Review Article - (2019) Volume 1, Issue 1
Role of Prostaglandins, Thromboxanes and Leukotrienes as Mediators in Inflammations
Received Date: Nov 02, 2019 / Accepted Date: Nov 08, 2019 / Published Date: Nov 17, 2019
Copyright: ©Eman Refaat Youness. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Eicosanoids are 20-carbon bioactive lipids derived from the polyunsaturated fatty acids metabolism, which can modify numerous biological processes comprising cell proliferation, migration and adhesion, vascular permeability, angiogenesis and inflammatory responses [1].
Introduction
Eicosanoids are 20-carbon bioactive lipids derived from the polyunsaturated fatty acids metabolism, which can modify numerous biological processes comprising cell proliferation, migration and adhesion, vascular permeability, angiogenesis and inflammatory responses [1].
Eicosanoid (from the Greek, eicosa for twenty) and eicosanoid-like molecules (communally labelled ‘eicosanoids’) denote small polar lipid metabolites created via comprehensive and variable oxidation of mainly 18 to 22 carbon polyunsaturated fatty acids (PUFA’s), comprising omega-6 fatty acids, like arachidonate, adrenic acid, &linoleic acid, & omega-3 fatty acids, such as docosapenaenoic acid (DPA), docosahexaenoic acid (DHA) & eiocosapentaenoic acid (EPA) [2].
Eicosanoids form one of the most complicated networks in the body & regulate numerous pathological & physiological processes, predominantly in different directions. They modify biological responses as vascular homeostasis, gastrointestinal integrity, platelet aggregation and kidney perfusion, and are also extremely implicated in the autoimmunity regulation, cancer, & inflammation. The arachidonic acid metabolites have duple roles in inflammation, anti-inflammatory & pro-inflammatory, and their production effect depending on several factors. Among the pro-inflammatory eicosanoids, prostaglandin E2 (PGE2) is considered a key mediator of different parts of inflammation, involving fever, inflammatory pain and swelling [3]. Thromboxane A2 (TXA2) and prostaglandin I2 (PGI2, known also as prostacyclin) control platelet aggregation, & leukotriene B4 (LTB4) is one of the most potent chemotactic agents. Whereas cysteinyl leukotrienes amend smooth muscle & vascular responses. Another eicosanoids are embroiled in the resolution phase of inflammation, as lipoxin A4 (LXA4) & cyclopentenone 15-deoxy Δ12, 14-prostaglandin J2 (15d-PGJ2), instead, they have been shown to supress inflammatory processes [4].
Eicosanoid Synthesis
Since the human body lacks the enzymes needed to synthesize the polyunsaturated fatty acids (PUFAs), α-linolenic (ALA) and linoleic acids (LA), diet is its only source. The rate of conversion of (PUFAs) into DHA (docosahexaenoic acid), eicosapentaenoic acid (EPA) and arachidonic Acid (AA), is decelerated because of the decreased activity of the Δ6 desaturase. That is why only 0.2–2% of the dietary ALA is converted into DHA and EPA, while the rest undergoes β-oxidation [5]. To bypass this step towards eicosanoid production, the simplest means is to increase the dietary intake of EPA and DHA. Cells also utilize PUFAs for de novo synthesize glycerophospholipids through the lysophospholipids remodeling or Kennedy pathway in the Lands cycle [6]. Glycerophospholipids are one of the components of the cell membranes and augment their properties and functions. The next step in eicosanoid synthesis is catalyzed by the phospholipase A2 (PLA2) with ATP and calcium ions as cofactors. This includes the hydrolysis at the sn-2 position of glycerophospholipids. The results of this reaction are free fatty acids and lysophospholipids [7].
Eicosanoid metabolites are resulting from the metabolism of arachidonic acid throughout three main branches: cytochrome P450monooxygenase, cyclooxygenase (COX), and the lipoxygenase (LOX) branches. Each of these branches results in specific eicosanoid metabolites having dissimilar biological effects [8] (Fig. 1).
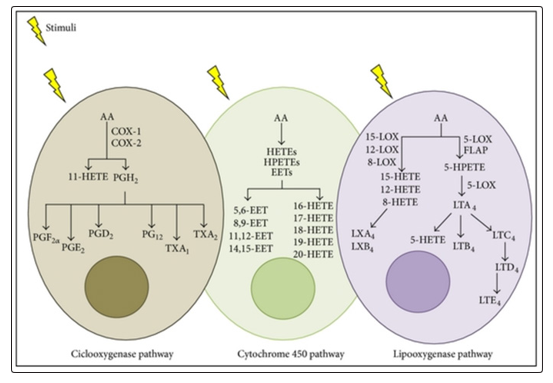
Figure 1: Eicosanoid synthesis pathways. Following cell stimulation, arachidonic acid (AA) could be metabolized via three enzymes:lipoxygenase (LOX), cyclooxygenase(COX), & cytochrome P450 (CYP 450). COX catalyzes AA in (prostaglandin) PGG2 & PGH2, & these areconverted into PGE2, PGD2, PGF2α, PG12, TXA1, & TXA2. The LOX pathway catalyzes AA into hydroxyl eicosatetraenoic acids (HETEs) & oppose hydro-peroxy-eicosatetraenoic acids (HPETEs).This pathway includes four
enzymes: 5-LOX, 8-LOX, 12-LOX, and 15-LOX. 5-LOXreacts with a 5-LOX-activating protein (FLAP), stimulating the binding of 5-LOX to AA. LTA4 hydrolases transform LTA4 into LTB4, and LTC4 synthase can convert LTA4 to LTC4, then it is then metabolized to LTE4and LTD4. 5-LOX synthetizes LXA4 & LXB4 using 15-HETE. CYP-450 pathway leads to the change of HETEs,including 16-, 17-, 18-, 19-, & 20-HETE and epoxy-eicosatrienoicacids(EETs): 5,6-, 8,9-, 11,12-, & 14,15-EET.
https://www.google.com/imgres?imgurl=https://www.researchgate. net/profile/Thais_Ayala/publication/273776955/figure/fig2/ AS:324638992289798@14544113
Because of external conversion of omega 6 and omega 3 families of fatty acids such as EPA, arachidonic acid (AA), DHA get produced, which are precursors of mediators of a group of compounds that have physiological significance (Fig.2). Eicosanoids as leukotrienes & prostaglandins are products of the AA & EPA metabolism. This influenced by cyclooxygenase (COX) leads to conversion into prostaglandin E2 that is an inflammatory mediator, prostacyclin I2 (PGI2), responsible for blood vessel dilation and thromboxane A2 (TXA2), vasospasm and activating blood platelet aggregation. Due to lipoxygenase (LOX) effect four leukotrienes series are formed which have an imperativerole in the development and maintenance of the inflammatory response. EPA (C20:5ω-3), get mobilized in a parallel behavior with the participation of the same enzymes i.e. lipoxygenase and cyclooxygenase, but its metabolic products are dissimilar i.e. series 3 of prostanoids and series 5 of leukotrienes of dissimilar properties mostly anti-inflammatory (PGE3, LTA5, LTB5, LTC5, LTD5), anti-aggregatory (TXA3) and vasodilative (PGI3) [9].
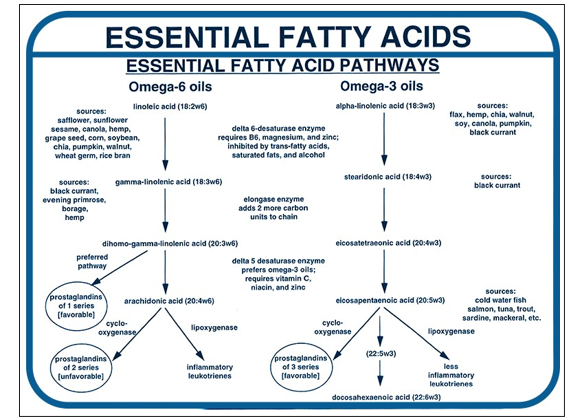
Figure 2: Metabolic pathway of a polyunsaturated fattyacid omega-6 and omega-3 series
http://www.healthproductsdistributors.com/wp/wp-content/ uploads/2017/12/FattyAcidDiagrams-for-blog-2.png
Biosynthesis of Prostanoids
Prostanoid embrace prostaglandins (PGs); PGD, PGE and PGF), thromboxane (TXA) and prostacyclin (PGI). These lipids are synthesized from the polyunsaturated fatty acids (PUFAs) dihomo-gamma-linolenic acid (DGLA, precursor of series 1 prostanoids), arachidonic acid (AA, precursor of series 2 prostanoids) & eicosapentaenoic acid (EPA, precursor of series 3 prostanoids). Among these precursors, AA is the most imperative and dominant in humans [10].
Prostanoids are universal lipids in animal tissues and organize a plenty of pathological and physiological processes, either within the cells in which they are made or in closely neighboring cells in response to certain stimuli. Under normal physiological conditions, prostanoids are entangled in the contraction & relaxation of smooth muscles, modulation of immune responses and regulation of blood clotting. Also implicated in maintenance of renal homeostasis, stimulation, inhibition of neurotransmitter release, regulation of gastrointestinal tract motility & secretion and protection of the gastrointestinal mucosa. Prostanoids are also implicated in many pathological conditions, as cancer, inflammation and cardiovascular disease [1].
Prostaglandins (PGs) were discovered in 1935 as a blood-pressure-lowering matter from the prostate gland secretion. Von Euler found that seminalvesicles and seminal fluid from many animals including men comprise a material that causes contraction of the smooth muscle of the uterus. He entitled this new substance as “prostaglandins” since they were initially supposed to be secreted from prostate gland [11]. In 1976, prostacyclin was discovered as a powerful repressor of function of platelet & as arobust vasodilator. PG12 & PGE1 together are compounds of endogenous source and their activities spread out through reacting via the same surface receptor. Their pharmacological characteristics were the motive that PGE1 act as the initial PG has been extensively used since 1973 for cardiovascular diseases treatment, generally in patients with superior peripheral vascular disease.
The cyclic pathway started via the prostaglandin synthase action, PGS (in addition named prostaglandin-endoperoxide synthetase). The enzyme retains dual actions, peroxidase & cyclooxygenase (COX). Two forms of the COX activity are found in humans, COX-1 and COX-2. PGS-1 (COX-1) is constitutively formed in kidney gastric, platelets, vascular endothelial cells & mucosa. PGS-2(COX-2) is stimulated and expressed in monocytes and macrophages due to inflammation. The first cause for COX-2 stimulation in macrophages & monocytes are interleukin-1 (IL-1) and platelet-activating factor (PAF). COX-1 and COX-2 spur arachidonic acid to be converted into PGG2 then, to PGH2 (Fig.3). The gene encoding COX-1 is specified as the PTGS1 gene and that encoding COX-2 is the PTGS2 gene. The PTGS1 gene is situatedat chromosome 9q33.2 and it is composed of sixteen exons that produce7 alternatively merged mRNAs that collectively encode six protein isoforms. PTGS2 gene lies at chromosome 1q31.1.It comprise 10 exons which encode a precursor protein of 604 amino acids [12].
After synthesis, prostanoids can cross the cell membrane via simple diffusion (poorly, due to their charged nature at physiological pH) or can be imparted out of the cell by members of the ABC transporter superfamily [13]. In the extracellular environment, prostanoids can fasten to their specific receptors for multiple intracellular pathways activation [14]. There are nine diverse receptors for the prostanoids: DP1 and DP2 receptors for PGD2; EP1, EP2, EP3 (splice variants) and EP4 receptors for PGE2; FP receptor for PGF2a; IP receptor for PGI2 & TP receptor for TXA2 [1].
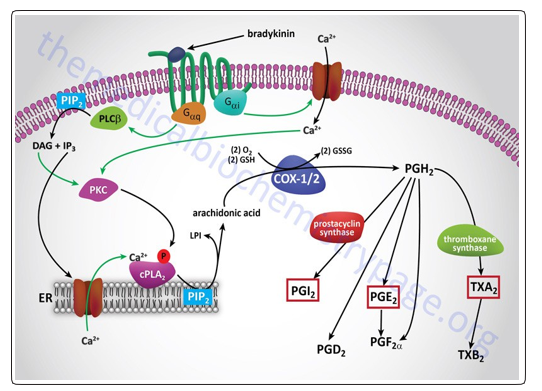
Figure 3: clinically pertinent prostaglandins &thromboxane ssynthesis from arachidonic acid
Plentiful stimuli (e.g. epinephrine, bradykinin & thrombin) stimulate PLA2 that splits arachidonate from cell membrane phospholipids. Receptor of bradykinin binds to Gq G-protein &Gi/0 producing augmention action of internal calcium and stimulation of PKC. Ca2+ ions and PKC phosphorylation activate cPLA2 that, when activated split arachidonate from PIP . Arachidonate is changed into PGH via COX-1 and COX-2 action. Prostacyclin (PGI2) is synthesized from PGH2 through the prostacyclin synthase action. Prostaglandin F synthase 1 changes PGH2 into PGF2α which also changes PGD2 into 9α, 11β-PGF2α, β. Thromboxane (TXA2) resulting from PGH2 through the thromboxane A synthase 1 action. TXB2 outcome from TXA2 degradation & play an important pate in the acetaminophen hepatotoxicity. The 3 mainly eicosanoids that have physiologically important cyclic are presented in boxes in red. Arrows in green indicate +ve effects. The subscript 2 in every particle denotes the number of carbon-carbon double bonds.
https://www.google.com/search?q=&source=lnms&tbm= isch&sa=X&ved=0ahUKEwjChaO12p_fAhWMsqQKHYAPAcgQ_ AUIDygC&biw=1280&bih=640#imgrc=yTDMO2ItA7VlkM
Synthesis of Leukotriene
This pathway begins via the arachidonate lipoxygenases action (LOXs).There are three forms, 12-LOX, 5-LOX and 15-LOX. The executive names for these enzymes are arachidonate 12-lipoxygenase, arachidonate 5-lipoxygenase, and arachidonate 15-lipoxygenase. ALOX5 gene encoded 5-LOX enzyme that is situatedat chromosome 10q11.21 & is contains 14 exons that produce five alternatively spliced mRNAs each of which encode a specific protein isoform. The 12-LOX enzyme is encoded by the ALOX12 gene that is situated at chromosome 17p13.1 & consists of 14 exons which encode a protein of 663 amino acids. ALOX15 gene encodes 15-LOX enzyme that is situatedat chromosome 17p13.2 &consists of 14 exons which encode a protein of 662 amino acids. 5-LOX generates to the leukotrienes. These leukotrienes are synthesized via numerous dissimilar types of cells comprising white blood cells, spleen, mast cells, brain, lung & heart. 15-LOX &12-LOX of activities are implicated in synthesis of lipoxins. (Fig. 4) [12].
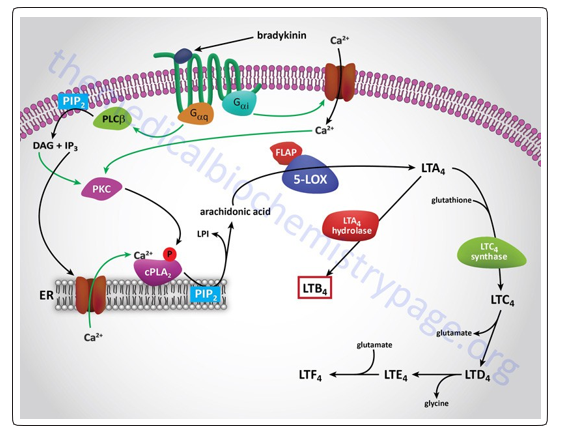
Figure 4: Synthesis of leukotrienes from arachidonic acid
Leukotrienes are known as LT. numerous stimuli (e.g. thrombin, bradykinin & epinephrine) stimulate PLA2 that split arachidonate from cellular membrane phospholipids .Receptor of bradykinin is bound to Gq G-protein activation and Gi/0 & increased intracellular calcium & activation of PKC. Ca2+ ions &PKC phosphorylation activate cPLA2 isoforms that, on activation, dissociates arachidonate from PIP2. 5-lipoxygenase enzyme stimulates the change of arachidonate, into5-hydroperoxy-eicosatetraenoic acid (5-HPETE) that suddenly decreases into 5-hydroxy-eicosatetraenoic acid (5-HETE), then into LTA4. LTA4 is converted to LTB4 in monocytes & neutrophils utilizing LTA4 hydrolase & it are unsettled. LTB4 is surrounded by a red box to show its importance as one of the greatest powerful inflammation-mediating lipids. Leukotrienes LTD4, LTC4, LTF4 andLTE4 are identified as the peptido-leukotrienes or cysteinylleukotrienes due to the presence of amino acids. These peptido-leukotrienes are ingredient of slow-reacting substance of anaphylaxis (SRSA). Arrows in green refer to positive effects. 4 in subscript in each particlerefers to the carbon-carbon double bonds number.
https://www.google.com/h?q=relevant+prostaglandins+and+t hromboxanes+from+arachidonate.&source=lnms&tbm=isch& sa=X&ved=0ahUKEwjChaO12p_fAhWMsqQKHYAPAcgQ_ AUIDygC&biw=1280&bih=640#imgrc=yTDMO2ItA7VlkM:
In inflammation, eicosanoids studies generally concentrate on the pathways of signals triggered via lipids that are generated through the COX enzymes.They collectively educe the signs of inflammation, comprising swelling, pain, redness,loss of function & heat [15].
The physiological role of the referred lipids was understood through the different impacts that they possess invarious tissues: for instance, the attaching of prostaglandin E2 (PGE2) to its related G-protein-coupled receptors (GPCRs), the family of EP receptor, in neurons produce pain accompanied by inflammations, while autocrine EP gesturing via PGE2 in macrophages (might be in another leukocytes) able to down regulate the tumour necrosis factor (TNF) production & up-regulate interleukin-10 production, causing reduction in inflammatory signalling [16].
While COX pathway of eicosanoids regulates an extensive series of processes, 5-LOX pathway is extraparticularly working throughout inflammations to encourage broncho-constriction [17] & leukocyte recruitment to places of tissue damage [18]. While the functions of 5-LOX-derived leukotrienes in allergy & asthma are mentioned, eventual biological assignments for the intermediate metabolites; 8-hydroperoxy - eicosatetraenoic acid (8-HPETE), 15-HPETE & 12-HPETE & their hydroxyl-eicosatetraenoic acid (HETE) yields, have not been identified, howeverothersmight beligands for PPAR-α (peroxisome proliferator-activated receptor-α) [19]& PPAR-γ [21], which encourage anti-inflammatory impacts & amend X receptor in liver (LXR; that controls homeostasis of cholesterol). Therefore, specific LOX enzymes metabolites might be anti-inflammatory, however the correlation among COX &LOX enzymes throughout inflammation embarrass unpretentious strategies to repress individual pathways used for therapeutic gain. In spite of a leading role for numerous of mono-hydroxylated fatty acids derived from LOX, created via a solitary enzyme, plentiful families of more complex tri-&di- hydroxylated fatty acids are generated via COX & LOX enzymes carrying out in grouping; they have amazingly specific & powerful linked to GPCRs which speed neutrophil clearance & clearance of bacterial, this switch on programmes of anti-inflammatory cytokine. The pathway of CYP includes a huge number of enzymes containing a haem iron. Numerous CYPs are synthesized in the liver. Also they are synthesized in other tissues wherever they deactivate and remove toxins & metabolites. In the eicosanoid pathway the most upstream CYPs transform arachidonate into epoxyeicosa-trienoicacids (EETs) or ω-HETE 2, that were assumed to be anti-inflammatory. Whereas the downstream diHETEs(that can be produced by soluble epoxide hydrolase (sEH)) assumed to be pro-inflammatory or inactive.
The systematic description of prostaglandin as well as leukotriene composition, natural receptors, biological functions and biosynthetic pathways has give rise to novel drug categories production in the form of analogues, likewise receptor & enzyme inhibitors. These therapies that target eicosanoids cause significant improvements in treatment of inflammatory symptoms comprising pain & swelling. Many chronic diseases, like atherosclerosis, arthritis & more life-threatening pathogenic diseases chiefly are not affected by the repression of eicosanoids. like numerous supplementary mediators’ derived from PUFAs throughout inflammation & infection have sustained to be known & characterized. Now there is asuperior focus on determining which lipid mediators ameliorate appointed sides of host protection and which mediate homeostasis return. Anti-inflammatory responses modulation, specific mediators of eicosanoid have been correlated with augmenting clearance of pathogen, antibody-mediated immune responses & clearance of neutrophil.
Leukotrienes and prostaglandins are generated rapidly following the beginning of inflammation & encourage the early stimulation of oedema from post-capillary venules. This is naturally occurs to mediate leukocyte & complement enrolment to a spot of acuteinjury, but the eicosanoid-mediated response might be morepowering & life-menacing to the host throughout septic or toxic shock for instance, prostaglandins formed by means of COX1 (also termed PTGS1) during inflammasome stimulation have been recently found to participate in exaggerated vascular leakage & in lethality. They comprise part of a major pro-inflammatory eicosanoid response to infection [21]. Also, binary suppression of COX2 (also termed PTGS2) & 5-LOX along with flavocoxid has been found to ameliorate survival in mice following caecal puncture & ligation, with a diminution in the circulating levels of leukotriene B4(LTB4) and PGE2, an augmentation in lipoxin A4 (LXA4) expression, and a reduction in liver & lung myeloperoxidase (MPO) levels [22].
Factors Influencing COX Activity
NSAIDs suppress eicosanoid synthesis through its action on COX-1 and COX-2. Aspirin differs from other NSAIDs due to its capability to acetylate COX-2 irreversibly and switch this enzyme to generate 15R-HETE instead, a substrate for the 5-LOX. This leads to the synthesis of 15-epi-lipoxinA4, known also as aspirin-triggered lipoxin (ATL) [23]. Celecoxib and rofecoxib reduce the activity of acetylated COX-2 and the synthesis of the anti-inflammatory 15(R)-epi-LXA4; therefore merging them with aspirin significantly augments the risk of the damage of gastric mucosa. These opposing interactions have not been observed following the administration of celecoxib and rofecoxib with the novel acetylsalicylate derivative NCX 4016 [24]. The acetylated COX-2 turn EPA into 18(R)-hydroxy-eicosapentaenoic acid (18R-HEPE) which is then transformed by 5-LOX into resolvin (Rv) E1 and RvE2. RvE1 exerts its anti-inflammatory activity after binding with the leukotriene BLT1 and ChemR23 receptors [25]. The synthesis of D-series Rv may take place via two pathways. One pathway depends on the aspirin-acetylated COX-2 and utilizes DHA as a substrate for 17(R)-hydroxy-DHA which is later converted to D4 by LOX to 17(R)-RvD1, identified as the aspirin-triggered RvD-series (AT-RvDs) [26]. The second pathway is independent of aspirin and produces resolvins similar to those of the first pathway. Clinical studies of the effect of EPA’s and DHA’s on the inflammatory reaction require beholding the fact that statins and aspirin, NSAIDs, all significantly change the profile of eicosanoid. Aspirin (ASA) encouraged acetylation of cyclooxygenase-2 (COX-2). This alters the enzyme specificity varying the result of the catalytic reaction with fatty acids leading to the so-called “aspirin-triggered (AT)” products instead [27] (Fig.5).
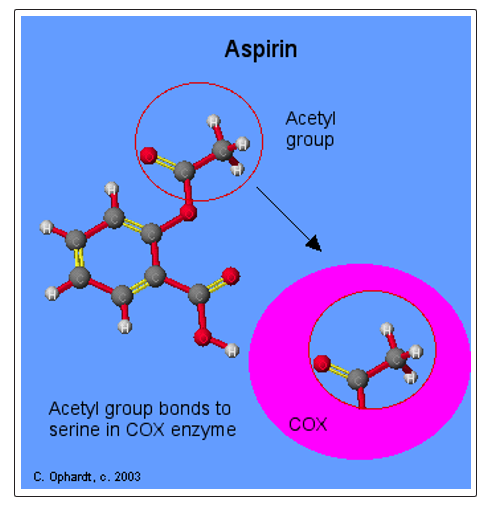
Figure 5: Aspirin induced acetylation of cyclooxygenase-2
http://www.elmhurst.edu/~chm/vchembook/
Factors Influencing the 5-, 12-, and 15-LOX Activity
Leukotrienes (LTs) created by 5-LOX play a key role in the pathogenesis of allergic rhinitis, asthma, and other diseases [28]. Repressors of this enzyme and the cysteinyl leukotriene receptor antagonists (e.g., zafirlukast, montelukast, pabilukast, and pranlukast) are used successfully to avoid the exacerbations of those diseases. The 5-LOX supressor are not useful to treat attacks of asthma because the inhibition of LT synthesis also inhibits the trans-cellular synthesis of the pro-resolving substances like protectins, resolvins, and maresins. Pergola et al. confirmed that the low levels of testosterone in women are the cause for their nearly twice higher level of 5-LOX products than in men. This explains why asthma symptoms control and treatment are more complicated in women than inmen [29].
Fatty Acid Functions
Omega 6 and omega 3 fatty acids play certain functions in the organisms and their neuro-functions are still being discovered. On the basis of unsaturated fatty acids conversions in the human body, their role in forming leukotrienes & prostanoids have been seen.
When they arise from ARA like PGE2, PGD2 or series 4 leukotrienes, they exhibit pro-inflammatory activity which is commonly described and known in the reports that argue the mechanism of non-steroidal anti-inflammatory drug action. Basic studies have revealed that EPA and DHA are useful for the human body applying these biological actions [30].
Effects of PUFA’s on anti-inflammatory activity
Omega 6 and omega 3 PUFA’s are incorporated into the cell membrane.They are released from membrane phospholipids and encompass substrates for eicosanoid synthesis as prostaglandins, thromboxanes, prostacyclins and leukotrienes. Eicosanoids resulting from arachidonic acid (omega 6) induce an inflammatory response by series 2 prostanoid synthesis through PGE2 may also enhance anti-inflammatory effect by increasing lipoxin production via stimulting 15LOX (lipoxygenase) [31].
Eicosanoids derived from arachidonic acid are accountable for vasoconstriction and pro-aggregation effect (TXA2 and TXB2) and proliferation of cancer cells (especially by colorectal, breast, prostate cancers [31]. Five series leukotriene and 3 series prostanoids arising from fatty acids of omega 3 series (mainly from EPA) have weaker inflammation inducing properties. This means that factors prompting infection, impairment or inflammation depends on the ofcell membranes composition. If there are appropriate proportions of omega 3 PUFA’s, the response to inflammatory factors is weaker. Production of resolvins, lipoxins as well as oxylipins from both groups of polyunsaturated fatty acids permit to extinct the on¬going inflammation or extreme tissue damage and development of numerous diseases whose pathogenesis is related to inflammatory diseases [33].
Omega 3 fatty acids derivatives may also have anti-thrombotic activities contradicting blood vessel narrowing and inhibiting carcinogenesis [9]. Omega 3 fatty acids have anti-inflammatory and anti-allergicactivity principally by the inhibition of exaggerated immuneresponse competing for mutual enzymes with omega 6 fatty acids in the metabolic pathway. They diminish the synthesis of pro-inflammatory compounds (PGE2, LTB4, TNF, and IL-1) as well as stimulate the synthesis of cytokines that possess anti-inflammatory actions (IL-2, TGF). Mitigation of inflammatory symptoms has been shown following administration of omega 3 acid preparations in case of autoimmune diseases as ulcerative colitis, rheumatoid arthritis, psoriasis, asthma and other autoimmune diseases. Also some reports found that they may lessen the course of inflammatory processes of the viral or bacterial origin [34] [9].
Ideal Eicosanoids
• Constrain aggregation of platelet -- this declinesthe risk for strokes &. heart attacks
• Reinforce vasodilation-- this diminishes the risk of increasing heart disease & hypertension.
• Prevent cellular proliferation -- this declines the probability of cancer development &other conditions that depending on cells dividing. Also this has several benefits in fighting viral infections comprising HIV. • Encourage immune response -- this helps us fight off viral & bacterial infections.
• Act as anti-inflammatory -- this modulates the mechanisms of inflammatory response in our body that helps avoid and/ or improve diseases as Crohn’s disease, ulcerative colitis & rheumatoid arthritis.
• Reduce pain transmission -- this assists the body overcome illnesses & prevents debilitation resulting from progressive conditions as osteoarthritis.
Detrimental Eicosanoids
• Reinforce aggregation of platelet -- this reinforcesstrokes &heart attacks.
• Encourage vasoconstriction -- this stimulates heart disease & hypertension.
• Help proliferation of cells -- this accelerates the cancer development and spread.
• Suppress immune response -- this minimizes defenses against inflammation & infections.
• Act as pro-inflammatory -- this encourages inflammatory illnesses, like arthritis & other autoimmune diseases.
• Increasing transmission of pain -- this weakens the body and plunders vitality & strength. Methods for estimation of eicosanoids
• Estimation of the selected eicosanoids via reversed-phase HPLC method using fluorescence detection.
• Also, eicosanoidscould be measured via Gas chromatography.
• Eicosanoids can be estimated by enzyme immunoassay (ELISA) kits for the quantitative determination of Prostaglandin E2 in plasma, urine, tissue culture media and other sample matrices.
Conclusions and Recommendation
Conclusions
• Eicosanoids are one of the greatest convoluted networks in our body that regulate several pathologi¬cal and physiological processes, often in opposing directions. They act as mediators for biological responses like gastrointestinal integrity, platelet aggregation & kidney perfusion, vascular homeostasis, and are also intensely included in cancer autoimmunity regu¬lation & inflammation.
• Eicosanoids comprise the thromboxanes (TX), prostaglandins (PG), leukotrienes (LT) and lipoxins (LX).TXs &PGs are together known as prostanoids.
• Prostaglandins were synthesized originally in prostate, leukotrienes from leukocytes and thromboxanes from platelets (thrombocytes) sotheir names were derived. Lipoxins are anti-inflammatory eicosanoids that synthesized via interactions of lipoxygenase (therefore the derivation of their name).
• Eicosanoids cause a broad range of biological effects on inflammatory responses (mainly thoseof the skin, eyes &joints), on function of reproduction (comprising induction of labour), andon the duration and intensity of fever & pain. They also inhibitthe secretion gastric acid, regulating blood pressure throughout constriction or vasodilation, and activating or inhibiting platelet aggregation &thrombosis.
• Among the pro-inflammatory eicosanoids, prostaglandin E2 (PGE2) acts as a key mediator of different types of inflammation, involving fever & swelling, inflammatory pain.
• The main actions of the series-2 prostaglandins & thromboxanes (chieflyTXA2&PGE2) are pro-inflammatory as the series-4 leukotrienes (mainly LTB4). Therefore, it makes sense that drugs that diminishthese compounds production would be beneficial incurtailment inflammation & the associated vascular pathologies.
Recommendations
• Additional studies are essential to classify the physiological roles of all eicosanoids & related PUFAs.
• Increase dietary intake of DHA and EPA as they are useful for the human body exerting the mechanism of non-steroidal anti-inflammatory drug action.
References
- Renata N G, Felipe C S, Alison C (2018) Eicosanoids and cancer. Clinics 73(suppl 1):e530s.
- Khanapure, S P, Garvey, D S, Janero et al., (2007) Eicosanoids in inflammation: biosynthesis, pharmacology, and therapeutic frontiers. Curr Top Med Chem 7: 311-340.
- Korotkova M, Jakobsson P J (2014) Persisting eicosanoid pathways in rheumatic diseases. Nature Reviews Rheumatology 10: 229-241.
- Stables M J, Gilroy D W (2011) Old and new generation lipid mediators in acute inflammation and resolution. Prog.Lipid Res. 50: 35-51.
- Gibellini F, Smith T K (2010) The Kennedy pathway-denovo synthesis of phosphatidylethanolamine and phosphatidylcholine. IUBMB Life 62: 414-428.
- Ha K D, Clarke B A, Brown W J (2012) Regulation of the Golgi complex by phospholipid remodeling enzymes. Biochim Biophysica Acta 182: 1078-1088.
- Szefel J, Kruszewski W J, Sobczak E (2015) FactorsInfluencing the Eicosanoids Synthesis in Vivo. BioMed Research International. Article ID 690692: 6.
- Schneider C, Pozzi A (2011) Cyclooxygenases and lipoxygenases in cancer. Cancer metastasis reviews 30: 277-94.
- Kaur K K, Allahbadia G, Singh M (2018) Synthesis and Functional Significance of Poly Unsaturated Fatty Acids (PUFA’s) in Body. Acta Scientific Nutritional Health 2: 43-50.
- Simopoulos A P (2016) An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients 8: 128.
- Von Euler U S (1934) An adrenaline-like action in extracts fromthe prostatic and related glands. J Physiol 81: 102-12.
- Michael W K (2018) Prostaglandins, Thromboxanes, Leukotrienes and Lipoxins. The MedicalBiochem.
- Reid G, Wielinga P, Zelcer N, van der Heijden I, Kuil A. et al., (2003) The human multidrug resistance protein MRP4functions as a prostaglandin efflux transporter and is inhibited by non-steroidal anti-inflammatory drugs. Proc Natl Acad Sci
- U S A 100: 9244-9.
- Kochel T J, Fulton A M (2015) Multiple drug resistance-associated protein 4 (MRP4), prostaglandin transporter (PGT), and 15-hydroxyprostaglandin dehydrogenase (15-PGDH) as determinants of PGE2 levels in cancer. Prostaglandins Other Lipid Mediat 116-117: 99-103.
- Lawrence T, Willoughby D A, Gilroy D W (2002) Anti-inflammatory lipid mediators and insights into the resolution of inflammation. Nat. Rev. Immunol 2: 787-795.
- Shinomiya S, Naraba H, Ueno A, Utsunomiya I (2001) Regulation of TNFα andinterleukin10 production by prostaglandins I2 andE2: studies with prostaglandin receptor-efficient miceand prostaglandin E2 receptor subtype-selectivesynthetic agonists. Biochem Pharmacol 61: 1153-1160.
- Samuelsson B. (1983): Leukotrienes: mediators of immediate hypersensitivity reactions and inflammation. Science 220, 568-575.
- Lammermann T, Afonso P V, Angermann B R, Wang J M, Kastenmüller et al., (2013) Neutrophilswarms requireLTB4 and integrins at sites of cell death in vivo. Nature 498: 371-375.
- Murakami K, Ide T, Suzuki M, Mochizuki T, Kadowaki T et al., (1999) Evidence for direct binding of fatty acidsand eicosanoids to human peroxisome proliferators activated receptor α. Biochem. Biophys.Res. Commun 260: 609-613.
- Jonathan J, Katrina J B, Dominik N M, Jens T (2014) Macrophages in homeostatic immune function. Front Physiol 5:146.
- Von Moltke J, Trinidad N J, Moayeri M, Kintzer A F, Wang S B et al., (2012) Rapid induction of inflammatory lipid mediators by the inflammasome in vivo. Nature 490: 107-111.
- Bitto A, Minutoli L, David A, Irrera N, Rinaldi M et al., (2012) Flavocoxid, a dual inhibitorofCOX-2 and 5-LOX of natural origin, attenuates the inflammatory response and protects mice from sepsis. Crit. Care 16: R32.
- Serhan C N (2002) Lipoxins and aspirin-triggered 15-epi-lipoxinbiosynthesis: an update and role in anti-inflammation and pro-resolution. Prostaglandins and Other Lipid Mediators 68-69: 433-455.
- Wallace J L, ZamunerS R, McKnight W, Dicay M , Mencarelli Aet al., (2004) Aspirin, butnot NO-releasing aspirin (NCX-4016), interacts with selectiveCOX-2 inhibitors to aggravate gastric damage and inflammation. American Journal of Physiology—Gastrointestinal and Liver Physiology 286: G76-G81.
- Arita M, Ohira T, Sun Y P, Elangovan S, Chiang N et al., (2007) Resolvin E1 selectively interacts with leukotriene B4receptor BLT1 and ChemR23 to regulate inflammation. Journal of Immunol 178: 3912-3917.
- Sun Y P, Oh S F, Uddin J (2007) Resolvin D1 and itsaspirin-triggered 17R epimer: stereochemical assignments, anti-inflammatory properties, and enzymatic inactivation. Journal of Biological Chemistry 282: 9323-9334.
- Fullerton J N, O’Brien A J, Gilroy D W (2014) Lipidmediatorsin immune dysfunction after severe inflammation. Trends in Immunol 35: 12-21.
- Hammarberg T, Provost P, Persson B, RË?admark O (2000)The N-terminal domain of 5-lipoxygenase binds calcium and mediates calcium stimulation of enzyme activity. The Journalof Biologic. Chem 275: 38787-38793.
- Pergola C, Rogge A, Dodt G, Northoff H, Weinigel C, et al., (2011) Testosterone suppresses phospholipase D, causing sex differences in leukotriene biosynthesis in human monocytes. The FASEB Journal 5: 3377-3387.
- Novak J Z (2009) Wielonienasyconekwasythuszczowe omega 3:asperktybichemicznefunkcjonalne I praktycyzne. FarmakoterPsychiatry Neurology 3-4: 127-146.
- Nowak J Z (2011) Inflammation: course and role of PUFAderivedlipid mediators in the solution of inflammatoryreaction”. MilitaryPharmacy Medicine 1: 20-30.
- Calder P C (2006) n-3Polyunsaturated fatty acids, inflammation and inflammatory disease”. American Journal of Clinical Nutrition 83: 1505-1519.
- Drevon C A (1992) Marine oils and their effects. Nutrit. Reviews50: 38-45.
- Simopoulos AP (2002) Omega 3 fatty acids in inflammation andautoimmunedisease. Journal of the American College of Nutrition 21: 495-505.



