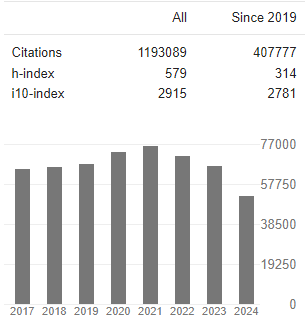
Research Article - (2025) Volume 4, Issue 3
Staining Tissues with Dyes Obtained from Safran, Curcumin and Henna Extract Instead of Hematoxylin and Eosin Used in Routine Tissue Staining
Received Date: Jul 30, 2025 / Accepted Date: Aug 29, 2025 / Published Date: Sep 05, 2025
Copyright: ©Â©2025 Alpaslan Gokçimen, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Tuncel, S., Gokçimen, A., Kalkan, N. S. (2025). Staining Tissues with Dyes Obtained from Safran, Curcumin and Henna Extract Instead of Hematoxylin and Eosin Used in Routine Tissue Staining. J Surg Care, 4(3), 01-13.
Abstract
In this study, it is aimed to investigate the potential use of saffron, curcumin, and henna extracts as alternatives to the routine histological staining method, hematoxylin and eosin (H&E), in tissue staining. In the study, liver, pancreas, and spleen tissues obtained from four-week-old male Wistar albino rats were subjected to standard tissue processing protocols, and staining was performed using natural extracts. The staining results obtained were compared with hematoxylin and eosin (H&E) staining. In liver tissues stained with saffron and curcumin, Kupffer cells were observed more prominently, while in tissue samples stained with stone henna, hepatocytes and nuclei were clearly distinguishable. The findings revealed that while natural extracts did not replicate the classic nuclear and cytoplasmic staining patterns of hematoxylin and eosin, they showed selective staining properties that may be useful in complementary or preliminary histological assessments.
Keywords
Natural Dyes, Curcumin, Saffron, Henna, Histological Staining, Hematoxylin and Eosin Alternatives
Introduction
Stone henna, commonly known as "black henna" or "German stone" among the public, is naturally found in solid (stone-like) form and is used after being crushed into powder. It consists of chemical compounds such as para-phenylenediamine (PPD). PPD produces a black color but may cause severe skin reactions such as allergic contact dermatitis. Its molecular formula is Câ??Hâ??Nâ??. Black henna is widely used, especially in hair dyeing and temporary tattoo applications; however, due to the presence of PPD, its use has been restricted in many countries. Curcumin (diferuloylmethane), extracted from Curcuma longa, is a polyphenol with both hydrophilic and lipophilic regions, enabling interaction with multiple cellular components [1,2].
Saffron (Crocus sativus) contains crocin and crocetin, carotenoid derivatives that are water-soluble and capable of forming ionic bonds with basic tissue components [3,4]. In order to examine animal and plant tissues under a light or electron microscope, they must be properly prepared and stained. For this purpose, various staining techniques are applied during the preparation of histological specimens [5,6]. One of the most commonly used methods in histology and pathology laboratories is hematoxylin and eosin (H&E) staining. Hematoxylin is a basic dye that stains basophilic structures; however, since it does not have staining properties on its own, it must be oxidized into its hematein form and supported by mordants to acquire staining capability. Eosin, on the other hand, is an acidic dye that stains acidophilic structures [7,8].
The conversion of hematoxylin into hematein, the active staining agent, occurs through two methods: natural and artificial oxidation. When hematoxylin undergoes natural oxidation to form hematein, it is exposed to air and sunlight for a period of 6 to 8 weeks [9,10]. In the process of artificial oxidation of hematoxylin to hematein, it must be treated with oxidizing agents such as sodium iodate, hydrogen peroxide, mercury oxide, and potassium permanganate [8,11]. Hematein, due to its anionic structure, is not able to stain tissues effectively, and efficient staining cannot be achieved unless an oxidizing mordant is used. This solution is known as hematoxylin [12]. The most commonly used and strong mordants for hematoxylin are metal cations such as chromium, iron, aluminum, molybdenum, lead, and tungsten [13,14].
When aluminum and potassium salts are used as mordants with hematein, a blue-purple reaction occurs, while iron salts, when used as mordants, result in a black-blue reaction [15]. Hematoxylin stains cell nuclei in blue and purple colors. Among the basophilic structures stained by hematoxylin are organelles with high RNA content, such as ribosomes and the granular endoplasmic reticulum. Eosin interacts with cytoplasmic proteins, staining connective tissue fibers, muscle cells, and erythrocytes as acidophilic structures in red and pink colors [16-19]. Hematoxylin and eosin, with their high contrast, enhance the morphological features of cells and are a widely preferred method in histopathological studies [20].
Hematoxylin is a natural dye obtained from the heartwood of the Haematoxylum campechianum tree, which takes its name from the city of Campeche in Mexico, and is widely used in histological staining Faisal and [14-23]. The cost of hematoxylin per gram ranges from 50 to 100 USD, and the increasing costs can create a budget burden in laboratories [24,25]. Eosin was discovered by Heinrich Caro in 1871 and was initially used as a textile dye, but today it is used in conjunction with hematoxylin in the hematoxylin and eosin staining protocol [26,17]. Although the cost of eosin is lower, considering the amount used and the quality, it can still be costly for laboratories [27]. In recent years, research has been conducted on alternatives such as environmentally friendly and cost-effective plant-based dyes that can be used instead of hematoxylin and eosin [28].
he aim of this study is to evaluate the feasibility of histological staining using plant extracts as an alternative to hematoxylin and eosin, which are routinely used for tissue staining, and thereby reduce laboratory costs. Henna (Lawsonia inermis) contains lawsone (2-hydroxy-1,4-naphthoquinone) as its active dye component, which interacts with proteins and possibly nucleic acids. Curcumin (diferuloylmethane), derived from turmeric, is a polyphenol with both hydrophilic and lipophilic properties, enabling varied cellular interactions. Saffron contains crocin and crocetin, water-soluble carotenoids responsible for its coloration and potential staining properties. For a comprehensive understanding of the traditional, chemical, and technological aspects of natural dyes, readers are referred to Dominique Cardon’s seminal work Natural Dyes: Sources, Tradition, Technology and Science [29].
Materials and Methods
Supply Experimental Animals
In the study, four male Wistar albino rats obtained from the Aydin Adnan Menderes University Laboratory Animal Facility and approved by the Aydin Adnan Menderes University Animal Ethics Committee (approval number: 64583101/2024/50) were sacrificed under anesthesia by cervical dislocation, with two rats procured in the first week and two in the second week. Subsequently, standard tissue processing, embedding, and sectioning procedures were performed.
Preparation of Extracts
All dye solutions were stored in amber-colored bottles at ambient room temperature (24–26 °C) in a light-protected cabinet to minimize photodegradation and oxidation, in accordance with recommendations for natural dye preservation [29]. Copper (II) sulfate (CuSOâ??) was added as a mordant to curcumin and saffron extracts to enhance dye affinity for tissue components by forming stable dye-metal complexes [30,31]. No mordant was used with henna.
Preparation of Henna Extract
Stone henna is a natural stone found in solid form in nature, known as "German stone" in many countries, and can be ground into powder for use. One gram of powdered henna was mixed with 20 ml of 96% ethanol and dissolved. The mixture was left at room temperature in a dark environment for 3–4 hours with intermittent stirring. At the end of the extraction, the liquid was filtered three times and stored in the dark for use in histological staining.
Preparation of Curcumin Extract
One gram of powdered curcumin was dissolved in 20 ml of 96% ethanol and left at room temperature in a dark environment for 5–6 hours with intermittent stirring. At the end of the extraction process, the mixture was filtered three times, and the resulting liquid was stored in the dark for use in histological staining [31,32].
Preparation of Saffron
Ground 0.5 g of saffron stigmas were dissolved in 20 ml of 96% ethanol and left at room temperature in a dark environment for 24 hours. After the extraction, the mixture was filtered twice, and the resulting liquid was stored in the dark for use in histological staining [3,4].
Combination of Extracts with Mordant
Combination of Saffron Extract with Mordant
The prepared saffron extract was mixed with 0.5 g of Copper (II) Sulfate (CuSOâ??). The mixed solution was filtered and stored in an appropriate container at room temperature in a dark environment for use in histological staining procedures.
Combination of Curcumin Extract with Mordant
The prepared curcumin extract was mixed with 0.5 g of Copper (II) Sulfate (CuSOâ??). The mixed solution was filtered and stored in an appropriate container at room temperature in a dark environment for use in histological staining procedures [31]. 2.6.3. Formation of Staining Groups The pH of each dye solution was not standardized in this study, and as previous research indicates, variations in pH can significantly alter dye-tissue binding and color development [14,33]. This limitation may have influenced staining outcomes and should be controlled in future experiments.
|
Tissues |
Staining Groups |
|||||||
|
Liver |
Hematoxylin |
Hematoxylin |
Henna |
Henna and |
Curcumin |
Curcumin, |
Saffron and |
Saffron, |
|
Pancreas |
and Eosin |
|
|
Hematoxylin |
and |
Hematoxylin |
Hematoxylin |
Hematoxylin |
|
Spleen |
|
|
|
|
Hematoxylin |
and Copper |
|
and Copper |
|
|
|
|
|
|
|
II Sulfate |
|
II Sulfate |
Histopathologic Studies
While initial observations were made at x10–x20 magnification, future studies should include higher magnification images (e.g., x40, x100) to allow for more accurate evaluation of nuclear detail and cytoplasmic staining, as recommended for histological assessment [16,17].
Examination of Liver, Pancreas, and Spleen Tissues Stained with Hematoxylin and Eosin
Following the hematoxylin and eosin staining procedure, examination of the liver tissue under a light microscope revealed that the histological structures appeared in their characteristic colors; the cell nuclei were observed in purple, and the cytoplasm appeared pink. As a result of the examination of the pancreas tissue, the histological structures were observed in their typical colors. The cell nuclei appeared blue-purple, the cytoplasm was pink, and the sinus acini and Langerhans islets were clearly distinguished. Examination of the spleen tissue showed that the histological structures were also observed in their characteristic colors. The cell nuclei were blue-purple, cytoplasm was pink, pulp was white, pulp was red, and trabecular structures were clearly detected (Figure 1).
Figure 1: Liver (A), Pancreas (B) and Spleen Tissue (C); (H&E; x20)
Examination of Liver, Pancreas, and Spleen Tissues Stained with Hematoxylin
In the liver tissue examined under a light microscope following the hematoxylin staining procedure, the cell nuclei appeared pale purple, while the cytoplasmic structures were not observed. As a result of the examination of the pancreas tissue, the cytoplasm of the acinar cells and Langerhans islets, as well as the connective tissue and blood vessel walls, could not be clearly observed. However, the cell nuclei appeared as pale blue. Upon examination of the spleen tissue, the red pulp, white pulp, and trabecular structures were clearly observed, although the central arteries were not distinctly visible (Figure 2).
Examination of Liver, Pancreas, and Spleen Tissues Stained with Curcumin and Hematoxylin
The dual staining of liver tissue, in comparison to routine hematoxylin and eosin staining, showed a transition of the pink color to yellow, light brown, and greenish tones, with this fading observed in both the nuclei and cytoplasm. In the dual staining, conducted differently from hematoxylin-eosin staining, Kupffer cells were observed clearly and intensely. Hepatocyte nuclei were observed in distinct dark blue, while their cytoplasm appeared in light brown and greenish tones. In the staining of liver tissue with curcumin and hematoxylin, the addition of curcumin to the hematoxylin stain resulted in a clearer observation of the Kupffer cells, which are part of the liver's mononuclear phagocytic system (Figure 3). When pancreas tissue was stained with curcumin and hematoxylin, the nuclei were stained dark brown and the cytoplasm light brown, and they were clearly distinguishable. As the cell walls and nuclei were more distinct, the Langerhans islets were observed more clearly (Figure 4). As a result of staining spleen tissue with the curcumin and hematoxylin method, the cell nuclei appeared blue-purple, while the red pulp, white pulp, and trabecular structures were distinctly visible (Figure 5).
Figure 3: Liver tissue, H&E; x20 (A), Hematoxylin; x20 (B), Curcumin and Hematoxylin; x20, x40 (C-D)
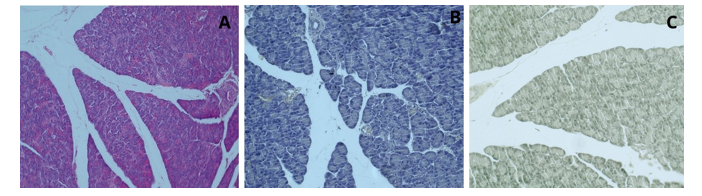
Figure 4: Pancreas Tissue, H&E; x20 (A), Hematoxylin; x20 (B), Curcumin and Hematoxylin; x20 (C)
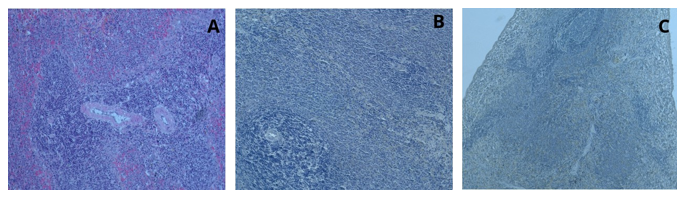
Figure 5: Spleen tissue, H&E; x20 (A), Hematoxylin; x20 (B), Curcumin and Hematoxylin; x10 (C)
Examination of Liver, Pancreas, and Spleen Tissues Stained with Curcumin, Hematoxylin, and Copper (II) Sulfate
The liver tissue stained with curcumin, hematoxylin, and Copper (II) sulfate was examined under a light microscope at 20x magnification. The cell nuclei and Kupffer cells were observed to appear pale yellow in color. On the other hand, in the group stained with curcumin and hematoxylin, Kupffer cells were observed with a more distinct coloration, whereas in the group stained with curcumin, hematoxylin, and Copper (II) sulfate, the Kupffer cells and cell nuclei appeared in pale yellow-brown tones (Figure 6).
Figure 6: Liver tissue, H&E; x20 (A), Hematoxylin; x20 (B), Curcumin and Hematoxylin; x20 (C), Curcumin, Hematoxylin and Copper II Sulfate x20 (D)
The staining of pancreatic tissue with curcumin, hematoxylin, and copper (II) sulfate resulted in pale blue nuclei and pale yellow cytoplasm. In the group stained with curcumin and hematoxylin, capillaries were not clearly visible, whereas in the group stained with curcumin, hematoxylin, and copper (II) sulfate, the capillaries became more distinct, appearing pale yellow to light brown.
In the pancreatic tissue stained with curcumin, hematoxylin, and copper (II) sulfate, observation under a light microscope at 40x magnification revealed that the sinusoid-like acini, zymogen granules, the connective tissue capsule of the pancreatic islet, and the endocrine cells within the islet were clearly distinguishable. In the pancreatic tissue stained with curcumin, hematoxylin, and copper (II) sulfate, a difference in contrast was observed compared to the tissue stained with curcumin and hematoxylin (Figure 7).
Figure 7: Pancreatic tissue, H&E; x20 (A), Hematoxylin; x20 (B), Curcumin and Hematoxylin; x20 (C), Curcumin, Hematoxylin and Copper II Sulfate x20, x40 (D-E),
Following the application of a staining protocol involving curcumin, hematoxylin, and copper (II) sulfate, splenic tissue sections revealed well-defined histoarchitectural components under light microscopy, including the dense connective tissue capsule, red pulp, white pulp, central arterioles, and trabecular structures.
Examination of the sections at ×20 magnification demonstrated that cell nuclei, which typically exhibit blue-purple hues with conventional hematoxylin staining, underwent a marked chromatic shift to dark brown. Furthermore, the central arterioles and trabeculae appeared distinctly delineated, and the tissue displayed an intense distribution of dark yellow coloration throughout the parenchyma. Notably, a comparative evaluation with tissues stained solely with curcumin and hematoxylin indicated that the inclusion of copper (II) sulfate markedly enhanced contrast, thereby facilitating improved visualization of structural details (Figure 8).
Figure 8: Spleen tissue, H&E; x20 (A), Hematoxylin; x20 (B), Curcumin and Hematoxylin; x20 (C), Curcumin, Hematoxylin and Copper II Sulfate x10, x20 (D-E)
Examination of Liver, Pancreas and Spleen Tissues Stained with Saffron and Hematoxylin
When liver tissue stained with safranin and hematoxylin was examined under a light microscope, the cell nuclei appeared distinct but pale blue in color, while the cytoplasmic areas were observed in a dark yellow to brownish hue (Figure 9).
Figure 9: Liver tissue, H&E; x20 (A), Hematoxylin; x20 (B), Saffron and Hematoxylin; x20 (C)
In the pancreatic tissue, the islets of Langerhans and sinusoid-like acini were clearly visible; the cell nuclei appeared in a distinct navy blue tone, while the cytoplasm was observed in a prominent dark yellowish-brown color. As a result of staining pancreatic tissue using the safranin and hematoxylin method, observation under a light microscope at 20x magnification revealed that the cell nuclei transitioned from a navy blue tone to pale blue, while distinct dark yellowish-olive tones were observed in the cytoplasm (Figure 10).
Figure 10: Pancreatic tissue, H&E; x20 (A), Hematoxylin; x20 (B), Saffron and Hematoxylin; x10-x20 (C-D)
In the splenic tissue, compared to the tissue stained only with hematoxylin, the red pulp, white pulp, central arteries, and trabeculae were clearly observed. In the splenic tissue stained with safranin and hematoxylin, observation under a light microscope at 20x magnification revealed the central artery and lymphatic nodule clearly; the nuclei appeared pale blue, while the surrounding cytoplasm was distinctly dark yellow in color (Figure 11).
Figure 11: Spleen tissue, H&E; x20 (A), Hematoxylin; x20 (B), Saffron and Hematoxylin; x10-x20 (C-D)
Examination of Liver, Pancreas, and Spleen Tissues Stained with Safranin, Hematoxylin, and Copper (II) Sulfate
After staining liver tissue with safranin, hematoxylin, and Copper (II) sulfate, examination under a light microscope revealed that the cell nuclei were brown in color and easily distinguishable. The cytoplasmic areas were observed in shades ranging from yellow to green. In contrast to tissues stained only with safranin and hematoxylin, Kupffer cells appeared pale with this staining method, and significant differences in contrast were observed (Figure 12).
Figure 12: Liver tissue, H&E; x20 (A), Hematoxylin; x20 (B), Saffron and Hematoxylin; x20 (C), Saffron, Hematoxylin and Copper II Sulfate x20 (D)
Examination of the pancreatic tissue revealed that the cell nuclei were blue in color, the boundaries of the serous acini were well- defined, and the zymogenic cells were clearly observed. These findings, when compared to pancreatic tissue stained only with safranin and hematoxylin, indicated that the combination of safranin, hematoxylin, and Copper (II) sulfate produced significant changes in contrast properties (Figure 13).
Figure 13: Pancreatic tissue, H&E; x20 (A), Hematoxylin; x20 (B), Saffron and Hematoxylin; x20 (C), Saffron, Hematoxylin and Copper II Sulfate x20 (D)
Examination of the splenic tissue revealed that the connective tissue capsule, red pulp, white pulp, and central artery were clearly observed. Upon examination of the splenic tissue under a light microscope at 20x magnification, the cell nuclei lost their pale blue-purple color and appeared in dark brownish-gray tones; the lymphatic nodule and central artery were clearly visible, and a dense yellow coloration was observed in the splenic tissue section. In the spleen tissue stained with safranin, hematoxylin, and Copper (II) sulfate, a significant difference in contrast was observed compared to the tissue stained only with safranin and hematoxylin (Figure 14).
Figure 14: Spleen tissue, H&E; x20 (A), Hematoxylin; x20 (B), Saffron and Hematoxylin; x20 (C), Saffron, Hematoxylin and Copper II Sulfate x10, x20 (D-E)
Examination of Liver, Pancreas, and Spleen Tissues Stained with Henna
After examination under a light microscope, it was observed that in the liver tissue stained solely with henna powder, all areas were stained brown; the portal area, hepatic cord structures, and hepatocytes were clearly distinguishable, and the cell nuclei were stained dark brown (Figure 15). After staining the pancreatic tissue solely with henna powder, examination under a light microscope with increased light intensity allowed the capillary vessels to be distinguished. Sinus acinuses were dark brown but not clearly distinguishable as borders. Islets of Langerhans could not be clearly observed. However, granules were clearly visible unlike hematoxylin and eosin staining (Figure 16). After staining the spleen tissue with henna alone, the central arteries and trabecular structures were more clearly selected by dimming the light source. Erythrocytes were observed as light brown. Cord structures, red pulp, white pulp, trabecular structures and luminal structures were visible (Figure 17).
Figure 15: Liver tissue, H&E; x20 (A), Hematoxylin; x20 (B), Henna; x20 (C)
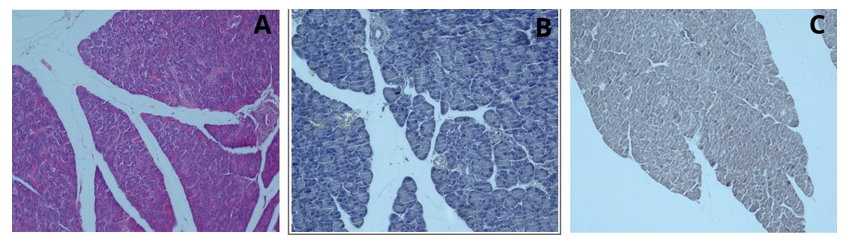
Figure 16: Pancreatic tissue, H&E; x20 (A), Hematoxylin; x20 (B), Henna; x20 (C)
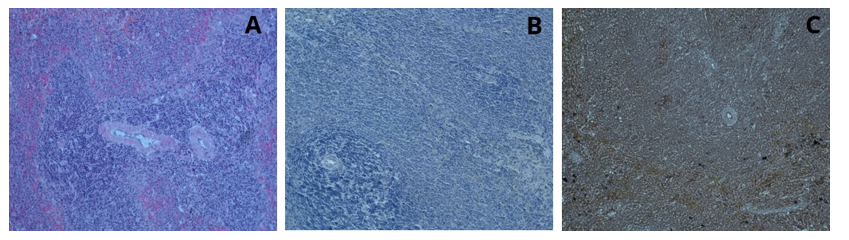
Figure17: Spleen tissue, H&E; x20 (A), Hematoxylin; x20 (B), Henna; x20 (C)
Examination of Liver, Pancreas and Spleen Tissues Stained with Henna and Hematoxylin
After staining the liver tissue with hematoxylin and henna powder, examination under a light microscope revealed that, unlike staining with henna (Figure 18). In the examination of the pancreatic tissue under the light microscope, unlike staining with henna powder alone, the sinusoidal vascular structures and sinusoid-like acini were clearly distinguishable. In the examination of the pancreatic tissue under the light microscope at 40x magnification, the boundaries of the sinusoid-like acini and the zymogenic granules were more clearly observed, and the borders of the pancreatic islets were distinctly visible (Figure 19). Examination of the splenic tissue revealed that the red pulp, white pulp, central arteries, cords, and venous sinuses were clearly distinguishable (Figure 20).
Figure 18: Liver tissue, H&E; x20 (A), Hematoxylin; x20 (B), Henna; x20 (C), Hematoxylin and Henna; x20 (D
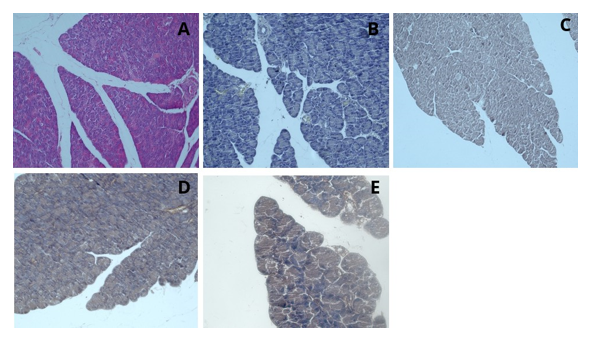
Figure 19: Pancreatic tissue, H&E; x20 (A), Hematoxylin; x20 (B), Henna; x20 (C), Hematoxylin and Henna; x20-x40 (D-E)
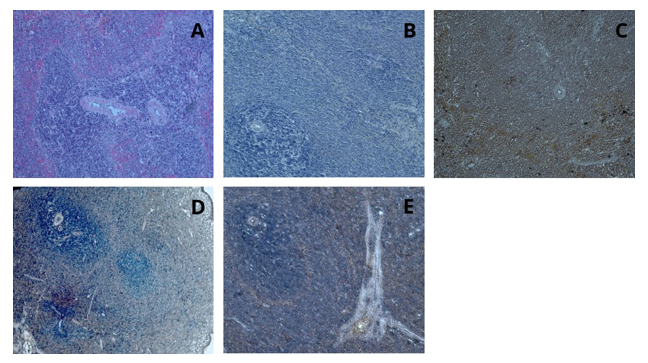
Figure 20: Spleen tissue, H&E; x20 (A), Hematoxylin; x20 (B), Henna; x20 (C), Hematoxylin and Henna; x10-x20 (D-E)
Discussion
In histological analysis, the visualization and differentiation of tissue structures under light or electron microscopy necessitate precise staining protocols. This is typically achieved by processing tissue sections obtained from animal or plant specimens using various dyes [5,6]. Among the most routinely employed staining techniques in histopathological laboratories is the hematoxylin and eosin (H&E) method. Hematoxylin is a basic dye that requires oxidative transformation into hematein to become functionally active [34,35]. This oxidation can occur naturally through exposure to atmospheric oxygen and light or be chemically induced using agents such as sodium iodate, hydrogen peroxide, mercuric oxide, or potassium permanganate.
Due to hematoxylin’s limited intrinsic affinity for tissue components, effective staining necessitates the incorporation of mordants—typically metal ions such as aluminum, iron, chromium, or molybdenum—which facilitate the formation of stable dye-tissue complexes [14,33]. In conventional H&E staining, hematoxylin preferentially binds to nuclear chromatin, rendering it blue to purple, while eosin stains the cytoplasmic and extracellular proteins various shades of pink due to its affinity for basic components [16,17]. However, the high cost and synthetic nature of these dyes, particularly hematoxylin—which poses potential toxicity risks in its oxidized form—have prompted a growing interest in natural, eco-friendly alternatives [36-38]. In this context, our study explored the staining efficacy of plant- derived extracts—namely curcumin, saffron, and henna (Lawsonia inermis)—as substitutes or supplements to conventional H&E staining, using liver, pancreas, and spleen tissues as experimental models.
Standard H&E staining produced the expected results, with nuclei appearing distinctly blue-purple and the cytoplasm pink. In contrast, tissues stained exclusively with hematoxylin exhibited clearly defined nuclei but lacked cytoplasmic contrast, impeding the evaluation of cellular architecture and function. Dual staining with hematoxylin and curcumin extract resulted in nuclei retaining their characteristic blue-purple hue, while the cytoplasm appeared in variable shades of yellow, light brown, or green. This suggests that curcumin, due to its polyphenolic structure, is capable of binding through both hydrogen bonding and electrostatic interactions; however, lipophilic properties are unlikely to be relevant following paraffin processing and dewaxing [39]. Of particular note was the enhanced visibility of Kupffer cells in hepatic tissue, indicating curcumin’s high affinity for certain macrophage populations.
A more refined staining contrast was observed with the implementation of a trichrome technique, involving sequential application of hematoxylin, curcumin extract, and Copper (II) sulfate. This mordant-mediated protocol substantially improved tissue differentiation, supporting previous assertions that mordant addition amplifies the staining capacity of natural dyes. Similarly, incorporation of saffron extract into the staining protocol produced nuclei with standard hematoxylin coloration and cytoplasm stained in dark yellow hues. The observed ionic interactions between saffron and basic cellular constituents align with the behavior of conventional eosin, suggesting that saffron may serve as a viable natural counterstain. Staining with henna extract yielded distinct nuclear staining, with hepatocyte nuclei appearing dark brown.
Given the known electrostatic interactions between hematoxylin and nucleic acids, these findings suggest that henna may also bind to DNA and RNA through a similar mechanism, highlighting its potential utility in nuclear visualization. Collectively, the data demonstrate that plant-based dyes such as curcumin, saffron, and henna not only provide satisfactory histological staining but also offer advantages in terms of biocompatibility, environmental safety, and economic feasibility. These natural alternatives represent a promising direction for sustainable histological practices, particularly in resource-limited settings where cost-effectiveness is paramount. Although the natural dyes did not reproduce the classic nuclear and cytoplasmic contrast seen with hematoxylin and eosin (H&E) staining, selective staining of certain structures such as Kupffer cells and pancreatic acini was observed, suggesting partial affinity depending on tissue type and dye characteristics [38,39].
Conclusions and Recommendations
As a result of this study, saffron, curcumin and stone henna extracts could be used in histological tissue staining instead of hematoxylin and eosin. In the light of the analysis, it was observed that these natural dyes showed high interest when used with mordants and successfully stained the tissues. In our study, we aimed to demonstrate the routine use of saffron, curcumin and stone henna extracts in laboratories as these dyes are non- toxic, low cost and easily accessible. In this context, the use of natural dyes such as saffron, curcumin and stone henna instead of traditional synthetic dyes offers a more sustainable alternative both economically and environmentally. Natural dyes need to be tested more extensively in various tissue types and with different histological methods, and their long-term stability and staining efficiency should be studied in detail. Furthermore, it is of great importance that extensive standardization studies and quality control procedures are established before the routine use of these dyes in clinical laboratories. This will enable more widespread and reliable use of natural dyes such as saffron, curcumin and stone henna in histological staining.
Ethical Consideration
Funding
This research received no external funding.
Conflict of Interest
The authors declare that there is no conflict of interest.
Ethical Approval
All experimental procedures involving animals were approved by the Animal Ethics Committee of Aydın Adnan Menderes University (Approval No: 64583101/2024/50) and carried out in accordance with institutional guidelines.
Informed Consent
Not applicable (N/A). This study involved no human participants.
Author Contributions
Sümeyye Tunçel: Experimental design and execution, data collection. Nur Sevim Kalkan: Data analysis, figure preparation, manuscript writing. Alpaslan Gökçimen: Supervision, manuscript revision, corresponding author.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request. The datasets are not publicly available in an open-access repository.
Acknowledgments
We would like to thank Aydin Adnan Menderes University for providing the laboratory infrastructure and technical support needed for this study.
References
- Avwioro, O. G., Bankole, J. K., Ogundipe, O. A., Odujoko,O. O., & Olabiyi, O. (2007). Curcumin and other natural dyes as histological stains. Nigerian Journal of Health and Biomedical Sciences, 6(1), 1–5.
- Kumar, A., Pandey, A., & Jain, S. (2014). Use of curcuma longa as histological stain for the demonstration of connective tissue fibres. International Journal of Health Sciences & Research, 4(1), 86–91.
- Hadizadeh, F., Mohajeri, S. A., & Seifi, M. (2010). Extraction and purification of crocin from saffron stigmas employing a simple and efficient crystallization method. Pakistan Journal of Biological Sciences, 13(14), 691.
- Rezaee-Khorasany, A., Razavi, B. M., Taghiabadi, E., Yazdi,A. T., & Hosseinzadeh, H. (2019). Effect of saffron (stigma of Crocus sativus L.) aqueous extract on ethanol toxicity in rats: A biochemical, histopathological and molecular study. Journal of ethnopharmacology, 237, 286-299.
- Black, J. (2012). Microbiology: Principles and exploration (8th ed. b.). John Wiley and Sons.
- Anderson, J., & Rolls, G. (2011). An introduction to Routine and special staining. Retrieved on August, 18, 2014.
- Avwioro, O. G. (2010). Histochemistry and tissue pathology, principles and techniques. Nigeria: Claverianum.
- Marshall, P. N., & Horobin, R. W. (1972). The oxidation products of haematoxylin and their role in biological staining. The Histochemical Journal, 4(6), 493-503.
- Lalor, G. C., & Martin, S. L. (1959). Studies on Haematoxylin and Haematein, the Colouring Principles of Logwood: I–Absorption Spectra of Pure Compounds in Various Solvents and a Spectrophotometric Method of Analysis for Haematoxylin and Haematein. Journal of the Society of Dyers and Colourists, 75(11), 513-517.
- Drury, R., and Wallington, E. (1980). Carleton's Histological Technique. Oxford: Oxford University Press.
- Clark, G. (1974). Comparison of various oxidants for alum hematoxylin. Stain Technology, 49(4), 225-227.
- Titford, M., & Bowman, B. (2012). What may the future hold for histotechnologists?. Laboratory Medicine, 43(suppl_2), e5-e10.
- Ali, F., and Orchard, G. (2017). Hematoxylin in history – the heritage of histology. JAMA Dermatol, 153(3):328.
- Titford, M. (2005). The long history of hematoxylin.Biotechnic & histochemistry, 80(2), 73-78.
- Gurr, E., & Florey, H. W. (1962). Staining animal tissues: practical and theoretical. (No Title).
- Bancroft, J. D., & Gamble, M. (2008). Theory and Practice of Histological Techniques (6th ed.). Churchill Livingstone/ Elsevier.
- Fischer, A. H., Jacobson, K. A., Rose, J., & Zeller, R. (2008). Hematoxylin and eosin staining of tissue and cell sections. Cold Spring Harbor Protocols, 2008(5), pdb.prot4986.
- Suvarna, K. S., Layton, C., & Bancroft, J. D. (2018). Bancroft's theory and practice of histological techniques E-Book. Elsevier health sciences.
- Kiernan, J. (2015). Histological and histochemical methods.Scion publishing ltd.
- Lowe, D., and Anderson, P. G. (2015). Pathology: A Modern Case Study. McGraw-Hill Education.
- Rojas-Sandoval, J., and Acevedo-Rodríguez, P. (2013). Haematoxylum campechianum (logwood). Forestry Compendium.
- Faisal, R. A., and Orchard, G. E. (2017). Hematoxylin in history – the heritage of histology. JAMA Dermatol, 153(3):328.
- Cook, H. C. (2015). Manual of Histological Demonstration Techniques. Butterworth Heinemann.
- Orchard, G. E. (2019). Haematoxylin in history – the heritage of haematoxylin. Journal of Histotechnology, 42(2), 89-95.
- Lendrum, A. C. (1947). A Note on Haematoxylin. Journal of Clinical Pathology, 1(1), 2-4.
- Cooksey, C. J. (2018 ). Quirks of dye nomenclature 10. Eosin Y and its close relatives. Biotechnic and Histochemistry, 93(3), 211-219.
- Titford, M. (2009). Histological stains and their diagnostic uses. Journal of Histotechnology, 32(4), 165–173.
- Singh, A., Singh, R., & Meena, R. (2020). Potential of natural dyes in histopathology: A review. Journal of Cytology and Histology, 11(2), 1–5.
- Cardon, D. (2007). Natural dyes. Sources, tradition, technology and science, 268.
- Kumar, S., Singh, N. N., Singh, A., Singh, N., & Sinha, R.K. (2014). Use of Curcuma longa L. extract to stain various tissue samples for histological studies. AYU (An International Quarterly Journal of Research in Ayurveda), 35(4), 447-451.
- Shet Verenkar, N. G., & Sellappan, K. (2021). Evaluation of natural dyes Curcuma longa and Nyctanthes arborâ?tristis with different mordants on plant tissues under fluorescence microscopy. Microscopy Research and Technique, 84(5), 902- 911.
- Rubina, M. P., Krishnan, A. M., Riyas Basheer, K. B., Mohammed Safeer, T. K., & Soumya, V. (2020). Assessment of staining quality of curcumin as a substitute for eosin in hematoxyline and eosin staining in histopathology. J Res Med Dent Sci, 8(5), 146-50.
- Clark, G. (1974). The experimental histologist (3rd ed.). Williams & Wilkins.
- Chan, J. K. (2014). The wonderful colors of the hematoxylin– eosin stain in diagnostic surgical pathology. International journal of surgical pathology, 22(1), 12-32.
- Marshall, P. N., & Horobin, R. W. (1972). The mechanism of staining with hematoxylin. Histochemie, 31(1), 31–36.
- Orchard, G. E. (2019). Plant-based histological stains: A sustainable alternative? Biotechnic & Histochemistry, 94(3), 173–180.
- Titford, M. (2009). Progress in the development of microscopical techniques for diagnostic pathology. Journal of Histotechnology, 32(1), 9–19.
- Singh, S., Dash, A. K., and Singh, R. (2020). Sustainable alternatives for hematoxylin: An approach towards green histology. Histochemistry and Cell Biology, 154(4), 393-403.
- Avwioro, O. G., Onwuka, S. K., Moody, J. O., Agbedahunsi, J. M., Oduola, T., Ekpo, O. E., & Oladele, A. A. (2007). Curcuma longa extract as a histological dye for collagen fibres and red blood cells. Journal of anatomy, 210(5), 600-603.