistulas into four distinct types, each defined by specific anatomical and pathological characteristics, objectives, and surgical approaches. By tailoring treatment plans to these classifications, clinicians can address the complexity of debridement, reconstruction, and infection control with greater precision. Additionally, the framework emphasizes the importance of advanced reconstructive strategies and multidisciplinary collaboration to restore function and prevent recurrence [15,16].
Proposed Treatment for Each Type in the Soares Classifi- cation
The Soares Classification delineates a structured paradigm for the surgical management of cutaneous fistulas in chronic osteomyelitis of the lower extremity. Each type introduces specific anatomical and surgical complexities that necessitate a tailored operative plan. The following section expands upon the treatment guidelines for each classification subtype, emphasizing the importance of meticulous surgical technique, strategic tissue handling, and interdisciplinary coordination.
Type 0
In Type 0 cases, the fistula is not associated with previous surgical scars, thereby facilitating a more straightforward surgical approach. The operative procedure is initiated with a fusiform incision strategically oriented along the natural skin tension lines and in accordance with regional anatomical considerations. This incision design allows for optimal exposure of the pathological tract and ensures comprehensive excision of necrotic and infected tissue while preserving surrounding healthy structures [14,17].
Figure 1 illustrates both the schematic and practical aspects of the Type 0 surgical approach. Image (a) presents a diagrammatic representation of the fusiform incision, emphasizing alignment with Langer’s lines to optimize wound closure and minimize tension. Image (b) displays the clinical application of this approach in a patient, highlighting the preoperative skin markings and the anatomical localization of the fistula. This dual representation underscores the integration of surgical planning and anatomical principles essential for successful outcomes in Type 0 cases.
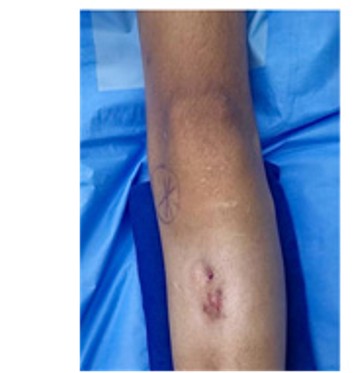
Figure 1: Schematic and clinical representation of a surgical approach for Type 0 fistulas in the lower limbs.
Legend: Clinical image of the lower limb demonstrating the fistula site and preoperative markings for the incision.
A fusiform incision is strategically performed over the fistulous tract, extending longitudinally in alignment with the skin’s natural tension lines and in accordance with standard anatomical and surgical landmarks of the lower limb. This approach enables optimal exposure of the infected site and surrounding tissues, facilitating a more comprehensive and controlled debridement. Following the skin incision, an atraumatic dissection of the subcutaneous tissue from the underlying fascia is carried out with the primary objective of preserving vascular supply and minimizing additional iatrogenic trauma. Tissue preservation at this stage is critical to promoting effective wound healing and mitigating postoperative complications, such as delayed epithelialization and cutaneous necrosis [16].
Subsequent to debridement, emphasis is placed on achieving a tension-free, multilayered closure, ensuring anatomical approximation of the wound edges. The use of meticulous suturing techniques-adapted to the biomechanical demands of the lower extremity-serves to enhance wound stability, prevent dehiscence, and promote uniform tissue regeneration. This is particularly relevant in the lower limb, where dynamic mechanical loads and mobility impose additional stress on surgical sites, thereby increasing the risk of healing disturbances [10,18].
In clinical scenarios where, primary closure is unfeasible or inadequate due to tissue loss or tension, the application of local advancement flaps is recommended. These flaps provide durable coverage, re-establish skin integrity, and offer protection to exposed structures, thus reducing the potential for secondary infection. Beyond functional recovery, advancement flaps contribute significantly to the aesthetic restoration of the limb and help preserve mobility by maintaining soft tissue elasticity and contour [19].
This comprehensive surgical protocol for Type 0 fistulas in chronic lower limb osteomyelitis integrates key principles of infection control, vascular preservation, and reconstructive planning. By combining precise incision techniques, gentle tissue handling, tension-free closure, and the judicious use of local flaps, this approach aligns with contemporary orthopedic surgical standards and addresses the multifaceted challenges posed by chronic infections in weight-bearing anatomical regions.
Type 1
Type 1 fistulas are typically located along previous surgical scars and are considered integral components of the infected complex. These scars frequently harbor colonized or devitalized tissue and, therefore, require thorough excision as part of the surgical management. The operative strategy begins with a fusiform incision that encompasses the entire extent of the prior scar and the fistulous tract. Scar tissue is systematically classified as biologically compromised and a potential nidus for persistent infection, necessitating its complete removal to reduce the microbial burden and eliminate potential reservoirs of reinfection [14,20].
Following the initial incision, a deep wedge-shaped debridement is performed, directed toward the underlying bone. This maneuver facilitates the radical excision of necrotic and infected soft tissue, while enabling direct access to osseous structures for evaluation and debridement. Adequate resection of both bone and surrounding infected tissue is critical to reducing the risk of chronicity and recurrence. To promote proper wound healing and minimize the incidence of dehiscence, deep tissue layers are approximated using durable absorbable or non-absorbable sutures, ensuring mechanical stability during the reparative process [21,22].
Consistent with the principles applied to Type 0 lesions, atraumatic dissection of the subcutaneous tissue from the underlying fascia is essential to preserving vascular integrity and mitigating iatrogenic trauma. This technique ensures sufficient perfusion to the surgical site, facilitating oxygenation, nutrient transport, and cellular regeneration, which are paramount for effective tissue healing. Moreover, preservation of vascular networks significantly reduces the risk of soft tissue necrosis and impaired wound closure [16].
A tension-free multilayer closure, particularly of the skin and deep fascial planes, is fundamental in preventing complications such as wound breakdown and reinfection. In most cases, adequate coverage can be achieved through the use of local advancement flaps, which not only restore cutaneous continuity but also enhance both functional and aesthetic outcomes. The employment of flaps additionally mitigates reinfection risk by providing well- vascularized tissue to the previously infected area [23].
The preoperative phase involves comprehensive planning with careful identification of chronic infection sites-typically involving bones such as the femur or tibia-and the precise demarcation of surgical access points. Advanced imaging modalities may assist in delineating the extent of osseous and soft tissue involvement, ensuring complete excision during surgery. During theintraoperative stage, wide surgical exposure enables complete debridement of infected bone and soft tissue. When required, orthopedic fixation devices (e.g., plates, rods, or external fixators) are employed to provide temporary or definitive stabilization, particularly in cases of structural compromise following debridement. This is followed by the creation of a biologically viable surgical bed, which is essential for subsequent reconstruction [24,25].
In the postoperative phase, emphasis is placed on ensuring structural integrity and minimizing tension along the closure site. The use of long-lasting sutures to secure deep layers, in combination with mechanical stabilization, contributes to enhanced healing dynamics and reduces the risk of recurrence.
Figure 2 illustrates the surgical strategy for managing Type 1 fistulas, which are commonly located on previous surgical scars. Image (a) presents a schematic depiction of the fusiform incision encompassing the entire scar and fistulous opening. Clinical images in (b) trace the operative sequence from the preoperative marking of the scar and fistula to the immediate postoperative result, evidencing the removal of contaminated tissues and reconstruction of the surgical field. This integrated approach- comprising wide excision, wedge-shaped debridement, tension- free closure, and vascularized flap coverage-constitutes a robust strategy for infection control and functional restoration in chronic osteomyelitis of the lower limb.
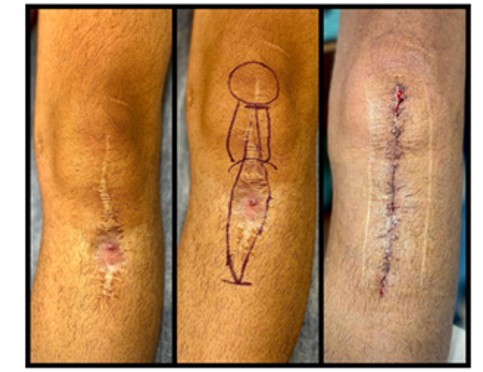
Figure 2: Management of Type 1 fistulas involving previous surgical scars in the lower limb
Legend: Clinical images demonstrating (from left to right): (1) the presence of the fistula on a previous scar, (2) preoperative planning
with markings for the fusiform incision, and (3) the immediate postoperative result showing closure after excision and debridement
The treatment of Type 1 fistulas, commonly located over previous surgical scars, follows a structured five-step protocol designed to address the anatomical and pathological complexities associated with chronic osteomyelitis in the lower limb. These fistulas are typically integrated into the infected surgical field and necessitate comprehensive excision and reconstruction to minimize recurrence and optimize functional outcomes.
The first step consists of performing a fusiform incision that encompasses the entirety of the previous surgical scar and the associated fistulous tract. This incision is planned in alignment with natural skin tension lines and regional anatomy to facilitate subsequent closure and reduce the risk of tension-related complications. The complete excision of scar tissue, which is often contaminated and represents a potential source of reinfection, is essential for decreasing bacterial load and establishing a clean surgical field [14,20].
The second step involves a deep wedge-shaped debridement, extending from the skin surface toward the bone. This maneuver enables the thorough removal of necrotic, infected, or non-viable tissue, thereby reducing the microbial burden and promoting the formation of a biologically active bed suitable for tissue regeneration. Accurate surgical planning ensures that both the osseous structures and surrounding soft tissues are properly addressed, reducing the likelihood of persistent infection [24,25].
In the third step, an atraumatic dissection of the subcutaneous tissue from the fascia is performed to preserve the vascular supply critical for tissue viability. By maintaining microvascular integrity, this technique facilitates effective oxygenation and nutrient delivery, which are essential for tissue repair and prevention of postoperative complications such as delayed healing or skin necrosis [16].
The fourth step is the tension-free closure of all anatomical layers using absorbable or non-absorbable sutures, selected based on the biomechanical demands of the region and the depth of each tissue plane. Proper approximation of the deep fascial, subcutaneous, and dermal layers enhances wound stability, reduces the risk of dehiscence, and supports uniform healing. In weight-bearing regions such as the lower limb, achieving tension-free closure is particularly important due to the mechanical stress imposed during ambulation [10,23].
Finally, when primary closure is unfeasible or when tissue coverage is insufficient, a local advancement flap is utilized. This reconstructive technique offers durable, vascularized coverage of the surgical site, restoring skin integrity and minimizing the risk of reinfection. In addition to protecting underlying structures, local flaps improve both functional and aesthetic outcomes, which are essential for preserving mobility and limb appearance [24].
In summary, the five-step protocol for Type 1 fistulas integrates key surgical principles-excision of contaminated tissue, radical debridement, vascular preservation, layered closure, and flap reconstruction-into a comprehensive framework. This approach enhances infection control, supports tissue regeneration, and aligns with best practices in orthopedic and reconstructive surgery for chronic osteomyelitis of the lower limb.
Type 2
Type 2 fistulas, which are anatomically distant from previous surgical scars, represent a considerable surgical challenge due to the extent of tissue loss and the complexity involved in achieving effective and durable wound closure. These cases often necessitate advanced surgical planning and specialized technical execution. The initial step of the treatment consists of the complete excision of the prior surgical scar (if present within the affected limb segment), the entire fistulous tract, and surrounding infected tissues. This comprehensive resection is fundamental to reducing bacterial load and eliminating potential reservoirs of reinfection. Moreover, it establishes a clean, biologically viable surgical field that serves as the foundation for subsequent reconstructive procedures [14,26].
Following excision, the resultant defect is carefully assessed in terms of its size, depth, and involvement of adjacent anatomical structures. This evaluation is critical to determine the feasibility of achieving a tension-free primary closure. In instances where direct closure is not possible-particularly in cases involving substantial tissue loss-early involvement of a plastic or microsurgical specialist is highly recommended. These professionals are equipped to perform complex reconstructions using a range of techniques, from local and regional flaps to fasciocutaneous, musculocutaneous flaps, or even free tissue transfer, depending on the extent and location of the defect [11,17].
A multidisciplinary approach is essential for the effective management of Type 2 fistulas. Collaboration among orthopedic surgeons, plastic surgeons, and microsurgeons ensures comprehensive care by addressing not only the mechanical and infectious components of the lesion but also its aesthetic and functional implications. Such an integrative strategy reduces the risk of complications including wound dehiscence, soft tissue necrosis, and recurrence of infection. Additionally, the use of advanced reconstructive techniques enhances vascularity, improves tissue integration, and minimizes the need for future revision surgeries-contributing significantly to the patient’s long- term recovery and quality of life [27].
The first operative step involves the meticulous removal of all devitalized and infected tissue, including the prior scar (if present), the fistulous tract, and surrounding zones of inflammation or fibrosis. This step is crucial not only for bacterial control, but also to prepare the surgical site for later stages of reconstruction [14,20]. The second step consists of an in-depth evaluation of the post- debridement defect, with particular attention to dimensions and potential for spontaneous closure. If closure cannot be achieved without tension, reconstructive strategies must be planned and executed promptly to avoid exposure-related complications [10,17].
In situations involving large or anatomically complex defects, the participation of plastic or microsurgical teams is indispensable. Techniques such as fasciocutaneous and musculocutaneous flaps offer excellent vascular support and mechanical stability to the reconstructed area. These flaps are especially suitable for weight- bearing regions or areas requiring high durability. Their use not only promotes effective healing but also helps restore functionality and limb contour [16,28].
The final stage of treatment is the implementation of the selected reconstructive technique, tailored to the individual anatomical and pathological context of the patient. Fasciocutaneous and musculocutaneous flaps are preferred due to their ability to provide resilient, well-vascularized coverage, which is critical for tissue regeneration and wound stability. When adequately planned and executed, these flaps ensure optimal functional and aesthetic outcomes, significantly reducing the risk of reinfection and structural failure over time [19,29].
Figure 3 illustrates the surgical management of Type 2 fistulas, characterized by their anatomical location away from previous surgical incisions and the presence of extensive tissue compromise. Image (a) schematically depicts the absence of prior scarring and highlights the area designated for resection. The clinical photographs in image (b) document the preoperative condition and the outcome following complete resection of the fistulous tract and associated tissue. This integrative and carefully planned surgical approach is crucial to reducing microbial burden, preventing recurrence, and achieving successful reconstruction for both functional recovery and limb aesthetics.
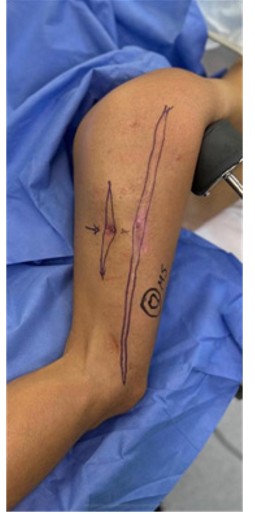
Figure 3: Surgical management of Type 2 fistulas located away from previous surgical scars.
Legend: Clinical images demonstrate the surgical site and preparation for excision
Achieving a tension-free closure across all anatomical layers- including fascia, subcutaneous tissue, and skin-is a critical determinant of successful surgical repair in Type 2 fistulas. This principle is essential for maintaining the structural integrity of the wound, reducing the risk of cutaneous necrosis, and preventing postoperative reinfection, especially in anatomically complex and weight-bearing regions such as the lower limb. A meticulous approach to closure, incorporating precise suture techniques and, when necessary, the use of advanced reconstructive flaps, contributes not only to the durability of the surgical outcome but also to its aesthetic quality. These measures highlight the importance of integrating high-level surgical proficiency with reconstructive expertise to achieve functional restoration and long- term success in the management of chronic osteomyelitis-related fistulas [10,23].
Type 3
Type 3 fistulas represent the most advanced and severe form of chronic osteomyelitis-related cutaneous complications in the lower limb. These cases are marked by extensive soft tissue loss, significant vascular compromise, and the absence of viable local tissue suitable for advancement or primary closure. The critical challenge in managing Type 3 lesions lies in the profound alteration of local vascularity, which significantly increases the risk of postoperative complications such as cutaneous necrosis, wound dehiscence, and persistent or recurrent infection [13,19].
Due to the complexity of these presentations, the management of Type 3 fistulas necessitates a comprehensive multidisciplinary approach. Coordination among orthopedic surgeons, plastic surgeons, and microsurgeons is indispensable to address the multifactorial challenges posed by these cases. This collaborative strategy is essential not only to ensure complete infection control through radical debridement but also to plan and execute advanced reconstructive procedures capable of restoring both structural integrity and functional capacity [16,25].
Preoperative evaluation must include a thorough assessment of tissue viability and vascular supply, utilizing both clinical examination and imaging techniques (e.g., Doppler ultrasound, CT angiography) to guide decision-making. In most cases, local flaps are contraindicated due to poor tissue quality and insufficient perfusion. As such, regional or free tissue transfer becomes the preferred option, often involving musculocutaneous or fasciocutaneous flaps harvested from well-vascularized donor sites. These techniques offer reliable coverage of extensive defects, promote effective tissue integration, and reduce the likelihood of reinfection.
Figure 4 illustrates the surgical management of Type 3 fistulas in the context of severe osteomyelitis. The schematic diagram in image (a) highlights the typical characteristics of these lesions, including substantial tissue destruction and impaired vascularization. The clinical image (b) demonstrates the intraoperative scenario, with visible areas of exposed bone and soft tissue loss. These cases exemplify the necessity of integrative care and advanced surgical planning. A successful outcome depends on precise excision of necrotic tissue, accurate evaluation of recipient bed vascularity, and the application of complex reconstructive strategies. These measures collectively enhance the probability of durable closure, functional recovery, and aesthetic preservation, while minimizing the risk of complications such as flap failure, reinfection, or delayed wound healing.
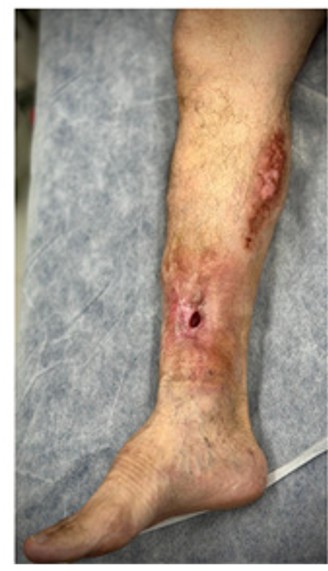
Figure 4: Surgical management of Type 3 fistulas in the lower limb, characterized by severe tissue loss and vascular compromise
Legend: (a) Schematic representation of a Type 3 fistula, highlighting the extensive tissue loss and the area of significant vascular compromise requiring comprehensive evaluation and planning. (b) Clinical image of the lower limb, illustrating the severity of tissue damage and the presence of the fistula, necessitating a multidisciplinary approach for surgical management.
The treatment process for Type 3 fistulas begins with a comprehensive evaluation of the defect, encompassing the extent of soft tissue loss, the viability of adjacent anatomical structures, and the degree of local vascular compromise. This detailed preoperative assessment is fundamental for guiding surgical decision-making, as it allows for the development of a patient-specific treatment plan that aligns with the biological and mechanical demands of the defect [8,13]. Identifying the precise characteristics of the lesion is essential not only for defining the reconstructive strategy but also for optimizing the functional and aesthetic outcomes of the intervention.
Once the planning phase is complete, surgical intervention proceeds with the complete excision of the fistulous tract and the radical removal of all necrotic or infected tissues. This step is crucial for reducing the bacterial load and creating a sterile, well- vascularized environment conducive to reconstruction. Given the extent of tissue loss commonly encountered in Type 3 cases, advanced reconstructive techniques are frequently required. These include the use of musculocutaneous or fasciocutaneous flaps, such as the anterolateral thigh flap or the reverse sural flap, which provide durable and vascularized tissue coverage. These flaps promote tissue regeneration, reestablish the anatomical contour, and significantly reduce the risk of recurrent infection, making them particularly advantageous in cases of complex lower limb osteomyelitis [20,23].
The final stage of treatment focuses on achieving a tension-free closure of the reconstructed area. This step is critical for maintaining tissue viability, ensuring the stability of the wound margins, and preventing complications such as dehiscence, necrosis, or structural failure. Tension-free closure techniques, when executed by teams with expertise in plastic and microsurgery, provide robust mechanical support while ensuring aesthetically acceptable results. This not only facilitates early mobilization and functional restoration but also contributes to long-term limb preservation and improved quality of life for the patient [9,13,15,16].
Surgical Tips and Tricks for Different Types of Fistulas The surgical management of skin fistulas associated with chronic osteomyelitis of the lower limb demands a structured and evidence- based approach, guided by the biomechanical demands of the region and the principles of soft tissue reconstruction. The Soares Classification stratifies these fistulas into four types (Type 0 to Type 3), each with distinct anatomical and pathological characteristics, requiring tailored surgical strategies to optimize both functional and structural outcomes.
In Type 0 fistulas, where soft tissue integrity and vascularity are preserved, the primary objective is to excise the fistulous tract with minimal disruption to surrounding tissues. A fusiform excision aligned with Langer’s lines is preferred, as it minimizes wound tension and promotes physiological wound healing. The use of monofilament absorbable sutures such as polydioxanone (PDS) for deep layer closure reduces the risk of dehiscence while maintaining tissue support. In addition, the removal of any subtle devitalized or fibrotic tissue is crucial to prevent recurrence and ensure complete infection control (Silva et al., 2024).
For Type 1 fistulas, which involve pre-existing surgical scars and moderate soft tissue compromise, the surgical approach includes a fusiform incision encompassing the entirety of the prior scar, allowing for complete excision of potentially colonized and fibrotic tissue. This method facilitates tension-free closure and prevents postoperative contracture formation. Monofilament sutures (PDS) are preferred for deeper layers due to their low inflammatory profile, while multifilament sutures, such as Vicryl, should be avoided in contaminated fields owing to their propensity for bacterial adherence and colonization (Almeida et al., 2022).
In Type 2 fistulas, the extent of tissue loss precludes primary closure and necessitates advanced reconstructive techniques. The use of fasciocutaneous or musculocutaneous flaps, such as the medial gastrocnemius flap or the anterolateral thigh (ALT) flap, offers robust vascularized coverage suitable for weight-bearing areas. In edematous or chronically inflamed regions, Prolene sutures are favored due to their low tissue reactivity and mechanical resistance. These cases frequently require microsurgical expertise to ensure flap viability, integration, and the restoration of both limb function and contour (Mendes, 2020).
Type 3 fistulas present the most complex surgical scenarios, characterized by extensive tissue destruction, exposed bone, and severely compromised vascular supply. A multidisciplinary team, typically including orthopedic, plastic, and microsurgeons, is essential for adequate management. For final coverage, microsurgical free flaps, such as the ALT flap or reverse sural artery flap, are employed to ensure durable soft tissue coverage, restore limb function, and preserve biomechanical integrity [21,30].
Ultimately, the success of surgical management depends on a patient-specific, multidisciplinary strategy that integrates precise debridement, appropriate flap selection, and tension-free closure techniques aligned with the mechanical and vascular characteristics of the defect. By tailoring each intervention to the biological status of the tissues involved, surgeons can improve healing trajectories, reduce complication rates, and restore both form and function in patients with chronic osteomyelitis of the lower limb.
Detailed Surgical Considerations
The surgical management of osteomyelitis-associated cutaneous fistulas in the lower limb requires a comprehensive and evidence- based approach, capable of addressing the intricate balance between infection control, soft tissue preservation, and functional reconstruction. A key principle in this context is the assessment of “skin stock”, a concept analogous to “bone stock” in joint prosthesis revisions. This evaluation emphasizes the identification and preservation of viable skin, while planning for the radical excision of all infected elements, including prior scars, fistulous tracts, and areas of devitalized or poorly vascularized skin with limited healing potential. Preoperative planning must anticipate the potential need for advanced closure techniques-such as musculocutaneous flaps or skin grafts-ensuring the surgical team is technically and logistically prepared. Inadequate planning often results in conservative debridement due to concerns over closure capacity, which may leave residual infection and compromise the therapeutic outcome. For this reason, treatment in specialized centers with multidisciplinary surgical teams substantially improves clinical results and reduces the burden on general orthopedic practitioners [21].
Debridement remains the cornerstone of effective fistula management in chronic osteomyelitis. It must be radical and comprehensive, targeting not only macroscopic necrosis but also biofilm-laden tissues. The use of wedge-shaped debridement allows direct access to infected bone, facilitates the removal of fibrotic adhesions, and helps restore mobility to previously fixed soft tissues. In parallel, dead space management must be executed with precision to avoid seroma formation and bacterial recolonization. This can be achieved using Baroudi sutures or closed-suction drainage systems, both of which contribute to preparing the wound bed for successful reconstruction [31].
During soft tissue reconstruction, special attention must be directed toward avoiding skin bridges, particularly those less than 4 cm in width, as these are highly susceptible to ischemia and necrosis due to insufficient vascular support. Even wider bridges may carry elevated risk, especially in areas affected by chronic infection. Therefore, tension-free closure remains a critical goal. This can be accomplished using a spectrum of reconstructive strategies, from local advancement flaps to musculocutaneous and microsurgical free flaps, depending on the extent of tissue loss and the vascular status of the surrounding area.
Figure 5 demonstrates significant linear tension across the skin bridge, which may exacerbate complications, especially in patients with chronic infection or impaired local vascularity. Optimal surgical management should prioritize tension-free closure, which can be achieved through various reconstructive techniques, including local advancement flaps, musculocutaneous flaps, or microsurgical free flaps, depending on the extent of tissue loss and the vascular integrity of the surrounding area.
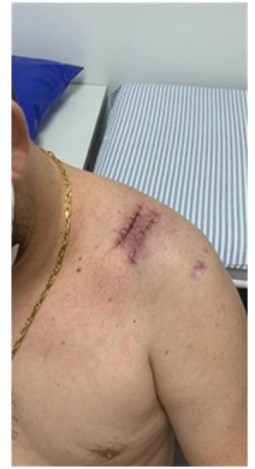
Figure 5: Postoperative image showing a narrow skin bridge following wound closure in the shoulder region.
Effective closure techniques must also include the excision of previous scars through fusiform incisions, release of fibrotic bands to enhance tissue mobility, and layered suturing to ensure a secure, watertight, and tension-free approximation of tissues. Long-lasting absorbable sutures, such as PDS, are recommended to provide durable support in deeper layers. When tension persists despite primary closure attempts, the use of advanced local flaps becomes necessary. In such scenarios, collaboration with plastic or microsurgical teams should be prioritized to ensure optimal outcomes, both in terms of wound stability and aesthetic restoration.
In cases of extensive soft tissue loss, particularly in weight-bearing regions of the lower limb, fasciocutaneous or musculocutaneous flaps become indispensable. The involvement of microsurgeons allows for precise planning and execution of complex reconstructions, which provide robust, vascularized coverage while preserving functional biomechanics. Furthermore, the integration of vacuum-assisted closure (VAC) therapy enhances wound bed preparation by reducing bacterial load, controlling exudate, and stimulating granulation tissue development. These advanced techniques, when combined with meticulous surgical planning and interdisciplinary collaboration, are essential to achieving durable, functional, and aesthetically favorable outcomes in the treatment of complex lower limb osteomyelitis [24,32].
Surgical Principles for Addressing the Debridement Complex
Debridement is a fundamental component in the surgical treatment of osteomyelitis-associated fistulas in the lower limb. Its primary objective is the radical removal of necrotic, infected, or biologically compromised tissues to halt the progression of infection and establish a favorable environment for tissue repair and regeneration. However, unlike routine soft tissue debridement, the surgical management of chronic osteomyelitis requires an expanded concept-referred to as the “debridement complex.” This concept includes all anatomical and pathological elements that sustain or propagate infection, necessitating their complete and strategic excision (Bancroft et al., 2007; Deng et al., 2014).
The debridement complex comprises a wide range of infected and structurally compromised tissues, including necrotic bone, scar tissue, cutaneous and sinus tract fistulas, biofilm-laden soft tissues, ischemic fascia and muscle, and infected orthopedic implants. Necrotic bone, in particular, must be aggressively resected, as it frequently harbors bacterial biofilms and serves as a persistent nidus for infection [14]. Similarly, scar tissue, characterized by diminished vascularity and poor healing potential, must be excised in its entirety to reduce the risk of recurrence. Cutaneous fistulas and sinus tracts require deep and circumferential removal to eliminate all channels of microbial dissemination. Infected or ischemic fascia and muscle must also be removed, while preserving viable, vascularized muscle whenever possible, as it plays a critical role in immunologic defense, tissue regeneration, and soft tissue coverage. Finally, biofilm-contaminated implants must be explanted to ensure comprehensive infection control [16,27].
A core element of surgical planning involves the preoperative assessment of “skin stock”-a term analogous to “bone stock” in revision arthroplasty-which refers to the availability, quality, and mobility of viable skin required for wound closure. Accurate evaluation of skin stock is essential to anticipate whether tension- free closure can be achieved following debridement. In cases of limited skin availability or extensive tissue loss, the use of fasciocutaneous or musculocutaneous flaps, as well as skin grafts, must be preemptively considered. Insufficient planning often leads to hesitant or incomplete debridement due to concerns about closure, resulting in residual infected foci that compromise the overall outcome. Collaboration with plastic and reconstructive surgeons at this stage is instrumental in ensuring optimal functional and reconstructive planning [19].
Intraoperatively, particular attention must be given to avoiding skin bridges, especially those narrower than 4 cm, which are prone to ischemia and necrosis due to insufficient perfusion. Even wider bridges (>6 cm) may fail in the context of chronic infection or subclinical vascular compromise. Consequently, wide fusiform incisions, planned according to the principles of relaxed skin tension lines and vascular anatomy, are recommended to facilitate adequate exposure and safe closure. The creation of skin bridges should be avoided whenever possible, and advanced flap techniques should be employed to achieve tension-free coverage [20].
Principles of wound closure in the context of osteomyelitis-related surgery should follow a standardized protocol aimed at minimizing complications. This includes the excision of previous scars using fusiform patterns, release of subcutaneous fibrosis to restore tissue mobility, dead space management using Baroudi sutures or closed suction drains, and layered closure using long-lasting absorbable sutures such as PDS to ensure a watertight and tension-free repair. When tension persists, local or regional flaps must be considered, and in more advanced cases, microsurgical free tissue transfer may be necessary to ensure durable closure [10,25].
In situations involving substantial tissue loss, the use of fasciocutaneous or musculocutaneous flaps is essential to provide vascularized, stable coverage capable of supporting tissue regeneration and mechanical load. In parallel, vacuum-assisted closure (VAC) therapy serves as a powerful adjunct, enhancing the wound bed by reducing microbial burden, managing exudate, and stimulating the development of granulation tissue. These modalities-when combined with precise surgical technique and interdisciplinary coordination-optimize healing trajectories and reduce the risk of reinfection, wound dehiscence, or flap failure [21,33].
In conclusion, the management of osteomyelitis-associated fistulas in the lower limb demands a comprehensive and anatomically guided surgical strategy. By thoroughly addressing the debridement complex, incorporating the principles of skin stock assessment, avoiding vascularly compromised skin bridges, and applying advanced reconstructive techniques, surgeons can achieve superior functional, structural, and aesthetic outcomes while minimizing recurrence rates and the need for future interventions [14,22].
Key Recommendations for Clinical Practice
The successful management of osteomyelitis-associated fistulas depends on a set of well-defined clinical recommendations that address both the pathological complexity of the condition and the imperative of individualized, patient-centered care. This section synthesizes core principles of best clinical and surgical practice, emphasizing the importance of comprehensive preoperative planning, the strategic resolution of intraoperative challenges, and the centralization of care within high-complexity, multidisciplinary centers. These guidelines aim to support clinicians in the delivery of high-standard, coordinated care by integrating accurate diagnostic assessment, evidence-based surgical strategies, and interprofessional collaboration, all of which are essential for optimizing outcomes and ensuring long-term therapeutic success.
a) Thorough Preoperative Planning
The foundation of effective treatment lies in meticulous preoperative planning, which must prioritize the complete excision of infected and nonviable tissues-including old surgical scars, cutaneous fistulas, and atrophic or devitalized skin with limited regenerative potential. In parallel, the surgical plan should anticipate the need for advanced reconstructive alternatives, such as skin grafts, fasciocutaneous flaps, or musculocutaneous flaps, depending on defect size and location. Optimal planning involves assembling a multidisciplinary team with experience in complex reconstructions and ensuring access to the specialized equipment required for precise execution. This comprehensive preparation is crucial for minimizing intraoperative uncertainties and enhancing surgical efficacy (Silva et al., 2024).
b) Addressing Common Challenges in Clinical Practice
A frequent barrier to effective treatment is the inadequate debridement of infected tissue, often driven by concern over the feasibility of wound closure. However, incomplete debridement perpetuates infection and leads to suboptimal clinical evolution. To overcome this, surgeons must adopt a strategy that prioritizes complete removal of infectious foci in combination with evidence- based closure techniques. By addressing these challenges preemptively and incorporating advanced reconstructive options into the operative plan, it is possible to reduce recurrence rates, promote healing, and significantly improve patient outcomes.
c) Centralized Care in Specialized Centers
The treatment of osteomyelitis-associated fistulas should ideally occur in specialized centers with dedicated infrastructure and multidisciplinary expertise. These institutions are uniquely equipped to handle the complexities of radical debridement and intricate soft tissue reconstruction. Furthermore, they provide an environment that fosters surgical precision and reduces the procedural burden often faced by general orthopedic teams. Centralized care also ensures access to innovative surgical technologies and standardized protocols, resulting in greater consistency and improved outcomes [34].
d) The Role of Specialized Centers in Complex Cases
In cases involving extensive tissue destruction, multisystemic involvement, or high reconstructive complexity, the role of specialized centers becomes even more critical. Beyond technical resources, these centers provide an ecosystem of collective clinical experience and interdisciplinary integration, which facilitates the development of comprehensive, tailored treatment plans. This approach allows for the effective incorporation of plastic, vascular, and microsurgical expertise, particularly in situations requiring flap coverage, staged procedures, or advanced infection control. The strategic advantages of these institutions directly translate into enhanced clinical results, lower complication rates, and improved patient quality of life.
Conclusion
The Soares Classification for cutaneous fistulas associated with chronic osteomyelitis constitutes a notable advancement in the clinical and surgical management of these complex conditions. By offering a structured framework for the evaluation of fistulous tracts and their anatomical relationships with surrounding tissues, the classification system facilitates precise preoperative planning, enabling a more rational and individualized therapeutic approach. This is particularly relevant in surgical contexts, where the integration of concepts such as the debridement complex and the strategic assessment of skin stock is essential to optimize outcomes and reduce complications.
Accurate and comprehensive debridement, guided by the principles of the debridement complex, plays a central role in the eradication of infection. The radical removal of necrotic bone, fibrotic tissue, fistulas, and biofilm-laden structures ensures the viability of adjacent tissues and establishes a biologically favorable environment for healing and reconstruction. Simultaneously, the preservation and judicious management of skin stock is critical in preventing ischemic complications, minimizing the risk of wound dehiscence and necrosis, and maintaining options for tension-free closure or advanced flap coverage.
By incorporating the Soares Classification into routine clinical and surgical workflows, clinicians are better equipped to tailor interventions according to the anatomical and pathological complexity of each case. This approach not only improves infection control but also contributes to the prevention of soft tissue-related complications, such as delayed healing and reoperation. As a result, this framework enhances overall surgical success, promotes functional and aesthetic recovery, and ultimately improves long- term patient outcomes and quality of life [35-47].
References
- Lima, A. L. L., Oliveira, P. R., Carvalho, V. C., Cimerman, S., & Savio, E. (2014). Recommendations for the treatment of osteomyelitis. Brazilian Journal of Infectious Diseases, 18, 526-534.
- Radcliffe, G. (2015). (iii) Osteomyelitis–a historical and basic sciences review. Orthopaedics and Trauma, 29(4), 243-252.
- WU, C. et al. (2017). Local antibiotic delivery for bone infection: challenges and strategies. Clinical Orthopaedics and Related Research, 475(7), 1705-1716.
- Calhoun, J. H., Manring, M. M., & Shirtliff, M. (2009, May). Osteomyelitis of the long bones. In Seminars in plastic surgery (Vol. 23, No. 02, pp. 059-072). © Thieme Medical Publishers.
- FODOR, L. et al. (2006). Complex tibial wounds and osteomyelitis. Journal of Plastic, Reconstructive & Aesthetic Surgery, 62(3), 435-440.
- SCHMITT, S. (2017). Advances in osteomyelitis imaging techniques. Radiology Clinics of North America, 45(7), 879- 886.
- HOTCHEN, A. et al. Surgical reconstruction techniques for tibial defects. Bone & Joint Journal, v. 99-B, n. 2, p. 128-136, 2017.
- LAM, T. et al. (2005). Clinical application of vacuum-assisted closure therapy in osteomyelitis. Clinical Orthopaedics and Related Research, 468(6), 1595-1602.
- GARCIA, A. et al. (2018). Advances in local antibiotic delivery in orthopedics. International Journal of Clinical Practice, 72(11), 1423-1431.
- Bancroft, E. A. (2007). Antimicrobial resistance: it's not just for hospitals. Jama, 298(15), 1803-1804.
- BANCROFT, E. et al. (2007). Biofilm resistance in osteomyelitis. Clinical Orthopaedics and Related Research, 464 (9), 124-130.
- DENG, C. et al. (2014). Advanced therapies in chronic osteomyelitis. World Journal of Orthopedics, 10(3), 189-199.
- Fang, C., Wong, T. M., Lau, T. W., To, K. K., Wong, S. S.,& Leung, F. (2017). Infection after fracture osteosynthesis– part I: pathogenesis, diagnosis and classification. Journal of Orthopaedic Surgery, 25(1), 2309499017692712.
- CALHOUN, J. H. et al. (2009). Clinical principles in the management of osteomyelitis. Journal of Bone and Joint Surgery – American Volume, 91 (3), 169-174.
- DENG, C. et al. (2014). Chronic osteomyelitis management:a surgeon's perspective. Orthopaedic Surgery, 6(3), 181-189.
- HEYMANS, O. et al. Management of chronic osteomyelitis with modern surgical techniques. Plastic and Reconstructive Surgery, v. 116, n. 7, p. 1996-2002, 2005.
- Panteli, M., & Giannoudis, P. V. (2016). Chronic osteomyelitis: what the surgeon needs to know. EFORT open Reviews, 1(5), 128-135.
- FODOR, L. et al. (2006). Innovations in flap surgery for osteomyelitis. Journal of Plastic Surgery and Hand Surgery, 44(8), 367-375.
- LAM, T. K. et al. (2005). Chronic osteomyelitis imaging updates. European Journal of Radiology, 78 (4), 323-331.
- Hotchen, A. J., McNally, M. A., & Sendi, P. (2017). The classification of long bone osteomyelitis: a systemic review of the literature. Journal of bone and joint infection, 2(4), 167- 174.
- LAM, T. K. et al. (2025). Innovative techniques in chronic osteomyelitis management. European Journal of Plastic Surgery, 31(4), 201-210.
- GARCIA, J. et al. (2018) Antimicrobial management in chronic osteomyelitis. International Journal of Antimicrobial Agents, 52(2), 207-215.
- FODOR, L. et al. (2008). Perforator flap coverage in complex limb wounds. Journal of Plastic Surgery and Hand Surgery, 42(6), 267-272.
- LAM, T. P. et al. (2005). Multidisciplinary management of osteomyelitis: clinical updates. Annals of Plastic Surgery, 55(6), 673-679.
- GARCIA, J. et al. (2018). Local and systemic antibiotics in osteomyelitis. International Journal of Antimicrobial Agents, 50(3), 301-309.
- Hotchen, A. J., McNally, M. A., & Sendi, P. (2017). The classification of long bone osteomyelitis: a systemic review of the literature. Journal of bone and joint infection, 2(4), 167- 174.
- Markakis, K., Faris, A. R., Sharaf, H., Faris, B., Rees, S., & Bowling, F. L. (2018). Local antibiotic delivery systems: current and future applications for diabetic foot infections. The International Journal of Lower Extremity Wounds, 17(1), 14-21.
- FODOR, L. et al. (2006). Perforator-based flaps for coverage of complex tibial wounds: a retrospective analysis of 25 cases. Journal of Plastic, Reconstructive & Aesthetic Surgery, 59(8), 882-887.
- GARCIA, J. et al. (2018). The role of vacuum-assisted closure in bone infections. European Journal of Orthopaedic Surgery & Traumatology, 27(5), 725-733.
- SHI, J. et al. (2023). Antibiotic delivery systems in bone infections. European Journal of Orthopaedic Surgery & Traumatology, 33(3), 155-165.
- SHI, J. et al. (2023). Vacuum-assisted closure therapy for osteomyelitis-related soft tissue defects. European Journal of Orthopaedic Surgery & Traumatology, 33(1), 23-33.
- SHI, X. et al. (2023). Recent trends in osteomyelitis treatment.International Orthopaedics, 47(1), 101-110.
- SHI, X. et al. (2023). Vacuum-assisted therapy in chronic bone infections. Journal of Orthopaedic Surgery and Research, 18(1), 1-10.
- Marais, L. C., Ferreira, N., Aldous, C., & Le Roux, T. L.B. (2014). The classification of chronic osteomyelitis. SA orthopaedic Journal, 13(1), 22-28.
- LIMA, R. F. et al. (2014). Chronic osteomyelitis: principles of treatment and complications. Brazilian Journal of Orthopedics, 49(6), 601-608.
- MARAIS, L. C. et al. (2015). Multidisciplinary osteomyelitis care. International Orthopaedics, 43(8), 1985-1993.
- MARAIS, L. C. et al. (2018). Strategies in osteomyelitis management. International Orthopaedics, 42 (7), 1895-1902.
- MARAIS, L. C. et al. (2015). The use of local antibiotics in chronic osteomyelitis: an overview of current treatment strategies. Acta Orthopaedica Belgica, 81(2), 182-190.
- MARAIS, L. C. et al. (2015). Vacuum-assisted therapy in chronic infections. World Journal of Orthopedics, 11(4), 233- 243.
- PANTELI, M. et al. (2016). The role of systemic antibiotic therapy in osteomyelitis. Journal of Bone and Joint Surgery – American Volume, 98(4), 234-240.
- RADCLIFFE, S. J. (2015). Advances in imaging for the management of osteomyelitis. Radiology Clinics of North America, 53(1), 39-49.
- SCHMITT, S. K. (2017). Advances in imaging for osteomyelitis. Clinical Radiology, 72(2), 124-131.
- Schmitt, S. K. (2017). Osteomyelitis. Infectious Disease Clinics, 31(2), 325-338.
- VERHELLE, F. et al. (2003). Flap-based coverage for osteomyelitis. Plastic and Reconstructive Surgery, 115(5), 1424-1431.
- VERHELLE, F. et al. (2003). Modern surgical approaches to bone infections. Plastic and Reconstructive Surgery, 126(6), 1928-1935.
- VERHELLE, F. et al. (2003) The use of perforator flaps in complex wound reconstruction. Plastic and Reconstructive Surgery, 112(6), 1623-1629.
- WU, H. et al. (2017). Two-stage management of Cierny- Mader type IV chronic osteomyelitis of the long bones. Injury, 48(5), 511-518.


