Research Article - (2023) Volume 6, Issue 1
Reproductive Function during Lactation
Received Date: Oct 02, 2023 / Accepted Date: Oct 23, 2023 / Published Date: Nov 23, 2023
Copyright: ©Â©2023 Rehan Haider. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Haider, R. (2023). Reproductive Function during Lactation. Toxi App Phar Insig, 6(1), 132-149.
Abstract
The phenomenon of locational amenorrhea, in which postpartum women experience temporary suppression of menstruation and fertility during breastfeeding, has long been recognized. This natural contraceptive effect is primarily attributed to elevated prolactin levels, which are stimulated by frequent suckling and inhibit the release of gonadotropin- releasing hormones and, subsequently, the menstrual cycle. However, the extent of lactational amenorrhea contraceptive efficacy varies among individuals owing to factors such as breastfeeding patterns, infant feeding behavior, and maternal health.
Recent research has shed light on the underlying mechanisms governing the interplay between lactation and reproductive functions. Studies have shown that, while exclusively on-demand breastfeeding offers a higher level of contraceptive protection, introducing supplementary foods and reducing breastfeeding frequency can shorten the duration of lactational amenorrhea, potentially leading to earlier ovulation and conception. Additionally, the hormonal milieu during lactation may not be entirely suppressive, because occasional instances of follicular development and ovulation have been reported before the return of regular menstruation. Understanding the intricacies of reproductive function during lactation is important not only for family planning but also for optimizing maternal and infant health. The duration of exclusive breastfeeding has been associated with various health benefits for both the mother and the child, including enhanced immune protection, cognitive development, and a reduced risk of certain chronic diseases. Lactational amenorrhea serves as a physiological contraceptive method, but its efficacy is influenced by individual variations and breastfeeding practices. Further research is needed to elucidate the complex hormonal interactions and refine guidelines for optimal family planning during the postpartum period. Balancing the benefits of extended breastfeeding with a desire for family planning requires a comprehensive understanding of the intricate relationship between lactation and reproductive physiology.
Keywords
Lactational amenorrhea, Reproductive function, Breastfeeding, Contraception Prolactin, Menstrual cycle, Gonadotropin- releasing hormone, Family planning, Ovulation Exclusive breastfeeding.
Introduction
Fertility
The understanding of the mechanisms underlying infertility and fertility return during lactation has increased over the last 30 years, with studies in both animal and human models. Much has been learned from comparing lactating and non-lactating hormonal physiologies, and from the study of associated brain peptides. Although we understand this further today, several significant questions remain.
Lactational Infertility
Lactation has been attributed to changes in the hypothalamic- pituitary-ovarian axis mediated by gonadotropin secretion. Frequent suckling of the breast causes changes in gonadotropin- releasing hormone (GnRH), which affects the anterior pituitary hormone and disorganizes the pulsatility and levels of luteinizing hormone (LH) and follicle-stimulating hormone (FSH), thereby disallowing rhythmic patterns that result in ovulation. Frequent suckling also results in high prolactin levels; however, the role of prolactin in fertility suppression is less clear.
Figure 20-1: Possible mechanisms of lactational amenorrhea: a nervous impulse from the nipple produces not only a rise in pro-lectin (1) but also changes in hypothalamic sensitivity to ovarian steroid feedback (2) and changes in gonadotropin-releasing hormone (GnRH) (3), leading to changes in the pituitary release of luteinizing hormone and follicle-stimulating hormone (4). Suckling may also stimulate the release of -endorphin (5), thus suppressing GnRH from the hypothalamus. (Redrawn from Winikoff B, Semeraro) P, Zimmerman M: Contraception during breastfeeding: a clinician’s sourcebook, New York, 1988, Population Council.)
Figure 20-1 illustrates the menstrual cycle and gonadotropic management. The key points include the following.
1. Follicular development was initiated by pituitary gonadotropin (FSH) levels.
2. persevered in increasing calls for FSH and estradiol from the growing follicle in response to the luteinizing hormone (LH), which is released in a pulsatile manner from the pituitary gland.
3. the
4. Follicle secretes progesterone (luteinization).
5. The oocytes were harvested 36 h later.
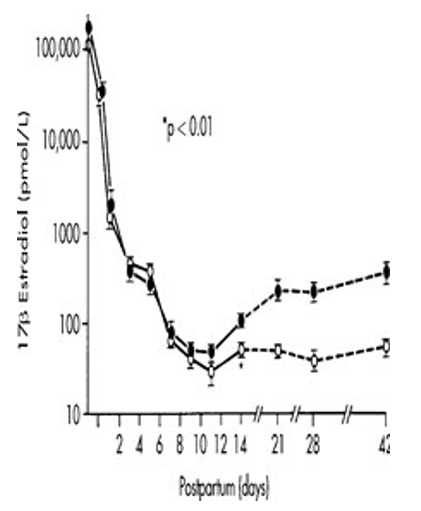
Figure 20-2: 17-Estradiol levels in postpartum period in lactating (open circles) and nonlactating (solid circles) women. Levels in lactating women vary with the intensity of suckling. (From Neville MC: Regulation of mammary development and lactation. In Neville MC, Neifert MR, editors: Lactation: physiology, nutrition, and breastfeeding, New York, 1983, Plenum.
The pulsatile release of GnRH from the hypothalamus stimulates LH release. In the cycling girl, estrogen increases GnRH secretion, and the combination of progesterone and estrogen decreases it.42.
However, postpartum duration is hormonally individualized with the aid of improved prolactin levels and occasional gonadotropin stages, resulting in anovulation and amenorrhea. Throughout breastfeeding, this kingdom can persist for an extended duration even though prolactin levels decrease over time. As currently understood, the pulsatile secretion of GnRH is altered via the suckling stimulus, influencing ovarian pastime.42, although the movement of prolactin on multiple goal-organ websites has often been cautioned as the cause of this ovarian quiescence, it seems that a suckling-brought-about alteration in hypothalamic GnRH production is the number one mechanism. Found that when pulsatility is restored in the course of lactation using exogenous pulsatility, GnRH and LH values increase and FSH levels decrease, while the pulsatile organization of both increases and the ovary responds accordingly.
The period of lactational amenorrhea depends on the frequency and intensity of suckling. In this study, open circles are lactating subjects and solid circles are non-actors postpartum.
Animal studies have shown that intense suckling inhibits the release of follicle-stimulating hormone (FSH) and luteinizing hormone (LH). In addition, when the nipple is stimulated while the milk ducts are tied off, the estrous and menstrual cycles are still suppressed. Se-lye and McKeown63 concluded that interruption of sexual cyclist.
During lactation, suckling is the result of suckling, and not the secretory activity of the mammary gland. This early animal study was a timely test. The greater the suckling stimulus in frequency and duration, the more consistent the suppression of ovulation 42 [1-4].
Gonadotropin levels in all postpartum women decreased during the first few weeks of the postpartum period, which substantiates the theory of postpartum ovarian refractoriness. In the first two weeks, postpartum, low levels of FSH were found in the urine and plasma. Estrogen excretion was low with a linear increase during the first Weeks. In a longitudinal study of 48 women, endocrine profiles were assessed using morning blood samples from the first postpartum month until the recovery of ovulation .Additional samples were drawn throughout 24 h at the end of the third postpartum month from 10 exclusively nursing women with amenorrhea. Levels of prolactin, LH, FSH, estradiol (E2), progesterone, cortisol, and dehydroepiandrosterone sulfate were measured. In response to suckling, there was a smaller increase in women who ovulated within six months postpartum than in those who did not. Suggested that this may explain some of the variability in the duration of lactational amenorrhea. The greater prolactin response to suckling associated with longer amenorrhea may result from a higher sensitivity to the breast- hypothalamus-pituitary system [5, 17].
Prolactin and Dopamine
There is a relationship between locational infertility and physiological hyperprolactinemia; however, its role in fertility suppression is not as clear in an extensive study of prolactin levels in lactating women, measured the pattern of prolactin secretion during suckling and the return of ovarian activity. Blood samples were drawn at 10-minute intervals for 24 h at 4 and 8 weeks, when weaning was initiated and suckling was reduced, at first menses, and in the follicular phase of the first menstrual cycle after weaning. Mothers fed their infants according to their usual pattern with no restrictions or alterations to replicate natural lactation [14, 67].
These data confirmed that prolactin levels did not decline significantly between the feeds with frequent suckling. When suckling became less frequent, prolactin dropped to baseline levels between feeds, but surged when suckling was initiated. The natural increase in prolactin at night was evident only after weaning. Prolactin also declined greatly in association with sucking after the return of menses. This occurred at 3.5–3.6 weeks postpartum in the present study. No relationship was observed between the duration of amenorrhea and plasma prolactin levels throughout the day, night, or lactation periods. The timing of the introduction of solids strongly correlated with the duration of amenorrhea. The authors concluded that no exact link exists between the release of prolactin during lactation and the duration of lactational infertility in breastfeeding women [67].
However, at least one study identified a differential response to different prolactins.
Another study explored the ability of parameters during pregnancy to predict the duration of amenorrhea followed up on 17 women at 34 and 38 weeks of gestation who were fully breastfed for at least 6 months. Free estradiol levels during pregnancy and the postpartum period were calculated. They found that the 10 women who experienced long lactational amenorrhea (greater than 6 months) had a different hormonal profile during pregnancy than those who experienced a short duration (less than 6 months) of lactational amenorrhea. At 38 weeks of gestation, women who experienced long lactational amenorrhea had twice as much prolactin, approximately half of the total estradiol, a significantly lower sex hormone-binding globulin concentration, and a similar free estradiol concentration as those who experienced short lactational amenorrhea. They concluded that at 38 weeks gestation, the higher ratio of prolactin to estradiol identified all women who would go on to experience a longer duration of their lactational amenorrhea, suggesting that the duration of lactational amenorrhea is conditional during pregnancy [7, 18].
Inhibition of dopamine secretion from the hypothalamus is associated with neural impulses from nipple stimulation during lactation. Normally, dopamine inhibits prolactin secretion; conversely, when dopamine is inhibited, prolactin levels rise. Two pathways of ovulatory inhibition are possible because of the increase in prolactin. One is a lack of responsiveness to oral steroids of the hypothalamic-pituitary axis of a lactating girl, leading to the non-pulsatile release of pituitary gonadotropins, FSH, and LH, which in turn result in absent or reduced ovarian activity. FSH may also be better at a few points; LH is non pulsatile [9 -12].
The presence of prolactin receptors in the ovary indicates that there may be a second mechanism contributing to infertility via an impaired ovarian response to gonadotropins determine 20-1).
The transition from amenorrhea to the ordinary menstrual cycle is one of the most challenging times during breastfeeding and the desire to use herbal family planning. Uncertainty regarding the onset of ovulation and the return of menses is difficult. The efficacy of a new postpartum transition protocol for maintaining pregnancy has been reported by an electronic hormonal fertility test (Clean Blue Fertility Screen; Swiss Precision Diagnostics, Geneva, Switzerland) was used to identify the fertility duration. It measures changes in urinary estrone-three-glucuronide from baseline and in urinary luteinizing hormone (LH) levels above a particular threshold. The tool has evolved to assist with conception and is similarly effective when used to avoid pregnancy. Internet teaching and charting protocols can help to prevent postpartum pregnancy during lactation.
Clinically, the onset of menstruation is a proxy for the return of fertility. The return of reproductive function varies depending on the period and degree of lactation. Most studies do not, in reality, document the pattern of breastfeeding, that is, whether or not the little one is fully or exclusively breastfed or is also receiving solid foods or supplemental bottles.20 At the end of the third month, the handiest 33% of fully lactating ladies have had a menstrual length, while 91% of no lactating ladies have had a minimum of one length13.
No longer all menstrual vaginal bleeds; not all bleeds comply with ovulation.21 In 72 completely breastfeeding women studied prospectively from 42 days postpartum, vaginal bleeding was recorded daily if it occurred. Approximately half of the women had a few bleeding episodes or were diagnosed between six and eight weeks postpartum. Those who experienced bleeding at the end of menstruation ovulated in advance of those who did not, but the differences were not well-defined. Seven women showed ovarian follicular improvement earlier than day 56, but neither bleeding nor follicular broadening was associated with ovulation in the first eight weeks. The authors stated that not going that far (bleeding earlier than 8 weeks in a breastfeeding girl) shows a return to fertility and, therefore, is not the return of menses.
In a prospective study, diagnosed the first postpartum ovulation using endometrial biopsy, basal body temperature, vaginal cytological evaluation, and cervical mucus in a group of 200 women. The dates of first ovulation, first menses, and nursing status were analyzed. None of the women demonstrated signs of ovulation before day 36 regardless of lactation. Of the 170 women who breastfed, 24 ovulated while completely nursing, 49 while partially nursing, and 97 after weaning [55].
Possibility of Conception
A non-lactating woman had a return of her period to 25 days at the earliest, a return of ovulation at 25–35 days, and a 5% chance of regaining fertility before six weeks postpartum.
The risk of ovulation during lactation was studied by in Baltimore and Manila. During the first 6 months postpartum, amenorrheic women had a low risk of ovulation (less than 10%) with partial breastfeeding and a 1% to 5% risk of exclusive breastfeeding with either frequent short feeds or infrequent longer feeds. This would have resulted in pregnancy rates of 2% and 1% [18, 20].
In a detailed study of 130 women in Chile, found the cumulative probability of pregnancy at the end of six months postpartum in women who were exclusively nursing and amenorrheic to be 1.8%. For exclusively nursing women who had a return to menses, it was 27.2% and for those who were partially nursing, it was 40.5% [16, 17].
Although many investigators continue to evaluate the effects of lactation on ovulation and menstruation, fundamental observations remain the same 68. Available data on the return of ovulation and menstruation can be summarized as follows60 [68].
|
Average Number of Feeds per Day |
Relative Risk |
|
0 |
1.0 |
|
1 |
0.62 |
|
2 |
0.43 |
|
3 |
0.28 |
|
4 |
0.19 |
|
5 |
0.12 |
|
6 |
0.08 |
|
7 |
0.05 |
|
8 |
0.04 |
|
9 |
0.02 |
|
10 |
0.01 |
TABLE 20-1: Relative Risk for Ovulation in Relation to Breastfeeding Frequency*
Breastfeeding episodes per day before ovulation
p < 0.0001.From Gray RH, Campbell O, The return of Ovarian function during lactation: results of studies from the United States and the Philippines In Gray R, Leridon H, and Spira A, editors: Biomedical and demographic determinants of reproduction, Oxford, 1993, Clarendon
I. Nursing mothers
A. Ovulation generally occurs before menses return and varies from 14% to 75%.
B. The longer the first menses are delayed, the more likely it is that the first cycle is ovulatory.
C. Continuous suckling and elevated prolactin levels lead to inadequate luteal function during the first cycle.
D. Exclusive breastfeeding: the first bleeding generally precedes ovulation return; if ovulation occurs, it is generally inadequate for conception [21].
II. Nonnursing mothers
A. The Earliest possible menstruation occurred 4 weeks postpartum.
B. Most women menstruate with the aid of 1/3 month postpartum.
C. go back to menstruation
1.6 weeks postpartum: 40%
2.12 weeks postpartum: 65%
3.24 weeks postpartum: 90%
The earliest possible ovulation was three½–5 weeks postpartum.
E. Ovular cycles occur in 50% of the patients with the first menstrual length postpartum.
F. Early postpartum ovulation can also arise overdue in the menstrual cycle: Shortening of the secretory segment and more tendency toward abnormal menses.
G. go back of ovulation
1.6 weeks postpartum: 15%
2.12 weeks postpartum: forty%
3.24 weeks postpartum: 75%.
III. Amenorrheic Non Nursing Mothers: Go Back To Ovulation
A.12 weeks postpartum: 20%
B. sixteen weeks postpartum: 40%.
Milk Composition during the Ovulatory Menstrual Cycle
Acute adjustments in the composition of milk throughout the ovulatory menstrual cycle in lactating women were studied by Hartmann and Prosser23 in women during lactational amenorrhea, taking oral contraceptives, and throughout the ovulatory menstrual cycle. Milk samples were collected from the breasts of each animal for 28 days.
Two acute changes were observed during the ovulatory menstrual cycle. For the 5 to 6 days before ovulation and the 6 to 7 days after ovulation, the sodium and chloride values changed from 4.6 mM Na and 11.1 mM Cl to 10.1 and 22.0, respectively, and lactose and potassium decreased. The concentrations of lactose, Cl, K, and Na remain relatively constant during lactational amenorrhea, ovulatory menstrual cycles, and in women taking oral contraceptives. The authors concluded that an increase in the permeability of the mammary epithelium was affected by changes related to ovulation. Perhaps the first acute change in composition is associated with the final stages of follicle maturation and the second with the regression of the corpus luteum during the ovarian menstrual cycle .66 Nutritional status had virtually no effect on amenorrhea except in extreme cases. In a study of Guatemalan women, maternal energy supplements did not shorten the duration of lactational amenorrhea; however, supplementation of breastfed infants shortened amenorrhea by reducing suckling. A difference exists between postpartum and nutritional amenorrhea: true nutritional amenorrhea is predictable based on the height/weight ratio; locational amenorrhea is hormonal, and when it occurs, nutrition has only a trivial role [31].
Breastfeeding and Birth Interval
Among Kung hunter-gatherers, long intervals pass between births, which has puzzled investigators because the tribes are well-nourished, have low fetal wastage, and do not employ contraceptives or prolonged abstinence. Kung only eats what they hunt and gather for. The farmers had no prior agricultural experience. They leaned and spared. They have late menarche (approximately 16 years of age), their first pregnancy at age 18, and early menopause at approximately 40 years, leaving 24 reproductive years during which they produce 4.4 children, which, with some perinatal deaths, replaces society. This compares to an industrial society, where productive years begin at 11 years and end at 51 years. Konner and Worthman35 report that the Kung has unusual temporal patterns of nursing characterized by highly frequent nursing bouts with short space between nursing’s. The Kung nurses worked several times an hour, with only 15 minutes at most between bouts, which lasted only 15–120 seconds each. Serum estradiol and progesterone levels are correspondingly low. Infants are always in the immediate proximity of their mothers until they are weaned at approximately 312 years of age during the gestation of a new sibling.
In Nigeria, the duration and frequency of Breastfeeding and postpartum amenorrhea are comparable in that Nigerians breastfeed for 16.5 months with a frequency of 4.5 times a day. The mean duration of amenorrhea was 12.5 months. Amenorrheic mothers who were lactating had lower serum estradiol and lactic dehydrogenase levels. A significant association was observed between hyperprolactinemia and amenorrhea. The incidence of amenorrhea decreased in parallel with the incidence of hyperprolactinemia [19, 25].
Suckling was the most important factor inhibiting the return to ovulation.49 during duration was the first factor studied in Edinburgh. Mothers who ovulated while breastfeeding had introduced two or more supplementary feeds per day and had reduced suckling to less than six times per day, with 60 min or less of suckling time per day. Basal prolactin level was < 600 mU/L. The mothers who did not ovulate until after 40 weeks postpartum breastfed the longest, suckled the most intensely, maintained night feeds the longest, and introduced supplement feeds the most slowly. The prolactin levels remained substantially greater than 600 mU/L [20].
Another review of the effects of hormonal contraceptives on lactation by Hull 43 concludes that a significant number of reports indicated a decrease in milk yield. The description of severe growth failure in nursing, even leading to “contraceptive marasmus,” in Egypt and Tunisia is a cause for concern.
Most large studies on birth interval and its relationship with feeding methods have been conducted in developing countries. However, Rosner and Schulman60 reported on 112 Orthodox Jewish women from metropolitan New York with 266 birth experiences. The women strictly adhered to biblical and rabbinic laws that prohibited birth control. They were well-nourished, middle-class, educated women who breastfed on demand (210 infants) for a mean duration of 10.7 months, with 177 infants receiving formula less than once per week. Significant positive correlations were found with the duration of lactational amenorrhea, which increased as the breastfeeding duration increased. Delays in starting solids, continuation of night feeding, and postponement of other liquid feeds were associated with prolongation of the birth interval. The investigators found a longer mean duration of lactational amenorrhea (8.6 months) and mean birth interval (22 months) than other studies because of the more intensive feeding patterns Contraception during Lactation [26-30].
Medical Eligibility Standards for Contraceptive Use
The WHO’s clinical eligibility standards for Contraceptive Use present tips for policymakers to help rationalize the provision of various contraceptives based on the most up- to-date records available for the protection of people with certain health situations. The document covers the subsequent circle of relative planning methods: low-dose blended oral contraceptives (COCs), mixed patch (P), combined vaginal ring (R), blended injectable contraceptives (CICs), progestin-most effective drugs (POPs), depot medroxyprogesterone acetate (DMPA), norethindrone enanthate (Internet-EN), levonorgestrel (LNG), etonogestrel (ETG) implants, emergency contraceptive tablets (ECPs), copper-bearing intrauterine gadgets (Cu IUDs), levonorgestrel-liberating IUDs (LNG-IUDs), copper IUD for emergency contraception (E-IUD), barrier techniques (BARR), fertility cognizance-primarily based methods (FAB), lactational amenorrhea method (LAM), coitus interrupts (CI), and lady and male sterilization (STER).
The purpose of this file is to provide insurance and selection makers and the scientific community with a hard and rapid set of hints that can be used to develop or revise extensive country guidelines on clinical eligibility standards for contraceptive use [77, 78].
Lactational Amenorrhea Method
The Bellagio Consensus Conference11 on breastfeeding as a family-making plan approach established that a mother who is absolutely or nearly simply breastfeeding her little one and remains amenorrheic might also have greater than 98% safety from pregnancy in the first six months postpartum. nine, This was changed and codified as a family planning technique for the subsequent year at a meeting of a global convention at George Washington University. The primary examination of the approach consistent with se determined that the pleasant girl in an examination of 422 center-beauty city women in Chile has grown to become pregnant because of the use of the LAM as the handiest method for pregnancy avoidance in the first six months, 13–27, with a protecting charge of 90–nine.5%. Menses as an indicator of ovulation have been studied, with data amassed not only at the onset of menses but also on urinary hormone assays. Among ladies who menstruated in advance of 6 months postpartum, 77% of cycles have been ovulatory, and the lag between an ovular first menses and the next over- state has emerged as 15.7 weeks. Then again, after Six months postpartum, the proportion of ovular first menses declined to 22%, and the lag to ovulation declined to 7.3 weeks. The mean interval between the first observed menses and ovulation was 8.4 weeks in the first 6 months and only 0.1 weeks after 6 months postpartum [25,30 ,13, 37].
A significant distinction should be made between token breastfeeding with early solids and more rigid feeding schedules and ad-lib breastfeeding around the clock with no solids until the infant is 6 months old.6The amount and frequency of sucking are closely related to continued amenorrhea. When a breastfed infant sleeps through the night at an early age, requiring no suckling for approximately 6 h at night, the suppressive effect on menses diminishes. It has also been shown that if an infant uses a pacifier rather than receiving non-nutritive sucking at the breast, ovulation suppression is diminished.The degree of fertility inhibition associated with breastfeeding has decreased remarkably since the time of hunter-gatherers. She points out that fertility rates vary; population and socioeconomic factors, urbanization, and nutrition influence not only breastfeeding patterns but also associated ovarian quiescence. Lactational amenorrhea can protect against pregnancy for the first six months, even in well-nourished women who are giving the infant some supplemental foods (Figure 20-7). The efficacy was remarkably good. Details the reestablishment of menses in breastfeeding women using LAM. Bellagio and Beyond: Breastfeeding and LAM in Reproductive Health was published in 1997 as a final report of many years of work worldwide involving the use of LAM.13 It was concluded that the efficacy of LAM is well- established in prospective studies. Policy support is still needed to institute an additional method that increases the family planning choices of postpartum women [37, 17, 10].
Studied a population of women in Baltimore in comparison to a group from Manila. Those in the Baltimore group were older, more educated, more frequently employed, and had fewer children. Women in Manila breastfed more frequently (at 10 weeks postpartum: 11.4 feeds in Manila versus 7.1 feeds in Baltimore). The mean duration of amenorrhea was 31.7 weeks, versus 26.3 weeks, and the mean delay before the first ovulation was 38 and 27 weeks (Manila and Baltimore, respectively). The frequency of suckling episodes was most strongly associated with ovulation in the Baltimore population, in which small declines in breastfeeding were sufficient to permit the return of ovarian activity. In contrast, women in Manila maintained high sucking rates even when solids were introduced. More Baltimore women (49%) than Manila women (31%) ovulated before six months. There was no simple algorithm, however, to predict ovulation [21, 69].
Reported the effects of age at the introduction of complementary foods to breastfed infants on the duration of lactational amenorrhea in Honduran women. Introducing food at 4 months significantly affected the likelihood of amenorrhea at 6 months, but not thereafter. However, this effect was not observed if the breastfeeding frequency was maintained. The most significant determinant of lactational amenorrhea was time spent breastfeeding (minutes per day), which was negatively associated with the infant’s energy intake from complementary foods [15].
In a large prospective study of the duration of lactic-national anovulation and amenorrhea in well-nourished Australian women—members of the Nursing Mothers’ Association of Australia who breastfed for a long time determined that breastfeeding by myself is not an effective form of contraception because all women resumed ovulation at the same time as breastfeeding. They compared breastfeeding women who had unprotected intercourse with breastfeeding girls who had unprotected sex only during lactational amenorrhea, and who adopted other contraceptive measures after the resumption of menstruation. The simplest 1.7% became pregnant during the primary 6 months of amenorrhea, followed by 7% after 365 days, and 13% after 24 months [65].
Subsequent research on this method has shown its high efficacy in many countries. Comparisons of follicular development and hormonal profiles are critical to expertise in lactational amenorrhea. There is a profound disassociation between follicular increase and follicular endocrine activity, which shows an alteration within the stimulus-reaction dating at the follicular stage, Serum FSH polymorphism for the duration of lactational amenorrhea was also studied by who concluded that FSH heterogeneity can be one of the critical factors contributing to incomplete follicular improvement and ovulation throughout lactational amenorrhea [72, 73, 74].
The herbal suppression of ovulation during early lactation and concomitant amenorrhea resulted in using one-of-a-kind or almost special breastfeeding seasoned, vide 98% or higher protection in opposition to pregnancy. Three situations are essential: amenorrhea, intense breastfeeding day and night time, and up to six months of exceptional breastfeeding postpartum. LAM can be used to time the introduction of any complementary approach (barrier, etc.; figure 20-9); it isn't just for users of natural own family planning [37-40]. 1.9 Natural Family Planning Although lactation provides protection early in the postpartum period, women who are not fully breastfeeding and are interested in avoiding conception should be informed of their options. If she does not want to use contraceptives, medications, or devices, she should be instructed on the external signs of ovulation, including the ovulation method, LH-releasing hormone, and home tests [31, 36].
Figure 20-9: Temperature, mucus, and cervical assessments during lactation to identify ovulation. (Courtesy National Family Planning of Rochester, New York
Pregnancy rates and fertility-related behavior of users of the ovulation method were studied prospectively in Kenya and Chile in groups of breastfeeding women. The rate of unplanned pregnancies was less than 1% in the first six months. The rate of unplanned pregnancies increased after menses began, and supplementary food was added to infants’ diets. The rates were compared with those of non-lactating women who also used the ovulation method to avoid pregnancy. Women breastfeeding followed the method closely, although clients who had not used the ovulation method before pregnancy had an increased incidence of unplanned pregnancies [38].
One of the difficulties in studying the use, effectiveness, and continuation of natural family planning methods is that terms have always been imprecise and markers have been different between studies.48 The method is effective when properly used (failure rate of 3.4%) but unforgiving when use is imperfect (failure rate of 84.2% in the first year).22 Unplanned pregnancy rates rise among breast-feeders after menses return compared with the rates for those who use thermal or secretion surveillance methods when not lactating. The increased pregnancy rate was related to poor compliance and understanding of the “rules” of the method [22].
The symptothermal method of fertility awareness during lactation has been studied in Canada. A special postpartum chart was designed to record morning temperatures, cervical mucus, and other signs of fertility or infertility on dates and postpartum days. Breastfeeding intensity was recorded. There were 54 breastfeeding experiences among the 47 women whose ages ranged from 20 to 39 years old. Parity ranged from 1 to 7, with an average of 3.3. The duration of full breastfeeding averaged 3.6 months (range,3 weeks–8 months). The duration of partial breastfeeding ranged from 2 months to 28 months, with an average of 8.8 months. These mothers found that, in general, they could accurately predict fertility during breastfeeding. During times of weaning or a change in suckling pattern, special caution was suggested, during which the mothers watched for signs of first ovulation [49, 50].
The effectiveness of periodic abstinence in lactating women shows that long periods of lactational infertility can be identified by either a lack of mucus or a continuous, unchanging mucus flow.
Cervical mucus accepts, filters, prepares, and releases sperm for successful transportation to the egg for fertilization. The advancing sperm must penetrate the mucous structure or the small interstices between the mucous macro molecules. The interstices were the largest in the peri ovulatory phase of the menstrual cycle. As ovulation resumes, irregular mucous patterns that are difficult to interpret occur, and, therefore, prolonged abstinence is required. The pregnancy rate using this method was 9.1 per 100. Because two-thirds of the 82 women studied were breastfeeding, many of the ensuing postpartum cycles may have been ovulatory or had an inadequate luteal phase, thus helping to maintain a low pregnancy rate [54].
Studies on cervical secretions alone (mucous patterns) during lactation have indicated that the same signs in mucus are reliable during lactation. Charting is carried out in the usual manner, and feeding is recorded. A woman who follows her postpartum pattern should be seen every 2 weeks for guidance until her pattern is well documented. The couple should make careful observations of when the infant sleeps through the night, the mother reduced breastfeeding frequency, the infant began solid foods, the infant began other liquids or bottles, and illness occurred in either mother or baby. Abstinence or LAM use was advised until the situation was clear. If there has been no previous ovulation or menstrual action when weaning begins, ovulation may occur quite quickly [75].
Although contraceptive methods such as barrier methods and “the pill” have a statistically better record in avoiding pregnancy, that is a moot point for a woman for whom these methods are not an option for religious or medical reasons.1 It is important that a clinician be as well informed about natural child spacing as possible so that the best advice can be provided. Ideally, the woman has used natural family planning before the pregnancy, so she is familiar with her patterns, but it is more urgent that she knows how to check her mucus, cervix, and temperature and is not trying to learn about her fertility signs during lactation. Natural family planning programs across the United States are gaining experience with lactating women using the ovulation method and are available to assist lactating women.28 Further information can be obtained from the National Office of Natural Family Planning, 8514 Brad moor Drive, Bethesda, MD 20817- 3810, (301) 897-9323 if no local office is available; http://www. boma-usa.org and http:// www.teenstar.org are the websites for the Billings Ovulation Method Association in the United States. Natural Family Planning International can be reached at www. nfpandmore.org or P.O. Box 11216, Cincinnati, OH 45211 [38- 50].
In a carefully designed study conducted in Chile by at the Pontificia Universidad Catolica de Chile Department of Obstetrics and Gynecology and by members of the faculty at the Johns Hopkins University School of Hygiene and Public Health, 419 postpartum women were enrolled in the Natural Family Planning Program and were taught how to record their observations. The purpose of this study was to define cervical mucus patterns concerning time since delivery, time of first bleeding, frequency of feeding, the introduction of supplements and solid foods, and time of weaning. The diaries of 110 women with detailed records were selected for critical evaluation; 49 were reported, and the preliminary observations were evaluated by Barker.1Two characteristics of mucus (sensation and observation) were charted each day, along with the women’s breastfeeding patterns. Only seven women had previously used natural family planning. None of the women menstruated before 4 months postpartum, while 10% had their first menses. By the fourth month, 50% of the women had mucus detected, and by the seventh month, 50% had their first menses. Mucus was observed approximately two months before the first menses. As women moved from total breastfeeding to partial and complete weaning, the duration of mucus episodes increased. The duration of mucus approached normal during weaning [55, 56].
Rural women exposed to breastfeeding education programs prenatally and postpartum breastfed more and used fewer bottles than the comparison group, which had no training on lactational infertility. However, no difference was seen in postpartum amenorrhea. Ovulation or pregnancy measures were not performed. Rural women tend to breastfeed optimally and naturally. Supplementary feeding affects the return of menses and ovulation, as demonstrated by a study conducted in rural China. There was a positive correlation between the start of solid food intake, the return of menses, and the first ovulation. A Cochrane review of lactational amenorrhea for family planning was provided by Van der who found 459 relevant studies, of which only 159 investigated the risk of pregnancy during lactation and 14 were included. The duration of lactational amenorrhea among women using LAM was very different between the populations studied. They could not determine if LAM made a difference beyond amenorrhea while fully breastfeeding [55, 71, 72, 76].
Touch Sensitivity and Ovulation
In the search for a simple method of identifying ovulation during lactation, urinary pregnanediol, estrogen, and breast sensitivity were measured in six breastfeeding and six bottle-feeding normal women. Two-point discrimination and touch sensitivity were also measured. The mean duration of amenorrhea among breast feeders was 24.3 weeks (range 14–35 weeks) and among bottle feeders was 7.5 weeks (range 6–14 weeks). However, these findings were not indicative of ovulation. Touch sensitivity tended to decrease over several months as lactation progressed. This change is gradual and difficult to detect because it has no practical value in determining ovulation [51-54].
Oral Contraceptives and Lactation
The significant issues related to lactation and the use of oral contraceptives are the potentially adverse effects of oral contraceptives on milk production, uterine involution, and the growth and development of breastfed infants.48 A single case of breast enlargement in a breastfed male infant whose mother began taking norethynodrel with ethinyl estradiol 3-methyl ether (Enovid) on the third day postpartum was reported. Breast enlargement begins in the third week of life. The mother noted that her milk was not as “rich” and started supplements in the second week. Nursing was discontinued at approximately 4 weeks of age, and the infant's breasts returned to normal within 2–3 weeks. The additional risks of thromboembolism, hypertension, and cancer to the mother have also been extensively discussed in the literature. These events occurred with early administration of high-dose products [70].
In a study of over 900 Latina women who had developed gestational diabetes during pregnancy, they were noted to develop postpartum diabetes if they were given a progesterone-only oral contraceptive compared with those who received combinations of oral contraceptives or non-hormonal contraception. This observation requires further follow-up. A Cochrane review of combined hormonal versus non-hormonal versus progestin- only contraception during lactation was inconclusive, and it was decided that existing trials were insufficient to establish any effect of hormonal contraceptive therapy on milk quantity and quality. The WHO recommendation regarding the use of progestin-only pills states that a delay of onset for 4 to 6 weeks postpartum is necessary. A depot medroxyprogesterone-only contraceptive providing highly effective, long-acting, reversible contraception, usually dosing every 12 weeks, was studied by concluded that it was not a significant risk for breastfeeding or the baby. The risk of pregnancy outweighed all other considerations [58, 59, 70].
Other Hormonal Contraceptive Use
The impact of the distribution of oral contraceptives on breastfeeding and pregnancy status in rural Haiti indicated that it did not alter breastfeeding patterns in women who began the pill at 8 to 9 months postpartum. Pregnancy prevalence also decreased as a result.64 Studies of progestin-only pills beginning at 6 weeks postpartum showed that progestin-only pills do not suppress gonadotropins or affect ovarian follicular development.
The contra-captive effect is believed to be mediated through local actions of the endometrium and cervix in normally menstruating women.50 Birth control for breastfeeding women remains open to discussion. A Cochrane review of the literature found that it was grossly inadequate, despite numerous articles published on hormonal and non-hormonal birth control utilized postpartum during breastfeeding. They found only five trials adequate for discussion, but the dropout rate of the participants was a concern. The authors found no major differences in infant growth or weight gain due to hormonal birth control. They stated that information is too limited to determine whether women should use hormonal birth control. However, the impact on breastfeeding and infants remains unclear [47].
Implants and Injections
Progestin-only products are recommended for contraceptive use during lactation (Norplant System, Depo-Provera injections and mini pills, and progestin once-only pills). The use of these methods has not been associated with adverse effects on infant growth or development, and may even increase the volume of milk (39, 62) (Table 20-5).
Injectable DMPA administered as a contraceptive in the immediate postpartum period was reported to be a safe and effective alternative method with no deleterious effects on the mother’s milk supply or the infant’s growth for women in New Delhi, India. Because the loss of milk supply after the injection has been documented, this study by of 100 women is significant. However, it is important to note that the injection was not administered until the milk supply was well-established. Some of the injections were not administered for 10 days. Thus, the establishment of a good milk supply before injection is important for the outcome of this study. Practitioners in lactation medicine know that depot injections and the insertion of intrauterine devices can affect milk supply, despite claims to the contrary. The mother must be warned of and allowed to make informed decisions. DMPA should not be used for at least 6 weeks if there is a history of depression.
In a study of the effects of levonorgestrel (Nor-plant) in breast milk on thyroid activity, compared infants of mothers with implants and those of women assigned to intrauterine device (IUD) use. Hormones (levonorgestrel) in breast milk significantly decreased the infants’ thyroid-stimulating hormone (TSH) levels at 3 months, and the TSH levels were even lower at 6 months of age. The higher the levonorgestrel levels, the lower the TSH levels in infants. It is recommended that the progestin- only method should not be initiated until 6 weeks postpartum, on the premise that the theoretical danger to infants from exogenous steroids has passed by this time. The practice of injecting Depo- Provera immediately after delivery can interfere with lactation. This is dependent on the dramatic natural decline in postpartum progesterone [2].
Steroids are not well bound in plasma and are not well conjugated by the liver or excreted by the immature kidneys of the neonate. Exogenous hormones may compete for receptor sites with natural hormones in the liver, brain, or other tissues. As an infant’s liver and kidneys mature, these issues disappear. Medications that contain estrogen suppress milk production. With the many alternatives available, it should never be necessary to discontinue breastfeeding to initiate contraception. The growth and development of breastfed infants whose mothers received implants of Norplant containing levonorgestrel or injections of norethindrone enanthate were studied and compared with those whose mothers used IUDs. The breastfeeding performance was similar. The infants were also similar in growth rate, development, and general health. Similarly, a group of mothers was given a vaginal ring that released 10 mg of “natural” progesterone every 24 hours, producing a maternal serum level of 4 ng/mL, which results in only a minimal amount in the milk and is not absorbed by the infant's gut. These infants also had normal growth and development and remained in good health [26, 10, 24, 63].
LH-releasing hormone agonists for contraception have also been tested in nine fully breastfeeding women beginning 6 weeks postpartum. They received 300 mg of an LH-releasing hormone agonist (buserelin) intranasally once a day for the duration of their breastfeeding. Urinary excretion of LH, estrone, and pregnanediol was compared with that of nine control breastfeeding women. No ovulation occurred in the treated group, and seven of the nine untreated control subjects had one to six. Ovulations. The LH-releasing hormone has the potential as a safe, acceptable method of contraception while breastfeeding, according to [19].
Home tests to monitor fertility are available; however, women must be tested during lactation and especially during the transition period when breastfeeding frequency and feeding duration are changing. Ovarian follicular dynamics can be accurately monitored through the non-instrumented analysis of daily estrone conjugates in urine samples at home.36 Readings can be affected by urine osmoclarity, either high or low, giving false positive or false negative results, respectively. Controlling the intake of fluids would guard against this. Two algorithms for initiating contraceptive treatment are shown in Figures 20-7 and 20-10 [56, 57].
Intrauterine Devices and Other Contraceptive Methods
Various alternatives to oral contraceptives exist and have different degrees of reliability (Table 20-6). The IUDs (95% to 98% effective), cervical caps and diaphragms (85% to 88% effective), condoms (80% to 85% effective), and vaginal suppositories, jellies, or creams (80% effective) have no known contraindication during breastfeeding because no chemicals are absorbed. The only contraceptive that is 100% effective is abstinence.
A study of 2271 postpartum women who had IUDs inserted between 1976 and 1981 and were followed for 6 to 12 months was reported, with careful attention to details of lactation. Data were analyzed separately for IUDs inserted immediately after birth (within 10 minutes of placental expulsion). The results of this analysis indicate that IUD insertion for breastfeeding women would be appropriate either immediately after delivery or much later (42 days or more postpartum). When inserted immediately postpartum, the delta loop and delta T were modified by adding projections of chromic sutures, which helped the device remain in the uterus. The sutures biodegrade in 6 weeks, leaving a standard device in place. These authors report that breastfeeding is not a contraindication to IUD insertion, with no increased expulsion rate. Conversely, the presence of an IUD has no adverse effect on lactation. The appropriate time for insertion should be selected to predate anticipated ovulation but guarantee patient compliance. A study showed that the use of the TCu- 380A IUD in breastfeeding women resulted in fewer insertion- related complaints and lower removal rates for bleeding and pain in the first 12 months postpartum than in non-breast feeders. No intrauterine perforations were reported in either group. Copper IUDs are usually smaller, so there is a little problem with the effects of a let-down on the uterus [24].
A group of 32 women hospitalized for uterine perforation necessitating Tran’s peritoneal IUD removal and a matched control group of 497 women who had worn IUDs uneventfully were compared. Of the women in the study, 97% were postpartum, compared with 68% of the control subjects. Of the parous study group, 42% were lactating, and of the parous control subjects, 7% were lactating when the IUD was inserted. The risk of perforation was 10 times greater in lactating women than in non-lactating women, unrelated to the time of the insertion postpartum. In another group hospitalized for difficult trans- cervical IUD removal, the risk was 2.3 times greater for lactating women.24 The authors recommend caution, not abandonment of the procedure, during lactation because they believe the IUD is the best form of artificial contraception during lactation. The ideal candidate is a woman who wants reversible contraception to space births or limits the size of the family, especially breastfeeding parous women in a monogamous relationship [20, 10].
The Technical Guidance and Competence Group of the United States Agency for International Development (USAID) and the WHO recommend IUD insertion immediately postpartum as soon as the placenta is removed, whether by vaginal or cesarean delivery. They point out that it must be done by a specially trained physician. From 10 minutes to 48 hours after delivery, however, the expulsion rate is high. If this window of opportunity is missed or no urgency exists, it is best to wait. Although immediate insertion is possible, a copper-T device may be safely inserted 4 or more weeks postpartum in breastfeeding women. Using the withdrawal technique minimizes the risk of perforation. Other IUDs should not be inserted until 6 weeks postpartum.
The IUD is a long-acting, reversible method of contraception with expulsion rates of 5–15 per 100 woman-years of use when used as a post-placental method immediately after a cesarean section. An IUD does not affect breastfeeding. These experiences were reported by Gold Stuck and Steyn from South Africa. The use of barrier and spermicidal products is an alternative contraceptive method during early lactation before hormonal methods or IUDs are introduced. These capital-dependent methods do not affect lactation. The lubrication supplied by way of the spermicidal- handled condoms has the advantage of contributing lubrication, while the hypes-estrogenic vagina is distinctly dry in association with lactation. A spermicide is more powerful than a condom but is good enough early postpartum, while relative infertility is a gift. A diaphragm cannot be properly outfitted for six to eight weeks postpartum, so it isn't always encouraged all through this era, with or without lactation [77, 78].
Abstinence
Many cultures and societies have taboos on sexual intercourse for nursing mothers as an effective manner of spacing youngsters. Generally, there aren't any scientific contraindications to sexual relationships in the course of lactation. In a look at contraceptive use in America, among white girls, 34% have been no longer sexually active in the first month postpartum, 12.5% within the second month, and 4.3% in the course o the third month. Among black girls in the survey, 25% were not sexually energetic in the first month, 8.1% in the second, and 4.2% in the third. Contraceptive use among those sexually active becomes absent in 16%, 12.2%, and 13.8% of the first, second, and third postpartum months, respectively, among whites. Among blacks, no technique was used by 27.3%, 22.7%, and 22.3% within the first, 2nd, and 1/3 postpartum months, respectively [59-66].
Sex and the Nursing Mother Sexual Assault Related To Sucking
If one examines normal grownup girls regarding the menstrual cycle, sexual sex, pregnancy, childbirth, and lactation, one observes that these events are all encouraged via the interaction of the same hormones not just estrogen, progesterone, testosterone, FSH, and LH, but also oxytocin and prolactin. The breast is thought to respond at some point at all these levels, enlarging before menstruation, all through pregnancy, earlier than orgasm, and at some stage in lactation. The nipples additionally respond during these phases. Furthermore, the uterus contracts in the course of childbirth, orgasm, and lactation. Body temperature rises at some point during ovulation, childbirth, orgasm, and death. As pointed out in Chapter 3, oxytocin is essential to the allow-down reflex at some stage in lactation. Oxy-toxin levels additionally rise at some stage in orgasms and exertions, and oxytocin causes the uterus to agree and the nipples to grow to be erect. File other similarities in ladies, together with sensory consistency with caption and emotional reactions, in the course of these activities [52, 54].
The Psycho-Physiological Similarities between Lactation and Coitus Are As Follows
1. The uterus contracts.
2. The nipples become erect.
3. Breast stroking and nipple stimulation occur.
4. The emotions experienced involve skin changes (vascular dilatation and raised temperature).
5. Milk let-down (or ejection) reflex can be triggered.
6. The emotions experienced may be closely allied.
7. An accepting attitude toward sexuality may be related to an accepting attitude to breastfeeding (and vice versa).
Women also report hot flashes in association with some feeds, especially at night. This phenomenon has been studied by who looked at the oxytocin effect. Initiation of breastfeeding-ing was accompanied by an increase in skin conductance resulting in increased skin temperature, especially of the breast. The pattern is similar to menopausal hot flashes [45].
Given the biological and hormonal similarities of lactation to the other events in the sexual cycle of adult women, it is not surprising that some women experience some form of sexual gratification during suckling on certain occasions. In a study of 111 par- parturient women, only 24 of which breastfed, Masters and Johnson46 reported that sexual arousal was experienced during suckling on some occasions. The exact incidence of this response is unknown, but it is thought to be uncommon. Nursing mothers may have an element of guilt surrounding these experiences and underreporting. That guilt may lead to early weaning in some cases. For some women, the breasts are highly erogenous. The handling and manipulation of the breast necessary during lactation by both mother and infant can, in the right but unpremeditated circumstances, be stimulating. The majority of women who enjoy breastfeeding have no feelings or responses to the stimulation of the breast that could be construed as sexual arousal, although they enjoy breastfeeding and the intimacy with their infant that it provides. The erotic response to nursing the infant has no significance in terms of being normal or abnormal. The decline of breastfeeding because of feelings of shame, modesty, embarrassment, and distaste has been reported and interpreted as indicating that breastfeeding is viewed as a forbidden sexual activity. For such women, any sexual allusions and excitement accompanying breastfeeding are not permissible and cause shame. Such attitudes are more common in lower social groups and need to be considered in counseling mothers about breastfeeding pre partum or when premature weaning takes place. Major changes in the number of women who breastfeed may not be possible until society can accept the breast in its relationship to nurturing the infant and as an object of less sexual ambivalence.
The sensuousness of breastfeeding has been the topic of discussion in women’s magazines as more has been written about women and their bodies. For the well-educated, well-read woman who breastfeeds her infant because she intellectually arrives at the decision, such discussions are an avenue of increased knowledge. Others may still be uncomfortable about breastfeeding if it is apt to be “pleasurable.” The physician may sense this discomfort in a patient prenatally through her responses to bodily changes during pregnancy. Cultural attitudes are an important part of this response and are deeply ingrained in an individual by the time she reaches the age of parenting. Professionals need to be sensitive to the patient and cautious about imposing cultural change on a patient while still being alert to the need for information and openness. A woman who experiences any erogenous reaction to breastfeeding, especially with an older child who may inadvertently roll the nipple while feeding, should not be criticized, but the phenomenon should be explained and discussed openly by the physician.
Sexual Activity of Nursing Mothers
A review of the limited data available on lactating women in the study by Masters and Johnson, does indicate that in their group of 111 postpartum women, the nursing mothers were more eager than no nursing mothers to resume sexual relations postpartum. The data were independent of the fear of pregnancy. They report that this interest was apparent 2 to 3 weeks postpartum. Individual reports through a questionnaire indicate that 30% of nursing mothers believed their sexual relationships were improved, and only 2.5% believed they were worse postpartum. The individual testimonies of nursing mothers indicate they had a better feeling about themselves as well as their relationships with their husbands and family in general. In a study of sexual behavior during pregnancy and lactation, the desire returned after 4 weeks for most women, long before they thought it was safe. The longer they had been married and the more children they had, the sooner the interest returned and the sooner they felt it was safe. No change in interest or enjoyment occurred with weaning [64, 47].
In studies of recovery after childbirth, the longer duration of breastfeeding (more than 5 months) has been associated with a longer duration of awareness of perineal damage (dyspareunia) during inter-course and a longer amenorrheic period in some women. High prolactin levels and decreased libido have also been observed in women with evidence of continued vaginal discomfort. In the clinical study of lactational amenorrhea in 422 women in Chile; however, recorded the incidence of intercourse to be one to two times a week, beginning 4 weeks postpartum [55,56].
The frequency of coitus during breastfeeding was studied at four sites: Birmingham, United Kingdom; Montreal, Canada; Sydney, Australia; and Manila, Philippines. 74 The frequency was lower than reported in other studies of married women, ranging from 4 to 30 episodes a month while averaging three to five times a month. Rates were not correlative to the number of children but were related to maternal age, being slightly more frequent in younger women. The resumption of coital activity in these populations was more variable, with a median of 8 weeks postpartum and 75% resuming activities by the end of the third postpartum month. Thus, “normal” encompasses a broad range. More general observations indicate that although some women may have increased interest in sexual relations while nursing, others may experience no interest at all for 6 months or so. Whether this results from the satiation of the mother’s needs for intimate relationships and stimulus through nursing, general fatigue, or fear of pregnancy is debatable.46 Sexual stimuli may trigger the ejection reflex, and milk ejection may hurt some men. A practical solution to spraying milk during love-making is feeding the infant or expressing some milk beforehand. The total knowledge of nursing and suckling as a biological phenomenon will help couples understand such reactions and thus avoid inappropriate psychological responses.
The conflict in some adult men over their role regarding the nursing mother’s breasts is usually a result of guilt or upbringing. There is no need to advice against fondling the lactating breast during lovemaking, although physicians have often imposed rigid restrictions on sexual activity in lactating women. No scientific basis for such a restriction exists, and no difference in the incidence of infection or mastitis is associated with such activity. Unusually restrictive protocols are often imposed on patients without medical indication.
It is helpful to discuss with lactating women that the hormonal effect on the vagina may be excessive dryness with an increase in dyspareunia. With the abrupt withdrawal of gonadotropins and ovarian hormones and elevation of prolactin at the time of delivery of the infant and placenta, the vaginal epithelium becomes thin and atrophic.57 normally, the vagina and ectocervix are lined with stratified squamous epithelium, which is multilayered and protective. It is also responsive to ovarian hormones. The greatest maturation and thickness occur around ovulation in response to peak estrogen secretion. During pregnancy, progesterone inhibits the maturation of the epithelial cells. The vaginal lining retains its thickness, but cells do not fully mature because the effect of progesterone overtakes the effect of estrogen on the epithelium and cervical mucus; both hormones are abundant during pregnancy.
The lowered ovarian hormones that cause vaginal dryness and a lack of cervical mucus during lactate production lead to discomfort during intercourse. The dryness responds to locally applied lubricants and tends to improve over time. A sudden change may reflect ovulation. The breast that is being stimulated by feeding frequently may not be as sensitive during lovemaking. Usually, this, too, is transient. Physicians should perhaps remind mothers that some adjustments to attend to fathers’ needs may be necessary.
Sexual Abuse and Baby Bottom
In most cases, sexual abuse or any type of abuse during childhood takes its toll for a lifetime. Sexual abuse during adult life, especially during a relationship that results in pregnancy, impact a mother’s ability to accept the pregnancy, endure the labor, and mother the child. The role of abuse is recognized as significant in all phases of women’s pregnancies and postpartum periods. Anthropologists have identified abuse as a significant thread in the lives of low-income women living in the most inhumane conditions in the world. Those studying childbirth issues and breastfeeding have noted a relationship between alleged inadequate milk and abuse, as well as an inability to breastfeed. Chin and Solomonik9 have analyzed the term “inadequate” not just as a description by the woman for her milk supply but as a metaphor for the lives of low-income women in the United States who have been identified in numerous statistical reports as the least likely to breastfeed.9 Chin and Solomonik9 note that everything about their lives is inadequate: education, income, health services, and life span. Their lives are saturated with violence, a lack of safety, and fear, even in their own homes. Chin and Solomonik9 suggest an agenda to explore the relationship between these social inadequacies and the forces that compel these women to choose the less optimal infant feeding by bottle. Childhood sexual abuse is not limited to families in poverty but has been reported in as many as 20% of children.56 Sexual abuse can have short- and long-term effects that manifest in various symptoms, including health problems, behavior problems, post- traumatic stress disorder, and interpersonal difficulties. Amnesia for the original abusive events can develop until the events of childbirth and subsequently putting the newborn to the breast trigger an anguished flashback. Kendall-Tackett described the long-term effects of sexual abuse, which she divided into five domains: emotional situations- As more information is brought forward, we learn of successful breastfeeding with mothers with a history of child abuse by a family member. Coles11 interviewed eleven women and identified four key themes: “enhancement of the mother-baby relationship; validation of the maternal body; splitting of the breasts’ dual role as maternal and sexual objects; and exposure and control when breastfeeding in public.” A case report published by Beck3 vividly described the original abuses, the victims’ labors and deliveries, and breastfeeding that triggered panic attacks, disassociation, and flashbacks. After extensive interviewing of 18 women who were sexually abused by family members before the age of 16, Coles and Jones (12) reported two key themes: safety issues for survivors and their babies in clinical encounters and wishing for better and safer clinical experiences because of feelings of pain, fear, blame, helplessness, and guilt in the health care environment. The authors suggest important steps to make the health care experience more tolerable for these women, who fear the intimacy of this care. Many of these suggestions are appropriate for all patients [3, 9, 11, 61].
Severe physical violence between intimate parts news during pregnancy is a major risk factor for early cessation of exclusive breastfeeding. This correlation has been made in many cultures around the world. Using a health services survey, the Conflict Tactics Scale, investigated premature cessation of breastfeeding in Brazil. They concluded that severe physical violence during pregnancy was an important risk factor for early cessation of exclusive breastfeeding. They suggest that healthcare workers who deal with lactating women need to be trained beyond the biological aspects of lactation to include the maternal, psy- geologic dimensions. Similar findings were reported from multiple African countries (Ghana, Kenya, Liberia, Malawi, Nigeria, Tanzania, Zambia, and Zimbabwe) by Misch and Yount. Partner violence was associated with early breastfeeding cessation. Screening for intimate partner violence victimization both prenatally and postpartum may mitigate the potential intergenerational effects of violence, as the authors recommend. Sexual assault has a pervasive negative effect on a new mother’s sleep quality and risk of depression report Kendall- Tackett, studied over 6000 moms with young infants using an online survey. Even though assault records had poor outcomes, the consequences were much less intense for the mothers who breastfed than those who bottle-fed or mixed-fed. Abuse of any kind has long-term effects. Breastfeeding seems to trigger repressed bottle-fed memories of the occasion and becomes tough for a few ladies. Surely, the manner isn't always completely understood. A health practitioner, starting during pregnancy and then postpartum, plays an essential role in assisting an affected person to cope. The difficulty may work unidentified until a pediatrician enters the image. in addition to screening for depression, a pediatrician has to be alert to a probable record of abuse, at the same time as carrying out care of the little one [29].
Nursing all through pregnancy and Tandem Nursing
Being pregnant can and does arise whilst a lady is lactating. While it does occur, it produces some questions. There’s no want to wean the first toddler from the breast, that's regularly ordered with the aid of a health practitioner. It’s far viable to lactate for the duration of being pregnant after which to have two infants at the breast postpartum. This is now sufficiently common to be called tandem nursing.
The quantity of nourishment provided to the first infant at the breast depends on age and other supplements. Whilst an infant at the breast is only a few months vintage while being pregnant takes place, there may be a few causes to maintain breastfeeding for the benefit of the little one until it is time to wean to solids and other drinks at 6 months of age or so. This toddler could be about 12 months old whilst the new little one arrives and, if still on the breast, might also have needed over the mother’s ability to offer. Issue has been expressed that the older infant will take plenty of the nourishment needed by the brand-new infant. In some societies, its miles believed that a suckling little one will “take the spirit” from the newly conceived fetus; consequently weaning is mandated once pregnancy is confirmed in these groups.
The milk produced straight away postpartum via the mom who by no means stopped nursing seems to be colostrum. The kangaroo has been discovered to have a teat with mature milk for the older offspring and a treat for the brand-new offspring which calls for significantly special nourishment. Such a provision no longer exists for humans. Mothers who need to hold both babies at the breast have proven that it can be achieved without any apparent impact on the nourishment of the brand-new infant. Counseling any such mother has to recall the mother’s sources to acquire adequate rest, nourishment, and psychological support to withstand the added demand, physically and mentally, on her.
If the first child will be older than 1 year of age when the new infant arrives, the need for physical nourishment is minimal, and continuation at the breast is more for security and psychological benefits. This is referred to as comfort nursing and may continue for several years (see Chapter 10). Abrupt weaning should be avoided, and consideration should be given to the impact of separation when the mother is confined during the birth of the new infant. This is an argument for 12-hour hospitalization for delivery for women who request it. The first few days of colostrum are most vital for the new infant, and the supply is not infinite; therefore, priorities need to be set concerning the older child. The new baby should be nursed first. Some older infants reject colostrum.
The growth rate of children weaned during a subsequent pregnancy was compared with that of children weaned at the same age from no pregnant mothers in a longitudinal study in Bhutan by Bohler and Bergstrom, who followed 113 children closely for the first 3 years of life. The period of overlap for lactation and pregnancy was 5 months (median), increasing by 1 week for each month of reduction in the birth interval. When a child stopped breastfeeding during the mother’s subsequent pregnancy, the growth rate was reduced during the last months before termination of breastfeeding compared with children weaned at the same age from no pregnant mothers and with children who continued to breastfeed.
In a study of 503 La Leche League members, Newton and Theotokatos53 reviewed breastfeeding during pregnancy practices and found that 69% of breastfeeding children weaned spontaneously when the mother became pregnant. Many of the children may have been at an age to wean even without an inter current pregnancy.
Moscone and Moore51 conducted a questionnaire survey of 57 women who were concurrently pregnant and breastfeeding. The main reasons given for continuing breastfeeding after conception involved the emotional needs of the breastfeeding child; 43% of the children continued to breastfeed throughout pregnancy and after the birth of a sibling. The main reason for mother-initiated weaning was breast and nipple pain. When the child weaned during the pregnancy, it occurred during the second trimester and seemed to be associated with diminished milk production. Three pregnancies terminated in spontaneous abortions (a rate higher than in the general population of 5%). The ages of the children at the onset of pregnancy varied from 4 to 42 months. The feeding pattern was one to eight times a day for less than 5 minutes to more than 30 minutes. A descriptive study of 2617 women in a prenatal clinic in Egypt revealed that 95% had previously breastfed and 25.3% conceived while breastfeeding; 4.4% conceived in the first 6 months, 15.1% while still amenorrheic, and 28.1% while exclusively breastfeeding. Only 4 pregnancies (1.5%) occurred when all the prerequisites for the LAM method were present. In Egypt, especially in rural areas, infants are breastfed for at least two years. Pregnancy while breastfeeding is common in Egypt.64.
Impact of Nursing during Pregnancy
Among rural Guatemalan women who were part of a nutrition supplementation trial, 253 of the 504 pregnant women had another pregnancy overlap while breastfeeding (50.2%); 41.4% of mothers with concurrent pregnancy and lactation continued to breastfeed into the second trimester and 3.2% into the third trimester. These “overlap” mothers received more supplements. The authors stated that overlap resulted in short recuperative periods (less than 6 months) requiring increased supplement intake and reduced maternal fat stores. The energetic stresses and short recovery time did not significantly affect fetal growth. It appeared that the mother buffers the energy stress, protecting fetal growth.44 Significant decreases in bone mineral density do occur during breastfeeding when calcium demands are the greatest.41 These changes are reversible and do not persist after a subsequent pregnancy, according to Prentice who studied calcium utilization and reproduction-related osteoporosis extensively. They indicate that extended periods of breastfeeding and closely spaced pregnancies are unlikely to have a lasting effect on bone mineral status and osteoporosis when a mother is healthy and nourished. Although short birth intervals and breastfeeding during pregnancy further deplete fat stores in a malnourished mother, healthy, well-nourished women fare well and replenish their stores during a subsequent pregnancy [42,43,44 56].
Infant Health
Although pregnancy during lactation can cause flavor and volume changes that lead to early weaning, milk still provides immunologic benefits. This is demonstrated by Bohler and Bergstrom4 among women in Bhutan, where abrupt weaning caused diarrhea, stunted growth, illness, and even death. Research in India showed that overlapping breastfeeding and pregnancy in a malnourished mother produced growth retardation in the older child. Healthy infants in the United States derive significant nutritional and immunologic benefits in the second year and beyond. The risk to a nurse depends on a child’s age, other diets, and the amount of human milk available.
Fetal Health
The nutritional status of a mother is key to adequate fetal growth. Varying results of fetal growth patterns are reported related to a mother’s nutritional status before pregnancy. A significant issue is the viability of the pregnancy. Breastfeeding stimuli trigger oxytocin release, and concern focuses on the potential for initiating uterine contractions and fetal loss. Studies of oxytocin sensitivities in pregnancy and the state of oxytocin receptors during early pregnancy are graphically illustrated in Figure 3-19. The uterus is insensitive until close to 40 weeks in most women. It is well documented that nipple stimulation can be as effective as intravenous Pitocin for inducing labor at term.
Risk of Fetal Loss or Preterm Labor
Retrospective studies of fetal loss and preterm loss suggest that breast stimulation could be the source in some women. The Miscarriage Clinic in London states that once a pregnancy is clinically detectable, breastfeeding should pose no added risk of pregnancy loss, and there is no reason to link breastfeeding and miscarriage. Most obstetricians, however, caution against sex during pregnancy in women with a history of fetal loss or premature birth. Breast stimulation is equally prohibited in such circumstances. The stimulus is not exactly equal. In addition, the nurses will be nursing several times per day. Twin pregnancies and other multiples are considered high-risk, and weaning is usually recommended if the mother is nursing.
The decision to continue nursing when a new pregnancy is normal with no factors for a high-risk pregnancy should rest on the comfort level of the mother and child. Breast pain can be improved by wearing a supportive bra and repositioning during breastfeeding. The decrease in the volume of milk is usually not remediable, but milk usually returns toward the end of pregnancy and is completely regenerated at delivery. [49].
In a study of 68 Peruvian women who breastfed during pregnancy and 65 who had not breastfed during the pregnancy, Marquis 44 reported that on day 2 postpartum women who breastfed had higher concentrations of lactose and lysozyme but lower lactoferrin than women who did not breastfeed. At 1 month, immunoglobulin A was lower among women who breastfed. The infants of the women who breastfed were five times as likely to have respiratory symptoms (p < 0.05) in these early weeks [44, 45].
Women who were 2 months pregnant and weaning their infants showed a progressive loss of secretory activity by the mammary gland, seemingly due to an inhibition of milk secretion that overrides the stimulus provided by the infants. These results were compared to the milk of women weaning without pregnancy [52, 53].
Many of the changes in child-rearing practices in recent years have increased freedom and responded to human needs. When carried to extremes, instant gratification becomes a right rather than a privilege. Sometimes a mother may need help seeing that she does not need to feel guilty if she decides to wean the older child. If it is only an occasional feeding or suckling experience for added security, especially when security is threatened by the arrival of a new infant, it may be tolerable in terms of endurance for the mother, and she may agree willingly. When, however, continuing nursing becomes a strain or is painful or stressful, she should feel free to stop. When a mother feels real resentment toward the older child who is nursing, it is time to wean gently but firmly. If such a situation can be anticipated, it is probably easier for an older child to be weaned before the delivery of the new infant.
Sore nipples are the most stressful symptom during pregnancy and may be the first sign of the new pregnancy as the hormonal milieu changes. There is no specific treatment, although having the toddler re-positioned at the breast may ease the discomfort. If the toddler is old enough to understand, asking him or her to nurse more gently or “more softly” may help. The soreness may last for the first trimester or the entire pregnancy until the new baby is born.
Decision: Sorting Out Personal Feelings
Little is in the medical literature about nursing during pregnancy and tandem nursing. A mother must make up her mind if there are no medical contraindications. Much depends on the age of a child, the nursing pattern, and the nutritional and emotional needs of the child. Medical indications to wean during pregnancy are uterine bleeding, signs of preterm labor, or failure to gain enough weight during pregnancy.
Tandem nursing for some women is too much “touching,” especially when the infant and the toddler are a year or more apart in age. Nursing twins or triplets presents a similar situation for some mothers.
As with any such decisions to wean, a physician should work this out in frank discussion with a mother (and father, if available) so that any misgivings, resentment, or feeling of failure can be dealt with openly. Many patients automatically suspect the physician as being antagonistic to breastfeeding if the physician suggests weaning. Even when the reason is purely but urgently medical, the discussion should be open and include options and alternatives and their risks. Weaning is part of a baby’s growing up, but it is sometimes part of a mother’s moving on as well.
While tandem nursing requires ordinary hygiene, it is usually not necessary to limit each child to one breast because any infections or colds have spread before the first symptom. There are a few precautions. If one child has thrush, assigning one breast may keep it under control. If the older child develops a herpetic lesion or cold sore, he or she must not nurse. The newborn could acquire a potentially fatal herpes infection.
The dilemma of tandem nursing and weaning an older child has been dealt with in other societies with various manipulations, such as painting the breast with pepper or bitter herbs to make it taste terrible. The mother may leave the child with other caregivers. The provision of love and affection during this difficult adaptation for the child is what makes the difference between a traumatic occasion and a step toward growing up. Equally important is the provision of some opportunity for a mother to express her concerns and doubts during the process to her physician, who should be neither judgmental nor unduly rigid in the medical care plan.
Research Method
In this section, you will describe the methods and approaches you used to conduct your research on reproductive function during lactation. This might include details about: Study Design: Explain whether it's a clinical study, an observational study, an experimental study, or another type. Justify why this design is suitable for your research question.
Participants: Describe the characteristics of the participants, such as age, health status, and any relevant criteria for selection.
Data Collection: Explain how you collected the data. This might involve surveys, medical examinations, hormone level measurements, etc.
Variables: Outline the independent and dependent variables in your study. For instance, the independent variable could be lactation, and dependent variables might include hormone levels, menstrual cycle changes, etc.
Procedures: Detail the procedures followed, including any interventions or treatments applied to the participants.
Data Analysis: Describe the statistical or analytical methods used to analyze the collected data. This might involve descriptive statistics, correlation analysis, t-tests, ANOVA, etc.
Results
In this section, you present the findings of your research. This could include:
Descriptive Data: Present basic descriptive statistics of the data, such as means, standard deviations, and frequencies.
Statistical Findings: Report the results of any statistical tests you conducted, indicating whether the differences or relationships observed are statistically significant.
Figures and Tables: Use graphs, charts, and tables to visualize your results. For example, you might show hormone level changes over time during lactation or any other relevant patterns.
Discussion
Here, you interpret the results and provide context for your findings:
Comparison to Previous Research: Discuss how your results compare to existing literature on reproductive function during lactation. Highlight similarities and differences.
Mechanisms and Explanations: Offer potential explanations for the patterns you observed. Discuss the biological, hormonal, or physiological mechanisms that might underlie these findings.
Implications: Talk about the implications of your findings. How might they contribute to our understanding of reproductive function during lactation? Are there practical applications or clinical implications.
Limitations: Address any limitations of your study, such as sample size, methodology constraints, or potential biases.
Conclusion
Restate Findings: Briefly recap the main findings of your study. Significance: Emphasize the importance of your findings in the broader context of reproductive science.
Future Research: Suggest areas for future research based on the gaps or questions that your study revealed. Remember that these sections should flow logically and provide a clear narrative about your research on reproductive function during lactation. If you have specific data or information to include, you can adapt the outline accordingly.
Acknowledgment
The completion of this research project would not have been possible without the Contributions and support of many individuals and organizations. We are deeply Grateful to all those who played a role in the success of this project.
We would also like to thank My Mentor. Naweed Imam Syed Prof. Department of Cell Biology at the University of Calgary and Dr. Sadaf Ahmed Psychophysiology Lab University of Karachi for their invaluable input and support throughout the research. Their insights and expertise were instrumental in shaping the direction of this project Declaration of Interest.
I at This Moment Declare That
I have no pecuniary or other personal interest, direct or indirect, in any matter that raises or may raise a conflict with my duties as a manager of my office Management Conflicts of Interest.
References
- Barker, D. C., Gray, R. H., Labbok, M. H., & Kambic, R. (1986). Use of natural family planning by breastfeeding women. Breastfeeding and Natural Family Planning. Ed. M. Shivanandan, Bethesda, MD: KM Associates, 23-31.
- Bassol, S., Nava-Hernandez, M. P., Hernandez-Morales, C., Trujillo-Macias, A. M., Lopez-Lozano, M. R., & Recio, R. (2002). Effects of levonorgestrel implant upon TSH and LH levels in male infants during lactation. International journal of gynecology & obstetrics, 76(3), 273-277.
- Beck, C. T. (2009). An adult survivor of child sexual abuse and her breastfeeding experience: A case study. MCN: The American Journal of Maternal/Child Nursing, 34(2), 91-97.
- Bøhler, E., & Bergström, S. (1996). Child growth during weaning depends on whether mother is pregnant again. Journal of Tropical Pediatrics, 42(2), 104-109.
- Brambilla, F., & Sirtori, C. M. (1971). Gonadotropin- inhibiting factor in pregnancy, lactation, and menopause. American Journal of Obstetrics and Gynecology, 109(4), 599-603.
- Brown, R. E. (1982). Breast-feeding and family planning: a review of the relationships between breast-feeding and family planning. The American journal of clinical nutrition, 35(1), 162-171.
- Campino, C., Torres, C., Rioseco, A., Poblete, A., Pugin, E., Valdés, V., ... & Seron-Ferré, M. (2001). Plasma prolactin/ oestradiol ratio at 38 weeks gestation predicts the duration of lactational amenorrhoea. Human reproduction, 16(12), 2540-2545.
- Chayen, B., Tejani, N., & Verma, U. (1986). Induction of labor with an electric breast pump. The Journal of reproductive medicine, 31(2), 116-118.
- Chin, N. P., & Solomonik, A. (2009). Inadequate: a metaphor for the lives of low-income women?. Breastfeeding Medicine, 4(S1), S-41.
- Cole, L. P., McCann, M. F., Higgins, J. E., & Waszak, C.S. (1983). Effects of breastfeeding on IUD performance.American journal of public health, 73(4), 384-388.
- Coles, J. (2009). Qualitative study of breastfeeding after childhood sexual assault. Journal of Human Lactation, 25(3), 317-324.
- Coles, J., & Jones, K. (2009). “Universal precautions”: Perinatal touch and examination after childhood sexual abuse. Birth, 36(3), 230-236.
- Cooney KA, Nahmias SR: Bellagio and past: breast-feeding and LAM in reproductive health, Washington, D.C., 1997, Institute for Reproductive fitness, Georgetown College.
- Delvoye, P., Demaegd, M., & Robyn, C. (1978). Serum prolactin, gonadotropins, and estradiol in menstruating and amenorrheic mothers during two years' lactation. American journal of obstetrics and gynecology, 130(6), 635-639.
- Dewey, K. G., Cohen, R. J., Rivera, L. L., Canahuati, J., & Brown, K. H. (1997). Effects of age at introduction of complementary foods to breast-fed infants on duration of lactational amenorrhea in Honduran women. The American journal of clinical nutrition, 65(5), 1403-1409.
- Diaz, S., Aravena, R., Cardenas, H., Casado, M. E., Miranda, P., Schiappacasse, V., & Croxatto, H. B. (1991). Contraceptive efficacy of lactational amenorrhea in urban Chilean women. Contraception, 43(4), 335-352.
- Diaz, S., Cardenas, H., Brandeis, A., Miranda, P., Schiappacasse, V., Salvatierra, A. M., ... & Croxatto, H.B. (1991). Early difference in the endocrine profile of long and short lactational amenorrhea. The Journal of Clinical Endocrinology & Metabolism, 72(1), 196-201.
- Eslami, S. S., Gray, R. H., Apelo, R., & Ramos, R. (1990). The reliability of menses to indicate the return of ovulation in breastfeeding women in Manila, The Philippines. Studies in family planning, 21(5), 243-250.
- Fraser, H. M., Dewart, P. J., Smith, S. K., Cowen, G. M., Sandow, J., & McNeilly, A. S. (1989). Luteinizing hormone releasing hormone agonist for contraception in breast feeding women. The Journal of Clinical Endocrinology & Metabolism, 69(5), 996-1002.
- Gray, R. H., Campbell, O. M., Apelo, R., Eslami, S. S.,Zacur, H., Ramos, R. M., ... & Labbok, M. H. (1990). Risk of ovulation during lactation. The Lancet, 335(8680), 25- 29.
- grey RH, Campbell O, Eslamic S, et al: The return of ovarian function all through lactation: in consequences of studies from us and the Philippines, Biomedical demographic determinants, Oxford, 1993, Clarendon.
- Gross, B. A. (1991). Is the lactational amenorrhea method a part of natural family planning? Biology and policy. American journal of obstetrics and gynecology, 165(6), 2014-2019.
- Hartmann, P. E., & Prosser, C. G. (1982). Acute changes in the composition of milk during the ovulatory menstrual cycle in lactating women. The Journal of Physiology, 324(1), 21-30.
- HEARTWELL, S. F., & Schlesselman, S. A. R. A. H. (1983).Risk of uterine perforation among users of intrauterine devices. Obstetrics & Gynecology, 61(1), 31-36.
- Bennett, P., Matheson, I., Notarianni, L. J., Kane, A., & Reinhardt, D. (1996). Monographs on individual drugs. In Drugs and Human Lactation (pp. 75-532). Elsevier Science BV.
- Howie, P. W., McNEILLY, A. S., Houston, M. J., Cook, A.,& Boyle, H. (1982). Fertility after childbirth: Infant feeding patterns, basal prl levels and post-partum ovulation. Clinical Endocrinology, 17(4), 315-322.
- Howie, P. W., McNEILLY, A. S., Houston, M. J., Cook, A.,& Boyle, H. (1982). Fertility after childbirth: post-artum ovulation and menstruation in bottle and breast feeding mothers.Clinical endocrinology, 17(4), 323-332.
- Kambic, R. T. (1991). Natural family planning use- effectiveness and continuation. American Journal of Obstetrics and Gynecology, 165(6 Pt 2), 2046-2048.
- Mezrani, C. (2005). Breastfeeding and the Sexual Abuse Survivor. Journal of Human Lactation, 21(3), 370-370.
- Kennedy, K. I., Labbok, M. H., & Van Look, P. F. A. (1996). Lactational amenorrhea method for family planning. International Journal of Gynecology & Obstetrics, 54(1), 55-57.
- Kennedy, K. I., Rivera, R., & McNeilly, A. S. (1989). Consensus statement on the use of breastfeeding as a family planning method. Contraception, 39(5), 477-496.
- Kennedy, K. I., Short, R. V., & Tully, M. R. (1997). Premature introduction of progestin-only contraceptive methods during lactation. Contraception, 55(6), 347-350.
- Kennedy, K. I., & Visness, C. M. (1992). Contraceptive efficacy of lactational amenorrhoea. The Lancet, 339(8787),227-230.
- Kent, J. C., Mitoulas, L., Cox, D. B., Owens, R. A., & Hartmann, P. E. (1999). Breast volume and milk production during extended lactation in women. Experimental physiology, 84(2), 435-447.
- Konner, M., & Worthman, C. (1980). Nursing frequency, gonadal function, and birth spacing among! Kung hunter- gatherers. Science, 207(4432), 788-791.
- Kurz, K. M., Habicht, J. P., & Rasmussen, K. M. (1991). Influences of maternal nutrition and lactation on length of post-partum amenorrhoea. Journal of tropical pediatrics, 37, 15-18.
- Labbok, M. H. (1983). Breast-feeding and contraception. The New England journal of medicine, 308(1), 51.
- Stover, J., Bertrand, J. T., & Shelton, J. D. (2000). Empirically based conversion factors for calculating couple-years of protection. Evaluation Review, 24(1), 3-46.
- Labbok, M. H., Perez, A., Valdes, V., Sevilla, F., Wade, K., Laukaran, V. H., ... & Queenan, J. T. (1994). The Lactational Amenorrhea Method (LAM): a postpartum introductory family planning method with policy and program implications. Advances in contraception, 10, 93-109.
- Labbok, M. H., Stallings, R. Y., Shah, F., Perez, A., Klaus, H., Jacobson, M., & Muruthi, T. (1991). Ovulation method use during breastfeeding: is there increased risk of unplanned pregnancy?. American journal of obstetrics and gynecology, 165(6), 2031-2036.
- Laskey, M. A., & Prentice, A. (1999). Bone mineral changes during and after lactation. Obstetrics & Gynecology, 94(4), 608-615.
- Lasley, B. L., Shideler, S. E., & Munro, C. J. (1991). A prototype for ovulation detection: pros and cons. American journal of obstetrics and gynecology, 165(6), 2003-2007.
- me, FertilSteril 55:529, 1991.
- Marquis, G. S., Penny, M. E., Zimmer, J. P., Díaz, J. M., & Marín, R. M. (2003). An overlap of breastfeeding during late pregnancy is associated with subsequent changes in colostrum composition and morbidity rates among Peruvian infants and their mothers. The Journal of nutrition, 133(8), 2585-2591.
- Marshall, W. M. (1993). Psychophysiological aspects of lactation.
- Masters W.H., Johnson W.E.: The Human Sexual Response, Boston, 1996, Brown.
- McCann, M. F., & Potter, L. S. (1994). Progestin-only oral contraception: a comprehensive review. Contraception, 50(6 Suppl 1), S1-S195.
- McNeilly, A. S. (1997). Lactation and fertility. Journal of Mammary Gland Biology and Neoplasia, 2, 291-298.
- McNeilly, A. S. (2001). Lactational control of reproduction. Reproduction, Fertility and Development, 13(8), 583-590.
- Merchant, K., Martorell, R., & Haas, J. (1990). Maternal and fetal responses to the stresses of lactation concurrent with pregnancy and of short recuperative intervals. The American journal of clinical nutrition, 52(2), 280-288.
- Moscone, S. R., & Moore, M. J. (1993). Breastfeeding during pregnancy. Journal of Human Lactation, 9(2), 83-88.
- Newton, N., & Newton, M. (1967). Psychologic aspects of lactation. New England Journal of Medicine, 277(22), 1179-1188.
- Newton N., Theotokatos M.: 503 women breastfeeding during pregnancy. Does a person have psychobiological mechanisms of the psyche? Proc Fifth Int Congr Psychcosom Obstet Gymco, 1979, p. 845, 1986.
- Nilsson, S., Mellbin, T., Hofvander, Y., Sundelin, C., Valentin, J., & Nygren, K. G. (1986). Long-term follow-up of children breast-fed by mothers using oral contraceptives. Contraception, 34(5), 443-457.
- Perez, A., Labbok, M. H., & Queenan, J. T. (1992). Clinical study of the lactational amenorrhoea method for family planning. The Lancet, 339(8799), 968-970.
- Prentice, J. C., Lu, M. C., Lange, L., & Halfon, N. (2002). The association between reported childhood sexual abuse and breastfeeding initiation. Journal of Human Lactation, 18(3), 219-226.
- Prosser, C. G., Saint, L., & Hartmann, P. E. (1984). Mammary gland function during gradual weaning and early gestation in women. Australian journal of experimental biology and medical science, 62(2), 215-228.
- Rodriguez, M. I., & Kaunitz,A. M. (2009).An evidence-based approach to postpartum use of depot medroxyprogesterone acetate in breastfeeding women. Contraception, 80(1), 4-6.
- Rosner, A. E., & Schulman, S. K. (1990). Birth interval among breast-feeding women not using contraceptives. Pediatrics, 86(5), 747-752.
- Saadeh R., Benbouzid D.: Breastfeeding and infant Spacing: The importance of statistics accumulating for Public health coverage, Bull Global Health Organ
- Sarkar, N. N. (2008). The impact of intimate partner violence on women's reproductive health and pregnancy outcome. Journal of Obstetrics and Gynaecology, 28(3), 266-271.
- Sas, M., Gellen, J. J., Dusitsin, N., Tunkeyoon, M., Chalapati, S., Crawford, M. A., ... & Pinol, A. (1986). An investigation on the influence of steroidal contraceptives on milk lipid and fatty acids in Hungary and Thailand. Contraception, 33(2), 159-178.
- Selye, H., & McKeown, T. (1934). The effect of mechanical stimulation of the nipples on the ovary and the sexual cycle. Surg Gynecol Obstet, 59, 886-890.
- Shaaban, O. M., & Glasier, A. F. (2008). Pregnancy during breastfeeding in rural Egypt. Contraception, 77(5), 350-354.
- Short, R. V., Renfree, M. B., Shaw, G., & Lewis, P. R. (1991). Contraceptive effects of extended lactational amenorrhoea: beyond the Bellagio Consensus. The lancet, 337(8743), 715-717.
- Tankeyoon, M., Dusitsin, N., Chalapati, S., Koetsawang, S., Saibiang, S., Sas, M., ... & Zegers, L. (1984). Effects of hormonal contraceptives on milk volume and infant growth: WHO Special Programme of Research and Development andResearch Training in Human Reproduction. Contraception, 30(6), 505-522.
- Tay, C. C. K., Glasier, A. F., & McNeilly, A. S. (1996). Twenty-four hour patterns of prolactin secretion during lactation and the relationship to suckling and the resumption of fertility hi breast-feeding women. Human Reproduction, 11(5), 950-955.
- Taylor RS: physiology of the vagina and cervix in breast- feeding ladies In Shivanandan M., editor: Breastfeeding and herbal circle of relatives planning Fourth global symposium on herbal family planning, Chevy Chase, Md., 1985, KM Companion Publishers
- Technical steering/Competence group USAID/WHO: circle of relatives making plans methods: New steering document XXIV, No. 2, Baltimore, Md., 1996, Johns Hopkins College of Public Health
- Lopez, L. M., Grey, T. W., Stuebe, A. M., Chen, M., Truitt,S. T., & Gallo, M. F. (2015). Combined hormonal versus nonhormonal versus progestin-only contraception in lactation. Cochrane Database of Systematic Reviews, (3).
- Van der Wijden C, Brown J, Kleijnen J: Lactational amenorrhea for the circle of relatives planning (assessment), issue three, 2009, The Cochran Library.
- Velasquez, E. V., Creus, S., Trigo, R. V., Cigorraga, S. B., Pellizzari, E. H., Croxatto, H. B., & Campo, S. (2006). Pituitary–ovarian axis during lactational amenorrhoea.II. Longitudinal assessment of serum FSH polymorphism before and after recovery of menstrual cycles. Human Reproduction, 21(4), 916-923.
- Velasquez, E. V., Trigo, R. V., Creus, S., Campo, S., & Croxatto, H. B. (2006). Pituitary–ovarian axis during lactational amenorrhoea. I. Longitudinal assessment of follicular growth, gonadotrophins, sex steroids and inhibin levels before and after recovery of menstrual cyclicity. Human Reproduction, 21(4), 909-915.
- Visness, C. M., Kennedy, K. I., Gross, B. A., Parenteau- Carreau, S., Flynn, A. M., & Brown, J. B. (1997). Fertility of fully breast-feeding women in the early postpartum period. Obstetrics & Gynecology, 89(2), 164-167.
- Wade, K. B., Sevilla, F., & Labbok, M. H. (1994). Integrating the lactational amenorrhea method into a family planning program in Ecuador. Studies in Family Planning, 162-175.
- Wei, L. I., & Yi, Q. I. U. (2007). Relation of supplementary feeding to resumptions of menstruation and ovulation in lactating postpartum women. Chinese Medical Journal, 120(10), 868-870.
- WHO clinical eligibility standards for contraceptive use, Geneva, 2004
- International Fitness Corporation mission force for Epidemiological research on Reproductive fitness: Progestogen-best contraceptives for the duration of lactation I and II Infant Improvement, Contraception 50:55, 1994.


