Research Article - (2025) Volume 10, Issue 2
Smoking and Secondhand Smoke Exposure Among Us Women: Analysis of NHANES Measures
2Professor University of Tennessee Health Science Center College of Nursing Memphis, TN,, USA
3Associate Professor; Biostatistics University of Arkansas for Medical Sciences College Public Health, USA
Received Date: Mar 06, 2025 / Accepted Date: Apr 08, 2025 / Published Date: Apr 15, 2025
Copyright: ©Â©2025 Natalie Pate Capps, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Capps, N. P., Rhoads, S. J., Barone, C. P., Rojo, M., Wright, P., et al. Smoking and Secondhand Smoke Exposure Among Us Women: Analysis of NHANES Measures. J Nur Healthcare, 10(2), 01-14.
Abstract
Purpose Active smoking and secondhand smoke exposure (SHSe) in women are leading causes of morbidity and mortality and have significant economic costs in the US. The purpose of this study is to compare methods of collecting smoke-exposure data and trends of exposure in a representative population of women in the US.
Design A probability sample of women from the National Health and Nutrition Examination Survey (NHANES) was analyzed.
Methods The sample included 13,396 women ≥20 years old. Data were analyzed using biochemical and self-report smoke exposure methods available in the NHANES. Rao-Scott chi-squared and logistic regression were used for analysis.
Results Women most at risk for active smoking were non-Hispanic white (18.81%) and Black women (20.74%) and women with incomes <$20,000 annually (28.36%). SHSe was more likely among Black women (6.18%). NHANES self-report measures demonstrated a similar upward trend as biochemical serum cotinine levels. Number of household smokers was the most sensitive self-report measure.
Discussion This study demonstrated a strong relationship between self-reported smoke exposure and serum cotinine values. Women who are non-Hispanic white, Black and low-income are particularly at risk for smoking and SHSe. In the absence of a gold-standard measure for SHSe, number of household smokers may be a good self-report measure.
Implications Providers must continue to assess smoking and smoke exposure, particularly in women. Gender tailored cessation and initiation reduction methods are essential.
Keywords
Behavior, Gender, Health Behaviors, Smoking, Hazardous Exposures, Secondhand Smoke
Introduction
Highly addictive tobacco and its derivative, environmental tobacco smoke, are major health burdens to smokers and non-smokers alike [1]. Each year in the United States (US), more than 480,000 deaths are attributable to active smoking and 41,000 deaths to secondhand smoke exposure (SHSe) [2]. Smoking and secondhand smoke exposure also present a significant economic toll. Healthcare costs associated with active smoking in the US have been estimated at
$4.6 billion annually [3]. The devastating health effects of active smoking have also been well established. Smoking has been linked to pulmonary disorders, coronary heart disease, decreased bone density, and countless cancers. Further, SHSe has been linked to greater likelihood of active smoking, nicotine dependency, and susceptibility to diseases associated with smoke exposure [4-6].
Smoking rates among women have traditionally been low compared to men; however, recent studies demonstrate a narrowing gap between male and female smoking rates. The risk of women dying from smoking has more than tripled in the last 50 years, now equaling the risk of men. For example, between the years 1959 and 2010, the risk of lung cancer—80% of which is attributable directly to smoking—increased tenfold among women [2]. Smoking remains the most preventable cause of death among women [7].
There has been a significant drop in smoking rates among women since the landmark Surgeon General’s Report in 1964. Despite this overall reduction, the decline has not been distributed equally, with smaller declines among rural women, women with socioeconomic disadvantages and sexual minorities [8-14]. Active smoking and SHSe prevalence are particularly high among racial and ethnic minority groups and low-income women, adding to the effects of health disparities surrounding smoke exposure [15-18].
Women have gender-unique risks of active smoking and SHSe. These include increased risk of cervical and breast cancers, increased relative risk of coronary disease in smoking women compared to smoking men, and increased risk of osteoporosis and premature menopause [19-22]. Additionally, there is a relationship between smoking, estrogen, and lung cancers. Women who use estrogen therapy and smoke have more than double the risk of adenocarcinoma of the lung than female smokers not using estrogen therapy [23]. The consequences of smoke exposure extend to infants of mothers who have smoked in pregnancy, including intrauterine growth restriction, complicated preterm delivery, and late term bleeding disorders [24,25]. It is reported that most women who smoke prior to pregnancy continue to smoke throughout their pregnancies and as a result have associated risks to themselves and offspring [26].
Based on the risks described above, it is imperative to understand which populations of women are most likely to be smoke exposed and how best to measure exposure. There is a dearth of analysis in the literature regarding gendered tobacco use and prevention trends. Additionally, there is need for comparative analysis of self-reported smoking and biochemical testing for both active and SHSe in women. The purpose of this study is (1) to compare methods of collecting smoke exposure data and (2) to identify trends of exposure in a representative population of women in the US who are over 20 years old.
Method
Design and Participants
Data from the National Health and Nutrition Examination Survey (NHANES) 2007–2016 were analyzed for this study. Five 2-year cohorts ranging from 2007–2016 provided a cross-sectional, nationally representative sample. This population represents the non-institutionalized civilian population in the US. All participants provided informed consent and the study was approved by the National Center for Health Statistics ethics review board. NHANES included an interview and a physical examination. The interviews were conducted in each participant’s home with the use of a facilitator. Participants also received a physical examination and biologic testing in a mobile examination center (MEC). This secondary data analysis was deemed exempt by the IRB Board, and the de-identified data is publicly available. The final sample consisted of 13,396 women aged 20 and older.
Measurement
Smoking assessment is most commonly assessed through self- report mechanisms or biochemical testing. The NHANES utilized both methods; this combination is a standardized mechanism of assessment. Secondhand smoke can be more difficult to assess than active smoking and there is no gold standard for secondhand smoke measurement [27,28]. Smoke exposure: Biochemical. Cotinine is the primary metabolite of nicotine and was the biochemical mechanism used for smoke-exposure analysis [29]. Cotinine has a longer half-life and higher concentrations for analysis than nicotine measurement. For example, the nicotine half-life is 0.5– 3 hours, versus a much longer cotinine half-life of 15–20 hours [30]. Due to longer stability, cotinine is generally the biochemical measure of choice in serum samples. Serum cotinine samples were collected in MEC’s and shipped, stored, processed, and analyzed at the Centers for Disease Control and Prevention (CDC). Serum vials were stored appropriately at -20C and analyzed with liquid chromatography/mass spectrometry. Detailed biochemical analysis information is available online through the CDC.
Smoke exposure: Self-report. Smoke exposure was measured using two different self-report questions during the home interview. The first smoke exposure question asked women if they currently smoked. If they responded “yes” to this question, they were classified as active smokers. In women who responded “no” to this question, cotinine levels were used to identify SHSe. Women who self-reported non-smoking status and had serum cotinine levels of 15ng/mL or greater were classified as SHSe. This dual method of self-report and biochemical analysis for SHSe status has previously been used to analyze NHANES data [31]. Further, the serum cotinine cutoff value of 15 ng/ml has been demonstrated in the literature in both NHANES analysis and other large representative samples [32-37]. Women who responded that they did not currently smoke and had serum cotinine values less than 15ng/mL were classified as non-smokers. Overall, women were classified as non-smokers, SHSe, or active smokers.
Number of household smokers: Self-report. A second smoke- exposure measurement question asked how many smokers were in each household. This question varied between survey cycles and responses were merged between cohorts to create a new variable. The variable describes the overall number of people who smoke inside the respondent’s home as 0, 1, 2, or ≥3.
Data Analysis
Statistical analyses were completed using SAS V. 9.4 (SAS Institute Inc, Cary, North Carolina, USA). To account for oversampling and complex design, analyses used survey weights. Calculated merged cohort weights for five survey cycles were used, as described in NHANES analysis guidelines. Further, weights were selected (either interview or MEC) as appropriate for the variables used in the analysis. Primary sampling units and clusters were also accounted for in analysis. No single variable had more than 10% missing data, and missing data were removed from analysis.
Survey means and standard errors (SE) were calculated for continuous variables. Rao-Scott chi squared analyses were utilized for categorical variables. Logistic regression was used to model the associations between cotinine values and smoke exposure and the associations between cotinine and number of household smokers. Regression models controlled for education, age, annual household income, and race/ethnicity. Associations between covariates and outcomes were confirmed through Rao-Scott chi squared as significant in the model (all p<.001). Statistical significance was set at p<0.01.
Results
A sample of 13,396 women ≥ 20 years old were primarily non- Hispanic white (41.7%), some college or an associate’s degree (31.3%), annual household income of more than $20,000 (73.3%), live in a two person household (28.2%), and mean age of 49 years old (Table 1). Smoke exposure was defined as non-smoker, SHSe, or active smoker. Smoking incidence was 80.0% non-smoker, 2.8% SHSe, and 17.1% active smoker. The second smoke-exposure measure found that most women had no smokers in their home (86.5%), followed by one (8.0%), two (4.3%) and three or more (1.2%) household smokers.
|
Demographics (n=13,396) |
|
|
Age |
|
|
M=49.3 SE=0.15 |
95% CI (49.01-49.61) |
|
Education |
|
|
|
Frequency Percent |
|
<9th grade |
1438 10.7 |
|
9-11th grade |
1867 13.9 |
|
High school graduate, GED or equivalent |
2869 21.4 |
|
Some college or associate’s degree |
4196 31.3 |
|
College graduate |
3026 22.6 |
|
Race/Ethnicity |
||
|
Mexican American |
2085 |
15.6 |
|
Other Hispanic |
1542 |
11.5 |
|
Non-Hispanic White |
5586 |
41.7 |
|
Non-Hispanic Black |
2744 |
20.5 |
|
Other race-including multiracial |
1439 |
10.7 |
|
Annual Household Income |
||
|
<$20,000 |
3524 |
23.2 |
|
$20,000 or greater |
9815 |
73.3 |
|
Don’t know/refused |
473 |
3.5 |
|
Total number in household |
||
|
1 |
1977 |
14.8 |
|
2 |
3771 |
28.2 |
|
3 |
2423 |
18.1 |
|
4 |
2195 |
16.4 |
|
5 |
1487 |
11.1 |
|
6 |
758 |
5.7 |
|
7 or more people in household |
785 |
5.9 |
Table 1
Biochemical: Mean Cotinine. The weighted mean cotinine level for all women was 43.21 ng/ml. There was an upward trend in mean cotinine values comparing self-reported non-smokers, SHSe, and active smokers. A similar upward trend in mean serum cotinine was demonstrated as the number of household smokers increased
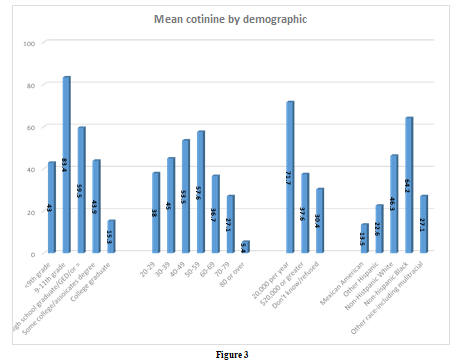
(Figures 1 & 2). Women with the highest mean cotinine levels were non-Hispanic Black (64.19ng/ml), 9th–11th grade education (83.42ng/ml), annual household income of <$20,000 per year (71.66ng/ml), and were aged 50–59 (57.56ng/ml) (Figure 3).
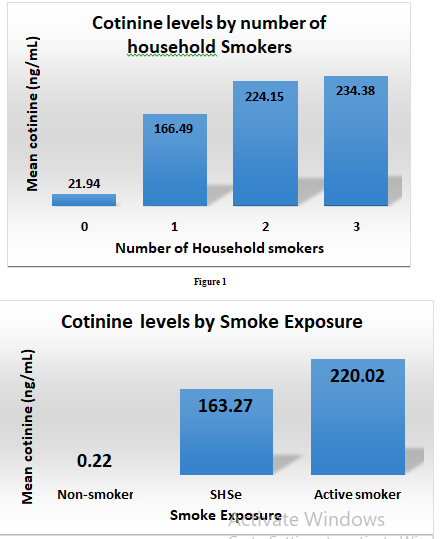
Self-report: Household smokers. The number of household smokers was significantly related to income, education, and race/ethnicity. Considering household smokers, women with annual household incomes <$20,000 were far more likely to live with one, two, or three or more smokers than their higher-income counterparts (Rao Scott χ2=171.51, 6 DF, p<.0001). Women who had a 9th–11th grade education had the highest number of household smokers, and there was an inverse trend between education and number of household smokers (Rao Scott χ2[12df, N=13,396] =315.75, p<.0001). One exception to this trend was women with education levels of less than 9th grade. These women had household smoker numbers similar to women with some college or an associate’s degree. Non-Hispanic Black women had the highest number of household smokers, followed by non-Hispanic white women (Rao Scott χ2=192.3, 12 DF, p<.0001) (Table 2).
|
Variable |
Number |
of household smokers |
|
Race/ethnicity |
0 |
1 2 3 |
|
Mexican American |
93.88% |
4.46% 1.55% 0.11% |
|
Other Hispanic |
93.87% |
3.76% 2.16% 0.21% |
|
Non-Hispanic White |
87.24% |
6.65% 4.77% 1.34% |
|
Non-Hispanic Black |
78.47% |
13.86% 6.08% 1.59% |
|
Other/includes multiracial |
91.91% |
4.75% 2.56% 0.78% |
|
|
|
Rao Scott χ2=192.3,12 DF, p<.0001 |
|
Education |
||
|
<9th grade |
87.81% |
6.78% 4.01% 1.40% |
|
9-11th grade |
73.65% |
13.09% 10.49% 2.78% |
|
High school/GED or = |
81.37% |
9.95% 6.97% 1.71% |
|
Some college/Associates |
88.01% |
7.01% 3.66% 1.24% |
|
College graduate |
96.14% |
2.66% 1.13% 0.08% |
|
|
|
Rao Scott χ2=315.75,12 DF, p<.0001 |
|
Annual household income |
||
|
<$20,000 |
77.90% |
13.51% 7.08% 1.51% |
|
$20,000 or greater |
89.27% |
5.74% 3.88% 1.10% |
|
Don’t know/refused |
90.77% |
6.15% 2.01% 1.07% |
|
|
|
Rao Scott χ2=171.51, 6 DF, p<.0001 |
Table 2
Self-report: Smoke exposure. Using the same demographic measures, there were similar findings in smoke-exposure prevalence. Women who had annual household incomes of
<$20,000 were more likely to be SHSe and active smokers (Rao Scott χ2=284.06, 4 DF, p<.0001). Additionally, women who had a 9th–11th grade education had the highest levels of active smoking and SHSe (Rao Scott χ2=356.84, 8 DF, p<.0001). Non-Hispanic white and Black women were the most likely to be active smokers, and non-Hispanic Black women were nearly three times more likely to be SHSe than the next highest racial/ethnic group (Rao Scott χ2=123.70, 8 DF, p<.0001) (Table 3).
Regression. Using logistic regression to analyze cotinine values by number of household smokers, and controlling for education, age, race, and annual household income, there was a 92.19 ng/ mL increase in cotinine value per household smoker (p<.0001). In this model, education, age, race/ethnicity, and annual household income were significantly related to serum cotinine values (all p<.0001) (Table 4). Race, in particular, was significantly related to cotinine values, with the highest level occurring in non-Hispanic white (t=33.85) and non-Hispanic Black (t=35.10) women compared to their Mexican-American counterparts (all p<.0001, 46df). Mexican-American women were chosen as the reference because they had the lowest cotinine values.
|
Variable |
Smoke exposure |
|
|
Race/ethnicity |
Non-Smoker |
SHSe Active Smoker |
|
Mexican American |
88.47% |
2.13% 9.40% |
|
Other Hispanic |
85.99% |
1.90% 12.10% |
|
Non-Hispanic White |
78.92% |
2.27% 18.81% |
|
Non-Hispanic Black |
73.08% |
6.18% 20.74% |
|
Other/includes multiracial |
84.44% |
1.61% 13.94% |
|
|
|
Rao Scott χ2=123.70, 8 DF, p<.0001 |
|
Education |
||
|
<9th grade |
82.62% |
2.59% 14.78% |
|
9-11th grade |
64.47% |
3.58% 31.95% |
|
High school/GED or = |
72.92% |
3.45% 23.63% |
|
Some college/Associates |
78.37% |
2.95% 18.68% |
|
College graduate |
91.39% |
1.37% 7.24% |
|
|
|
Rao Scott χ2=356.84, 8 DF, p<.0001 |
|
Annual Household Income |
||
|
<$20,000 |
68.26% |
3.38% 28.36% |
|
$20,000 or greater |
81.95% |
2.46% 15.58% |
|
Don’t know/refused |
84.76% |
3.62% 11.62% |
|
Rao Scott χ2=284.06, 4 DF, p<.0001 |
||
Table 3
|
Cotinine by smoke exposure |
||||
|
|
Point estimate |
SE |
t value |
p value |
|
Intercept |
-11.528710 |
3.99737881 |
-2.88 |
0.0060 |
|
Smoke exposure |
||||
|
Non-smoker |
0.000000 |
0.000000 |
|
|
|
SHSe |
163.997826 |
8.83464975 |
18.56 |
<.0001 |
|
Active smoker |
218.350404 |
2.91930070 |
74.80 |
<.0001 |
|
Education |
||||
|
<9th grade |
0.000000 |
0.000000 |
|
|
|
9th-11th grade |
-1.742275 |
2.84434387 |
-0.61 |
0.5432 |
|
High school or equivalent |
-8.722539 |
3.35150012 |
-2.60 |
0.0124 |
|
Some college or Associates |
-11.713259 |
3.13300204 |
-3.74 |
0.0005 |
|
College graduate |
-12.699196 |
2.81545125 |
-4.51 |
<.0001 |
|
Age
Race/ethnicity |
0.28218 |
0.031649 |
8.92 |
<.0001 |
|
Mexican American |
0.000000 |
0.000000 |
|
|
|
Other Hispanic |
4.972023 |
1.30216833 |
3.82 |
0.0004 |
|
Non-Hispanic white |
14.060002 |
1.51613789 |
9.27 |
<.0001 |
|
Non-Hispanic black |
21.257919 |
2.06706848 |
10.28 |
<.0001 |
|
Other race includes multiracial |
8.461178 |
2.34085474 |
3.61 |
0.0007 |
|
Annual household income |
-1.908905 |
2.06671348 |
-0.92 |
0.3605 |
|
Note: The degrees of freedom for the t test is 46 |
||||
Table 4
A second regression analyzing cotinine values by smoke-exposure status, controlling for the same covariates, found a 164.00 ng/mL increase in serum cotinine values between non-smokers and SHSe. Additionally, a 218.35 ng/mL increase in cotinine levels was demonstrated between non-smokers and active smokers (all 46df, p<.0001). In this model, education, age, and race/ethnicity were significantly related to serum cotinine (all p<.0001). Additionally, non-Hispanic white (t=14.06) and non-Hispanic Black (t=21.26) women had the highest levels of serum cotinine levels (all p<.0001, 46df) (Table 5).
|
Cotinine by number of household smokers |
||||
|
|
Point estimate |
SE |
t value |
p value |
|
Intercept |
45.116258 |
6.03128776 |
7.48 |
<.0001 |
|
Number of household smokers
Education |
92.187125 |
6.42188386 |
14.36 |
<.0001 |
|
<9th grade 9th-11th grade High school or equivalent Some college or Associates College graduate |
0.000000
7.887169
-5.578904
-12.013380
-28.552456 |
0.00000000
4.88461785
4.48405811
4.15370101
3.18505064 |
1.61
-1.24 -2.89 -8.96 |
0.1132
0.2197 0.0058 <.0001 |
|
Age
Race/ethnicity |
-0.280903 |
0.0517819 |
-5.42 |
<.0001 |
|
Mexican American Other Hispanic Non-Hispanic white Non-Hispanic black Other race includes multiracial |
0.000000
11.917934
33.849662
35.102772
22.503018 |
0.00000000
2.80748867
3.34069171
5.30968954
4.74425155 |
4.25
10.13 6.61 4.74 |
0.0001
<.0001 <.0001 <.0001 |
|
Annual household income |
-12.878587 |
2.86049766 |
-4.50 |
<.0001 |
|
Note: The degrees of freedom for the t test is 46 |
||||
Table 5
Discussion
This study demonstrated a strong relationship between self- reported smoke exposure and serum cotinine values. Both self-report questions regarding smoking status and number of household smokers were verified biochemically through mean serum cotinine values. These serve as a good proxy for active and SHSe assessment when biochemical validation is not present.
This study confirms previously identified risk factors for increased smoke exposure, including income, race/ethnicity, and education. Additionally, SHSe was nearly three times higher among non- Hispanic Black women (6.2%) than the next highest racial/ethnic group (2.3%) and represents a significant finding. Non-Hispanic Black women may be a population of interest on which to focus smoking and smoke exposure reduction campaigns.
This study also found higher rates of one, two, and three or more household smokers among non-Hispanic white and Black women. The prevalence was particularly high among non-Hispanic Black women with one or two household smokers. Living with a smoker is a potent predictor of subsequent lifetime tobacco use and is an important consideration for all individuals living with a smoker [38]. Again, this demonstrates an assessment and intervention opportunity among this subpopulation of women.
Women with annual household incomes less than $20,000 had higher levels of active smoking and SHSe than women with higher incomes. Low income was a powerful predictor of active smoking and SHSe in women. Many studies have indicated the relationship between income and active smoking and SHSe; however, this study demonstrates the relationship, specifically in women. A large meta-analysis of men and women found a significant inverse relationship between cigarette smoking and income level [39]. Another study of men and women found dose of active smoking was significantly related to income level. This study found incomes of light smokers were $10,000 more annually than heavy smokers [40]. Overall, smoking initiation and cessation are closely tied to income. The lowest cessation rates across all income cohorts are among individuals living below the poverty level. Further, the lowest smoking initiation rates are among individuals with incomes ≥400% above the poverty level [41]. Income plays a significant role in the risk both of active smoking and of SHSe among women.
Controlling for race/ethnicity, income, education, and age, there was an increase in serum cotinine levels commiserate with self- reported smoke exposure. This relationship has previously been documented in the literature among both genders [42-44]. This analysis provides some level of cross validation, within these NHANES cohorts, that women self-reporting smoke exposure have serum cotinine levels that correspond with such reporting. Using the smoke-exposure discovery method of “number of household smokers” provided the most detailed picture of cotinine levels based on self-reported exposure. This may be the ideal measure when considering household smoke exposure using self-report. These data are unique and fill a demonstrated gap in analysis of smoke exposure, specifically in women.
Despite these findings, there are continued problems with measuring SHSe. These issues persist both within and outside the NHANES data. Because long-term SHSe may be a clinically significant factor in disease yet is difficult to assess, SHSe measurement remains a problem. Prior to 2013–2014, NHANES only assessed SHSe through number of household smokers. This proxy measures current SHSe but not long-term. Long-term measures might include antenatal, childhood, or previous, but not current SHSe. Examples may be a parent who smoked, a mother who smoked while pregnant, or long-term exposure from a housemate that is not currently occurring. NHANES does ask a question about antenatal smoke exposure, but this has very poor response rates [30]. Understanding and mitigating low response rates, as well as considering assessment of childhood and other long-term SHSe methods, are important pieces of SHSe assessment. With the emergence and understanding of epigenetic influences and genetic plasticity at differing intervals in life, it is important to better assess the role of smoke exposure throughout the lifespan [45].
Limitations
This study is limited by the constraints of self-report data. To address this concern, the study used dual reporting measures, including cotinine levels. Additionally, it does not assess longitudinal or historical smoke exposure or smoke exposure outside the home. This question was added to NHANES, as discussed above, yet has not been demonstrated to have high response rates; therefore, issues surrounding long-term measures persist. There are standard concerns regarding long-term recall of childhood incidents and exposures that continue to make this limitation difficult to address.
Implications
The identified trends of increased active smoking and SHSe in some women are concerning—specifically, the high prevalence of active smoking among non-Hispanic Black and white women and low-income women. Most worrisome is the significantly higher prevalence of SHSe among non-Hispanic Black women. Higher rates of active smoking and smoke exposure among some minority and low-income women may further exacerbate health disparities that already exist within these populations. It is imperative to identify and implement targeted smoking cessation and smoke exposure reduction interventions.
When considering possible interventions, the available data indicates that smoking bans are the most effective in reducing active smoking and exposure among women [46]. Further, a large study reviewing National Survey of Drug Use and Health (2006–2013) found decreased rates of smoking cessation among non-Hispanic Black girls as they grew older, compared to other races/ethnicities [47]. Given the increased prevalence of active smoking and SHSe among non-Hispanic Black women, targeted cessation and exposure reduction may be ideal. New calls have arisen to consider female-tailored smoking cessation interventions, including motivational, contextual, and harm-reduction methods [48,49]. When considering possible interventions, it is important to ensure that each is gender specific and clinically appropriate. Measuring SHSe continues to be difficult. Starting with the 2013– 2014 survey cycles, NHANES began collecting more detailed information about SHSe at work, in cars, and in social situations. This type of analysis has been recommended in the literature [27]. Once data from more recent NHANES cycles are released, it would be useful to analyze those data that provide more detailed smoke exposure data in women.
Much of what is known about smoking and SHSe prevalence is primarily gender blind [16]. This is one of only a few studies to examine US women’s smoke exposure with both biochemical and self-report measures. Smoke exposure, both active and SHSe, remains a significant problem for women in the US. Low-income and non-Hispanic white and Black women are at highest risk for active smoking and SHSe. This study also provides an introductory comparative analysis of NHANES self-report smoking data and biochemical testing. In particular, this study identified an increase in mean cotinine levels with each additional household smoker. The number of household smokers may be a simple way to grossly analyze dose of smoke exposure in the home. Dose of smoke exposure may be a clinically important assessment tool and best practices should be examined further. Despite the SHSe assessment barriers described above, it is critically important to understand the role of smoke exposure and disease. NHANES provides a good opportunity to analyze these relationships.
References
1.Dai, X., ... & Gakidou, E. (2024). Health effects associated with exposure to secondhand smoke: a Burden of Proof study. Nature medicine, 30(1), 149-167.
2.US Department of Health and Human Services. (2014). In: National Center for Chronic Disease Prevention and Health Promotion (US) Office on Smoking and Health. The Health Consequences of Smoking—50 Years of Progress: A Report of the Surgeon General.
3. Yao, T., Sung, H. Y., Wang, Y., Lightwood, J., & Max, W. (2018). Healthcare costs attributable to secondhand smoke exposure at home for US adults. Preventive medicine, 108, 41-46.
4. Hou, W., Chen, S., Zhu, C., Gu, Y., Zhu, L., & Zhou, Z. (2023). Associations between smoke exposure and osteoporosis or osteopenia in a US NHANES population of elderly individuals. Frontiers in endocrinology, 14, 1074574.
5. Office, U. S. P. H. S. (2020). The Health Benefits of Smoking Cessation. In Smoking Cessation: A Report of the Surgeon General [Internet]. US Department of Health and Human Services.
6. Okoli, C. T., & Kodet, J. (2015). A systematic review of secondhand tobacco smoke exposure and smoking behaviors: Smoking status, susceptibility, initiation, dependence, and cessation. Addictive behaviors, 47, 22-32.
7. Higgins, S. T. (2024). Behavior change, health, and health disparities 2024: Smoking and other tobacco use among women and girls. Preventive Medicine, 188, 108155.
8. Cepeda-Benito, A., Doogan, N. J., Redner, R., Roberts, M. E., Kurti, A. N., Villanti, A. C., ... & Higgins, S. T. (2018). Trend differences in men and women in rural and urban US settings. Preventive medicine, 117, 69-75.
9. Doogan, N. J., Roberts, M. E., Wewers, M. E., Stanton, C. A. , Keith, D. R., Gaalema, D. E., ... & Higgins, S. T. (2017). A growing geographic disparity: rural and urban cigarette smoking trends in the United States. Preventive medicine, 104, 79-85.
10. Higgins, S. T., Erath, T., & Chen, F. F. (2024). Examining US disparities in smoking among rural versus urban women of reproductive age: 2002–2019. Preventive Medicine, 185, 108054.
11. Nighbor, T. D., Doogan, N. J., Roberts, M. E., Cepeda-Benito, A., Kurti, A. N., Priest, J. S., ... & Higgins, S. T. (2018). Smoking prevalence and trends among a US national sample of women of reproductive age in rural versus urban settings. PloS one, 13(11), e0207818.
12. Kurti, A. N., Redner, R., Lopez, A. A., Keith, D. R., Villanti, A. C., Stanton, C. A., ... & Higgins, S. T. (2017). Tobacco and nicotine delivery product use in a national sample of pregnant women. Preventive medicine, 104, 50-56.
13. Lopez, A. A., Redner, R., Kurti, A. N., Keith, D. R., Villanti, A. C., Stanton, C. A., ... & Higgins, S. T. (2018). Tobacco and nicotine delivery product use in a US national sample of women of reproductive age. Preventive Medicine, 117, 61-68.
14. Hinds, J. T., Russell, S. T., & Weinberger, A. H. (2024). Smoking cessation among sexual minority women: Differences in cigarette quit ratios across age, race/ethnicity, and sexual orientation. Preventive Medicine, 188, 108035.
15. Agaku, I. T., Odani, S., Okuyemi, K. S., & Armour, B. (2020). Disparities in current cigarette smoking among US adults, 2002–2016. Tobacco control, 29(3), 269-276.
16. Amos, A., Greaves, L., Nichter, M., & Bloch, M. (2012). Women and tobacco: a call for including gender in tobacco control research, policy and practice. Tobacco control, 21(2), 236-243.
17. Andrews, J. O., Mueller, M., Dooley, M., Newman, S. D., Magwood, G. S., & Tingen, M. S. (2016). Effect of a smoking cessation intervention for women in subsidized neighborhoods: a randomized controlled trial. Preventive medicine, 90, 170-176.
18. Nguyen-Grozavu, F. T., Pierce, J. P., Sakuma, K. L. K., Leas, E. C., McMenamin, S. B., Kealey, S., ... & Trinidad, D. R. (2020). Widening disparities in cigarette smoking by race/ethnicity across education level in the United States. Preventive medicine, 139, 106220.
19. Jiang, H., Livingston, M., Room, R., Gan, Y., English, D., & Chenhall, R. (2019). Can public health policies on alcohol and tobacco reduce a cancer epidemic? Australia's experience. BMC medicine, 17, 1-14.
20. Johnson, K. C., Miller, A. B., Collishaw, N. E., Palmer, J. R., Hammond, S. K., Salmon, A. G., ... & Turcotte, F. (2011). Active smoking and secondhand smoke increase breast cancer risk: the report of the Canadian Expert Panel on Tobacco Smoke and Breast Cancer Risk (2009). Tobacco control, 20(1), e2-e2.
21. Huxley, R. R., & Woodward, M. (2011). Cigarette smoking as a risk factor for coronary heart disease in women compared with men: a systematic review and meta-analysis of prospective cohort studies. The Lancet, 378(9799), 1297-1305.
22. Dalmau, R. (2021). Women and tobacco, a gender perspective. E-Journal of Cardiology Practice, 20(3).
23. Stapelfeld, C., Dammann, C., & Maser, E. (2020). Sexâ? specificity in lung cancer risk. International journal of cancer, 146(9), 2376-2382.
24. Bonello, K., Figoni, H., Blanchard, E., Vignier, N., Avenin, G., Melchior, M., ... & Ibanez, G. (2023). Prevalence of smoking during pregnancy and associated social inequalities in developed countries over the 1995–2020 period: A systematic review. Paediatric and Perinatal Epidemiology, 37(6), 555- 565.
25. Bar-Zeev, Y., & Solt, I. (2018). Interventions for supporting women to stop smoking in pregnancy. Harefuah, 157(12), 783-786.
26. Wang, X., Lee, N. L., & Burstyn, I. (2023). Smokers’ utilization of quitting methods and vaping during pregnancy: an empirical cluster analysis of 2016–2018 Pregnancy Risk Assessment Monitoring System (PRAMS) data in seven US states. BMC pregnancy and childbirth, 23(1), 306.
27. Chapman Haynes, M., St. Claire, A. W., Boyle, R. G., & Betzner, A. (2018). Testing and refining measures of secondhand smoke exposure among smokers and nonsmokers. Nicotine and Tobacco Research, 20(2), 199-205.
28. Merianos, A. L., Mahabee-Gittens, E. M., Stone, T. M., Jandarov, R. A., Wang, L., Bhandari, D., ... & Matt, G. E. (2023). Distinguishing exposure to Secondhand and Thirdhand Tobacco smoke among US children using machine learning: NHANES 2013–2016. Environmental science & technology, 57(5), 2042-2053.
29. Ko, K., Kwon, M. J., Yang, S. H., Moon, C. J., Lee, E. H.,
Woo, H. Y., & Park, H. (2016). Evaluation of serum cotinine cut-off to distinguish smokers from nonsmokers in the Korean population. Annals of Laboratory Medicine, 36(5), 427.
30. Centers for Disease Control and Prevention (CDC). (2009). National Health and Nutrition Examination Survey Data. Hyattsville, MD: US Department of Health and Human Services, Centers for Disease Control and Prevention, 2003– 2004.
31. Mason, J., Wheeler, W., & Brown, M. J. (2015). The economic burden of exposure to secondhand smoke for child and adult never smokers residing in US public housing. Public Health Reports, 130(3), 230-244.
32. Caraballo, R. S., Giovino, G. A., Pechacek, T. F., & Mowery, P. D. (2001). Factors associated with discrepancies between self-reports on cigarette smoking and measured serum cotinine levels among persons aged 17 years or older: Third National Health and Nutrition Examination Survey, 1988– 1994. American journal of epidemiology, 153(8), 807-814.
33. Heller, W. D., Scherer, G., Sennewald, E., & Adlkofer, F. (1998). Misclassification of smoking in a follow-up population study in southern Germany. Journal of clinical epidemiology, 51(3), 211-218.
34. Pirkle, J. L., Flegal, K. M., Bernert, J. T., Brody, D. J., Etzel, R. A., & Maurer, K. R. (1996). Exposure of the US population to environmental tobacco smoke: the Third National Health and Nutrition Examination Survey, 1988 to 1991. Jama, 275(16), 1233-1240.
35. Kim, S. (2019). Overview of cotinine cutoff values for smoking status classification. Neuroscience of Nicotine, 419- 431.
36. Seccareccia, F., Zuccaro, P., Pacifici, R., Meli, P., Pannozzo, F., Freeman, K. M., ... & Giampaoli>, S. (2003). Serum cotinine as a marker of environmental tobacco smoke exposure in epidemiological studies: the experience of the MATISS project. European journal of epidemiology, 18, 487-492.
37. Slattery, M. L., Robison, L. M., Schuman, K. L., French, T. K., Abbott, T. M., Overall, J. C., & Gardner, J. W. (1989). Cigarette smoking and exposure to passive smoke are risk factors for cervical cancer. Jama, 261(11), 1593-1598.
38. O'Loughlin, J., O'Loughlin, E. K., Wellman, R. J., Sylvestre, M. P., Dugas, E. N., Chagnon, M., ... & McGrath, J. J. (2017). Predictors of cigarette smoking initiation in early, middle, and late adolescence. Journal of Adolescent Health, 61(3), 363- 370.
39. Casetta, B., Videla, A. J., Bardach, A., Morello, P., Soto, N., Lee, K., ... & Ciapponi, A. (2017). Association between cigarette smoking prevalence and income level: a systematic review and meta-analysis. Nicotine & Tobacco Research, 19(12), 1401-1407.
40. Chen, A., Machiorlatti, M., Krebs, N. M., & Muscat, J. E. (2019). Socioeconomic differences in nicotine exposure and dependence in adult daily smokers. BMC Public Health, 19, 1-10.
41. Jeon, J., Cao, P., Fleischer, N. L., Levy, D. T., Holford, T. R., Meza, R., & Tam, J. (2023). Birth cohortâ??specific smoking patterns by family income in the US. American journal of preventive medicine, 64(4), S32-S41.
42. Braverman Bronstein, A., Lomelín Gascón, J., Eugenio González, C. I., & Barrientos-Gutierrez, T. (2018). Environmental tobacco exposure and urinary cotinine levels in smoking and nonsmoking adolescents. Nicotine and Tobacco Research, 20(4), 523-526.
43. Lindsay, R. P., Tsoh, J. Y., Sung, H. Y., & Max, W. (2016). Secondhand smoke exposure and serum cotinine levels among current smokers in the USA. Tobacco control, 25(2), 224-231.
44. Zhang, Z., Li, Z., Zhang, X., Ye, W., Chen, J., Wang, L., ... & Li, Z. (2023). Association between secondhand smoke and cancers in adults in the US population. Journal of Cancer Research and Clinical Oncology, 149(7), 3447-3455.
45. Feinberg, A. P. (2007). Phenotypic plasticity and the epigenetics of human disease. Nature, 447(7143), 433-440.
46. Bird, Y., Kashaniamin, L., Nwankwo, C., & Moraros, J. (2020, January). Impact and effectiveness of legislative smoking bans and anti-tobacco media campaigns in reducing smoking among women in the US: a systematic review and meta-analysis. In Healthcare (Vol. 8, No. 1, p. 20). MDPI.
47. Thompson, A. B., Mowery, P. D., Tebes, J. K., & McKee, S. A. (2018). Time trends in smoking onset by sex and race/ ethnicity among adolescents and young adults: findings from the 2006–2013 National Survey on Drug Use and Health. Nicotine and Tobacco Research, 20(3), 312-320.
48. Jayakumar, N., Chaiton, M., Zhang, B., Selby, P., & Schwartz, R. (2020). Sex differences in use of smoking cessation services and resources: a real-world study. Tobacco Use Insights, 13, 1179173X20901500.
49. Lee, A., Lee, K. S., Lee, D., Ahn, H., Lee, H. K., Kim, H., ... & Seo, H. G. (2021). The utilization of national tobacco cessation services among female smokers and the need for a gender- responsive approach. International Journal of Environmental Research and Public Health, 18(10), 5313.



