Research Article - (2023) Volume 2, Issue 1
Stabilizing Effect of Vitamin C on Sulforaphane Level in A Controlled-Fermented Broccoli Sprouts
2Family Practice Dr. Kleiker, Am Wacholder 36, Herzo genrath-Aachen, Germany
Received Date: Jul 01, 2023 / Accepted Date: Jul 24, 2023 / Published Date: Aug 08, 2023
Copyright: ©Â©2023 Anne-Marie Kosi-Kupe, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Kosi-Kupe, A. M., Kleiker, H. (2023). Stabilizing Effect of Vitamin C on Sulforaphane Level in A Controlled-Fer-mented Broccoli Sprouts. World J Radiolo Img, 2(1), 08-14.
Abstract
This study investigates the sulforaphane levels in broccoli sprouts samples produced under a controlled-lactic acid fermentation. Such process includes combining broccoli sprouts, a cruciferous vegetable, with twelve selected strains of lactic acid bacteria: Lactobacilli, Streptococci, and Bifidobacteria to form a mixture that undergoes lactic acid fermentation, transforms glucosinolate within the cruciferous vegetable to sulforaphane, and yield a fermented mixture that contains sulforaphane, and subsequently from that fermented mixture manufacturing nutritional food item that can be ingestable and sustainable for humans. pH decrease within the lag phase is the critical and decisive moment for the end product sulforaphane stability.
Purpose Various sulforaphane powders, pills, and probiotics are commercially available. However, sulforaphane powder mixed in water is only stable over a short period and must be consumed as soon as possible, often within thirty minutes of mixing. Accordingly, there is a desire for a convenient and durable source of sulforaphane capable of delivering the nutritional benefits of sulforaphane, aside from the direct consumption of fresh broccoli sprouts. This study provides a process that answers that question and addresses the sulforaphane availability in post-oro-maxillofacial surgery patients.
Methods Lactic acid fermentation of broccoli sprouts triggered by twelve homo- and heterofermentative Lactobacilli strains. Early addition of the vitamin C at lag phase; Cell cultures grow in MRS-broth then on MRS-agar followed by a sulforaphane evaluation using HPLC method.
Results A mixture of twelve selected hetero- and homofermentative Lactobacilli strains employed during controlled-lactic acid fermentation enable the transformation of glucosinolate from broccoli sprouts to a stable sulforaphane level of 0.130 mg/g with a sustainable nutritional value. The mixture's Lactic acid bacteria (LAB) count remained high (2500000 cells/g), and we detected no coliform bacteria. The early addition of vitamin C is essential to stabilizing of sulforaphane content of this study.
Conclusion This study yields a nutritional and microbiological stable sulforaphane-containing product ingestible by humans whose sulforaphane levels survived the processing mechanism and can sustain daily dietary requirements in vitamins, minerals, proteins, and carbohydrates.
Keywords
Sulforaphane Level, Broccoli Sprouts, Lactic Acid Bacteria, Sulforaphane Bioavailability
Abbreviations
LAB: Lactic acid bacteria
HPLC: High-performance liquid chromatography
MRS-agar (broth): de Man, Rogosa and Sharp-agar
B: Bifidobacterium
ITCs: Isothiocyanates
HoLAB: Homofermentative Lactic Acid Bacteria
HeLAB: Heterofermentative Lactic Acid Bacteria
Introduction
Sulforaphane is known as a long-lasting antioxidant, who’s anti-cancerogenic effects were confirmed by in-vitro studies conducted by John Hopkins University School of Medicine [1-11] and confirmed by multiple publications [12-14]. Sulforaphane is obtained from cruciferous vegetables (the family of Brassicaceae, formerly Cruciferae), non-limiting examples of which include brussels sprouts, cabbages, cauliflower, broccoli particularly broccoli sprouts). A series of encyclopedias have extensively reported on cruciferous vegetable origin, genetics, and chemistry, especially how glucoraphanin (a glucosinolate) reacts with myrosinase (an enzyme present in the plant cell) to form sulforaphane [15-19,11]. The protective effect of these micronutrients is due to the inhibition of phase-I carcinogen- bioactivating enzymes and induction of phase-II detoxifying enzymes [5-10]. The protective effect of glucoraphanin is thought to be due to sulforaphane, an isothiocyanate metabolite formed from glucoraphanin by the enzyme myrosinase [3-5,20,21]. The reaction between myrosinase and glucosinolate occurs in the gut after glucosinolate has been made available by chewing such that sulforaphane is produced after the swallowing event occurs.
Storage, processing, and cooking can change ITCs formation from glucosinolate and affect the cruciferous vegetables' anticancer activity [12]. Intake of raw cruciferous vegetables provides two to nine times the number of ITCs in humans compared with similar intakes of their cooked counterparts due to heat-inactivating myrosinase, which reduces sulforaphane formation [13,14,22,23].
Human intestinal microflora also possesses myrosinase activity, able to partially hydrolyze ingested glucosinolates which is an advantage to this study with the use of Lactobacilli strains. Gut microflora is mainly responsible for converting ingested glucosinolates to their cognate isothiocyanates [14].
Despite their nutritional and health benefits, including being a source of sulforaphane, broccoli sprouts have certain disadvantages that have limited their use. For instance, broccoli sprouts tend to spoil quickly, and contamination with coliform bacteria is a well-known safety issue, resulting in unwanted additives often accompanying broccoli sprouts. Several studies have elucidated microbial fermentation's positive impact on product shelf life and the protective properties of lactic acid fermentation. Balows et al., 1991 also highlighted the prokaryotic ecophysiology, characterization, and applications [24-46].
Apart from providing an edible fermented new product capable of delivering sulforaphane's nutritional benefits (figure 1), this study offers ways to avoid drawbacks associated with the consumption of cruciferous vegetables, especially broccoli sprouts.
Materials and Methods
Processing Brief Description'
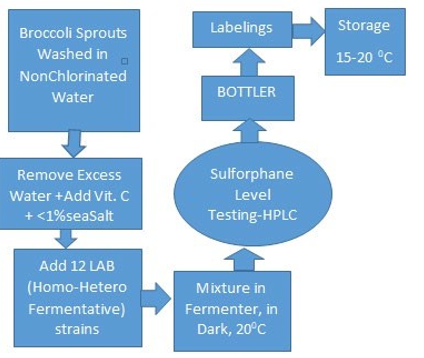
Figure 2: represents a flow diagram of a process for probiotics broccoli sprouts.
As outlined in figure 2, we sanitize broccoli seeds before being planted to grow broccoli sprouts. As used herein, broccoli sprouts will refer to new growth from seed germination, a plant that is a few (e.g., three to four) days old. After harvest, the sprouts are washed in purified water and dried to reduce the presence of bacteria on the sprouts. The sprouts are then combined with LAB strains and ingredients (<1% NaCl) which trigger a lactic acid fermentation that transforms glucosinolate contained in the sprouts into sulforaphane-containing product. Fermentation produces sulforaphane without requiring chewing or physically breaking up the sprouts to make the glucosinolate available and without the requirement for contacting and reacting the sprouts with myrosinase. The broccoli sprouts are combined with twelve strains of Lactobacilli, which are part of the lactic acid bacteria (LAB) group and known as probiotics. The novelty of this study is the addition of vitamin C at the earlier stage (5-10 minutes) during LAB growth lag phase.
The fermentation has been successfully conducted in fermenters under anaerobic and dark environments (i.e., shielded from and without natural and artificial lighting) at room temperatures of about 20oC.
Labs and Methods
In this particular study, organic broccoli sprouts germinated over three days were washed three times in non-chlorinated purified water, after which the following twelve selected strains of lactobacilli were added: B. bifidum, B. lactis, B. infantis, B. longum, L. acidophilus, L. brevis, L. bulgaricus, L. paracasei, L. plantarum, L. rhamnosus, L. salvaricus, and Streptococcus thermophilis from “Nature” were used as starter cultures.
These chosen strains are known for their physiological benefits and antimicrobial capabilities. Sea salt <1% and, especially, vitamin C added to this mixture create an early acidic pH suitable for lactic acid fermentation/Reference samples without vitamin C, conducted anaerobically in a dark environment at about 200C for seven days in handmade 3-layered wood-ceramic-glass fermenter (figure 3).
LAB-Analyses: By CMMEF, 4th Ed.; MRS-Agar (broth); E. coli & Coliform (by Petrifilm-AOAC 991.14)
Sulforaphane Content Evaluation: By Solid Phase Extraction (SPE)-High-Performance Liquid Chromatography (HPLC) by Han D.et al.
Minerals and Vitamins Evaluation: Calcium by ICP (AOAC 984.27); Iron by ICP; Vitamin A-Beta Carotene (Analyst 1984 109:48), Vitamin C (HPLC)- 53 mg Vitamin C in 113 g sprouts
Figure 3: Three- layered Fermenter Technology (external layer:woody#2, middle layer: ceramic#4, internal layer: glass#1;door opening#3; reduced air circulation #5)
Results
The process from this study yields a microbiologically safe and durable natural formula that contains sulforaphane before being ingested by an individual and is capable of delivering benefits to the individual when ingested.
After seven days at these conditions described above, the mixture was found to be fermented, and the mixture tested positive (0.130 mg/g) for sulforaphane. The Lactic acid bacteria (LAB) count of the mixture remained high (2500000 cells/g), pH-decrease from 5.0 to 3.9 (Figure 4) and no coliform bacteria was detected. Following storage of the fermented mixture in a refrigerator over a two-month period, coliform bacteria were not detected either. After six months of storage in a refrigerator, the amount of sulforaphane dropped to 50% (0.0651 mg/g), which is a good result given the instability of that phytochemical.
As such, this study processing method results in a broccoli sprouts mixture containing sulforaphane-probiotics with notable nutritional benefits, enriched with vitamins and minerals, which contribute to answering world challenges regarding food scarcity and sustainability.
|
Total fat |
12.7g/100g |
|
Saturated fat |
0.12 g/100g |
|
Trans fat |
0.12g/100g |
|
Sodium |
568mg/100g |
|
Total carbohydrate |
5.2g/100g |
|
Dietary fibers |
2.11g/100g |
|
Sugars |
1.6g/100g |
|
Protein (f=6.25) |
2.88g/100g |
|
Calcium |
38.3 mg/100g |
|
Iron |
0.77mg/100g |
|
Vitamin A |
662 IU/100g |
|
Vitamin C |
30.11 mg/100g |
|
Sulforaphane |
0.130 mg/g (triplicate samples) |
|
LAB |
2500000 cells/g |
|
E.coli/Coliform |
Not detected |
Table 1: Nutritional and microbiological evaluation of fermented broccoli sprouts
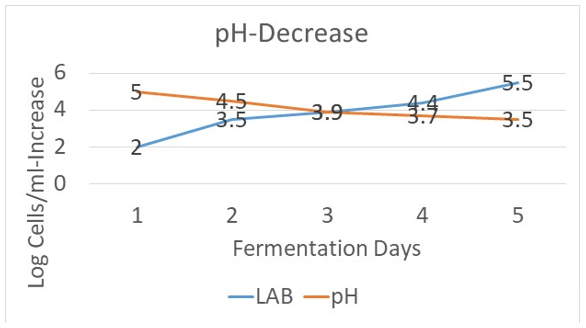
Figure 4: pH-Decrease within lag-phase corrreponding to LAB Increase (log cell count/ml)
Conclusion and Discussion
The broccoli sprouts combined with twelve specific LAB, as described in this study yielding to the current new product, have been found to trigger a lactic acid fermentation that transforms glucosinolate contained in the sprouts to sulforaphane. Selected Lactobacilli (HeLAB and HoLAB) strains employed by the process in this study yield a sulforaphane-containing product and enhance the stable sulforaphane levels [13,14,22,23].
Lactic acid Fermentation from this study produces sulforaphane without requiring chewing or otherwise physically breaking up the sprouts to make the glucosinolate available and without the requirement for contacting and reacting the sprouts with myrosinase outside the human GI (Gastrointestinal)-tract.
The end product from this study can remain in the form of a consumer food item that can be directly ingested by an individual.
In addition to consumer food items, this study fermented products may be processed and packaged for use by medical personnel and nutritionists, in a variety of industries and practices, for example, as a pill, powder, for use in pharmaceuticals, nutraceuticals, preventive medicines, food processors, etc. This fermented product can particularly benefit individuals with digestive or other specific health issues. The shelf-life of this fermented product is up to two mounts between 10oC to 15oC temperature.
While this study has focused on twelve LAB strains, new studies could use other forms of the mixture; for example, though twelve specific strains of Lactobacillus were used in this study, hypothetically, it may be possible to omit or add some of the used strains of lactobacilli, and yet yield a fermentation that may produce a suitable fermented mixture that contains a sustainable amount of sulforaphane. In addition, it is foreseeable that multiple cruciferous vegetables could also be combined and fermented. We should keep in mind that the processing of Brassica vegetables has negatively impacted the contents of glucosinolates and has reduced their health-protective capabilities [12]. The other question remains on the impact of the ratio of homo- versus heterofermentative strains on the stabilization of sulforaphane level. On the contrary, it is fair to affirm that this study’s vitamin C addition at the lag phase in three-layered fermenter is the turning and essential point of this research paper’s contribution. It brings some hope regarding the end product’s sulforaphane levels and stability, which may contribute positively to food sustainability challenges and opportunities. You can grow it in your environmentally friendly backyard, and its scope provides countless applications [5-10]. Even though the daily value for sulforaphane is still unknown, its amount in this paper’s results is encouraging for consumers with oro-maxillofacial chewing issues. One could investigate monthly radiological and imaging evaluations of sulforaphane using this study’s new processing method.
Acknowledge
This study’s HPLC, minerals, and vitamin testing were supported by the Merieux Laboratories, USA.
Author & Co-Author Contributions
The manuscript was conceived, conducted and written by the author and reviewed by the co-author. Both agreed on the last draft.
Conflict of interest
The author declares no conflict of interest.
References
- Fahey, J. W., & Talalay, P. (1995). The role of crucifers in cancer chemoprotection.
- Farnham, M. W., Stephenson, K. K., & Fahey, J. W. (2000). Capacity of broccoli to induce a mammalian chemoprotective enzyme varies among inbred lines. Journal of the American Society for Horticultural Science, 125(4), 482-488.
- Fahey, J. W., Zhang, Y., & Talalay, P. (1997). Broccoli sprouts: an exceptionally rich source of inducers of enzymes that protect against chemical carcinogens. Proceedings of the National Academy of Sciences, 94(19), 10367-10372.
- Fahey, J. W., Stephenson, K. K., & Talalay, P. (1998). Glucosinolates, myrosinase, and isothiocyanates: three reasons for eating Brassica vegetables.
- Shapiro, T. A., Fahey, J. W., Wade, K. L., Stephenson, K. K., & Talalay, P. (1998). Human metabolism and excretion of cancer chemoprotective glucosinolates and isothiocyanates of cruciferous vegetables. Cancer epidemiology, biomarkers & prevention: a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology, 7(12), 1091-1100.
- Talalay, P. (1999). The war against cancer: new hope. Proceedings of the American Philosophical Society, 143(1), 52-72.
- Talalay, P., Fahey, J. W., Holtzclaw, W. D., Prestera, T., & Zhang, Y. (1995). Chemoprotection against cancer by phase 2 enzyme induction. Toxicology letters, 82, 173-179.
- Zhang, Y., Kensler, T. W., Cho, C. G., Posner, G. H., & Talalay, P. (1994). Anticarcinogenic activities of sulforaphane and structurally related synthetic norbornyl isothiocyanates. Proceedings of the National Academy of Sciences, 91(8), 3147-3150.
- Zhang, Y., Talalay, P., Cho, C. G., & Posner, G. H. (1992). A major induceÃÃÂ??ÂÃÂ?ÂÂr of anticarcinogenic protective enzymes from broccoli: isolation and elucidation of structure. Proceedings of the national academy of sciences, 89(6), 2399-2403.
- Perocco, P., Bronzetti, G., Canistro, D., Valgimigli, L., Sapone, A., Affatato, A., ... & Abdel-Rahman, S. Z. (2006). Glucoraphanin, the bioprecursor of the widely extolled chemopreventive agent sulforaphane found in broccoli, induces phase-I xenobiotic metabolizing enzymes and increases free radical generation in rat liver. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis, 595(1-2), 125-136.
- Augustin L.S.A., Nishi S. Encyclopedia of cancer. Diet and cancer. (3) 2019, 471-500
- Barba, F. J., Nikmaram, N., Roohinejad, S., Khelfa, A., Zhu, Z., & Koubaa, M. (2016). Bioavailability of glucosinolates and their breakdown products: impact of processing. Frontiers in nutrition, 3, 24.
- Getahun, S. M., & Chung, F. L. (1999). Conversion of glucosinolates to isothiocyanates in humans after ingestion of cooked watercress. Cancer Epidemiology Biomarkers & Prevention, 8(5), 447-451.
- Conaway, C. C., Getahun, S. M., Liebes, L. L., Pusateri,D. J., Topham, D. K., Botero-Omary, M., & Chung, F. L. (2000). Disposition of glucosinolates and sulforaphane in humans after ingestion of steamed and fresh broccoli. Nutrition and cancer, 38(2), 168-178.
- Holst B., Fenwick G.R. Encyclopedia of food sciences and nutrition (2) 2003,2922-2930
- Puzio, P. S., & Grundler, F. M. W. (2000). Isolation And Application Of Nematode Induced Promoters, Genes And Proteins From Arabidopsis thaliana. In Developments in Plant Genetics and Breeding (Vol. 6, pp. 169-175). Elsevier.
- Takeda, M., & Kainosho, M. (2012). 1.12 Labeling Techniques. Comprehensive Biophysics, 199.
- Craige Trenerry V., Rochfort S. J. Comprehensive natural products II (9) 2010, 595-628
- Bischoff, K. L. Glucosinolates. Nutraceuticals: Efficacy, Safety and Toxicity.
- Osman, A. G., Chittiboyina, A. G., & Khan, I. A. (2016).Cytoprotective role of dietary phytochemicals against cancer development via induction of phase II and antioxidant enzymes. In Advances in molecular toxicology (Vol. 10, pp. 99-137). Elsevier
- Bartnik M., Facey P.C.Pharmacognosy-Fundamentals. Glycosides- Applications and Strategies, 2017, 101-1611
- Rouzaud, G., Young, S. A., & Duncan, A. J. (2004). Hydrolysis of glucosinolates to isothiocyanates after ingestion of raw or microwaved cabbage by human volunteers. Cancer Epidemiology Biomarkers & Prevention, 13(1), 125-131.
- Guo, R. F., Yuan, G. F., & Wang, Q. M. (2013). Effect of NaCl treatments on glucosinolate metabolism in broccoli sprouts. Journal of Zhejiang University Science B, 14, 124- 131.
- Andres, C. (1977). Controlled fermentation reduces salt usage, improves pickle quality and profitability. Food Process, 38(12), 50.
- Ashenafi, M., & Busse, M. (1991). Production of tempeh from various indigenous Ethiopian beans. World Journal of Microbiology & Biotechnology, 7(1), 72-79.
- Ashenafi, M., & Busse, M. (1991). Development of microorganisms during cold storage of pea and chickpea tempeh and effect of Lactobacillus plantarum on storage microflora. Journal of the Science of Food and Agriculture, 56(1), 71-78.
- Ashenafi, M., & Busse, M. (1989). Inhibitory effect of Lactobacillus plantarum on Salmonella infantis, Enterobacter aerogenes and Escherichia coli during tempeh fermentation. Journal of Food Protection, 52(3), 169-172.
- Ashenafi, M., & Busse, M. (1991). Development of microorganisms during cold storage of pea and chickpea tempeh and effect of Lactobacillus plantarum on storage microflora. Journal of the Science of Food and Agriculture, 56(1), 71-78.
- Atanda, O. O., & Ikenebomeh, M. J. (1991). Microbiological quality of nono. World Journal of Microbiology and Biotechnology, 7, 89-91.
- Beever, R. E., & Bollard, E. G. (1970). The nature of the stimulation of fungal growth by potato extract. Microbiology, 60(2), 273-279.
- Amor, K. B., & Fields, M. L. (1990). The production of niacin by microorganisms isolated from fermenting corn meal. Journal of food protection, 53(10), 881-882.
- BUCKENHUÊSKES, H., Omran, H., & Gierschner, K. (1990). Lactic acid fermentation of whole white cabbage. Acta alimentaria (Budapest), 19(2), 157-163.
- Buckenhüskes, H. J. (1993). Selection criteria for lactic acid bacteria to be used as starter cultures for various food commodities. FEMS microbiology reviews, 12(1-3), 253-271.
- Buckle, K. A., & Kartadarma, E. (1990). Inhibition of bongkrek acid and toxoflavin production in tempe bongkrek containing Pseudomonas cocovenenans. Journal of Applied Microbiology, 68(6), 571-576.
- Burns, R. A. (1987). Protease inhibitors in processed plant foods. Journal of food protection, 50(2), 161-166.
- Ceemat-Cirad. From traditional techniques to semi-industrial production: Cassava-Gari, Div. Gén. et technol. aliment. France, 197
- Chen, K. H., McFeeters, R. F., & Fleming, H. P. (1983). Complete heterolactic acid fermentation of green beans by Lactobacillus cellobiosis. Journal of Food Science, 48(3), 967-971.
- CHEN, K. H., McFeeters, R. F., & Fleming, H. P. (1983).Stability of mannitol to Lactobacillus plantarum degradation in green beans fermented with Lactobacillus cellobiosus. Journal of Food Science, 48(3), 972-974.
- Chen, K. H., McFeeters, R. F., & Fleming, H. P. (1983). Fermentation characteristics of heterolactic acid bacteria in green bean juice. Journal of Food Science, 48(3), 962-966.
- Chen, T., & Ho, C. T. (1989). Past, present, and future of Chinese fermented food products. Food Reviews International, 5(2), 177-208.
- Samah, O. A., Ibrahim, N., Alimon, H., & Karim, M. A. (1993). Comparative studies on fermentation products of cocoa beans. World Journal of Microbiology and Biotechnology, 9, 381-382.
- Adachi, S. (1992). Lactic acid bacteria and the control of tumours. In The Lactic Acid Bacteria Volume 1: The Lactic Acid Bacteria in Health and Disease (pp. 233-261). Boston, MA: Springer US.
- Akinrele, I. A., Makanju, A., & Edwards, C. C. (1969). Effect of soya flour on the lactic fermentation of milled corn. Applied microbiology, 17(1), 186.
- Amor, K. B., & Fields, M. L. (1990). The production of niacin by microorganisms isolated from fermenting corn meal. Journal of food protection, 53(10), 881-882.
- Soh, A. L. A., Ralambotiana, H., Ollivier, B., Prensier, G., Tine, E., & Garcia, J. L. (1991). Clostridium thermopalmarium sp. nov., a moderately thermophilic butyrate-producing bacterium isolated from palm wine in Senegal. Systematic and applied microbiology, 14(2), 135- 139.
- Andersson, R. (1986). Lactic acid fermentation of vegetables- an energy conserving technique giving tasty products with good keeping qualities. Mjoelksyraferrmentering av groensaker-en energisnaal teknik som leder till goda och haallbara produkter.
- Yamane, H., Konno, K., Sabelis, M., Takabayashi, J., Sassa, T., Oikawa, H., ... & Lui, H. W. (2010). Comprehensive Natural Products II. Chemistry and Biology.
- Andersson, R. (1985). Nitrate reduction during fermentation by Gram-negative bacterial activity in carrots. International Journal of Food Microbiology, 2(4), 219-225.
- Trüper, H. G., Dworkin, M., Harder, W., & Schleifer, K.H. (1992). The prokaryotes: a handbook on the biology of bacteria: ecophysiology, isolation, identification, applications (Vol. 3). A. Balows (Ed.). New York: Springer- Verlag.
- Stein, M. A. C. F., Kulay, L. A., & Borzani, W. (1991). Semi-continuous lactic fermentation of whey by Lactobacillus bulgaricus. World Journal of Microbiology and Biotechnology, 7(4), 470-474.
- Gangopadhyay, H., & Mukherjee, S. (1971). Effect of different salt concentrations on the microflora and physico-chemical changes in sauerkraut fermentation. J Food Sci chemical changes in sauerkraut fermentation. J Food Sci Technol Mysore.
- Goodman, M. T., Kolonel, L. N., Wilkens, L. R., Yoshizawa,C. N., Le Marchand, L., & Hankin, J. H. (1992). Dietary factors in lung cancer prognosis. European Journal of Cancer, 28(2-3), 495-501.
- Gupta, Y. P. (1987). Anti-nutritional and toxic factors in food legumes: a review. Plant foods for human nutrition, 37, 201-228.
- Hoover, D. G., & Steenson, L. R. (Eds.). (2014). Bacteriocins of lactic acid bacteria. Academic Press.
- Hosono, A., Wardojo, R., & Otani, H. (1989). Microbial flora in dadih, a traditional fermented milk in Indonesia. Lebensmittel-Wissenschaft+ Technologie, 22(1), 20-24.
- KARKI, T., ITOH, H., & KOZAKI, M. (1983). STUDIES ON MICROORGANISMS AND THEIR ROLE IN GUN- DRUK FERMENTATION. 2. CHEMICAL-CHANGES OCCURRING DURING GUNDRUK FERMENTATION.2. 2 FLAVOR COMPONENTS. LEBENSMITTEL-WIS-SENSCHAFT & TECHNOLOGIE, 16(4), 203-208.
- Kassaye, T., Simpson, B. K., Smith, J. P., & O'connor, C.B. (1991). Chemical and microbiological characteristics of Ititu. Milchwissenschaft.
- KATO, T., SHIGA, I., & TERASAWA, K. (1986).Preservation of okara (soybean residue from soy mash) by lactic acid fermentation. Nippon Shokuhin Kogyo Gakkaishi, 33(12), 837-841.
- KAZANAS, N., & Fields, M. L. (1981). Nutritional improvement of sorghum by fermentation. Journal of Food Science, 46(3), 819-821.
- Kjaer, A., & Christensen, B. (1958). ISOTHIOCYANATES. 30. GLUCOHIRSUTIN, A NEW NATURALLY OCCURRING GLUCOSIDE FURNISHING (-)-8-METHYLSULPHINYL-OCTYL ISOTHIOCYANATE ON ENZYMIC HYDROLYSIS. ACTA CHEMICA SCANDINAVICA, 12(5), 833-838.
- Khachik, F., Bertram, J. S., Huang, M. T., Fahey, J. W., & Talalay, P. (1999). Dietary carotenoids and their metabolites as potentially useful chemoprotective agents against cancer. In Antioxidant food supplements in human health (pp. 203- 229). Academic Press.
- Lee, C. H., Souane, M., & Rhu, K. H. (1988). Effects of prefermentation and extrusion cooking on the lactic fermentation of rice-soybean based beverage.
 , 20(5), 666-673.
, 20(5), 666-673. - Mattick, A. T. R., & Hirsch, A. (1944). A powerful inhibitorysubstance produced by group N streptococci. Nature, 154(3913), 551-551.
- Noda, F., Hayashi, K., & Mizunuma, T. (1980). Antagonism between osmophilic lactic acid bacteria and yeasts in brine fermentation of soy sauce. Applied and Environmental Microbiology, 40(3), 452-457.
- Orlov, N.P.; Gul’tyaeva, V.P.; Belokhonova, A.V. Fermentation of cabbage in containers using pure lactic acid bacteria cultures. Tovarovedenie 15, 34-36, 1988.
- Ovcharova, T.P.; Maslennikova, N.M. Use of sorbic acid combined with lactic fermentation to improve the quality of Sauerkraut and cucumbers. Trudy, Vsesoyuznyi Nauchno- issledovatel'skii Institut Konservnoi i ovoshchesushil' noi Promyshlennosti 14, 61-67, 1971.
- Piard, J. C., & Desmazeaud, M. (1992). Inhibiting factors produced by lactic acid bacteria. 2. Bacteriocins and other antibacterial substances. Le lait, 72(2), 113-142.
- Pool-Zobel, B. L., Münzner, R., & Holzapfel, W. H. (1993). Antigenotoxic properties of lactic acid bacteria in the S. typhimurium mutagenicity assay.
- Procházka, Z. (1959). Isolation of sulforaphane from hoary cress (Lepidium draba L.). Collection of Czechoslovak Chemical Communications, 24(7), 2429-2430.
- Rasic, J. L., Bogdanovic, G., & Kerenji, A. (1984). Anti- cancer properties of lactic acid-fermented beetroot juice. Fluss Obst, 51(1), 25-28.




