Research Article - (2026) Volume 4, Issue 1
Re-understanding of Wave-particle Duality
Received Date: Dec 02, 2025 / Accepted Date: Dec 30, 2025 / Published Date: Jan 06, 2026
Copyright: ©2026 Chun Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Wang, C. (2026). Re-understanding of Wave-particle Duality. Eng OA, 4(1), 01-13
Abstract
The author has worked with high-resolution transmission electron microscope for many years, which provides the most direct evidence of wave-particle duality. This relates to Crookes and J. J. Thomson's cathode rays tubes to determine that electron beams are of particle nature. This also relates to Röntgen's discovery that when an electron beam in the cathode rays tube strikes the glass wall, it produces X-rays with extremely high penetrating power. This behavior is consistent with Maxwell-Lorentz’s electrodynamics that electromagnetic waves must be produced whenever electricity carriers alter their velocity. The wave-particle duality proposed by de Broglie refers to accelerated electron beam, rather than single electron. De Broglie emphasized that the crystallographic diffraction pattern produced by his electron beam was completely consistent with the crystallographic diffraction pattern of von Laue by X-rays, therefore, he proposed that electrons possess wave-particle duality. This is actually the selected area electron diffraction pattern in transmission microscope. It is X-rays, not the waves of the electrons themselves to produce diffraction pattern. He extended this to the wave-particle duality of all matter and light. Schrödinger directly introduced de Broglie's electron beam behavior into electron wave in atoms. It was just at this stage that illogical reasoning occurred, making the quantum theory increasingly difficult to understand.
Keywords
HR-TEM, Cathode Rays’ Tube, X-rays, Wave-Particle Duality, X-rays Diffraction
Introduction
The wave-particle duality of electrons was first proposed by de Broglie in 1923 [1-3]. He bombarded a single crystal with an electron beam and obtained the same transmission diffraction pattern as von Laue's X-ray bombardment of a single crystal. From this, he proposed that electrons themselves possess wave properties. Subsequently, he extended this view to the fact that everything possesses wave-particle duality, even including neutral matter. This paper argues that when an electron beam bombards a single crystal, X-rays are generated first, and the diffraction pattern is subsequently formed. It is this difference that led de Broglie to introduce a series of seemingly illogical concepts, such as electron wave packets and neutral matter waves. Modern quantum mechanics is primarily based on de Broglie's theory, particularly Schrödinger's atomic structure model, which describes electrons as standing waves that form electron clouds within atoms, which appear according to probability. This article, however, argues that electron beams are particle-like, not wave-like, and that X-rays produced by electron beams are wave-like, not particle-like. These are two different concepts. Neutral atoms and molecules do not exhibit matter waves. In order to understand this important physical concept, we must review its origins and development.
The existence of the smallest negative particles in matter, the electrons, was first definitely recognized by Crookes by using a cathode rays tube in 1870 [4]. Applying high voltage (from a few kilovolts to about 100 kilovolts) between the cathode and anode, some kind of rays emitted from the cathode (usually tungsten filament), and made themselves visible during striking the residual gases remaining in the partially evacuated tube. Crookes identified that these cathode rays were of material nature (particle matter) shown by the shadow cast by the metal Maltese cross on the fluorescence of the glass wall of the tube. Experiments showed further that the rays could be deflected by both magnetic and electric fields, proving they were negatively charged particles and not a form of electromagnetic wave.
This was the very initial recognition of the electrons. Since then, the electrons have become a concrete reality of the everyday life.
In 1897, J. J. Thomson used a device similar to Crookes' cathode rays tube, but evacuated to a high vacuum [5]. The electron beam was filtered into a thin line through two mutually perpendicular slits. He obtained the velocity of the electron beam and the charge- to-mass ratio of the electrons by the deflection in the electric and magnetic fields. His experiment further confirmed that the electron is a particle, not a wave.
The electric force bending the negatively charged electron is eE (e=electron charge, E=electric field strength). Introducing a magnetic field perpendicular to the electric field to repel the rays back to the zero-deflecting position, the magnetic force Hev (H=magnetic field strength, v= velocity of the electron) should be equal to the electric force. eE=Hev. Then, v=E/H, two macroscopic detectable values can determine the speed of the ultra-fast electron.
At this period of time, many scientists were studying cathode rays tubes. In this process, von Lenard introduced a new idea: “Could the electron beam be lead outside the high-vacuum glass tube to carry out direct experiments with them?” In 1893, he made a cathode rays tube with an airtight seal of very thin aluminum leaf (3 µm to 5 µm) at the bottom. As the cathode rays reached Lenard’s “aluminum window”, it was found that they passed through it and continued their course in the air outside the tube [6]. The rays could be deflected by magnetic field and so on, therefore, they had the same characteristics as the one in the tube. These are the transmitted electrons in our modern transmission microscope.
Lenard pointed out that “This study had also provided a great deal of other information, some of which is now general knowledge, on X-rays and radioactivity, as well as a deeper understanding of electricity and matter.” The rays could be made visible, by sheets of paper coated with certain ketone, platinum cyanide, or an alkaline-earth phosphor that are capable of becoming luminous without heat. By the way, these phosphorescent materials emit light because they contain a large number of double bonds, triple bonds and lone pairs of electrons. In my recently published papers, "New understanding of atomic structure," and “The movement of two parallel unpaired electrons in benzene and aromatic compounds”, I pointed out that the π-orbits of unpaired electrons in the double and triple bonds have weakly bonded to the atomic nuclei and can easily expand and contract under additional energy, thus emitting light [7,8].
Lenard further pointed out that aluminum, silver, and gold of equal thickness have different absorption rates of the rays. The lighter the metal, the lower its absorption rate. This is the principle of particle imaging in transmission electron microscopy. The permeability of the electron beam coming out of the window is very low. For example, a completely light permeable ½ mm thick rectangular quartz plate is not permeable to the electron beam.
In 1895, Röntgen discovered X-rays using cathode rays tube [9]. His attention was attracted by a glowing fluorescent screen that was too far from the tube to be affected by the cathode rays. The rays had extraordinary penetrative power and could make the screen glow, even when cardboard, wood, copper and aluminum were placed in the way. To date, the man-made X-rays have made immeasurable contributions to humanity. Lenard commented that “It is generally considered to be a good example of a lucky discovery. But, given the tube, the fact that the attention of the observer was already turned from the interior to the outside of the tube, and the presence of phosphorescent screens outside the tube because of the purpose of the tube, it appeared to me that this discovery had of necessity to be made at this stage of development.”
Since the discovery of the mysterious X-rays, it had attracted the attention of many contemporary great scientists, exploring its intrinsic nature, developing definite rays and expanding its range of applications. First of all, the true essence of the rays must be clear. Was this the radiation of particle nature or wave nature? If it was wave, was it longitudinal wave or transverse wave? Which methods could be used to determine the wavelength?
At first, Righi’s convincing observation in 1896 confirmed that the X-rays do not carry with them a negative charge that had shown the untenability of the theory that they were extremely fast cathode rays [10].
On the basis of a hypothesis expounded as early as 1896 by Stokes and Wiechert, it was believed that when cathode rays particles collided with matter, they were obstructed, creating a disturbance that lasted for a period of identical duration to that in which the electron was impeded. This period of time multiplied by the speed of light equaled the wavelength of X-rays.
In 1897, after Dorn successfully determined the proportion of blocked electron energy converted into X-rays, W. Wien was able to calculate the impulse width. According to his calculations, the impulse width was approximately 10-10 cm (wavelength).
In 1904, Charles Barkla had shown that X-rays could be polarized [11]. Polarization was a strong argument for a transverse wave.
Sommerfeld wrote in a paper in Annalen der Physik in early 1912 that now the “proof of diffraction would be a sort of a keystone” for the wave theory of X-rays [12].
In 1912, von Laue made an epoch-making discovery of the diffraction of X-rays by crystals [13]. When a narrow beam of X-rays passing through triclinic copper sulfate single crystal (CuSO4), von Laue immediately obtained undoubted evidence of regular diffraction. This demonstrated that X-rays could be described as transverse waves (0.02 Å, the wavelength of the X-rays is of a smaller order than the dimensions of the atomic structure 1 Å.), the same nature just like visible light (400-700 nm) and ultra violet light (100-400 nm). The other important contribution of von Laue confirmed the natural spacing between the atoms in the crystals (theoretically inferred by Bravais in 1850). But, the subsequent interpretation of the diffraction pattern seemed to be in trouble [14].
Contemporary scientists, W. Bragg and L. Bragg, father and son, had been aroused great curiosity by the strong evidence of von Laue. They conducted reflection diffraction on the single crystal surfaces of NaCl, KCl, LiF and CaF2 and obtained very regular patterns [15]. How to interpret these patterns was a huge challenge. Von Laue suggested that this might be accounted for by the existence, in the X-ray beam, of certain wavelengths alone; and that a diffracted beam only appeared when conditions were right for diffracting these wavelengths. The ingenious young Bragg considered this fact in a very different way. He thought that perhaps we ought to look for the origin of this selection of certain directions of diffraction in the peculiarities of the crystal structure, and not in the constitution of the X-ray beam. The idea suddenly leapt into his mind that Laue’s spots were due to the reflection of X-ray pulses by sheets of atoms in the crystal.
The diffraction could be treated as the reflection of the planes composed of ordered atoms. The pulses reflected by successive planes built up a wave train, which analysis showed to be composed by the wavelengths given by the formula:
nλ = 2d sin ϑ
In this expression, n is the integer, λ is the wavelength of the X-rays, d is the spacing of the planes, and ϑ is the glancing angle at which the X-rays are reflected.
If the nλ=2dsinϑ condition is satisfied, constructive interference of the electromagnetic waves (wave crest in phase) occurs and bright spots appear on the detecting screen.
The selected area electron diffraction (SAED) pattern obtained with TEM is actually the transmission diffraction pattern of von Laue. Whether transmission or reflection, the bright spots are formed through the maximum interference of the waves of the output X-rays.
X-rays are reflected by the shells of electrons surrounding their nuclei. The electron shells are basically spherical. When X-rays hits the surface of the sphere, the reflected X-rays penetrates the sample or emits out the sample surface, depending on the relative position between the incident X-rays and the surface of the shell. Anyway, the von Laue diffraction patterns are essentially the same as the Bragg diffraction patterns.
Most atoms have several electron shells. The closer to the nucleus, the harder the electron shell, and Rayleigh elastic total reflection will occur. No matter which shell totally reflects X-rays, it can be used to determine the position of atoms in the crystal. That's why Bragg supposed that the diffraction took place at the center of each atom, as if the whole effect were localized there.
Thanks to Bragg's equation, researchers can now determine the structure of a crystal by X-rays irradiating it and examining the lattice pattern produced by the reflected rays.
By 1924, when de Broglie proposed the wave-particle duality, scientists could easily measure the velocity v of the electron beam and the produced wavelength λ of the X-rays. De Broglie pointed out that since the particles and waves cannot be independent, it must be possible to establish the corresponds.
λ= h/p, p=m0v
In the formula, λ-the wavelength of X-rays, h-Plack constant, p-particle momentum of the electron, m0-electron rest mass, v-velocity of electron beam.
De Broglie's chain of reasoning: “The determination of the stable motions of the electrons in the atom involves whole numbers, and so far the only phenomena in which whole numbers were involved in physics were those of interference and of eigen-vibrations. That suggested the idea to me that electrons themselves could not be represented as simple corpuscles either, but that a periodicity had also to be assigned to them too. It is necessary to introduce the corpuscle concept and the wave concept at the same time. In other word the existence of corpuscles accompanied by waves has to be assumed in all cases.”
This reasoning process is perplexing! Bohr’s stable motions of the electrons in the atom shows no vibration or interference, only the unstable excited electron emits light during falling back to ground state. How could one so hastily assert that the existence of corpuscles accompanied by waves must be assumed in all cases?
As de Broglie once said, “Let us in fact imagine an indefinite cloud of electrons all moving at the same velocity in the same direction”. (This is actually an electron beam!).
Here, de Broglie made no mention of how X-rays are generated. When a known velocity v of the electrons impinging on the crystal, transmitted X-rays are immediately produced. In the time of his experiments, the size of a single crystal was certainly many times larger than the penetration depth of an electron beam, so the transmission diffraction pattern he obtained must have been caused by X-rays, and therefore is the von Laue diffraction pattern.
At this point, de Broglie used an accelerated electron beam to determine the wavelength of the electron. According to his theory material particles are always linked with a system of travelling waves, a «wave-packet», forming the constituent parts of matter and determining its movements. This is the first and most important step in the problems that will arise in quantum mechanical model of atoms later.
Schrödinger in 1926 [16-19], introduced de Broglie's electron beam behavior into the electron wave in atoms. It was just at this stage that illogical reasoning occurred, making the quantum theory that developed from it increasingly difficult to understand. Schrödinger's atomic model fully embraces wave-particle duality, treating the electron as a three-dimensional standing wave around the nucleus. According to de Broglie formula, λ=h/m0v, this wavelength of an electron must be smaller than the size of an atom, approximately one Å. The velocity of an electron in an atom must then be 108¸ m/s, circling the Earth four or five times per minute. Is it possible that, under such high speeds, electrons would appear around the atomic nucleus in probability?
In 1933, Ruska built the first electron transmission microscope [20-22]. He found that short coils could act as lens (instead of optical lens) to focus electron beam, to enlarge the images etc. Through continuous improvements, modern transmission microscopes have made it commonplace for people to see atoms. Its fundamental principles are the Lenard cathode rays tube with aluminum windows, and J. J. Thomson cathode rays tube to deflect electron beam by magnetic field. In our specific operations, different images can be obtained in the same area at the same magnification, demonstrating that transmission microscopy can most directly reveal the wave-particle duality. This will be discussed in detail in the discussion section.
In 1929, Otto Stern reported in a short note the reflection of He atoms from a cleaved NaCl crystal surface with well-resolved diffraction peaks [23]. Thereby he provided the first direct evidence that the incident atoms of helium as massive particles had wave properties. Their experimental curves do not show any constructive or destructive interferences of the waves. Their proposal also does not conform to Maxwell-Lorentz’s electrodynamics that electromagnetic waves must be produced whenever electricity carriers alter their velocity.
This article first details Röntgen's discovery of X-rays, emphasizing its crucial role in subsequent electron beam research. The second part introduces von Laue's groundbreaking discovery, which proved that X-rays are transverse waves with extremely short wavelengths and verified the correctness of lattice theory. The third part introduces the extremely simple rule governing the arrangement of atoms in a crystal lattice, discovered by the genius scientist L. Bragg. The fourth part introduces Ruska’s ingenious use of cathode rays and his invention of the first transmission electron microscope. After that, the author presents a renewed understanding of wave-particle duality using modern high- resolution transmission electron microscopy. Part Five delves into de Broglie's wave-particle duality and the experimental evidences he cited to support this theory. Finally, Part Six introduces Stern's experiment: the phenomenon of a beam of neutral small atoms being reflected by a crystal. He considered this evidence for matter waves. Stern invented the atomic beam device, and his experimental results consistently aimed to approximate the then- emerging quantum theory, neglecting simpler principles, such as the spatial quantization of neutral silver atoms. This is precisely a major problem currently facing the scientific community: the failure to thoroughly test whether experimental results conform to the logical reasoning chain of quantum theory.
The quantized and non-quantized “quantum phenomena” will be discussed in detail in another article.
Methodology
The re-understanding of wave particle duality is rooted in a rethinking of existing experimental facts. A strict chain of reasoning to link fundamental principles with evidence is maintained throughout the proposal.
The high-resolution transmission electron microscope (HRTEM) used in this work is Jeol 2010. The emitted electrons from tungsten filament are accelerated about one meter long, with 200 kV. The technical resolution is about 0.24 Å. The final electron velocity is about 2x108 m/s.
The program tools like “CrystalMaker” for analysis were used in this work.
Results and Discussions
Röntgen Discovery of X-rays
In nature, there is almost no detectable X-rays. This manmade radiation, discovered by Röntgen in 1895, has made enormous contributions to mankind in many ways and its far-reaching significance is still incalculable. It is necessary to provide a detailed account of his findings here, in order to clarify some confusing scientific concepts, especially wave-particle duality.
Röntgen’s apparatus for the new type of radiation (X-rays or Röntgen-rays) was Crookes’ or Thomson’s cathode rays tube. The sufficiently evacuated glass tube was enveloped pretty tightly with black carton. In the completely dark room, Röntgen took a piece of paper screen painted with barium tetracyanoplatinate BaPt(CN)4 close to the discharging tube and observed the lighting up of the screen, even if the painted side was facing away to the tube. At the distance of two meters apart from the tube, the screen could still be lighted up. The wrapped black carton did not allow UV, sunlight or arc light to pass, as shown schematically in Figure 1. Therefore, the high penetrability of the unknown light indicated a new sort of radiation.
Figure 1: The Illustration Depicts a Highly Penetrating, Man-made, Mysterious Radiation. It can Penetrate Black Carton and Illuminate a Phosphorescent Screen from a Distance. The Tightly Wrapped Black Carton did not allow UV, Sunlight or arc Light to Pass.
The screen painted with luminescent material BaPt(CN)4 makes the invisible light to become visible. The reason is that the painting compound contains a lot of π-electrons in the double and triple bonds and contains a lot of lone pairs of electrons around the platinum. These electrons belong to active electrons that are easily excited. Under the irradiation of X-rays, they absorb energy and are excited to higher energy level. They emit visible light during falling back to the ground state. The electron configurations of double bond, triple bond and lone pair are newly introduced in my paper “New understanding of atomic structure” [7]. After that, Röntgen tested various materials of different densities, different thicknesses. For example, aluminum of 15 mm thick still allowed the X-rays to pass, but greatly reduced the fluorescence. Ebonite several centimeters thick was transparent. Platinum 0.2 mm thick allowed some rays to pass. Lead 1.5 mm thick was practically opaque. Wood 20 mm was transparent. More meaningfully, he also checked the hand of his wife. The transparencies of flesh and bone were very different, which made tremendous contribution to medical science worldwide, as shown in Figure 2
Figure 2: Röntgen Tested the Penetrability of Various Materials. Of Particular note is the Significant Difference in Transparency Between the Flesh and Bones of the Human hand, which has made a Tremendous Contribution to Medical Science.
The following list in Figure 3 was Röntgen’s original data. It vividly showed that the transparencies of metals were greatly different. The tendency was that the higher the density, the lower the transparency.

That is why the most commonly used target materials for Compton’s inelastic scattering experiments were lightweight materials such as graphite, aluminum and plastics. The X-rays must pass through the target, otherwise the scattered X-rays cannot be detected [24].
The X-rays observed by Röntgen was created by an electron beam (cathode rays) striking on the glass tube wall. Lenard and Röntgen both confirmed that cathode rays and X-rays are two completely different things. In a magnetic field, the former deflects while the latter remains unchanged. Are X-rays beams of uncharged particles or waves? If they are waves, are they longitudinal or transverse waves? As mentioned in the introduction, people initially determined that X-rays were transverse waves with a wavelength of approximately 10-10 cm. These conclusions still needed to be verified by experiments.
Von Laue X-rays Tramsmission Crystallography
In 1912, Friedrich, Knipping and Laue published a paper “Interferenz-Erscheinungen bei Röntgenstrahlen” [25]. This made an epoch-making discovery to unmask the transverse wave nature of X-rays and proved the space-lattice structure, which was theoretically inferred by Bravais
Figure 4: Max Von Laue Obtained the First Diffraction Pattern on a Single Crystal (Triclinic CuSO4), Revealing the Transverse Wave Nature of X-Rays and Proving the Space-Lattice Structure, Which Bravais Theoretically Deduced.
Figure 4 is the transmission pattern from copper sulphate presented by von Laue in his Nobel lecture.
Why did he choose a relatively complex crystal for his experiment? Prior to this, he was seeking a mathematical presentation of the lattice theory. The fact that the lattice constant in crystals is of an order of 10-8 cm was sufficiently known from the analogy with other interatomic distances in solid and liquid substances, and, in addition, this could easily be argued from the density, molecular weight and the mass of the hydrogen atom which, just at that time, had been particularly well determined. The order of X-ray wavelengths was estimated by Wien and Sommerfeld at 10-9 cm. Thus, the ratio of wavelengths and lattice constants was extremely favorable if X-rays were to be transmitted through a crystal. They expected the crystal to contain copper, which would allow them to emit secondary characteristic X-rays to determine the location of the copper within the crystal. According to Barkla, heavy metals with atomic weights between 50 and 100 are the main sources of secondary radiation. The atomic weight of copper is 63.5, therefore, it is hoped that copper’s secondary radiation sources can be detected.
The ordered black spots on the photographic plate didn’t change by increasing the distance between detector and crystal. This may indicate that each secondary ray produces a spot of light, which emerges from the crystal as a parallel beam. This eliminates the possibility of spots formed by secondary radiation, instead, it's diffraction. It further confirmed by diamond transmission pattern. Barkla pointed out that carbon has difficulty emitting secondary radiation [26]. In my recently published article, "New understanding of atomic structure," I calculated, based on the Bohr model, that the energy required for an electron in the 1s shell to be excited and emit secondary radiation is much greater than that in other shells [7]. Therefore, a correct understanding of atomic structure can provide a reasonable explanation for experimental results.
Figure 5 is a very good illustration of the relationship between the primary X-rays in the center of the von Laue photo and the surrounding diffraction X-rays. Laue transmission patterns are composed by discrete spots related to individual diffraction events. Constructive interference can only occur when the primary rays are at specific angle to the spherical shell of the atom. This will be discussed in more detail in the next section, Bragg diffraction.
The von Laue transmission method is undoubtedly very useful for determining the quality of single crystals [27].
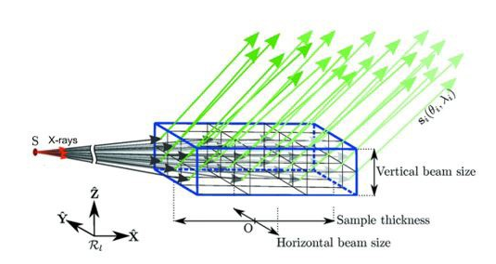
Figure 5: This Figure Is Based on a Schematic Diagram of Von Laue Diffraction and X-Ray Transmission Through a Bulk Single Crystal Drawn by Arnaud Et Al. [25]. Green Arrows Represent Diffracted Beams, and Black Arrows Represent Transmitted Incident Beam
Bragg X-rays Diffraction Crystallography
Contemporary scientists, W. Bragg and L. Bragg, father and son, had been aroused great curiosity by the strong evidence of von Laue. They conducted reflection diffraction on the single crystal surfaces of NaCl, KCl, LiF and CaF2 and obtained very regular patterns [15]. How to interpret these patterns was a huge challenge. The ingenious young Bragg considered this fact in a very different way. He found that the bright spots of the diffraction could be treated as the reflection of the planes composed of ordered atoms. The pulses reflected by successive planes built up a wave train, which analysis showed to be composed by the wavelengths given by the formula:
nλ = 2dsinϑ
In this expression, n is the integer, λ is the wavelength of the X-rays, d is the spacing of the planes, and ϑ is the glancing angle at which the X-rays are reflected.
If the nλ=2dsinϑ condition is satisfied, constructive interference of the electromagnetic waves (wave crest in phase) occurs and bright spots appear on the detecting screen, as shown in Figure 6.
If the nλ=dsinϑ condition is satisfied, destructive interference of the electromagnetic waves occurs, as shown in Figure 7. This is why Bragg equation must be 2d
Figure 6: Bragg Law. If λ=2dsinϑ is satisfied, Constructive Interference of the electromagnetic waves. (The Wave Crests are in Phase) Occurs and Bright Spots Appear on the Detecting Screen.
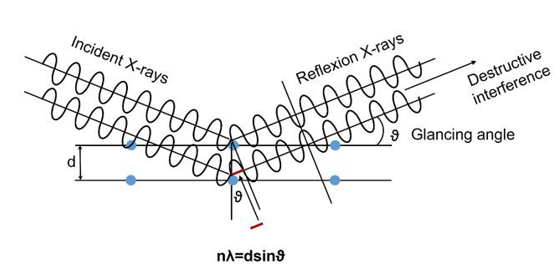
Figure 7: If λ=dsinϑ is satisfied, then the Electromagnetic Waves. Will Undergo Destructive Interference (The Wave Crests are Out of Phase), and the Wave Amplitudes Will Cancel Each other Out, So No Signal Can be Detected.
The selected area electron diffraction (SAED) pattern obtained with TEM instrument is actually the transmission diffraction pattern of von Laue. Whether transmission or reflection, the bright spots are formed through the maximum interference of the waves of the output X-rays.
X-rays are reflected by the shells of electrons surrounding their nuclei. For example, there is no electron shell around the hydrogen in the water molecule, so hydrogen does not produce X-rays reflection. The electron shells are basically spherical. When X-rays hits the surface of the sphere, the reflected X-rays penetrates the sample or emits out the sample surface, depending on the relative position between the incident X-rays and the surface of the shell. In explaining diffraction patterns, von Laue and L. Bragg differed. Von Laue believed that only X-rays with specific wavelengths were reflected by atoms in primary X-rays, and that interatomic thermal vibrations caused interference. Bragg, on the other hand, directly used monochronic secondary characteristic X-rays with specific wavelengths to irradiate the crystalline sample. He argued that interference was caused by the constructive and deconstructive interference of the light waves themselves. From von Laue's experimental setup, we can see that they inserted an anticathode crystal into the electron beam. The resulting monochromatic secondary characteristic X-rays penetrated copper sulfate. Because the experimenters and the theoretical interpreters separated, this detail was overlooked. Furthermore, the thermal vibration frequency is much, much lower than the frequency at which the X-rays hit the atoms; the diffraction pattern is already completed before the next thermal vibration occurs. Therefore, L. Bragg, without considering thermal vibrations, was able to derive a simple and practical formula.
Modern X-rays structural analysis has been greatly advanced by means of computers. No matter how complex molecules like DNR, Ribson etc., as long as they can form a single crystal, in which the atoms are arranged in regular manner, the position of each atom can be determined by an X-rays diffractometer. The principle used is the simple Bragg formula. How important is simplicity to science. There are really some simple secrets hidden in nature.
Figure 8 shows the typical modern X-rays tubes used in common laboratories. A heavy metal block is inserted into the path of the electron beam. Electrons emitted from the cathode are accelerated by the heavy metal block as an anode and strike on it, and then X-rays is released. More often, the heavy metal block is called anticathode.
The kinetic energy of the incident electron beam must be greater than the energy of the characteristic radiation, because part of the kinetic energy is converted into thermal energy and enters the anticathode. The accelerating voltage between the cathode and the anode varies from 20 kV to 150 kV, depending on the desired wavelength. The exact wavelength at which X-ray is emitted depends on the chemical elements. X-rays sources of various wavelengths have been discovered to be in the range of 0.01 nm to 10 nm. Commonly used Cu anticathode can emit 1.54 Å monochromatic X-rays (KαLine), (Cr 2.29 Å, Fe 1.94 Å, Co 1.79 Å, Mo 0.71 Å, Ag 0.56 Å). With the definite wavelength of the X-rays, many properties of the substance can be accurately explored. The larger the atomic number (Z), the shorter the characteristic wavelength and the higher the energy. By plotting the technical data of these commercially available X-ray tubes into a diagram, the Siegbahn K-series curves are immediately obtained
Figure 8: Monochromatic Beam of X-Rays of Known Wavelength λ is the
Decisive Role to Determine the Dimensions of the Unit Cell.
Transmission Electron Microscopy
In 1933, Ruska ingeniously used the whirl-shaped magnetic field generated by short coils to deflect the direction of the electron beam, which served the same purpose as the deflection of light by a convex lens in an optical microscope. Despite the significant advancements in modern TEM technology, its fundamental principles remain the same as they were ninety years ago.
The electron gun (electron source) generates electron beam by emitting electrons through heating a material (e.g., tungsten or LaB6 filament) and accelerating them with a high accelerating voltage. For example, tungsten filament can withstand temperatures of over 3,000°C, and each atom can release six unpaired electrons. Therefore, it is the best choice for an electron gun. In our experiment, the electron beam was accelerated at 200 kV, eventually reaching a velocity of 2.108 m/s (2/3 of the speed of light). At such a high speed, the electrons gained enormous momentum, like bullets, capable of penetrating 100-nm-thick samples. Electrons themselves carry negative electric fields, and the high-speed movement of electrons generates magnetic fields that surround the electron beam, as shown in Figure 9. This state has never been mentioned before. Such electromagnetic field strikes specimen, electromagnetic waves create, namely X-rays.
Figure 9: The Electron Beam in TEM is the Same as Cathode Rays Tube. The 200 kV Accelerating Voltage Enabled the Electron Beam to Reach Two-Thirds the Speed of Light. The Thin Specimen (< 100nm) Allows the Electrons Pass Through, Which is the Same as Lenard’ Aluminum Window. The Electron Beam Impinges the Specimen Creates Immediately X-Rays, Which is the same as Röntgen Radiation, which can Penetrate Depth Over 500 nm of a Core- Shell Spheres.
On the basis of a hypothesis expounded as early as 1896 by Stokes and Wiechert this impulse was believed to consist of a disturbance when the cathode-ray particle, i.e. a forward-rushing electron, is impeded on colliding with molecules of matter. This disturbance or impulse was maintained for a period of identical duration to that in which the electron was impeded. This period of time, multiplied by the speed of light, was described as the impulse width, a quantity which, if the nature of the X-rays were the same as that of the light rays, would coincide with the wavelength. This involves the relationship between wavelength and the speed of the electron beam. This explanation goes a step further than Maxwell-Lorentz's electrodynamics, that electromagnetic waves must be produced whenever electricity carriers alter their velocity
Passing through the TEM specimen, which is ca. 100 nm thick, the penetrating electrons and X-rays were mixed together. During the TEM operation, we can adjust the ratio of the electron beam to the X-rays to obtain two different images. This fact demonstrates that particles and waves are two separable properties, rather than the wave-particle duality of electrons being a single entity as de Broglie believed, thus extending to the wave-particle duality of neutral matter. The following example further illustrates this behavior. A core-shell sphere had a diameter of about 500 nanometers, which was much larger than the penetration depth of the electron beam (100 nm). By focusing, only one dark solid disk appeared on the screen; in the case of defocusing, the inclusions in the sphere could be distinguished. The contrast of the TEM micrograph can only be explained with the X-rays. When electromagnetic waves pass through domains of different densities, the wave propagation velocities and the wave phases are different.
Figure 10: HRTEM Atomic Resolution Images of TiO2 on a LaAlO3 Substrate. The Corresponding SAED Pattern of the TiO2.
The TEM images in Figure 10 were from an experiment I did previously. The left micrograph reveals the epitaxial growth of TiO2 (100) single crystal on the surface (001) of LaAlO3 single crystal substrate. The micrograph in the lower right corner is the corresponding selected area electron diffraction pattern. Both of the images were obtained by defocusing and fully opening the aperture. Whether transmission or diffraction, the two images should be formed by X-rays (Refer to Fig. 5). Ruska, the TEM inventor, once pointed out that it is best to allow electrons to arrive at the sample synchronously, which will improve the resolution. This is beneficial for X-rays to be in phase, but it doesn't help with electron beam imaging. Currently, in an effort to improve TEM resolution, researchers are continuously increasing the accelerating voltage. This may be a misguided direction. I spoke with Professor Frank about this. He visited University of Freiburg in June 2024. He is a 1987 Nobel laureate in Chemistry and also uses TEM to analyze their work. His collaborating TEM production unit, Cornell University, was preparing to increase the voltage from 200 kV (resolution 0.24 Å) to 600 kV. I told him that this would only make it easier to burn up the samples. I suggested that defocusing and widening the aperture should be used to increase the ratio of the X-rays. He accepted this view and said that many of their images were actually defocused. I also talked to him about how even if we could achieve atomic resolution, we still wouldn't be able to explain the properties. I believe that science urgently needs to overcome a hurdle: studying the motion of electrons within molecules. For example, covalent bonds, double bonds, lone pairs of electrons, free radicals, and their superimposed magnetic fields, etc.
Because the electron beam in a transmission electron microscope has very high energy, it can excite inner-shell electrons of atoms to release K, L, and M series second characteristic X-rays, thereby enabling elemental analysis. This is exactly the same as performing elemental analysis directly using X-rays. In fact, X-rays are generated as soon as the electron beam strikes the sample. In our previous teaching, we believed that high-energy X-rays and electron beams would knock out electrons in the 1s shell, and the electrons in the outer shell would fall into the inner shell one after another, releasing characteristic radiation. This is a completely wrong interpretation. The energy required to knock out an electron from a 1s shell is extremely high, and it would cause all outer electrons to be knocked out, resulting in an electron avalanche. The most reasonable explanation should be that the several electron shells were simultaneously excited and simultaneously released energies, so the K-, L-, M-series occurred simultaneously.
Electron avalanches are something we encounter in our TEM lab. A Geiger-Müller tube can be used to detect X-rays leaks under the control panel. The principle is that applying positive and negative electrodes puts the inert gas atoms into a metastable state. When the incident energy reaches a certain level, all electrons separate from the atomic nucleus, forming an electron avalanche and emitting a signal. Because X-rays have greater penetrating power, leakage can occur, while electron beams do not. This fact also indicates that, in transmission electron microscopy, electron beams (particles) and X-rays (waves) are two different things, not unified entities on the electrons as de Broglie believed.
De Broglie Wave-particle Duality related Experiments
By 1923, when de Broglie proposed wave-particle duality λ=h/ m0v, scientists were able to easily measure the velocity v of the electron beam and the generated X-rays wavelength λ. The faster the speed, the shorter the wavelength. De Broglie first experimental evidence was that the diffraction pattern formed by an electron beam directly passing through a crystal was the same as the diffraction pattern formed by von Laue using X-rays to transmit through a crystal. Therefore, electrons, in addition to their particle nature, also possess wave-like property. The previous sections have clearly demonstrated that when an electron beam strikes a solid, it immediately produces X-rays. Then it is not an inherent wave property of electron itself. He also cited the electron transmission ring-diffraction pattern on polycrystalline thin layers by G. P. Thomson and other, similar to the Debye-Scherrer ring pattern using X-rays.
This section provides an in-depth analysis of G. P. Thomson's experiments. A narrow beam of cathode rays was transmitted through a thin film of metal (platinum, gold, aluminum, copper etc.). The scattered beam showed a pattern of rings. The accelerating voltage were between 10 kV and 80 kV. The film thicknesses varied from 10 nm to 100 nm. Undoubtedly, the rays penetrating the sample include X-rays and electron beams. This is exactly the same as in transmission electron microscopy and is similar to the Lenard aluminum window (X-rays and electron beam mixed together).
It should be mentioned that G. P. Thomson also made an important discovery to explain many experimental phenomena. He reported that the ring diffraction pattern were deflected equally with those electron beam which had passed through holes in the film. Thus, he believed that the pattern is due to electrons which have preserved unchanged the property of being deflected by a magnet [28,29].
From our current perspective, the electron beam passing through the sample is also surrounded by electromagnetic fields, just like the incident electron beam, as shown in Figure 9. X-rays are electromagnetic waves. When these two types of rays mix together, the movement of the electron beam will inevitably cause the X-rays to move as well. This is why short coils in TEM experiments can simultaneously amplify both the electron beam image and the X-ray image.
The same analysis can explain the pattern of the electron beam passing through the double slits. Lenard points out that the electron beams do not cross because of electrostatic repulsion. The interference pattern should be formed by X-rays.
Another experiment considered strong evidence of electron waves is the pattern produced by Davisson and Germer when they bombarded the surface of a nickel single crystal with an electron beam [30], as shown in Figure 11. There is a serious misunderstanding here. They used the accelerating voltages of 54 volts. Based on the relationship between mature electron energy and wavelength, eE = hν = hc/λ, the wavelength of the electron beam is
λ (nm) = 1240/E(eV)
Their electron wavelength was 23 nm. The electron beam wavelength in our TEM (200 kV) is 0.0062 nm or 0.062 Å. Bombarding the atoms in a nickel single crystal on the (111) surface with this type of electron beam does not produce a wave- like diffraction pattern. Instead, it involves the elastic or inelastic scattering of electrons from the atoms. Davisson claimed that they used this experimental result to obtain the electron wavelength of 1.67 Å. The energy of the incident electron beam must be greater than the energy of the reflected electron beam, meaning that a shorter incident wavelength can produce a longer reflected wavelength. How credible is the fact that they obtained an electron wavelength of 1.67 Å using an incident beam of 23 nm to support the theory of wave-particle duality? In fact, this method involves determining the arrangement of the atoms on the surface
Figure 11: Electron Beam at Accelerating Voltage 54 V was Deflected Rather Than Diffracted by Nickel Atoms. there is No Constructive and Destructive Interference of Wave Behavior.
De Broglie's citation of those experimental evidences to support his wave-particle duality of electrons is untenable. Furthermore, he even extends his theory directly to neutral matter waves. Therefore, the quantum mechanics theory, density function theory, and molecular orbital theory developed from this are all questionable.
Otto Stern and Matter Waves
From our analysis above, we can see that magnetic and electric fields exist around the electron beam; the higher the speed, the stronger the magnetic field, which is the basis for the generation of X-rays. The particle nature of the electron and the wave nature that it generates are thus combined.
In 1930, Stern used his invention to generate a beam of He atoms, which he then used to strike a LiF single crystal. The resulting peak was considered to be a neutral matter wave, as shown in Figure 12. The wavelength calculated from the lattice constant of the crystal has the value λ=h/mv required by de Broglie for various m and v. This line of reasoning is illogical. There is no electromagnetic field around the neutral atom beam, so it does not generate electromagnetic waves. This peak phenomenon is simply due to elastic or inelastic scattering caused by very small He atoms hitting large atoms in the crystal. This is particle-like, not wave- like, and will not produce constructive or destructive interference.
Soon after, it was discovered that the intensity of the scattered beam resonated with the energy of the incident helium atoms. These peaks were quickly interpreted as a sign that the incident atoms were in a bound state at the crystal surface: the incident helium atoms were temporarily trapped in a potential well above the crystal lattice. As shown in Figure 12, He atom is generally neutral, but its 1s shell surface is negatively charged. When it impacts the surface of a LiF ionic crystal, the positive ion Li attracts the He atom while the negative ion F repels it. The structure or configuration of atoms is a real entity. These facts cannot be ignored when studying the interactions between atoms.
In fact, the matter beams usually consist of very small atoms or molecules. The reason for generating the diffraction pattern should take into account the counteraction of the crystal field. The atoms in the crystal lattice will bounce back the high-speed atoms or molecules in the matter beam. Based on the diffraction evidence alone, we are unable determine the wave nature of the neutral particles.
Figure 12: Neutral Helium Atoms Impact on LiF Single Crystal. The Resulting Pattern is from the Deflection Rather Than Diffraction by Li and F Atoms, Not From Matter Waves.
Conclusion
De Broglie believed that the diffraction patterns formed when an electron beam strikes a single crystal originate from the wave nature of electrons. This article argues that the electron beam first generates X-rays when striking a single crystal, and only then do diffraction patterns form. It is this difference that led De Broglie to introduce a series of illogical concepts, such as electron wave packets and extending wave–particle duality to neutral matter. The quantum scientific system based on De Broglie's wave–particle duality contains many inherently contradictory theories. The most basic atomic model is considered to be composed of electron clouds. The author hopes that the scientific community can fundamentally examine those theories with significant contradictions; the purpose of this article is merely to provoke further discussion.
Wave-particle duality sometimes mix together and sometimes separate (Lenard). These are two concepts: electron beam and X-rays, not that electrons themselves possess two properties. The formula λ= h/p proposed by de Broglie is a simple way to estimate the wavelength of the X-rays created by electron beam at certain velocity. Under the experimental conditions at the time, most accelerating voltages were 30 kV, and electron speeds were 1/3 the speed of light. Schrödinger's atomic model fully embraces wave-particle duality, treating the electron as a three-dimensional standing wave around the nucleus. Therefore, this wavelength must be smaller than the size of an atom, approximately one Å. The velocity of an electron in an atom would then be 10⸠m/s, circling the Earth four or five times per minute. Is it possible that, under such high speeds, electrons would appear around the atomic nucleus according to probability?
Can we pass on such Schrödinger’s theory to future generations? This is a questioning of the soul of every scientist [28-32].
Conflicts of Interest
There are no conflicts to declare.
Acknowledgement
The author would like to thank Dr. Thomas VON LARCHER, Senior Editor, Research Publishing, Springer Nature. He took great effort to provide the author with a series of guidance, which enabled the author to comprehensively improve her ability to complete scientific papers.
Funding: Not Applicable.
Clinical trial number: not applicable.
Ethics, Consent to Participate, and Consent to Publish declarations: not applicable.
References
- De Broglie, L. (1923). Radiation----Waves and Quanta.Comptes rendus, 117, 507-510.
- De Broglie, L. (1924). On the theory of quanta. Doctoral thesis, A translation of : RECHERCHES SUR LA TH´ EORIE DES QUANTA (Ann. de Phys., 10e s´erie, t. III (Janvier-F´evrier 1925).
- De Broglie, L. (1929). The wave nature of the electron. Nobellecture, 12, 244-256.
- Crookes, W. (1878). On the illumination of lines of molecular pressure, and the trajectory of molecules. Philosophical Transactions of The Royal Society, 170, 135-164.
- Thomson, J. J. (1897). Cathode Rays. Philosophie Magazin 5th Series, 44(269), 293-316.
- Lenard, P. (1906). On cathode rays. Nobel Lecture in Physics.105-134.
- Wang, C. (2025). New Understanding of Atomic Structure.Adv Envi Man Rec, 8(3), 01-18.
- Wang, C. (2025). The Movement of Two Parallel Unpaired Electrons In Benzene and Aromatic Compounds. Adv Envi Man Rec, 8(3), 01-26.
- Röntgen, W. C. (1896). On a new kind of rays. Science, 3(59), 227-231.
- Righi, A. (1896). The propagation of electricity in gases traversed by X-rays. Mem. Accad. Sci. Bologna, Ser. 5, 6, in Lenard Nobel lecture 1906, page 120.
- Barkla, C. G. (1904). Polarisation in Röntgen rays. Nature, 69(1794), 463-463.
- Sommerfeld,A. (1912). Über die Beugung der Röntgenstrahlen.Annalen der Physik, 343(8), 473-506.
- Von Laue, M. (1920). Concerning the detection of X-ray interferences. Nobel Lecture in Physics 1914, 347-355.
- Bravais, A. (1850). Les systemes formes par des pointes distribues regulierement sur un plan ou dans l'espace. J. Ecole. Polytech., 1-128.
- Bragg, W. L. (1922). The diffraction of X-rays by crystals.Nobel Lecture in Physics 1915. 370-382.
- Schrödinger, E. (1926). Quantisierung als EigenwertproblemI. Annalen der Physik, 79, 361-376.
- Schrödinger, E. (1926). Quantisierung als EigenwertproblemII. Annalen der Physik, 79(6), 489-527.
- Schrödinger, E. (1926). Quantisierung als EigenwertproblemIII. Annalen der Physik, 80(13), 437-490.
- Schrödinger, E. (1926). Quantisierung als EigenwertproblemIV. Annalen der Physik, 81(18), 109-139.
- Knoll, M., & Ruska, E. (1932). Das elektronenmikroskop.Zeitschrift für physik, 78(5), 318-339.
- Ruska, E. (1935). The electron microscope as ultra- microscope. Research and Progress, 1, 18-19.
- Ruska, E. (1986). The development of the electron microscope and of electron microscopy. Nobel Prize in Physics Lecture, 355-380.
- Stern, O. (1929). Beugung von Molekularstrahlen am Gittereiner Krystallspaltfläche. Naturwissenschaften, 17(21), 391-391.
- Compton, A. H. (1927). X-rays as a branch of optics. Nobel Prize in Physics Lecture, 174-190.
- Friedrich, W., Knipping, P., & Laue, M. (1912). Interferenz- Erscheinungen bei Röntgenstrahlen. Sitz.ber. Bayer. Akad. Wiss., 311-322.
- Eckert, M. (2012). Max von Laue and the discovery of Xâ?ray diffraction in 1912. Annalen der Physik, 524(5), A83-A85.
- Arnaud, A., Guediche, W., Remacha, C., Romero, E., & Proudhon, H. (2020). A laboratory transmission diffraction Laue setup to evaluate single-crystal quality. Applied Crystallography, 53(4), 914-926.
- Thomson, G. P., & Reid, A. (1927). Diffraction of cathode rays by a thin film. Nature, 119(3007), 890-890.
- Thomson, G. P. & Reid, A. (1927). Diffraction of CathodeRays by Thin Films of platinum. Nature. 120(3031), 802.
- Davisson, C., & Germer, L. H. (1927). The scattering of electrons by a single crystal of nickel. Nature, 119(2998), 558-560.
- Estermann, I., & Stern, O. (1930). Beugung vonmolekularstrahlen. Zeitschrift für Physik, 61(1), 95-125.
- Toennies, J. P. (2021). Otto Stern and Wave-Particle Duality. Chapter 23, page 519-545. In Book Molecular beams in Physics and Chemistry. Editors, Friedrich, B. & Schmidt- Böcking, H.



