Research Article - (2025) Volume 8, Issue 4
Quantum Mechanics and Classical Mechanics Can Be Used Together
Received Date: Aug 04, 2025 / Accepted Date: Sep 02, 2025 / Published Date: Oct 13, 2025
Copyright: ©2025 Runsheng Tu. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Tu, R. (2025). Quantum Mechanics and Classical Mechanics Can Be Used Together. Adv Theo Comp Phy, 8(4), 01-07.
Abstract
Quantum mechanics and classical mechanics are incompatible, and the highly controversial concepts of "uncertainty" and "non realism" are deeply rooted old ideas. Their existence has seriously hindered the development of physics. It is the obligation of explorers to clarify the reasons, methods, application examples, and significance of combining quantum mechanics with classical mechanics. By analyzing the composition, structure, and function of the Schrödinger equation, it can be concluded that the equation itself is a product of the organic combination of classical mechanical laws and wave functions. Prove that the Schrödinger equation is an organic synthesis of the classical mechanical law T+V=E and the wave function (including its differential). Export the Schrödinger equation with gravitational potential energy that can describe macroscopic systems. Pure mathematical methods cannot limit the magnitude of the mass m in the Schrödinger equation. These three facts determine that classical mechanics and quantum mechanics can be used simultaneously to describe the same object that is not limited by mass (providing multiple successful computational examples of "simultaneous use"). These research conclusions can lead to conceptual innovation, theoretical innovation, methodological innovation, and knowledge renewal in physics.
Keywords
Schrödinger Equation for Gravitational Potential, Quantum Mechanics, Classical Mechanics, Compatibility, The Noumenon of Wave Function, Electron Spin Magnetic Moment
Introduction
Recently, both Nature and Scientific American have reported content that is detrimental to existing quantum mechanics. This article will introduce sufficient reasons why quantum and classical can be combined. 1-3 We used the following methods to demonstrate the reason for combining quantum mechanics and classical mechanics. Firstly, establish the Schrödinger equation with gravitational potential energy that can describe macroscopic systerns. 4-9 Secondly, the Schrödinger equation contains a classical mechanical potential energy function, which is closely related to the "force" of physical reality (realistic) and the laws of mechanics in classical mechanics. 10 Thirdly, we cannot use mathematical methods to determine the upper limit of the mass m in the Schrödinger equation, so there is no reason to believe that the fundamental equation of quantum mechanics (Schrödinger equation) cannot simultaneously or separately describe microscopic particles and macroscopic objects.
Because when m changes from microscopic to sufficiently large, the object described by the Schrödinger equation transitions from microscopic to macroscopic (i.e., from "quantum" to "classical"). Fourthly, the Schrödinger equation is a product of the combination of the Viry theorem T + V = E and the wave function in classical mechanics. Fourthly, the non-point wave element electronic structure model can explain the source of electron spin magnetic moment. This result supports both certainty and reality. Fifth, multiple computational examples combining quantum mechanics and classical mechanics can be provided (i.e., it can provide multiple successful application examples. 11-15.
Since we acknowledge that quantum mechanics and classical mechanics can be used together, we acknowledge the validity of using quantum mechanics and classical mechanics to calculate the same object (i.e., this article does not completely deny the validity of the existing mathematical logic system of quantum mechanics, but rather denies the absolutization of the explanatory system of quantum mechanics). This combination or simultaneous use does not involve the transition of particle properties from 'quantum' to 'classical' through the process of quantum decoherence. 4-9
The Structural and Functional Differences Between the Schrödinger Equation for Earth's Revolution and the Original Schrödinger Equation
The differences referred to in the title of this section include differences in form, usage methods (and/or scope), and meanings. Schrödinger initially established the Schrödinger equation for hydrogen atoms. Among them, the time dependent Schrödinger equation (one-dimensional) is
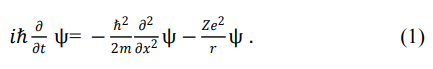
Refs. 7-11 provide the Schrödinger equation for gravitational potential energy. Taking the ideal orbital motion system of the Earth as an example, its form is
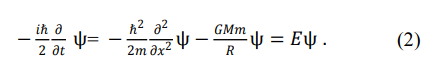
There are significant differences between the first and third terms of Eqs. (1) and (2). Equation (2) can be used to describe the ideal orbital motion of planets such as Earth. Eq. (1) cannot be used to describe the motion of planets in the solar system.
There are significant differences between equation (1) and the first and third terms of equation (2). The calculation results of the two terms after the equal sign of equations (1) and (2) reflect the relationship between kinetic energy and potential energy. (1) The formula does not have a coefficient of -1/2, which cannot guarantee that the Schrödinger equation reflects the or Viry's theorem of T + V = E. Equation (2) can be used to describe the ideal orbital motion of planets such as Earth. Equation (1) cannot be used to describe the motion of planets in the solar system (even replacing the electrodynamic non potential energy function with a gravitational potential energy function is not feasible).
The Fundamentals of Mathematics-Physics and Application Examples of the Combination of Quantum Mechanics and Classical Mechanics for Use
In the introduction, five reasons were mentioned for combining quantum mechanics with classical mechanics. This section specifically introduces several reasons: the fundamental equation of quantum mechanics is a product of the combination of classical mechanics formulas and wave functions; In mathematics, we cannot determine the finite upper limit of the mass m in the Schrödinger equation; We compared the computational results of the "quantum" method and the "classical" method for the macroscopic Earth and microscopic hydrogen atoms. That is, taking the macroscopic and microscopic hydrogen atoms as an example, the consistency of the calculation results between quantum mechanics methods and classical mechanics methods has been achieved.
The Composition and Structure of the Schrödinger Equation Reflecting Its Function
In classical mechanics, potential energy originates from the central force field in a way that conforms to the laws of classical mechanics. The V of the Schrödinger equation cannot be independent of the central force field. The dimensional theorem in classical mechanics determines
T + V = E. (3)

obtain Eq. (2). The term 'replacement' referred to here is actually logical deduction, rather than being written based solely on intuition (see Supplementary for the derivation process of E's operator. The derivation process of kinetic energy operator is similar to this). It can be seen that the Schrödinger equation is a product of the combination of classical mechanical laws and wave functions. This also determines that the Schrödinger equation can simultaneously or separately describe microscopic and macroscopic objects (i.e., it determines that quantum mechanics can be combined with classical mechanics to use).
Being able to use them in combination or simultaneously does not necessarily mean that they must be used in combination or simultaneously. Therefore, 'the existence of situations where they cannot be used in combination or simultaneously' does not deny that in some cases they can be used in combination or simultaneously (i.e., does not deny the theme of this article).
Since the same m in the Schrödinger equation cannot be math- ematically determined as an upper limit (the Schrödinger equa- tion with gravitational potential energy that can directly describe macroscopic systems can be established), the objects described by the Schrödinger equation are not limited to microscopic objects. Microscopic objects conform to quantum mechanics, while mac- roscopic objects conform to classical mechanics. In this way, the fundamental equations of quantum mechanics can simultaneously describe both microscopic and macroscopic objects, and quan- tum mechanics and classical mechanics can be combined for use. Whether quantum mechanics or the microscopic world is uncer- tain and non realistic is a long-standing topic of debate. The above analysis conclusion of this article can at least compress the scope of application of "uncertainty" and "non reality". The statement 'application examples 10-14 can be found (where Ref. 11 is a sum-mary of this calculation method)' seems to be beneficial for ending this debate (at least it can increase the discussion value of the 'the- oretical analysis results' mentioned above).
Comparison of Quantum Mechanics Calculation Results and Newtonian Mechanics Calculation Results for the Earth's Orbital System
After establishing the Schrödinger equation with gravitational potential energy, there are three methods to calculate the energy of the Earth's orbital system: Newtonian mechanics method; Calculation method of one-dimensional gravitational potential energy Schrödinger equation under deterministic background; Calculation method of three- dimensional Schrödinger equation under uncertainty background. The reason is that the Schrödinger equation is the fundamental equation of quantum mechanics, and the use of deterministic concepts and macroscopic gravitational potential energy is equivalent to the use of classical mechanics. In addition, in the planetary model of Earth's revolution, gravitational potential energy is caused by the central gravitational field. Below, we will use these three methods to calculate the ideal Earth's revolution and hydrogen atoms.
The combination of classical mechanics and quantum mechanics refers to the ability to simultaneously or separately describe the same object using classical mechanics and quantum mechanics. We can provide multiple such examples (in addition to the examples in this article, there are also many examples in Refs. 11-15) and compare the solution of Eq. (2) in a deterministic context with the results of Newtonian mechanics calculations. The following lists the solutions of the Schrödinger equation for hydrogen atoms under deterministic and uncertain backgrounds. For Eqs. (1) and (2), the solution under deterministic background is applicable to the planetary model (i.e., classical mechanical system), and r is taken as a constant value. In the process of solving the Schrödinger equation under uncertainty, r takes values sequentially in the entire space from zero to infinity. This is not suitable for macroscopic planetary models (because the ideal Earth's orbit, r, is determined and the Earth will not run to places extremely close or far from the Su, and it is impossible to reach every point in infinite space).
According to Newton's laws of motion, the ideal state for the Earth to move around the Sun is a uniform circular motion. In this case, the magnitude of the centripetal force of the Earth is

(where the energy is the kinetic energy of the Earth). On the premise of acknowledging that the Earth has a definite motion
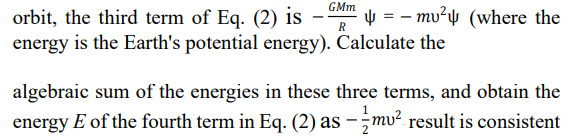
with the conclusion calculated using Newtonian mechanics. This indicates that Eq. (2) as the Schrödinger equation for the Earth's revolution is completely correct and practical. Please note that in υ = λν, ν is the frequency of the de Broglie wave. Its relationship with the energy of de Broglie waves is E = hν. In Ref. 16, it was demonstrated that the phase velocity and group velocity of de Broglie waves are consistent, thus avoiding the difficulty of their phase velocity being greater than the speed of light.

also has a solution, and the distribution of energy level quantization and the square of the membrane of ψ in space can be obtained. However, for macroscopic planetary systems, the concepts of orbit or energy quantization, uncertainty, and probability density are impractical. This indicates that although the combined use of quantum mechanics and classical mechanics is not dependent on whether the described system is a microscopic system, this combination is still conditional.
Several Calculation Methods for Hydrogen Atoms
There are three methods for calculating the energy of hydrogen atoms: Bohr's old quantum theory method; In the context of deterministic planetary models, one-dimensional Schrödinger equation calculation method; In the context of uncertainty, the three-dimensional Schrödinger equation calculation method. According to the method definition in the introduction, the latter two methods belong to the simultaneous or combined use of quantum mechanics and classical mechanics.
This is consistent with the calculation method using the Schrödinger equation in the context of planetary models (or deterministic backgrounds). It is actually a method of simultaneously using (or mixing for use) quantum mechanics and classical mechanics. The specific approach is to establish a one-dimensional partial differential Schrödinger equation, and take a finite definite value for the distance r. This approach satisfies the structure of the planetary model. This method is different from the mathematical calculations in Bohr's planetary model method in pure classical mechanics. For the energy of the ground state hydrogen atom, Bohr calculated it using pure classical methods, while this article calculated it using the Schrödinger equation. The similarity between the two is that they both use the planetary model or the determinacy of classical mechanics.
Eq. (2) is the algebraic sum of the time derivative of the wave function and the Hamiltonian operator acting on the wave function, which is obviously also the Schrödinger equation, which is clearly also the Schrödinger equation. Eq. (2) can be successfully applied to classical mechanical systems. This proves that the Schrödinger equation can describe classical mechanical systems (including planetary model systems). We restore the gravitational potential energy function in Eq. (2) to the electromagnetic interaction potential

of the equation is equal to − 1312 kJ/mol. This method is obviously not Bohr's old quantum theory method, but one of the quantum mechanics methods (using deterministic concepts in quantum mechanics). It is this method that determines that we can simultaneously use quantum mechanics and classical mechanics to describe a constrained system (i.e., we can combine quantum
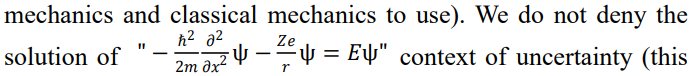
solution is quantized with a value of E = −1312/n2 kJ/mol). Bohr used old quantum theory to calculate the hydrogen atom in planetary models, mainly using classical electrodynamics methods. The calculated energy value of the ground state hydrogen atom is the same as the calculation result using the Schrödinger equation under deterministic background.
It is this method that determines that we can simultaneously use quantum mechanics and classical mechanics to describe a constrained system (i.e., we can combine quantitative mechanics and classical mechanics to use). The Schr ö dinger equation (especially the steady-state Schrödinger equation) consists of two parts: the potential energy function part and the partial differential part of the wave function. The partial differentiation of wave functions clearly belongs to the category of wave mechanics methods (quantum mechanics methods). It is difficult for us to exclude the expression and calculation of potential energy functions from the scope of classical mechanics methods. It is not difficult to see that as long as one is not absolutely bound by the concept of uncertainty (i.e. r takes a certain value), using the Schrödinger equation is equivalent to using both classical that the rejection of classical mechanical methods (i.e., the belief that the two are incompatible) by quantum mechanical methods stems from a conceptual idea rather than mathematical logic The derivation and application of the Schrödinger equation for gravitational potential energy more intuitively reflect this point.
Taking hydrogen molecules as an example, references used a new method that combines classical mechanics and quantum mechanics: Using classical electrodynamics to establish the mechanical equilibrium skeleton structure of hydrogen molecules, then establishing the Schrödinger equation for this structure, and using the Schrödinger equation and classical mechanics to calculate the bond length and dissociation energy of hydrogen molecules. There are multiple examples of calculations for diatomic molecules like this. 12 There are over a hundred examples of quantum mechanics calculations under deterministic backgrounds. 13
A Ring Electronic Structure Model that Can Qualitatively and Quantitatively Explain the Source of Electron Spin Magnetic Moments
For a hundred years, the specific form of electron spin explaining the source of electron spin magnetic moment has not been well done. The mystery of electron spin magnetic moment is a big dark cloud looming over physics. References 14-16 support the "solid ring electronic structure model". Refs. 4-9 established a wave ring electronic structure model. After using m = E/c2, there is a logical connection between these two electronic structure models. The solid-state ring particle structure ring model is just a small step forward from the point particle model. The wave ring, or wave element particle structure model, is a revolutionary step forward.
Based on the experimental fact that high-energy photons decay into electrons and anti electrons, I assume that electrons are composed of wave rings. Specifically, electrons are composed of circularly polarized photons with clockwise rotation. And anti electrons are composed of circularly polarized photons with anti rotation. A basic plane polarized photon is decomposed into a clockwise circularly polarized photon and an counterclockwise circularly oscillating photon. The momentum and energy of waves are also divided into two parts simultaneously. In this case, the energy and momentum of basic circularly polarized light are only half of those of plane polarized light, but the wavelength and frequency are the same. This determines the following relationships:

If the magnetic moment of an electron rotating counterclockwise on a horizontal plane is considered positive, then flipping such an electron loop 180 degrees results in a negative spin magnetic moment for the electron. If only these two values are taken, then

Please note that! The direction of the electron spin magnetic moment obtained from the experiment is not exactly two opposite directions, but multiple directions. For planar electromagnetic waves, pplane=2mec. For plane polarized light and de Broglie waves, the momentum Hermitian operator is

For basic circularly polarized light,pcircular = mec. This determines that the momentum operator of the intrinsic motion of electrons has an additional coefficient of 1/2 compared to the ordinary momentum Hermitian operator

The correlation functions of basic circularly polarized light and plane polarized light can both take the form of
Eq. (6).
y(x, t)= Ae-i 2p (nt - x / l ) . (7)
However, compared to the planar polarized light before decomposition, the amplitude A, energy value h v, and momentum value p of the decomposed circularly polarized light differ by two times. When calculating its partial derivatives, there is no difference in form between the results obtained.
Now let's derive the electron spin operator. For wave functions, we are accustomed to using Eq. (7). However, we assume in Ref. 9-12 that electrons are composed of fundamentally circularly polarized light. Therefore, when deriving the electron spin magnetic moment operator, the intrinsic motion operator of the electron must use Eq. (8). Considering the relationship between basic circularly polarized light and electrons, which is represented by the operator

It is not difficult to see that we have quantitatively determined the relationship between the self magnetic moment of an electron and its composition, structure, and internal movement mode. That is, the rigorous logic reveals that the spin magnetic moment of electrons originates from their intrinsic motion. The method is to break through the constraints of the standard model material structure theory and the point particle material structure theory to establish the wave element material structure theory. By replacing the

In fact, the orbital magnetic moment of the extranuclear electrons of hydrogen atoms during orbital motion is 274 times that of the spin magnetic moment of free electrons. 9-12 This is a basic assumption. With it, it is convenient to use planetary models in atomic and molecular systems (that is, to combine classical mechanics with quantum mechanics). Based on this assumption, it can be predicted that conducting Stern Gerlach experiments using alpha particle beam, electron beam, and hydrogen atom beam will gradually increase the experimental effect. The quantitative ratio of the splitting effect is "proton (alpha particle): electron: hydrogen atom"=1:1836:1836 × 274.
The ring electron model (wave element electronic structure model) discussed in this section is the foundation of the planetary hydrogen atom model. It is also one of the theoretical foundations for the combination of classical mechanics and quantum mechanics methods for hydrogen atoms. The reason is that in this model, the motion path of circularly polarized photons inside the electron is determined (In the centroid coordinate system). Since the internal motion path of an electron is determined, its external motion has more reason to conform to determinism. That is, the electronic structure model supports determinism and realism. This is beneficial for the combination of "quantum" and "classical".
Conclusion
The research work introduced in this article has formed a complete chain of evidence: firstly, theoretically, the Schrödinger equation for gravitational potential energy applicable to the combination of classical mechanics and quantum mechanics can be derived (Established the Schrödinger equation that can describemacroscopic mechanical systems, intuitively expressing that classical mechanics and quantum mechanics can be combined for use); Secondly, it can provide a convenient wave element electronic structure model for the combination of classical mechanics and quantum mechanics, and this model can successfully explain the source of the burst spin magnetic moment; Thirdly, many successful examples of the combination of classical mechanics and quantum mechanics have been found. The first point is the mathematical evidence of quantum mechanics. The second point is the theoretical evidence of material structure, which is also a theoretical advantage (solving the problem of the source of electron spin magnetic moment that cannot be solved by other theories). The third point is practical evidence (or theoretical application evidence). These contents are referred to as positive evidence chains.
By exporting and using the Schrödinger equation of gravitational potential energy, it intuitively reflects that the Schrödinger equation itself does not exclude the joint use of quantum mechanics and classical mechanics. This article supports the validity of planetary models, which still have a certain range of applicability in quantum mechanics. Taking hydrogen atoms as an example, a planetary model that only wants to obtain the deterministic ground state energy of the system is still applicable. If one wants to obtain quantized results, one can reject the planetary model in classical mechanical equilibrium systems.
This article does not deny the existing mathematical formal system of quantum mechanics, but suggests the conditional use of concepts such as uncertainty, state superposition, and coherence in existing interpretations of quantum mechanics. From another perspective, the research results of this article indicate that using methods compatible with classical mechanics and quantum mechanics can obtain accurate computational results.
The combination of quantum mechanics and classical mechanics can at least demonstrate that quantum mechanics is not only applicable to microscopic objects, but also to macroscopic objects that conform to classical mechanics and are deterministic and realistic. So much so that quantum mechanics can only use the name wave dynamics. Among them, the use of deterministic concepts, locality concepts, and reality concepts in wave dynamics is fatal to the existing quantum mechanics interpretation system. It will also lead to the emergence of new theories and computational methods within the existing mathematical framework of quantum mechanics. Does this mean that the Scientific Revolution of this century is about to break out?
Discussion
Readers who have accepted existing physics theories generally choose negative answers to the following questions. The author has provided a detailed analysis of these issues. Please ask the reader to choose a new answer after reading these analyses.
Has the Author Achieved the goal of Combining Classical Mechanics Methods with Quantum Mechanics for use? Can the Number of Successful Application Instances be Large Enough to Exclude Randomness?
Section 3 of this article introduces two computational examples that combine classical mechanics and quantum mechanics to use. Refs. 4 introduces a detailed method for this joint use (taking a hydrogen molecule as an example). Ref. 5 introduces several computational examples of small diatomic molecules (all of which are joint examples of classical mechanics and quantum mechanics). Ref. 8 introduces over 100 calculation examples of atoms (or ions), all of which are under deterministic background. If there are more than three calculation examples of the same type, coupling factors can be excluded. Moreover, we have provided theoretical support for this method. For example, the Schrödinger equation, which can be used to describe classical mechanical systems, was derived, and a ring electronic structure model supporting atomic molecular planetary models was established.
We do not completely deny the existing mathematical formal system of quantum mechanics, but suggest modifying the existing explanatory system of quantum mechanics. Therefore, existing examples of quantum mechanics applications are not counterexamples to the conclusions of this article. In this way, the old theory of quantum mechanics cannot be used as a standard to deny the successful application of the theory presented in this article.
The successful application of the combination of classical mechanics and quantum mechanics cannot be explained by coinci- dence. If you want to choose the negative answer to question 6.3, you must logically prove that the calculation method provided by the author has logical errors. It is not a fair approach to deny the conclusions of this article by using the phrase 'not in line with the existing theoretical framework of physics'.
What is the Significance of Comparing the Results of Classical Mechanics Calculations and Quantum Mechanics Calculations for the Same System?
In Section 3, we made the above comparison. If the calculation results of classical mechanics are consistent with those of quantum mechanics, it can indicate that the Schrödinger equation for gravitational potential energy is correct and effective. It can also indicate that there is still a market for planetary models and determinism in quantum mechanics. Combining quantum mechanics with classical mechanics for use is clearly a new approach to quantum mechanics.
What are the Similarities Between the Conclusion that Classical Mechanics and Quantum Mechanics can be Combined to use and the Conclusion that Energy Changes are Discontinuous (energy quantization) in the Past?
It is necessary to compare whether classical mechanics and quantum mechanics can be combined with the high- frequency formula and low-frequency formula of blackbody radiation (by interpolating the two). Wien and Rayleigh-Jeans proposed blackbody radiation formulas for different frequency bands. Before Planck, no one thought that the two could be combined (i.e., no one thought of interpolating between the two). Before the author of this article, no one had thought of combining quantum mechanics with classical mechanics for use. The "union" discussed in this article is very similar in form to Planck's union (interpolation between the two). The result of Planck interpolation and its explanation (energy quantization) is a new perspective with significant importance. The author of this article also provided an explanation for the result to combine use of classical mechanics and quantum mechanics, stating that quantum mechanics and classical mechanics can be compatible, and there is still a certain market for determinacy in the microscopic world. This explanation is also a completely new perspective. That is to say, in terms of the novelty of the viewpoint, the author's behavior in this article is similar to Planck's behavior back then.
In the field of science, the initial reactions (not widely accepted in a timely manner) caused by the two are also very similar. At the beginning of Planck's proposal of energy quantization, the physics community basically denied it. Both Planck himself strongly denied his proposal of energy quantization. At present, the number of people who reject the new viewpoint that "classical mechanics and quantum mechanics can be used together" far exceeds the number of people who affirm it.
References
1.Carroll, S. (2025). Why even physicists still don’t understand quantum theory 100 years on. Nature. 638, 31-34.
2.Lee Billings. (2025). Quantum Physics Is on the Wrong Track, Says Breakthrough Prize Winner Gerard ’t Hooft. Breakthrough Prize Winner Gerard ’t Hooft Says Quantum Mechanics Is ‘Nonsense’. Scientific American. 34(2), 104.
3.Gibney, E. (2025). Physicists disagree wildly on what quantum mechanics says about reality, Nature survey shows. Nature. 643, 1175-1179.
4.Runsheng Tu. (2025). Research Progress on the Schrödinger Equation that Can Describe the Earth's Revolution and its Applications, London Journal of Research in Science: Natural & Formal. 25(1), 17-28.
5.Runsheng Tu. (2025). Derive Schrödinger equation from F=ma. Vixra.
6.Tu, R. (2024). Establishing the Schrödinger Equation for Macroscopic Objects and Changing Human Scientific Concepts. Adv Theo Comp Phy, 7(4), 01-03.
7.Tu, R. (2025). Research Progress on the Schrödinger Equation of Gravitational Potential Energy. Adv Theo Comp Phy, 8(1), 0107.
8.Tu. R. (2025). Schrödinger-Tu Equation: A Bridge Between Classical Mechanics and Quantum Mechanics. Int J Quantum Technol, 1(1), 01-09.
9.Runsheng Tu. (2025). Research Progress on the Schrödinger Equation that Can Describe the Earth's Revolution and its Applications, London Journal of Research in Science: Natural & Formal, 25(1), 17-28.
11.Tu, R. (2025). A New Theoretical System Combinating Classical Mechanics and Quantum Mechanics. Adv Theo Comp Phy, 8(2), 01-06.
12.Tu, R. (2014). Some Successfull Applications for Local- Realism Quantum Mechanics: Nature of Covalent- Bond Revealed and Quantitative Analysis of Mechanical Equilibrium for Several Molecules. Journal of Modern Physics. 5(6), 309-318.
13.Tu, R. (2016). The principle and application of experimental method for measuring the interaction energy between electrons in atom. International Journal of Scientific Reports, 2(8), 187–200.
14.Tu, R. (2024). A Wave-Based Model of Electron Spin: Bridging Classical and Quantum Perspectives on Magnetic Moment. Adv Theo Comp Phy, 7(4), 01-10.
15.Tu, R. (2024). A Review of Research Achievements and Their Applications on the Essence of Electron Spin. Adv Theo Comp Phy, 7(4), 01-19.
16.Tu, R. (2024). Solving the problem of source of electron spin magnetic moment. Physical Science International Journal, 28(6), 105-110.