Research Article - (2025) Volume 4, Issue 1
Recent Advancements and Persistent Challenges in the Management of Patients with Takotsubo Syndrome
Received Date: Dec 19, 2024 / Accepted Date: Jan 23, 2025 / Published Date: Jan 27, 2025
Copyright: ©Â©2025 Ioannis Leontsinis, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Leontsinis, I., Mantzouranis, E., Sakalidis, A., Ntalakouras, I., Dri, I., et al. (2025). Recent Advancements and Persistent Challenges in the Management of Patients with Takotsubo Syndrome. Int J Clin Med Edu Res, 4(1), 01-11.
Abstract
Background: Takotsubo Syndrome (TTS) is an increasingly recognized condition, initially perceived as benign, but now associated with considerable morbidity and mortality. Its pathophysiology remains elusive and despite advancements in understanding, management strategies remain inconsistent and lack support from randomized clinical trials.
Aims: This review aims to consolidate current evidence on the pathophysiology, diagnostic algorithms, treatment approaches, and prognosis of TTS, while highlighting the ongoing debates and challenges in its management.
Methods: A comprehensive review of recent literature was conducted, focusing on key studies, registries, and consensus guidelines.
Results: TTS is predominantly observed in postmenopausal women and is often triggered by emotional or physical stressors. The pathophysiology likely involves catecholamine surges, microvascular dysfunction, and inflammation. Diagnosis relies on imaging modalities like echocardiography and cardiac MRI, with the InterTAK criteria being the most widely accepted. Acute management focuses on stabilizing patients based on hemodynamic status, avoiding harmful interventions, and carefully selecting pharmacotherapy, despite mixed evidence for long-term benefit. Prognosis varies significantly, with worse outcomes associated with physical stress triggers, male gender, and comorbidities. Recurrence rates remain low but concerning, with inconsistent data regarding the effectiveness of the neuro-hormonal blockade medications and other therapies in preventing recurrence.
Conclusion: TTS poses significant clinical challenges due to its variable prognosis and lack of standardized treatment. Current management is largely symptomatic and supportive, with decisions tailored to individual risk profiles.
Keywords
Takotsubo Syndrome, Acute Cardiovascular Care, Acute Heart Failure, Sympathetic Nervous SystemIntroduction
TTS is an acute cardiac condition that closely mimics acute coronary syndrome at presentation and is characterized by transient and spontaneously reversible systolic dysfunction not directly attributable to unstable coronary artery disease [1,2]. This syndrome may involve different segments of the left ventricle or, in some cases, both the left and right ventricles, with severity ranging from mild presentations to severe cases complicated by cardiogenic shock or even death. Approximately two-thirds of cases are preceded by a stressful trigger, whether emotional or physical, leading to alternative designations such as "broken heart syndrome" or "stress cardiomyopathy." Additionally, the term "neurologically stunned myocardium" has been employed in earlier reports, highlighting the frequent association with central nervous system pathologies as predisposing factors. Another designation, "apical ballooning syndrome," reflects the characteristic left ventricular morphology during the acute phase.
Sato introduced the term "Takotsubo" to describe this typical apical variant, which has since become the most widely accepted term [3]. The term "cardiomyopathy" has fallen out of favour due to insufficient evidence supporting an intrinsic structural pathology directly affecting the myocardium. Instead, the term "syndrome" is more commonly used, acknowledging the variability in clinical presentation, severity, precipitating factors, and prognosis. Irrespective of the changing terminology, it is clear that this condition has drawn significant scientific attention due to its rising prevalence, a course more troublesome than initially believed, and the uncertainties surrounding its pathogenesis [2]. Despite these concerns, treatment approaches lack the support of randomized clinical trial data, leading to varied management strategies largely influenced by local expertise. This review seeks to present the latest findings on the pathophysiology, diagnostic criteria, treatment options, and prognosis of TTS, while emphasizing recent evidence and ongoing debates in the management of this increasingly recognized cardiovascular disorder.
Epidemiological Trends
TTS was initially identified in Asian populations, but subsequent reports have confirmed that it affects individuals across all ethnicities and age groups [1,3,4]. It predominantly occurs in postmenopausal women, with female representation ranging from 72.3% to 90.4% in various registries [5-10]. Although older age appears to correlate with a higher incidence of TTS, the underlying mechanisms predisposing women and elderly individuals to the syndrome remain unclear. The rising incidence of TTS hospitalizations is largely attributed to enhanced recognition and diagnosis of the condition [2,6,11].
TTS shares clinical and prognostic features with the acute coronary syndrome (ACS) population, with most cases presenting with chest pain, dyspnoea, or palpitations, while cardiac arrest occurs in 1.5% of patients [1,2,8,12,13]. However, significant differences exist regarding traditional cardiovascular risk factors. Khaloo et al. demonstrated that the prevalence of hypertension in TTS patients was comparable to that in those with STEMI and NSTEMI (55% vs. 55% vs. 43%, respectively), but TTS patients exhibited lower rates of diabetes mellitus (DM) and chronic kidney disease (CKD) (21% vs. 42% vs. 40% for DM and 10% vs. 27% vs. 38% for CKD, respectively) [14]. Similarly, Isogai et al. reported lower incidences of DM (17% vs. 28%), hypercholesterolemia (25% vs. 43%), and smoking (10% vs. 13%) among TTS patients compared to those with ACS [15]. Consistent findings have been reported in the InterTAK registry (DM 14%, hypercholesterolemia 31%, prior coronary artery disease 15%, smoking 20%), the SWEDHEART registry (DM 10%, hypercholesterolemia 25%, smoking 16%), the GEIST registry (DM 19%, hypercholesterolemia 42%, smoking 22%), and the RETAKO registry (DM 16%, hypercholesterolemia 52%, smoking 13%, CKD 5%) [8,13,16,17].
Interestingly, TTS registries tend to include fewer obese patients compared to the prevalence of obesity in the general population [6,18]. Several hypotheses have been proposed to explain these observations supporting an "obesity paradox" in TTS, though none have been conclusively validated. For instance, patients with TTS are more likely to have concurrent chronic or acute conditions affecting various organ systems, including the central nervous, respiratory, psychiatric, or endocrine systems [8]. Cancer is also increasingly recognized as a comorbidity in TTS survivors. Not only do a significant proportion of TTS registry patients have a history of cancer, but emerging evidence suggests that TTS patients are at an elevated risk of developing cancer during follow¬up compared to matched controls [2,19].
Pathophysiological Pathways Currently Under Consideration
The precise aetiology of TTS remains elusive, with several hypotheses having been proposed, some of which have since been abandoned due to insufficient evidence. Given its resemblance to acute myocardial infarction (AMI), early investigations focused on ischemic mechanisms. Hypotheses such as epicardial spasm and spontaneously recanalized thrombus were initially explored but are largely disregarded today. Although some cases report spontaneous multivessel spasm, this theory is insufficient as electrocardiogram (ECG) and echocardiogram findings often persist longer than the spasm itself. Additionally, multivessel spasm does not account for reverse (basal) TTS, particularly in patients who develop this atypical variant after previously experiencing the apical (typical) form [20]. Epicardial vessel thrombosis, identified via intravascular imaging in older case series, is not a consistent finding and also fails to explain reverse or midventricular TTS [21,22]. Notably, pathological studies reveal distinct patterns of myocardial injury between TTS and AMI, with contraction band necrosis observed in TTS versus coagulation necrosis in AMI [23]. Another debated concept is left ventricular outflow tract obstruction, which is frequently observed in the typical form of TTS but is generally considered a complication rather than a causative factor [2,24,23].
Microvascular and macrovascular dysfunction are common findings in TTS patients. An increased coronary angiography-derived index of microcirculatory resistance in TTS patients compared to those with unobstructed vessels in STEMI has been reported [25]. The slow coronary flow phenomenon has also been described in TTS [26]. In a small study, TTS patients exhibited reduced coronary flow reserve, which correlated with impaired myocardial perfusion reserve as assessed by magnetic resonance imaging (CMR), reflecting increased resting but reduced stress myocardial perfusion flow. Notably, perfusion reserve inversely correlated with myocardial oedema and regional wall motion abnormalities, suggesting that acute myocardial oedema and altered blood flow contribute to contractile dysfunction in TTS [27]. However, there is no definitive evidence to support that vascular flow impairments are a causative mechanism rather than a bystander phenomenon secondary to hypocontractility.
The role of the autonomic nervous system in TTS has gained significant attention in recent decades. A considerable number of cases are preceded by intense emotional or physical stress, leading to the hypothesis that TTS is mediated by sympathetic nervous system activity [2,12,23,28-31]. The link between stress and TTS is further supported by cases occurring during pheochromocytoma crisis, thyrotoxicosis, and catecholamine infusion. Functional imaging studies provide compelling evidence, showing increased metabolic activity in brain regions associated with emotion and autonomic regulation—such as the hippocampus, limbic system, basal ganglia, nucleus coeruleus, thalamus, hypothalamus, and brainstem—during the subacute phase of the syndrome [2,29,32,33]. Additionally, growing evidence suggests autonomic imbalance following the acute event [34-36], though a direct causative relationship remains unproven. Microneurography studies have documented changes in sympathetic activity during the disease course, with lower muscle sympathetic nerve activity (MSNA) recorded in the subacute phase compared to matched controls, followed by further reduction over time [37]. Compared to chronic heart failure patients, where sympathetic overactivity is maladaptive, TTS survivors showed higher bursts per minute, highlighting potential conflicting interpretations that warrant further study [38].
The role of β-adrenergic receptors (β-AR) in TTS pathology is also under investigation. Embryologically, sympathetic innervation is more pronounced in the basal segments of the left ventricle, while β-AR density is higher in the apical segments. In response to excess catecholamines, myocytes may switch from the usual G-stimulatory pathway, involving protein kinase A (PKA) phosphorylation leading to increased intracellular calcium concentration, to an inhibitory pathway that results in negative inotropy. While this theory partially explains the typical apical form of TTS, it does not account for midventricular or basal variants, especially in patients with recurrent events affecting different regions of the left ventricle. Moreover, cases of TTS induced by inotropes like milrinone, which bypass β-AR and increase cyclic AMP through phosphodiesterase-3 inhibition, challenge this theory further. Despite uncertainties regarding its primary role, this cardio-inhibitory mechanism may serve a protective function against mechanical and oxidative stress, ischemia, and mitochondrial dysfunction—factors that are pro-arrhythmic and pro-inflammatory [2,12].
Inflammation is another area of interest in TTS pathophysiology. Animal models suggest a key role for cellular immunity, which can be directly triggered by sympathetic stimulation, leading to a pro-inflammatory cascade and the release of interleukins 6 and 10—findings that require validation in human studies [2,12,23,39]. Additionally, reports of TTS triggered by deactivated viral vaccines or immune checkpoint inhibitors lend further support to the role of inflammation, though the stress associated with these conditions might also drive sympathetic activity. The variability in inflammatory response duration among patients may explain the persistence of mild heart failure symptoms in some individuals.
More recent studies suggest a link between chronic stress and inflammation in TTS. It is over two centuries ago since Dr. Everard Holm first correlated stress-induced sudden cardiac death with pathological evidence of coronary atherosclerosis [40]. Subclinical inflammation related to chronic mild stress may contribute to both TTS and autonomic imbalance [41]. TTS survivors experience elevated stress levels during the subacute phase, impacting their quality of life [42,43]. Cattaneo et al. reported increased functional integration between brain regions involved in interoceptive pain awareness and negative emotional states, further highlighting the interaction between stress and cardiovascular dysfunction [35].
Although the pathophysiology of TTS remains incompletely understood, current theories converge on a triad of sympathetic overdrive, impaired coronary vascular physiology, and inflammation (Figure 1). Genetic, hormonal, behavioural, and aging factors may also contribute, though evidence remains limited [39].
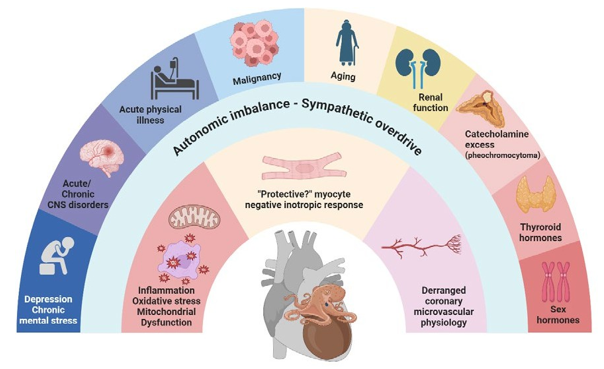
Figure 1: Emerging Pathophysiological Pathways in Takotsubo Syndrome. Image created with biorender.com
Gender Differences in Takotsubo Syndrome
Takotsubo syndrome (TTS) was initially thought to exclusively affect women, as early case reports primarily described female patients. Contemporary registry data confirm a striking female predominance, with women comprising 80–90% of all TTS cases. Notably, most affected women are postmenopausal, with these patients more likely to recall an emotional rather than a physical stressor preceding the event. Conversely, in younger patients, men are more frequently affected, and TTS is more commonly triggered by an acute physical illness.
The influence of circulating oestrogen on β-AR expression has been suggested as a potential mechanism underlying this gender disparity [2]. Animal models have shown that typical TTS can be induced in ovariectomized mice, with hormone replacement therapy attenuating the severity of the condition. However, similar findings have not been replicated in human studies. While multiple hypotheses have been explored to explain the observed gender differences, there is insufficient evidence to conclusively support a causative role for oestrogen deficiency in TTS.
Diagnostic Algorithm for Takotsubo Syndrome
The early recognition and the diagnostic approach to TTS has evolved significantly over the past decades. The InterTAK diagnostic criteria, developed through international consensus, are currently the most widely accepted diagnostic tool [23]. TTS is classified into four subtypes based on the segments of the left ventricle (LV) affected:
• Typical (Apical) Type: The most common form, accounting for over 80% of cases, involving apical and mid-segmental akinesia or hypokinesia.
• Midventricular Type: Representing 14.6% of cases, this subtype involves only the mid-segments of the LV.
• Basal Type: Rare, occurring in 2.2% of cases, characterized by hypokinesis or akinesis of the LV base.
• Focal Type: This more recently described subtype is characterized by localized systolic dysfunction without circumferential involvement.
Right ventricular involvement can occur in any subtype.
Unlike earlier criteria, the InterTAK criteria allow the coexistence of significant epicardial coronary artery disease, provided that the acute systolic dysfunction cannot be directly attributed to this condition. Additionally, previously excluded conditions, including acute neurological events (e.g., intracerebral haemorrhage) and pheochromocytoma, are now recognized as potential triggers for TTS [23].
Diagnosis requires demonstrating reversible systolic dysfunction and excluding acute myocarditis, preferably through magnetic resonance imaging (CMR). Various imaging modalities support this diagnostic process:
Echocardiography
The cornerstone for initial evaluation and follow-up, echocardi¬ography assesses systolic dysfunction, valve abnormalities, and complications. Contrast-enhanced echocardiography can detect intraventricular thrombi and left ventricular strain imaging high-lights regional wall motion abnormalities.
Cardiac Magnetic Resonance (CMR)
Imaging Superior to echocardiography for characterizing tissue characteristics, CMR typically reveals reversible damage (e.g., oedema) with minimal permanent scarring. CMR also better visualizes complications and right ventricular involvement. Guidelines from the European Association of Cardiovascular Imaging (EACVI) and the Japanese Society of Echocardiography recommend CMR within two months of the acute event to confirm the diagnosis. [44]. In clinical practice, the later a CMR is performed following the acute episode the higher the chances that typical changes (i.e, wall motion abnormality accompanied by oedema) may have resolved.
Coronary Angiography and Left Ventriculography
Coronary angiography, often with left ventriculography, is usually performed at presentation. When angiography shows non-obstructive coronary artery disease and ventriculography reveals features consistent with TTS (e.g., apical, midventricular, or basal wall motion abnormalities), the temptation to diagnose TTS is strong. However, it is recommended to adhere to the full diagnostic and follow-up algorithm until LV systolic function recovers and alternative diagnoses, such as myocarditis or myocardial infarction with non-obstructive coronary arteries (MINOCA), are excluded. CMR or echocardiography-derived strain imaging have shown promise in differentiating TTS from acute myocardial infarction, although larger studies are needed to validate these findings [45,46].
Additional Imaging and Diagnostic Tools
Coronary Computed Tomography Angiography (CCTA)
Useful in cases with low suspicion of true myocardial infarction, suspected TTS recurrence, or when an invasive procedure is contraindicated [44].
Nuclear Imaging
Techniques such as 123I-metaiodobenzylguanidine (MIBG) scintigraphy and FDG PET scanning can detect persistent abnormalities in sympathetic neurotransmission and glucose metabolism, respectively, beyond the resolution of systolic dysfunction, aiding in the diagnosis of delayed presentations [44].
Electrocardiography (ECG) and Biomarkers
Approximately 30% of TTS patients present with a normal ECG. ST elevation is also observed in around 30%, while other ECG abnormalities include QT prolongation, ST depression, and T wave inversion. Global T wave inversion, sparing only leads aVR and V1, has been suggested as characteristic ECG of TTS [47]. However, none of these ECG patterns are specific enough to definitively diagnose or exclude TTS. The InterTAK score integrates clinical and ECG data to estimate the probability of TTS at presentation [48].
Serum biomarkers lack sufficient specificity for TTS diagnosis. Although cardiac troponin and creatine phosphokinase (CPK) are typically elevated, the rise is disproportionately low compared to the degree of systolic dysfunction, as would be expected in myocardial infarction. In contrast, natriuretic peptides are often markedly elevated, reflecting the severity of systolic dysfunction, sometimes disproportionate to the clinical presentation. Other biomarkers such as microRNA, pro-inflammatory cytokines and copeptin are still under investigation [49,50].
Prognostic Tools and Severity Risk Factors in Takotsubo Syndrome
Short-term Prognosis and Complications
Initially considered a benign and self-limiting condition, TTS is now recognized for its substantial short- and long-term mortality and morbidity. Short-term complications are common and can be severe, occurring in about 35-45% of cases. Evidence of acute heart failure is observed in up to 35-45% of cases and may be complicated by cardiogenic shock in 10-20% of cases. Supraventricular and ventricular arrhythmias are not uncommon (i.e., atrial fibrillation (5-15%), ventricular tachycardia/ventricular fibrillation (4%), torsades de pointes (less than 2%), bradyarrhythmias/asystole (less than 2%). Left ventricular thrombus and peripheral embolism are observed in 2-5% of patients. Free wall or intraventricular septum rupture, tamponade, and pleural effusion are considered rare [2]. Despite progress in recognizing the severity of TTS, no disease-specific treatments have proven effective in mitigating these risks [10,12,51-53].
Long-term Prognosis and Mortality
Long-term mortality in TTS is primarily non-cardiac and reaches approximately 5-6% per patient-year [15,54]. The risk of any major cardiovascular event is about 10% per patient-year, a rate comparable to that seen in acute coronary syndromes (ACS) [55,56,51]. The recently published SWEDHEART study results report a 2% short-term mortality and a 15% long-term mortality, emphasizing that the TTS associated risk remains high despite any progress in cardiovascular care [13]. A study by Khaloo et al. found that while overall mortality in TTS is lower than in type I myocardial infarction (MI), TTS patients are more likely to develop cardiogenic shock and ventricular tachycardia than those with type II MI [14]. Interestingly, TTS cases share similar prognosis with patients experiencing true MI with not obstructive coronary arteries, whereas other MINOCA cases such as myocarditis or MINOCA with no scar in CMR present better outcomes [45].
Risk Factors Influencing Prognosis
Several sex-related factors may affect clinical outcome. Women, especially those who develop TTS after emotional stress, typically have a less severe course and better prognosis. [51]. Men, who are more likely to experience TTS after physical stress (e.g., acute CNS injury), face worse outcomes both during hospitalization and after discharge. TTS is more common in women but tends to be more fatal in men [57-59] .
Regarding other risk factors that have been proposed, The German-Italian-Spanish Takotsubo registry identified several predictors of long-term mortality, including advanced age, male gender, history of diabetes mellitus, malignancy, neurological disorders, chest pain on admission, and the use of angiotensin-converting enzyme inhibitors (ACEI) or angiotensin receptor blockers (ARB) [60]. In addition, patients with compromised renal function, high temperature or dyspnoea on admission, cardiogenic shock or thyroid dysfunction tend to present worse outcomes too [16,17,61-63]. ST changes on admission have been associated with higher complication rates especially in the context of ST elevation [66,55]. Microvascular dysfunction, indicated by high index of microcirculatory resistance (IMR) values or slow coronary flow, is also linked to worse outcomes [25,26].
With respect to arrhythmic risk, the generation of arrhythmias in TTS is thought to be linked to catecholamine surges, mechanical stretching of the atria and ventricles, and QT prolongation. QT prolongation may result from inflammation, epinephrine action, ischemia, oxidative stress, or oedema [6,9,12]. However, the association between QT prolongation and poor prognosis is debated. While some studies report no significant correlation, others suggest a higher burden of arrhythmias fatal or not, in patients with prolonged QT interval [64,65].
Prognostic Tools
A CMR-based tool, known as the PE2RT score, has been developed to predict outcomes in TTS. This score, derived from a retrospective cohort of 79 patients, incorporates the presence of pericardial and/or pleural effusion, right ventricular systolic dysfunction, and/or intraventricular thrombi to predict mortality and complications [66].
Quality of Life and Recurrence
Long-term impairment of quality of life is a concern for many TTS survivors, affecting both functional status and emotional well-being. Physical impairment in TTS mimics that seen in heart failure with preserved ejection fraction (HFpEF), although the prevalence and underlying pathophysiology are not fully understood [35,41,43,57,67,68]. Recurrence rates of TTS are reported to be around 1.8% per patient-year. The RETAKO investigators found a 4% recurrence rate within the first year of follow-up, with recurrent episodes being more common in patients with no identifiable stressor [69].
Current Treatment Practices of Takotsubo syndrome
The treatment approach for TTS is largely based on non-randomised, retrospective or observational data and obeys to a "first, do no harm" (primum non nocere) order. Since there is no evidence based standardized treatment protocol, management strategies vary across institutions and countries. The 2018 international consensus document outlines a pragmatic, individualized treatment plan focusing on symptom severity and patient needs. This can be summarised in figure 2, discussed in the following section:
Management of Acute Heart Failure (AHF)
Mild AHF
Patients with mild symptoms require short-term monitoring. Treatment may include renin-angiotensin system inhibitors (RASi) and beta-blockers, unless contraindicated. Non-selective beta-blockers are often preferred for their vasodilatory effects, although selective beta-blockers are recommended when left ventricular outflow tract obstruction (LVOTO) is present [2,12,70,71]. In cases of symptomatic LVOTO, hydration (if pulmonary congestion allows) and short-acting selective beta-blockers are recommended, while diuretics, vasodilators (e.g., nitrates), and intra-aortic balloon pumps should be avoided [70].
Moderate AHF
These patients should be monitored in a high-dependency unit, with diuretics as first-line therapy for symptom control.
Severe AHF or Cardiogenic Shock
Catecholamine or milrinone administration is generally avoided due to the risk of exacerbating the condition. Mechanical circulatory support is preferred, tailored to the patient's specific needs [70]. Patients with biventricular impairment may require extracorporeal membrane oxygenation (ECMO), while those with isolated left ventricular (LV) dysfunction may benefit from LV venting devices (e.g., intra-aortic balloon pump (expect for concomitant LVOTO), Impella, tandem heart or similar). Non randomized data support superiority of the Impella device [72,73].
Management of Arrhythmias and Thromboembolism
Medications or electrolyte disorders that may prolong QT interval are strongly discouraged. Tachyarrhythmia management is individualized. Direct current cardioversion and beta-blockers together with prompt anticoagulation when indicated are generally preferred. Temporary pacing can be considered in cases of symptomatic bradyarrhythmias. The implantation of permanent devices although not popular among TTS survivors [6,74], is generally not advised. The option of wearable automatic defibrillator can be considered for secondary sudden death prevention in rare cases waiting for cardiac function recovery and the completion of the diagnostic process. Systematic thromboembolism related to intraventricular thrombi more common due to the regional wall akinesia carries significant burden of morbidity, mortality and disability among TTS survivors. Anticoagulation treatment is generally advised in cases of severe LV impairment [70].
Long-Term Therapy and Recurrence Prevention
Despite the sympathetic overdrive in TTS, registry data have been inconsistent regarding the benefit of beta-blockers post-discharge. While studies like SWEDEHEART, Scotish and RETAKO found no significant benefit other studies showed improved outcomes with beta-blocker use especially in patients who presented with cardiogenic shock [13,75-78]. However, beta-blockers do not appear to reduce the risk of recurrence [17]. ACE inhibitors (ACEi) have been associated with better outcomes and reduced recurrence rates, supported by findings from the SWEDEHEART registry [13,70]. However, the Scottish registry found a neutral impact of these therapies [76]. The role of statins is debated, with conflicting data regarding their benefit in TTS [79]. Antiplatelet therapy has not been related with improved prognosis and can be considered in patients that coronary artery disease is revealed [80]. On the other hand, parenteral anticoagulation during hospitalization was found to be related with improved outcomes, likely reducing thromboembolic burden [13]. The BrokenHEART study is currently investigating the potential benefits of direct oral anticoagulant apixaban and adenosine infusion followed by dipyridamole administration for TTS patients [81].
In cases complicated by acute heart failure, it is reasonable that TTS management should align with standard heart failure (HF) guidelines. This includes considering foundational HF therapies like ARNI/RASi, MRAs, beta-blockers, and SGLT2 inhibitors, although there is still no robust, randomized evidence to support their use specifically in TTS. Since mental health seems to play a significant role in TTS, behavioural therapy is promising and increasingly recognized as part of the treatment approach [82].
Overall, TTS management remains a dynamic field with ongoing studies and evolving treatment strategies. A tailored approach considering the patient's specific clinical scenario is critical, given the variability in presentations and outcomes.
Figure 2: Treatment Approach in Takotsubo Syndrome. Image created with biorender.com
LVOTO: Left Ventricular Outflow Tract Obstruction, RAS-i: Renin-Angiotensin System Inhibitors, ARNI: Angiotensin Receptor/ Neprilysin Inhibitor, SGLT-2i: Sodium-Glucose Co-Transporter-2 Inhibitors, MRAs: Mineralocorticoid Receptor Antagonists, IABP: Intra-Aortic Balloon Pump, ECMO: Extracorporeal Membrane Oxygenation, EF: Ejection Fraction, AF: Atrial Fibrillation, CAD: Coronary Artery Disease, HTN: Hypertension, ACE: Angiotensin Converting Enzyme.
Conclusion
Takotsubo Syndrome remains a puzzling cardiovascular condition. Its intricate pathophysiology likely involves the neuro-hormonal brain-heart axis, alterations in coronary vascular function, and inflammatory activation, yet it is still not fully understood. Once thought to be a benign disorder, it is now recognized as being associated with substantial morbidity and mortality, necessitating prompt risk assessment and effective preventive and therapeutic measures—both of which are currently lacking. The prognosis of TTS varies widely and is influenced by factors such as the nature of the triggering stressor, patient comorbidities like renal impairment, thyroid dysfunction, malignancies, neurological disorders, or diabetes, as well as the presence of complications. Management strategies currently focus on stabilizing acute complications through personalized care tailored to hemodynamic requirements, avoiding triggers like catecholamines, and cautiously using treatments such as beta-blockers and RAS inhibitors, despite uncertain long-term benefits. Evidence remains mixed, particularly regarding the effectiveness of these therapies in preventing recurrence. While some studies suggest improved outcomes with ACE inhibitors, beta-blockers, anticoagulants, and statins, others show neutral results, highlighting the lack of consensus in post-discharge care. Until more definitive data becomes available, managing TTS will rely on vigilant patient monitoring, customized treatment approaches, and addressing the psychological factors that often play a critical role in this condition.
Funding Information
No funding was received with regards to this manuscript.
Competing Interests
The authors declare that they have no competing interests.
References
- Ghadri, J. R., Wittstein, I. S., Prasad, A., Sharkey, S., Dote, K., Akashi, Y. J., ... & Templin, C. (2018). International expert consensus document on Takotsubo syndrome (part I): clinical characteristics, diagnostic criteria, and pathophysiology. European heart journal, 39(22), 2032-2046.
- Omerovic, E., Citro, R., Bossone, E., Redfors, B., Backs, J., Bruns, B., ... & Lyon, A. R. (2022). Pathophysiology of T akotsubo syndrome–a joint scientific statement from the Heart Failure Association T akotsubo Syndrome Study Group and Myocardial Function Working Group of the E uropean Society of Cardiology–Part 1: overview and the central role for catecholamines and sympathetic nervous system. European journal of heart failure, 24(2), 257-273.
- Sato, H. T. H. U. T. (1990). Tako-tsubo-like left ventricular dysfunction due to multivessel coronary spasm. Clinical aspect of myocardial injury: from ischemia to heart failure, 56-64.
- Vazirani, R., Rodriguez-Gonzalez, M., Castellano-Martinez, A., Andrés, M., Uribarri, A., CorbI-Pascual, M., ... & RETAKO Investigators. (2024). Pediatric takotsubo cardiomyopathy: A review and insights from a National Multicentric Registry. Heart failure reviews, 1-12.
- Chong, T. K., Chen, J., Lyu, L., Wei, Y., Liu, Y., Wu, L., ... &Liu, K. (2023). Clinical characteristics and outcome correlates of Chinese patients with takotsubo syndrome: Results from the first Chinese takotsubo syndrome registry. International journal of cardiology, 387, 131129.
- Isogai, T., Matsui, H., Tanaka, H., Makito, K., Fushimi, K., & Yasunaga, H. (2023). Incidence, management, and prognostic impact of arrhythmias in patients with Takotsubo syndrome: a nationwide retrospective cohort study. European Heart Journal: Acute Cardiovascular Care, 12(12), 834-846.
- Alashi, A., Isaza, N., Faulx, J., Popovic, Z. B., Menon, V., Ellis, S. G., ... & Desai, M. Y. (2020). Characteristics and outcomes of patients with takotsubo syndrome: incremental prognostic value of baseline left ventricular systolic function. Journal of the American Heart Association, 9(16), e016537.
- Templin, C., Ghadri, J. R., Diekmann, J., Napp, L. C., Bataiosu,D. R., Jaguszewski, M., ... & Lüscher, T. F. (2015). Clinical features and outcomes of takotsubo (stress) cardiomyopathy. New England Journal of Medicine, 373(10), 929-938.
- El-Battrawy, I., Santoro, F., Stiermaier, T., Möller, C., Guastafierro, F., Novo, G., ... & Akin, I. (2020). Prevalence, management, and outcome of adverse rhythm disorders in takotsubo syndrome: insights from the international multicenter GEIST registry. Heart failure reviews, 25, 505-511.
- Almeida Junior, G. L. G. D., Mansur Filho, J., Albuquerque,D. C. D., Xavier, S. S., Pontes, Á., Gouvêa, E. P., ... & Giani,A. (2020). Registro multicêntrico de takotsubo (Remuta)–aspectos clínicos, desfechos intra-hospitalares e mortalidade a longo prazo. Arquivos Brasileiros de Cardiologia, 115, 207-216.
- Cammann, V. L., Szawan, K. A., Stähli, B. E., Kato, K.,Budnik, M., Wischnewsky, M., ... & Templin, C. (2020). Age-related variations in Takotsubo syndrome. Journal of the American College of Cardiology, 75(16), 1869-1877.
- Lyon, A. R., Citro, R., Schneider, B., Morel, O., Ghadri, J. R., Templin, C., & Omerovic, E. (2021). Pathophysiology of takotsubo syndrome: JACC state-of-the-art review. Journal of the American College of Cardiology, 77(7), 902-921.
- Petursson, P., Oštarijaš, E., Redfors, B., Råmunddal, T., Angerås, O., Völz, S., ... & Omerovic, E. (2024). Effects of pharmacological interventions on mortality in patients with Takotsubo syndrome: a report from the SWEDEHEART registry. ESC heart failure, 11(3), 1720-1729.
- Khaloo, P., Ledesma, P. A., Nahlawi, A., Galvin, J., Ptaszek,L. M., & Ruskin, J. N. (2023). Outcomes of Patients with takotsubo syndrome compared with type 1 and type 2 myocardial infarction. Journal of the American Heart Association, 12(18), e030114.
- Isogai, T., Yoshikawa, T., Ueda, T., Yamaguchi, T., Imori, Y., Maekawa, Y., ... & Takayama, M. (2019). Apical Takotsubo syndrome versus anterior acute myocardial infarction: findings from the Tokyo Cardiovascular Care Unit network registry. European Heart Journal: Acute Cardiovascular Care, 8(1), 86-95.
- Arcari, L., Musumeci, M. B., Stiermaier, T., El-Battrawy, I., Möller, C., Guerra, F., ... & Santoro, F. (2020). Incidence, determinants and prognostic relevance of dyspnea at admission in patients with Takotsubo syndrome: results from the international multicenter GEIST registry. Scientific reports, 10(1), 13603.
- Almendro-Delia, M., Núñez-Gil, I. J., Lobo, M., Andrés, M., Vedia, O., Sionis, A., ... & García-Rubira, J. C. (2018). Short-and long-term prognostic relevance of cardiogenic shock in takotsubo syndrome: results from the RETAKO registry. JACC: Heart Failure, 6(11), 928-936.
- Madias, J. E. (2022). “Obesity paradox” and takotsubo syndrome. InternationalJournalof Cardiology Cardiovascular Risk and Prevention, 15, 200152.
- Desai, R., Desai, A., Abbas, S. A., Patel, U., Bansod, S.,Damarlapally, N., ... & Kumar, G. (2020). National prevalence, trends and outcomes of takotsubo syndrome in hospitalizations with prior history of mediastinal/intrathoracic cancer and radiation therapy. International Journal of Cardiology, 309, 14-18.
- Shams, Y. (2014). Acute cardiac sympathetic disruption in the pathogenesis of the takotsubo syndrome: a systematic review of the literature to date. Cardiovascular Revascularization Medicine, 15(1), 35-42.
- Ibanez, B., Navarro, F., Cordoba, M. P. M. A., M-Alberca, P., & Farre, J. (2005). Tako-tsubo transient left ventricular apical ballooning: is intravascular ultrasound the key to resolve the enigma?. Heart, 91(1), 102-104.
- Delgado, G. A., Truesdell, A. G., Kirchner, R. M., Zuzek,R. W., Pomerantsev, E. V., Gordon, P. C., & Regnante, R. A. (2011). An angiographic and intravascular ultrasound study of the left anterior descending coronary artery in takotsubo cardiomyopathy. The American journal of cardiology, 108(6), 888-891.
- Ghadri, J. R., Wittstein, I. S., Prasad, A., Sharkey, S., Dote, K., Akashi, Y. J., ... & Templin, C. (2018). International expert consensus document on Takotsubo syndrome (part I): clinical characteristics, diagnostic criteria, and pathophysiology. European heart journal, 39(22), 2032-2046.
- El Mahmoud, R., Mansencal, N., Pilliére, R., Leyer, F., Abbou, N., Michaud, P., ... & Dubourg, O. (2008). Prevalence and characteristics of left ventricular outflow tract obstruction in Tako-Tsubo syndrome. American heart journal, 156(3), 543-548.
- Schweiger, V., Gilhofer, T., Fang, R., Candreva, A., Seifert, B., Di Vece, D., ... & Templin, C. (2023). Coronary microvascular dysfunction in Takotsubo syndrome: an analysis using angiography-derived index of microcirculatory resistance. Clinical Research in Cardiology, 1-9.
- Stepien, K., Nowak, K., Karcicska, A., Horosin, G., del Carmen Yika, A., Lenart, J., ... & Zalewski, J. (2024). Coronary slow-flow phenomenon in Takotsubo syndrome: the prevalence, clinical determinants, and long-term prognostic impact. International Journal of Molecular Sciences, 25(2), 1297.
- Couch, L. S., Thomas, K. E., Marin, F., Terentes-Printzios, D., Kotronias, R. A., Chai, J., ... & OxAMI Study Investigators. (2024). The role of coronary blood flow and myocardial edema in the pathophysiology of takotsubo syndrome. Cardiovascular Imaging, 17(7), 835-837.
- Wittstein, I. S., Thiemann, D. R., Lima, J. A., Baughman, K. L., Schulman, S. P., Gerstenblith, G., ... & Champion, H. C. (2005). Neurohumoral features of myocardial stunning due to sudden emotional stress. New England journal of medicine, 352(6), 539-548.
- Templin, C., Hänggi, J., Klein, C., Topka, M. S., Hiestand, T., Levinson, R. A., ... & Jäncke, L. (2019). Altered limbic and autonomic processing supports brain-heart axis in Takotsubo syndrome. European heart journal, 40(15), 1183-1187.
- Christensen, T. E., Bang, L. E., Holmvang, L., Skovgaard,D. C., Oturai, D. B., Søholm, H., ... & Hasbak, P. (2016). 123I-MIBG scintigraphy in the subacute state of Takotsubo cardiomyopathy. JACC: Cardiovascular Imaging, 9(8), 982-990.
- Brazdil, V., Kala, P., Hudec, M., Poloczek, M., Kanovsky, J., Stipal, R., ... & Brazdil, M. (2022). The role of central autonomic nervous system dysfunction in Takotsubo syndrome: a systematic review. Clinical Autonomic Research, 32(1), 9-17.
- Santoro, F., Selvaggi, P., D’apollo, R., Martino, T., Veronese, M., Carapelle, E., ... & Brunetti, N. D. (2024). Neurometabolic Features of Takotsubo Syndrome: A Brain 18F-FDG PET Case Control-Prospective Study. Cardiovascular Imaging.
- Dichtl, W., Tuovinen, N., Barbieri, F., Adukauskaite, A., Senoner, T., Rubatscher, A., ... & Steiger, R. (2020). Functional neuroimaging in the acute phase of Takotsubo syndrome: volumetric and functional changes of the right insular cortex. Clinical Research in Cardiology, 109, 1107-1113.
- Ortak, J., Khattab, K., Barantke, M., Wiegand, U. K., Baensch, D., Ince, H., ... & Bonnemeier, H. (2009). Evolution of cardiac autonomic nervous activity indices in patients presenting with transient left ventricular apical ballooning. Pacing and Clinical Electrophysiology, 32, S21-S25.
- Cattaneo, M. M., Pravatà, E., Provenzi, M., Moccetti, M., Kaelin, A., Sudano, I., ... & Cattaneo, M. (2020). Role of the central autonomic nervous system intrinsic functional organisation and psychosocial factors in primary microvascular angina and Takotsubo syndrome. Open heart, 7(2), e001315.
- Stepniewska, A., Budnik, M., Krzemieski, K., Niewiadomski, W., GÃÃÃÃÂ?????siorowska, A., Opolski, G., ... & Kowalik, R. (2019). Impaired hemodynamic response to tilt, handgrip and Valsalva manoeuvre in patients with takotsubo syndrome. Autonomic Neuroscience, 220, 102555.
- Sverrisdóttir, Y. B., Schultz, T., Omerovic, E., & Elam,M. (2012). Sympathetic nerve activity in stress-induced cardiomyopathy. Clinical Autonomic Research, 22, 259-264.
- Vaccaro, A., Despas, F., Delmas, C., Lairez, O., Lambert, E., Lambert, G., ... & Pathak, A. (2014). Direct evidences for sympathetic hyperactivity and baroreflex impairment in Tako Tsubo cardiopathy. PloS one, 9(3), e93278.
- Arcari, L., Musumeci, M. B., & Cacciotti, L. (2022). The Swiss cheese model in takotsubo syndrome. European Heart Journal-Case Reports, 6(6), ytac235.
- Ardell, J. L., & Armour, J. A. (2011). Neurocardiology: structure-based function. Comprehensive Physiology, 6(4), 1635-1653.
- Frank, N., Herrmann, M. J., Lauer, M., & Förster, C. Y. (2024). Exploratory Review of the Takotsubo Syndrome and the Possible Role of the Psychosocial Stress Response and Inflammaging. Biomolecules, 14(2), 167.
- Lazzeroni, D., Ciraci, C., Sommaruga, M., Oggioni, C., Saccò, M., Ziveri, V., ... & Galli, F. (2022). Perceived anxiety, coping, and autonomic function in takotsubo syndrome long after the acute event. Life, 12(9), 1376.
- Berg, E., Agewall, S., Brolin, E. B., Caidahl, K., Cederlund,K., Collste, O., ... & Lyngå, P. (2023). Health-related quality-of-life up to one year after myocardial infarction with non-obstructive coronary arteries. European Heart Journal-Quality of Care and Clinical Outcomes, 9(6), 639-644.
- Citro, R., Okura, H., Ghadri, J. R., Izumi, C., Meimoun, P., Izumo, M., ... & EACVI Scientific Documents Committee Bertrand Philippe Donal Erwan Dweck Marc Galderisi Maurizio Haugaa Kristina H Sade Leyla Elif Stankovic Ivan Cosyns Bernard Edvardsen Thor. (2020). Multimodality imaging in takotsubo syndrome: a joint consensus document of the European Association of Cardiovascular Imaging (EACVI) and the Japanese Society of Echocardiography (JSE). European Heart Journal-Cardiovascular Imaging, 21(11), 1184-1207.
- Cau, R., Muscogiuri, G., Palmisano, V., Porcu, M., Pintus, A., Montisci, R., ... & Saba, L. (2024). Base-to-apex Gradient Pattern Assessed by Cardiovascular Magnetic Resonance in Takotsubo Cardiomyopathy. Journal of Thoracic Imaging, 39(4), 217-223.
- Ahmed, T., Mangino, A. A., Lodhi, S. H., Gupta, V., Leung,S. W., & Sorrell, V. L. (2024). Simplified echocardiographic assessment of regional left ventricular wall motion pattern in patients with takotsubo and acute coronary syndrome: The Randomized Blinded Two-chamber Apical Kinesis Observation (TAKO) Study. Current Problems in Cardiology, 102731.
- Laurence, G., Vasiliu, A., Blommaert, D., Fabien, D., Benoit, B., Claude, H., & Antoine, G. (2022). Typical dynamic electrocardiographic changes in Takotsubo syndrome. Acta cardiologica, 77(2), 146-152.
- Ghadri, J. R., Cammann, V. L., Jurisic, S., Seifert, B., Napp,L. C., Diekmann, J., ... & Bax, J. J. (2017). A novel clinical score (InterTAK Diagnostic Score) to differentiate takotsubo syndrome from acute coronary syndrome: results from the International Takotsubo Registry. European journal of heart failure, 19(8), 1036-1042.
- Schweiger, V., Di Vece, D., Cammann, V. L., Koleva, I., Würdinger, M., Gilhofer, T., ... & Templin, C. (2024). Cardiac biomarkers for diagnosing Takotsubo syndrome. European heart journal, 45(25), 2254-2258.
- Rallidis, L., Kosmas, N., Iordanidis, D., Kiouri, E., Zapantiotis, D., Samiotis, E., ... & Filippatos, G. (2022). The role of peak NT-proBNP/peak cardiac troponin T ratio obtained within 3 days of admission to discriminate Takotsubo from acute coronary syndrome. European Heart Journal, 43(Supplement_2), ehac544-1464.
- Ghadri, J. R., Kato, K., Cammann, V. L., Gili, S., Jurisic, S., Di Vece, D., ... & Templin, C. (2018). Long-term prognosis of patients with Takotsubo syndrome. Journal of the American College of Cardiology, 72(8), 874-882.
- Santoro, F., Mallardi, A., Leopizzi, A., Vitale, E., Rawish, E., Stiermaier, T., ... & Brunetti, N. D. (2021). Current knowledge and future challenges in Takotsubo syndrome: part 2— treatment and prognosis. Journal of Clinical Medicine, 10(3), 468.
- Stiermaier, T., Eitel, C., Desch, S., Fuernau, G., Schuler, G., Thiele, H., & Eitel, I. (2016). Incidence, determinants and prognostic relevance of cardiogenic shock in patients with Takotsubo cardiomyopathy. European Heart Journal: Acute Cardiovascular Care, 5(6), 489-496.
- Pogran, E., Abd El-Razek, A., Gargiulo, L., Weihs, V., Kaufmann, C., Horváth, S., ... & Huber, K. (2022). Long-term outcome in patients with takotsubo syndrome: A single center study from Vienna. Wiener klinische Wochenschrift, 134(7), 261-268.
- Xenogiannis, I., Vemmou, E., Nikolakopoulos, I., Nowariak,M. E., Schmidt, C. W., Brilakis, E. S., & Sharkey, S. W. (2022). The impact of ST-segment elevation on the prognosis of patients with Takotsubo cardiomyopathy. Journal of Electrocardiology, 75, 60-65.
- Stiermaier, T., Moeller, C., Oehler, K., Desch, S., Graf, T., Eitel, C., ... & Eitel, I. (2016). Long-term excess mortality in takotsubo cardiomyopathy: predictors, causes and clinical consequences. European Journal of Heart Failure, 18(6), 650-656.
- Abusnina, W., Elhouderi, E., Walters, R. W., Al-Abdouh, A., Mostafa, M. R., Liu, J. L., ... & Mamas, M. A. (2024).Sex Differences in the Clinical Outcomes of Patients With Takotsubo Stress Cardiomyopathy: A Meta-Analysis of Observational Studies. The American journal of cardiology, 211, 316-325.
- Abuelazm, M., Saleh, O., Hassan, A. R., Ahmad, S., Albarakat,M. M., Abdalshafy, H., ... & Paul, T. K. (2023). Sex difference in clinical and management outcomes in patients with Takotsubo syndrome: a systematic review and meta-analysis. Current Problems in Cardiology, 48(4), 101545.
- Titus, A., Sattar, Y., Patel, N., Taha, A., Sandhyavenu, H., Gonuguntla, K., ... & Balla, S. (2023). In-Hospital Outcomes of Takotsubo Cardiomyopathy During the COVID-19 Pandemic: Propensity Matched National Cohort. Current Problems in Cardiology, 48(5), 101598.
- Pätz, T., Santoro, F., Cetera, R., Ragnatela, I., El-Battrawy, I., Mezger, M., ... & Stiermaier, T. (2023). Trigger-Associated Clinical Implications and Outcomes in Takotsubo Syndrome: Results From the Multicenter GEIST Registry. Journal of the American Heart Association, 12(14), e028511.
- Vazirani, R., Delia, M. A., Blanco-Ponce, E., Martín-García,A. C., Fernández-Cordón, C., Uribarri, A., ... & RETAKO Investigators. (2024). Renal impairment and outcome in Takotsubo syndrome: Insights from a national multicentric cohort. International Journal of Cardiology, 405, 131971.
- Aweimer, A., El-Battrawy, I., Akin, I., Borggrefe, M., Mügge, A., Patsalis, P. C., ... & Dietrich, J. W. (2021). Abnormal thyroid function is common in takotsubo syndrome and depends on two distinct mechanisms: results of a multicentre observational study. Journal of Internal Medicine, 289(5), 675-687.
- Kimura, A., Yoshikawa, T., Isogai, T., Tanaka, H., Ueda, T., Yamaguchi, T., ... & Takayama, M. (2020). Impact of body temperature at admission on inhospital outcomes in patients with takotsubo syndrome: insights from the Tokyo Cardiovascular Care Unit Network Registry. European Heart Journal: Acute Cardiovascular Care, 9(7), 703-710.
- Pinho, A. I., Oliveira, C., Santos, L. D., Marques, C., Cabrita, A., Dias, P., ... & Rodrigues, R. A. (2024). QT interval prolongation in Takotsubo syndrome: a frightening feature with no major prognostic impact. Monaldi Archives for Chest Disease.
- Del Buono, M. G., Damonte, J. I., Moroni, F., Ravindra, K., Westman, P., Chiabrando, J. G., ... & Abbate, A. (2022). QT prolongation and in-hospital ventricular arrhythmic complications in patients with apical ballooning takotsubo syndrome. Clinical Electrophysiology, 8(12), 1500-1510.
- Isaak, A., Bratz, J., Kravchenko, D., Mesropyan, N., Eckardt, I., Bischoff, L. M., ... & Luetkens, J. A. (2023). A novel and simple cardiac magnetic resonance score (PE2RT) predicts outcome in takotsubo syndrome. European Radiology, 33(8), 5498-5508.
- Scally, C., Rudd, A., Mezincescu, A., Wilson, H., Srivanasan, J., Horgan, G., ... & Dawson, D. K. (2018). Persistent longterm structural, functional, and metabolic changes after stress-induced (Takotsubo) cardiomyopathy. Circulation, 137(10), 1039-1048.
- Nowak, R., Fijalkowska, M., Gilis-Malinowska, N., Jaguszewski, M., Galaska, R., Rojek, A., ... & Fijalkowski,M. (2017). Left ventricular function after takotsubo is not fully recovered in long-term follow-up: A speckle tracking echocardiography study. Cardiology Journal, 24(1), 57-64.
- Fernández-Cordón, C., Núñez-Gil, I. J., de Miguel, I. M., Pérez-Castellanos, A., Vedia, O., Almendro-Delia, M., ... & Martínez-Selles, M. (2023). Takotsubo syndrome, stressful triggers, and risk of recurrence. The American Journal of Cardiology, 205, 58-62.
- Ghadri, J. R., Wittstein, I. S., Prasad, A., Sharkey, S., Dote, K., Akashi, Y. J., ... & Templin, C. (2018). International expert consensus document on Takotsubo syndrome (part II): diagnostic workup, outcome, and management. European Heart Journal, 39(22), 2047-2062.
- Aimo, A., Pelliccia, F., Panichella, G., Vergaro, G., Barison, A., Passino, C., ... & Camici, P. G. (2021). Indications of beta-adrenoceptor blockers in Takotsubo syndrome and theoretical reasons to prefer agents with vasodilating activity. International Journal of Cardiology, 333, 45-50.
- von Mackensen, J. K., Zwaans, V. I., El Shazly, A., Van Praet,K. M., Heck, R., Starck, C. T., ... & Wert, L. (2024). Mechanical circulatory support strategies in Takotsubo syndrome with cardiogenic shock: a systematic review. Journal of Clinical Medicine, 13(2), 473.
- Napp, L. C., Cammann, V. L., Jaguszewski, M., Szawan, K. A., Wischnewsky, M., Gili, S., ... & Templin, C. (2020). Coexistence and outcome of coronary artery disease in Takotsubo syndrome. European heart journal, 41(34), 3255-3268.
- Stiermaier, T., Rommel, K. P., Eitel, C., Möller, C., Graf, T., Desch, S., ... & Eitel, I. (2016). Management of arrhythmias in patients with Takotsubo cardiomyopathy: Is the implantation of permanent devices necessary?. Heart Rhythm, 13(10), 1979-1986.
- Raposeiras-Roubín, S., Núñez-Gil, I. J., Jamhour, K., Abu-Assi, E., Conty, D. A., Vedia, O., ... & RETAKO Investigators. (2023). Long-term prognostic impact of beta-blockers in patients with Takotsubo syndrome: Results from the RETAKO Registry. Revista Portuguesa de Cardiologia, 42(3), 237-246.
- Rudd, A. E., Horgan, G., Khan, H., Gamble, D. T., McGowan, J., Sood, A., ... & Dawson, D. K. (2024). Cardiovascular and noncardiovascular prescribing and mortality after Takotsubo: comparison with myocardial infarction and general population. JACC: Advances, 3(2), 100797.
- Silverio, A., Parodi, G., Scudiero, F., Bossone, E., Di Maio, M., Vriz, O., ... & Citro, R. (2022). Beta-blockers are associated with better long-term survival in patients with Takotsubo syndrome. Heart, 108(17), 1369-1376.
- Lau, C., Chiu, S., Nayak, R., Lin, B., & Lee, M. S. (2021).Survival and risk of recurrence of takotsubo syndrome. Heart, 107(14), 1160-1166.
- Novo, G., Arcari, L., Stiermaier, T., Alaimo, C., El-Battrawy, I., Cacciotti, L., ... & Santoro, F. (2024). Statin therapy and outcome in Takotsubo syndrome patients: Results from the multicenter international GEIST registry. Atherosclerosis, 389, 117421.
- D'Ascenzo, F., Gili, S., Bertaina, M., Iannaccone, M., Cammann, V. L., Di Vece, D., ... & Templin, C. (2020). Impact of aspirin on takotsubo syndrome: a propensity score-based analysis of the InterTAK Registry. European journal of heart failure, 22(2), 330-337.
- Omerovic, E., James, S., Erlinge, D., Hagström, H., Venetsanos, D., Henareh, L., ... & Redfors, B. (2023). Rationale and design of BROKEN-SWEDEHEART: a registry-based, randomized, parallel, open-label multicenter trial to test pharmacological treatments for broken heart (takotsubo) syndrome. American Heart Journal, 257, 33-40.
- Olsson, E. M., Norlund, F., Rondung, E., Humphries, S. M., Held, C., Lyngå, P., ... & Tornvall, P. (2022). The e-mental health treatment in Stockholm myocardial infarction with non-obstructive coronaries or Takotsubo syndrome study (E-SMINC): a study protocol for a randomised controlled trial. Trials, 23(1), 597



