Review Article - (2020) Volume 2, Issue 1
Struvite as a Non-Equilibrium Solid Phase in Electrolytic Systems (Aqueous Media)
2Department of Analytical Chemistry, Cracow University of Technology, Cracow, Poland
Received Date: Dec 09, 2019 / Accepted Date: Dec 20, 2019 / Published Date: Jan 24, 2020
Copyright: ©Tadeusz Michalowski. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Struvite (MgNH4 PO4 •6H2 O) is not the equilibrium solid phase when introduced into pure water or into an aqueous CO2 solution, with or without KOH added as the pH-modifying component. In some instances, the dissolution process consists of several steps where different solid phases are formed. It results from a simulating procedure, where the entire physicochemical knowledge on the system in question is involved in the iterative computer program. The results of calculations are put in context with formation of struvite renal calculi in urinary tracts.
Keywords
Struvite, Equilibrium Analysis, Computer Simulation, Renal Calculi
Notations
pri – precipitate of i–th kind (i = 1,...,5) with molar concentration [pri];
pr1 = MgNH4 PO4 , pr2 = Mg3 (PO4 )2 , pr3 = MgHPO4 , pr4 = Mg(OH)2 , pr5 = MgCO3 ;
Kspi – solubility product for pri (i= 1,..., 5);
pC0 = – logC0 ; pCCO2 = – logCCO2, pCb = – logCb ;
ppri = – log[pri].
Precipitates pri are written in bold letters
Introduction
Struvite (magnesium ammonium phosphate hexahydrate, MgNH4PO4•6H2O, written as MgNH4PO4 (for brevity) and denoted later as pr1, is of great importance from medical, tech- nical, environmental and agricultural viewpoints. Particularly, it is the main component of kidney stones (renal calculi) developed in human and animal urinary tracts infected by bacteria producing urease (urea amidohydrolase). Although clinical occurrence and presentation of kidney stones has already been described in the Aphorisms of Hippocrates, its behavior in aqueous media was not known until recently sufficiently well, as is seen on example of struvite [1].
This paper follows the article issued 2006 and provides the novel approach to simulated dissolution of pure struvite in aqueous media containing CO2 (CCO2 mol/L) and KOH (Cb mol/L) [2]. It is proved that pr1 is not a stable solid species in such media. The approach is based on charge and concentration balances, together with expressions for equilibrium constants, involving all physicochemical knowledge on the system in question [2]. The calculations were made with use of DELPHI iterative computer program.
Some Remarks
Struvite (pr1) is not the equilibrium solid phase (C0, mol/L) when introduced into pure water or aqueous solution of CO2, modified (or not) by free strong acid HB or base MOH. Although the fact that NH3 evolves from the system obtained after leaving pure pr1 in pure water [2].
3pr1 = pr2 + HPO4 -2 + 2NH3 +1 + NH3 (1)
has already been known at the end of 19th century (see [2-6])the solubility (s, mol/L) of pr1was commonly calculated from the approximate formula s = (K sp1 )1/3 (see e.g. [7]), based on an
In the past, it was noticed that mixing MgCl2 and NH4H2PO4 solutions, at the molar ratio 1:1 of the salts, gives a two-phase system containing an excess of ammonium species remaining in the solution, and the precipitate that “was not struvite, but was probably composed of magnesium phosphates” [7]. Such inferences were confirmed later on the basis of X-ray diffraction analysis of the crystallographic structure of the solid phase thus obtained [7, 8].
It was also stated [8,9] that the precipitation of pr1 requires a significant excess of ammonium species, e.g. Mg:N:P = 1:1.6:1. The co-precipitation of other phosphate species can occur on the stage of pr1 washing [3].
Formulation of Struvite + Aqueous Solution System
We refer first to the system where pure struvite (pr1) is introduced into aqueous solution with dissolved CO2 (CCO2 mol/L) + KOH (Cb mol/L) + HCl (Ca mol/L); initial (t=0) concentration of pr1 in the system equals C0 mol/L. The species involved in such systems are interrelated in the corresponding concentration (Eqs. 2, 4, 6) and charge (Eq. 5) balances:
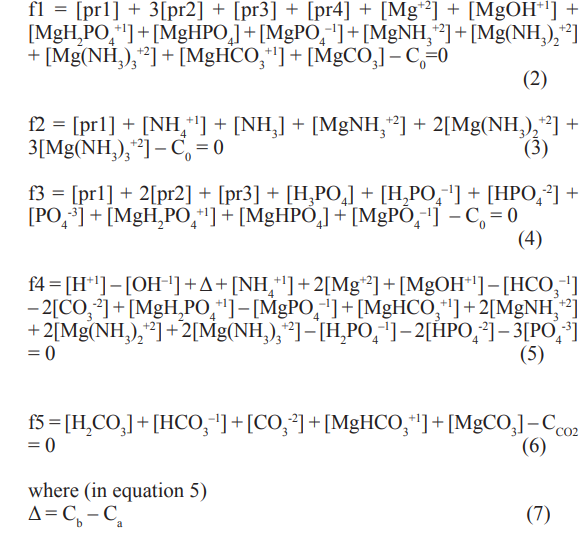
and [pri] is the concentration of pri (see the list of notations). On defined stage of pr1 dissolution, concentrations of some (or all) solid phases are assumed zero. To check it, the qi values for different potentially perceptible species pri (i=1,..., 5) were ‘peered’ in computer program applied for this purpose.
The concentration of MgCO3, i.e. [pr5], has not been included in the concentration balances specified above. Simply, at any case considered below, pr5 does not exist as the equilibrium solid phase,as results from preliminary calculations.
The manner of calculations was similar to one presented in [2]. The equilibrium data are collected in Table 1. At the start for calculations, four or five fundamental/independent variables were chosen, namely

This way, we were confirmed that the solid species pri is (or is not) formed in the system, i.e. logqi = 0 or logqi < 0.
Table 1: The logK values for equilibrium constants (K) related to the system tested; K = Kspi (i = 1,...,5) – solubility product of the i–th precipitate (pri); pKw = 14.0 for Kw= [H+1][OH-1].
|
Reaction |
logK |
Reaction |
logK |
|
|
Mg+2 + OH–1 = MgOH+1 |
2.57 |
Mg+2 + HCO –1 = 3 MgHCO +1 3 |
1.16 |
|
|
NH + H+1 = 3 NH +1 4 |
9.24 |
Mg+2 + CO –2 = 3 MgCO3 |
3.4 |
|
|
H+1 + PO –3 = 4 HPO –2 4 |
12.35 |
Mg+2 + NH = 3 MgNH +2 3 |
0.24 |
|
|
2H+1 + PO –3 = 4 H PO –1 2 4 |
19.55 |
Mg+2 + 2NH = 3 Mg(NH )2+2 3 |
0.2 |
|
|
3H+1 + PO –3 = 4 H3PO4 |
21.70 |
Mg+2 + 3NH = 3 Mg(NH ) +2 3 3 |
-0.3 |
|
|
H+1 + CO –2 = 3 HCO –1 3 |
10.33 |
MgNH PO = Mg+2 4 4 + NH +1 + PO –3 4 4 |
–12.6 |
K=Ksp1 |
|
2H+1 + CO –2 = 3 H2CO3 |
16.71 |
Mg (PO ) = 3Mg+2 3 4 2 + 2PO –3 4 |
–24.38 |
K=Ksp2 |
|
Mg+2 + H PO –1 2 4 = MgH PO +1 2 4 |
0.45 |
MgHPO = Mg+2 + 4 HPO –2 4 |
–5.5 |
K=Ksp3 |
|
Mg+2 + HPO –2 4 = MgHPO4 |
2.91 |
Mg(OH) = Mg+2 + 2 2 OH–1 |
–10.74 |
K=Ksp4 |
|
Mg+2 + PO –3 = 4 MgPO –1 4 |
4.8 |
MgCO = Mg+2 + 3 CO –2 3 |
–5.17 |
K=Ksp5 |

The Struvite Dissolution – Graphical Presentation
The results of calculations, referred to Ca = 0, (pC0, pC CO2 , pCb) =(3, 4,T) and (pC0, pCCO2, pCb) = (2, 4, 2), are presented in Figures 1 and 2, respectively



are presented in Figure (2c).
All the reaction notations specified above involve predominating species of the related systems. All them were formulated on the basis of the related speciation plots (Figure 2b) and confronted with the related plot in Figure (2c). Particularly, OH–1 ions participate the reactions (15) and (17) as substrates and then pH of the solution decreases during the dissolution process on the stages 1 and 3 (see Figure 2c). On the stage 2 we have pH â?? constant (see Eq. 16 and Figure 2c). A growth in concentration of NH and HPO -2 is also reflected in the reactions (15)-(17) notation.
Struvite Versus Kidney Stones
The urinary system is one of the most important regulatory systems for body’s waste removal (excretion). It consists of two kidneys that clean the body’s fluids, as well as the bladder and urethra, which store and eliminate waste.
An infection of urinary tract may concern bladder and kidney [10]. Kidney infection, if it occurs, usually follows a bladder infection. Symptoms from a bladder infection include pain while urination, and frequent urination. Rarely, the urine may appear to be bloody (hematuria). This infection is more common in women than men, and occurs more often at the age of 16 to 35 years.
The most common cause of infection is uropathogenic bacteria Escherichia coli (E. Coli) from the gut. After entering the bladder via the urethra, E. Coli are able to attach to the bladder wall and form a biofilm that resists the body’s immune response. When the kidneys, bladder, or ureters become inflamed by this infection, formation of bladder stones may occur. The stones can move through ureters and get out from urethra with urine. Some kidney stones can also get stuck in urinary tract and block urine from getting through. The flow of large kidney stones can be very painful in this case. Kidney stone disease is known as urolithiasis [11]. At present, urolithiasis is one of the leading urological diseases and, irrespective of the method of removal of kidney stones; there are serious complications and relapses.
Formation of kidney stones deposits is relatively common ailment in human’s organisms. A few percent of people in the world experience this ailment in their lifetime [12]. The list of known people suffering from this ailment is long [13, 14].
The kidney stones are formed mainly from crystallized, moderately soluble calcium and/or magnesium salts [15, 16]. Formation of the kidney stones results from combination of genetic and environmental factors [17]. Nearly 80 – 90% of urolithiasis cases involve concretions composed of calcium oxalates: whewellite and weddellite, and 10–15% cases involve struvite stones [18].
Struvite stones are almost always caused by urinary tract infections. Struvite stones are formed in presence of urease-producing bacteria (most commonly Proteus and Klebsiella), resulting from the infection [19-24]. Ureases are metalloenzymes catalyzing the hydrolysis of urea into carbon dioxide and ammonia [25-29] (NH2)2CO + H2O = CO2 + 2NH3 (18)
This reaction occurs effectively at pH ca. 7, where [NH4 +1]/[NH3 ] >> .
Normal urine is under-saturated with ammonium and phosphate species and, therefore, the solubility product of struvite is not crossed.The stone formation (crystallization) occurs only from supersaturated solution. Reaction (18) does not occur in presence of acetohydroxamic acid (CH3CO-NH-OH), used in drugs as the irreversible urease inhibitor, in combination with antibiotics. Some inhibiting substances (e.g. citrate) are able to prevent the stone formation in urine. An essential role in the inhibiting plays pH of the solution.
To diagnose this ailment accurately in hospital (radiology department), and provide the most effective treatment, the following diagnostic techniques are used: computerized tomography (CT) with X-ray scans for adults, or ultrasound bladder scan (sonogram) to monitor kidney stones in children/young people or an unborn baby in pregnant women.
Another option is cytoscopy, where a cytoscope (a specialized tube with camera at the end) is inserted into the body through the urethra in order to view the inside of the bladder and urethra.
On the treatment step, non-invasive techniques: lithotripsy or extracorporeal shockwave lithotripsy (ESWL) are most frequently applied [30-36]. Shock waves break up the stones into smaller sand-like fragments which can then pass on their own.
Final Comments
The paper concerns the behavior of struvite MgNH4PO4•6H2O introduced into either pure water or an aqueous solution of CO2, with or without KOH applied as the pH-modifying component. A simulating procedure, involving the entire physicochemical knowledge on the system in question, enables us to state that struvite is not the equilibrium solid phase in such systems. Struvite is one of the main components of infection stones, i.e. renal calculi developed in hu-man and animal urinary tracts infected by bacteria producing urease.
References
- Athanasios Diamandopoulos, Pavlos Goudas, Dimitrios Oreopoulos (2009) Thirty-six Hippocratic Aphorisms of Nephrologic Interest. American Journal of Kidney Diseases 54: 143-153.
- MichaÅ?owski T, Pietrzyk A (2006) A thermodynamic study of struvite+water system, Talanta 68: 594-601.
- Beilstein F, Grosset Th (1890) Ueber die Bestimmung der freien Schwefelsaure in der Schwefelsauren Thonerde. Zeitschrift für Analytische Chemie 29: 73-78.
- MichaÅ?owska Kaczmarczyk A M, MichaÅ?owski T (2015) Solubility product challenge. Analytical and Bioanalytical Chemistry 407: 1789-1791.
- MichaÅ?owska Kaczmarczyk A M, MichaÅ?owski T (2015) Solution to the solubility product challenge. Analytical and Bioanalytical Chemistry 407: 4877-4878.
- MichaÅ?owska-Kaczmarczyk A M, Spórna-Kucab A, MichaÅ?owski T (2017) Solubility products and solubility concepts. Descriptive Inorganic Chemistry Researches of Metal Compounds. Akitsu T (Ed.) InTech. Chap 5: 93-134.
- Doyle J D, Parsons S A (2002) Struvite formation, control andrecovery. Water Research 36: 3925-40.
- Stratful I, Scrimshaw M D, Lester J N (2001) Conditions influencing the precipitation of magnesium ammonium phosphate. Water Research 35: 4191-4199.
- MichaÅ?owski T (1982) Solubility diagrams and their use ingravimetric analysis. Chem Anal 27: 39-49.
- Urinary tract infection https://en.wikipedia.org/wiki/Urinary_ tract_infection.
- Sokol E, Nigmatulina E, Maximova N, Chiglintsev A (2005) CaC2O4·H2O spherulites in human kidney stones: morphology, chemical composition, and growth regime. European Journalof Mineralogy 17: 285-295.
- Schwaderer A L, Wolfe A J (2017) The association between bacteria and urinary stones. Annals of Translational Medicine 5: 32.
- List of kidney stone formers.
- Ostad E, Wise G J (2005) Celestial bodies and urinary stones: Isaac Newton (1641-1727) - health and urological problems. BJU INTERNATIONAL 95: 24-26.
- Amimanan P, Tavichakorntrakool R, Fongngern K, Sribenjalux P, Lulitanond A, et al. (2017) Elongation factor Tu on Escherichia coli isolated from urine of kidney stone patients promotes calcium oxalate crystal growth and aggregation. Scientific Reports 7: 2953.
- Bladder stone. https://en.wikipedia.org/wiki/Bladder_stone.
- https://en.wikipedia.org/wiki/Kidney_stone_disease.
- Han H, Segal A M, Seifter J L, Dwyer J T (2015) Nutritional Management of Kidney Stones (Nephrolithiasis). Clinical Nutrition Research 4: 137-152.
- Melekos M D, Naber K G (2000) Complicated urinary tractinfections. Int J Antimicrobial Agents 15: 247-256.
- Woolfrey B F, Quall C O, Fox J M (1981) Evaluation of the repliscan system for Enterobacteriaceae identification. Journal of Clinical Microbiology 13: 58-61.
- Collins C M, D Orazio S E F (1993) Bacterial ureases: structure, regulation of expression and role in pathogenesis. Molecular Microbiology 9: 907-913.
- Mobley H LT, Island M D, Hausinger R P (1995) Molecular biology of microbial ureases. Microbiol Rev 59: 451-480.
- Burne R A, Chen Y Y M (2000) Bacterial ureases in infectious diseases. Microbes and Infection 2: 533-542.
- Flannigan R., Ho Choy W, Chew B, Lange D (2014) Renal struvite stones-pathogenesis, microbiology, and management strategies. Nature Reviews Urology 11: 333-342.
- Mc Lean R J C, Nickel J C, Cheng K J, Costerton J W (1988) The ecology and patho-genicity of urease-producing bacteria in the urinary tract. CRC Crit Rev Microbiol 16: 37-79.
- Mobley H L T, Island M D, Hausinger R P (1995) Molecular biology of microbial ureases. Microbiol Rev 59: 451-480.
- Hassan S S M, El–Bahnasawy R M, Rizk N M, Nashwa M (1997) Potentiometric determination of salicylhydroxamic acid (urinary struvite stone inhibitor) based on the inhibition of urease activity. Analytica Chimica Acta 351: 91-96.
- Hesse A, Heimbach D (1997) Causes of phosphate stone formation and the importance of metaphylaxis by urinary acidification: a review. World J Urol 17: 308-315.
- Babic Ivancic V, Kontrec J, Brecevic L (2004) Formation and transformation of struvite and newberyite in aqueous solutions under conditions similar to physiological. Urol Res 32: 350-356.
- Shockwave lithotripsy. https://www.youtube.com/ watch?v=3KnPA4k9jo4.
- Chaussy C, Brendel W, Schmiedt E (1980) Extracorporeally induced destruction of kidney stones by shock waves. Lancet 2: 1265-8.
- Chaussy C, Schmiedt E, Jocham D, Brendel W, Forssman B (1982) First clinical experience with extracorporeally induced destruction of kidney stones by shock wave. Journal of Urology 127: 417-20.
- Chaussy C, Schmiedt E, Jocham D, Eisenberger F, Walther V, et al. (1982) Extracor-poreal shock wave lithotripsy-new aspect in the treatment of kidney stone disease. Basel Karger.
- Chaussy C, Schmiedt E, Jocham D, Schüller J, Brandl H, etal. (1984) Extracorporeal shock-wave lithotripsy (ESWL) fortreatment of urolithiasis. Urology 23: 59-66.
- Extracorporeal Shock Wave Lithotripsy (ESWL).
- Tailly G G (2013) Extracorporeal shock wave lithotripsy today. Indian Journal of Urology 29: 200-207.



