Review Article - (2025) Volume 10, Issue 1
Quantum Dots for Margin Visualization in Non-Melanoma Skin Cancers: Advances in Nanotechnology for Cutaneous Surgical Precision
2University of Utah College of Health, Salt Lake City, UT, USA
Received Date: May 01, 2025 / Accepted Date: Jun 02, 2025 / Published Date: Jun 11, 2025
Copyright: ©Â©2025 Andres D Parga, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Parga, A. D., Ray, B. (2025). Quantum Dots for Margin Visualization in Non-Melanoma Skin Cancers: Advances in Nanotechnology for Cutaneous Surgical Precision. Int J Nanotechnol Nanomed, 10(1), 01-08.
Abstract
Background: Non-melanoma skin cancers, including basal cell carcinoma and squamous cell carcinoma, are the most prevalent malignancies worldwide. Achieving histologically clear margins is critical for recurrence prevention, yet conventional techniques often fall short in precision and real-time intraoperative guidance.
Objective: To evaluate quantum dots as emerging nanotechnology platforms for surgical margin visualization in NMSC, highlighting their physicochemical properties, targeting strategies, imaging modalities, safety profiles, and translational barriers.
Methods: A comprehensive review of literature from 2011 to 2025 was conducted, encompassing quantum dot design, bioconjugation techniques, tumor-targeting ligands, imaging integration with dermatoscopes and microscopy, and preclinical/ clinical applications in cutaneous oncology.
Results: Quantum dot-based systems offer unprecedented spatial resolution (as low as 20–100 μm) for tumor margin delineation due to their exceptional brightness, photostability, and multiplexing capability. Functionalized quantum dots targeting biomarkers such as EGFR and integrins (αvβ3, αvβ6) demonstrate tumor-specific accumulation both in vivo and ex vivo. Comparative studies show that quantum dots outperform traditional dyes like ICG in photostability and specificity. Biodegradable alternatives, including carbon, silicon, and CuInSe-based quantum dots, are mitigating toxicity concerns. Integration with time-gated imaging, AI-assisted classification, and real-time surgical guidance systems further enhances clinical utility.
Conclusion: Quantum dots represent a transformative innovation in the precision resection of non-melanoma skin cancers. While challenges remain in safety, delivery, and regulatory approval, ongoing advancements in biocompatibility and intelligent imaging integration position quantum dots as pivotal tools for the next generation of fluorescence-guided dermatologic surgery.
Keywords
Quantum Dots, Non-Melanoma Skin Cancer, Surgical Margin Visualization, Fluorescence-Guided Surgery, EGFR Targeting, Nanotechnology, Multiplex Imaging, Cutaneous Oncology, Bioconjugation
Introduction
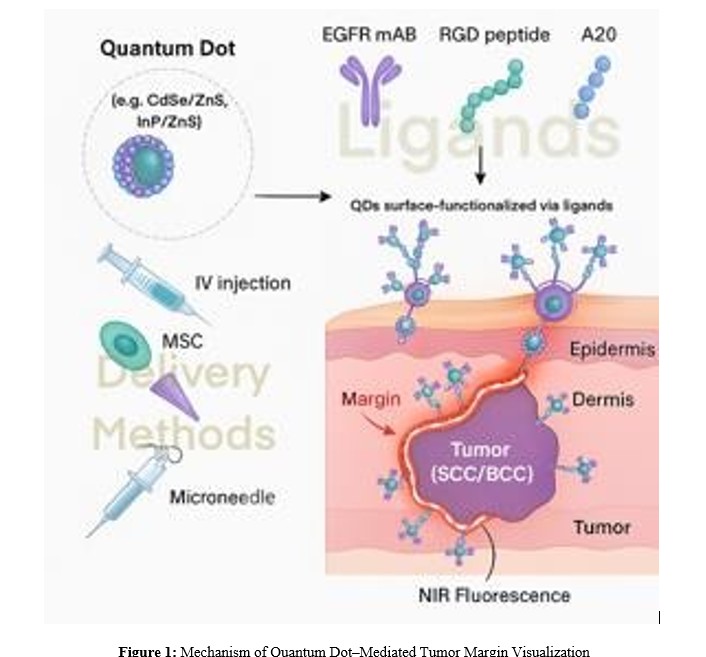
Non-melanoma skin cancers (NMSCs), primarily basal cell carcinoma (BCC) and squamous cell carcinoma (SCC), constitute over 95% of all skin cancers and are the most commonly diagnosed malignancies worldwide [1]. While BCC is typically indolent, SCC carries a higher risk of local invasion and metastasis, with rising incidence projections across aging and sun-exposed populations [1,2]. The primary treatment for NMSC remains surgical excision, where complete removal with histologically negative margins is critical for curative outcomes. Despite advances in surgical dermatology, margin delineation during excision remains challenging. Conventional approaches such as frozen section analysis, dermoscopy, and Mohs micrographic surgery (MMS) offer varying levels of precision but suffer from drawbacks including sampling errors, procedural complexity, high costs, or limited intraoperative visualization [3,4]. There is an unmet need for real-time, high-resolution tools that can reliably highlight tumor borders during excision, especially in cosmetically and functionally sensitive areas. Quantum dots (QDs), semiconductor nanocrystals with tunable fluorescence, represent a transformative platform for surgical margin visualization. Their unique optical characteristics include size-dependent emission wavelengths, high quantum yield, narrow emission spectra, broad excitation profiles, and exceptional photostability [5,6]. These properties allow for deep tissue penetration using near-infrared (NIR) fluorescence, multiplex imaging of tumor markers, and minimized background autofluorescence, features particularly advantageous for intraoperative guidance in cutaneous oncology (Figure 1). In the context of NMSC, QDs can be functionalized with monoclonal antibodies, peptides (e.g., RGD or A20FMDV2), or aptamers targeting overexpressed biomarkers such as EGFR, αvβ3, and αvβ6 integrins [7-10]. These conjugates enable real- time fluorescent labeling of tumor margins, even within complex tissue environments or 3D tumor spheroids [11]. Furthermore, innovations in QD delivery, such as microneedles, stem cell vehicles, and liposomal encapsulation, enhance tumor selectivity while minimizing systemic exposure [12,13]. Emerging preclinical evidence now supports the feasibility of QD-guided margin visualization in NMSC, offering spatial resolution in the range of 20–100 µm [3,4]. This review synthesizes current data on QD formulations, targeting ligands, delivery strategies, and clinical translation potential, and highlights how QDs may improve intraoperative precision and long-term oncologic outcomes in cutaneous oncology.
Schematic illustration of a quantum dot (QD) platform for surgical margin visualization in non-melanoma skin cancers. QDs (e.g., CdSe/ZnS, InP/ZnS) are surface-functionalized with ligands such as EGFR monoclonal antibodies, RGD peptides, or A20 peptides for selective binding to tumor-associated markers. These nanoconjugates can be delivered via intravenous injection, mesenchymal stem cell carriers, or microneedle arrays. Upon binding to tumor margins, QDs emit near-infrared (NIR) fluorescence, enabling real-time intraoperative guidance and precision excision.
Physicochemical Properties of Quantum Dots
QDs are semiconductor nanocrystals, typically ranging from 2 to 10 nanometers in diameter, whose optical and electronic properties are dictated by their size and composition. Their design can be customized to meet the demanding requirements of surgical oncology, especially for real-time, high-contrast visualization of non-melanoma skin cancer (NMSC) margins.
Composition and Core–Shell Architecture
Most QDs used for biomedical imaging are composed of a heavy metal core such as cadmium selenide (CdSe), cadmium telluride (CdTe), or indium phosphide (InP), surrounded by a stabilizing shell like zinc sulfide (ZnS) or zinc selenide (ZnSe). This core– shell architecture enhances photostability and reduces toxic metal ion leakage [5,6]. For example, QD800 nanoparticles composed of a CdSe/CdTe core with a ZnS shell have been successfully conjugated to EGFR antibodies for in vivo imaging of SCC margins [8]. To improve safety, cadmium-free alternatives like ZnCuInSe/ZnS have emerged, offering comparable brightness and longer circulation times with reduced cytotoxicity [10]. Surface functionalization further tailors QDs for biomedical use. Common coatings include polyethylene glycol (PEG), sulfobetaine polymers, and amphiphilic lipids, which increase water solubility, reduce aggregation, and enable ligand attachment [7,14]. The net surface charge, especially positive zeta potentials, can influence non-specific cellular uptake, as demonstrated in 3D models of HNSCC spheroids [11].
Photostability and Fluorescence Quantum Yield
A defining feature of QDs is their superior photostability. Unlike traditional organic dyes that rapidly photobleach, QDs can emit strong fluorescence signals for extended imaging periods, even under prolonged excitation. This property enables continuous intraoperative visualization without signal loss [6,15]. Quantum yield, the efficiency of photon emission following excitation, can exceed 50% for many core–shell QDs, making them among the brightest fluorophores available for biomedical use [5,16]. This brightness is preserved even in tissue-dense environments, facilitating deeper signal penetration when operating in the near- infrared (NIR) range (~700–800 nm). For instance, QD800 and ZnCuInSe/ZnS QDs exhibit high signal-to-noise ratios when applied to SCC tumor models, enhancing visualization of tumor– stroma interfaces [9,10].
Multiplexing Capacity for Multi-Marker Imaging
QDs can be engineered to emit distinct colors based on their core size and composition, allowing simultaneous detection of multiple tumor biomarkers in a single imaging session (Table 1). This multiplexing capability is especially advantageous in tumors with heterogeneous cellular populations or complex microenvironments. For example, Hu et al. utilized a panel of QDs (QD565–705) to map EGFR, E-cadherin, and vimentin expression in SCC tissues, achieving predictive accuracy for lymph node metastasis with area-under-the-curve (AUC) values >0.90 [4]. The broad excitation spectrum and narrow emission peaks of QDs also support spectrally resolved imaging systems, enabling precise spatial localization of multiple targets without signal overlap. When paired with computational deconvolution and time-gated imaging, QDs can outperform traditional dyes in both clarity and diagnostic value [5,7].
|
QD Type |
Core/Shell |
Emission Peak |
Targeting Ligand |
Imaging Modality |
Unique Features |
|
QD800 (CdSe/ CdTe-ZnS) |
Near-infrared QD |
800 nm |
EGFR, RGD |
In vivo optical fluorescence |
High tumor-to- background ratio |
|
CdSe/ZnS (PEG- coated) |
Visible red QD |
625 nm |
None (MSC carrier) |
Ex vivo fluorescence imaging |
MSC delivery; low toxicity, tumor-homing |
|
ZnCuInSe/ZnS |
Cadmium-free NIR QD |
750 nm |
A20 peptide |
Time-gated microscopy |
Deep tissue imaging, antifouling polymer shell |
|
CdSe/CdS/ZnS |
Visible/NIR QD |
610 nm |
A20 peptide |
Confocal imaging |
Uptake driven by zeta potential |
|
QD565–705 (varied) |
Multiplex panel |
565–705 nm |
EGFR, vimentin |
Immunohistofluorescence (IHF) |
Multiplex margin labeling in FFPE tissue |
Table 1: Summary of Quantum Dot Design Features and Imaging Characteristics
Targeting Strategies for Skin Cancer Margins
NMSCs, including BCC and SCC, often overexpress specific surface biomarkers such as epidermal growth factor receptor (EGFR), integrins (e.g., αvβ3, αvβ6), nucleolin, and folate receptor-α, which provide a foundation for molecularly targeted imaging strategies [5,17,18]. Among these, EGFR is particularly relevant in SCC and has been effectively targeted using monoclonal antibody–conjugated QDs for real-time intraoperative imaging of tumor margins [4,8]. Bioconjugation techniques play a critical role in enabling specific tumor targeting. QDs can be functionalized with peptides (e.g., iRGD, RGD, A20FMDV2), antibodies (e.g., anti-EGFR, anti-HER2), or aptamers (e.g., AS1411 against nucleolin), facilitating either receptor-mediated endocytosis or high-affinity binding to tumor-associated antigens [7,11,16] (Table 2). In a notable example, αvβ3-integrin-targeting QDs conjugated to RGD peptides demonstrated selective tumor accumulation in head and neck squamous cell carcinoma, offering a clear analog for cutaneous SCC targeting. Similarly, EGFR-targeted QDs successfully delineated tumor boundaries in vivo within minutes of administration [8,9]. These approaches reflect the broader dichotomy between passive and active targeting. Passive targeting relies on the enhanced permeability and retention (EPR) effect that allows nanoscale agents like QDs to accumulate within tumor tissue due to leaky vasculature and poor lymphatic drainage [7]. However, passive accumulation alone may be insufficient for high-resolution margin detection. Active targeting, achieved through ligand-mediated recognition of tumor biomarkers, offers superior specificity and is more effective for distinguishing tumor periphery during surgical resection [12,17]. Emerging delivery platforms such as skin-derived mesenchymal stem cells (MSCs) have also shown promise. These cells naturally home to tumor sites and can transport QDs into the tumor microenvironment without compromising viability or migration, demonstrating a novel delivery method with potential applications in margin visualization [12]. Additionally, multifunctional QDs designed for simultaneous imaging and therapy (theranostics) have been explored in epithelial tumors, further highlighting the translational potential of these systems for NMSC [5,16]. Together, these strategies suggest that bioconjugated QDs, whether peptide-, antibody-, or aptamer-linked, offer a robust and tunable platform for precise visualization of tumor margins (Table 3). This is especially important for superficial tumors like NMSC, where maximizing tissue conservation without compromising oncologic control remains paramount.
|
Biomarker |
Relevance to NMSC |
Targeting Ligand Used |
|
EGFR |
Overexpressed in SCC |
Monoclonal antibody |
|
αvβ3 integrin |
Neovasculature in SCC |
RGD peptide |
|
αvβ6 integrin |
Overexpressed in SCC |
A20FMDV2 peptide |
|
E-cadherin |
Tumor suppression loss |
Anti–E-cadherin mAb |
|
Vimentin |
EMT marker |
Anti-vimentin mAb |
|
Folate receptor |
Expressed in SCC/BCC |
Folic acid, aptamer |
Table 2: Biomarkers and Targeting Strategies for Quantum Dot Conjugation in NMSC
|
Study |
QD Type |
Emission (nm) |
Targeting Ligand |
Tumor Model |
Application |
Key Findings |
|
Yang et al., 2011 |
CdSe/CdTe-ZnS (QD800) |
800 |
Anti-EGFR mAb |
Buccal SCC (mouse) |
In vivo fluorescence imaging |
Specific margin labeling, 15 min onset, signal peaks at 1h, declines by 8h |
|
Huang et al., 2013 |
CdSe/ZnS (QD800) |
800 |
cRGDfC (αvβ3 integrin) |
HNSCC angiogenic vessels |
In vivo angiogenesis visualization |
Targeted vasculature, high TBR, NIR imaging through skin |
|
Hu et al., 2016 |
QD565–705 |
565–705 |
EGFR, E-cadherin, vimentin |
HNSCC FFPE specimens |
Multiplexed biomarker mapping |
Predictive of LNM; QD-IHF superior to IHC; AUC > 0.90 |
|
Yakavets et al., 2020 |
ZnCuInSe/ZnS |
750 |
A20 peptide (αvβ6 integrin) |
FaDu HNSCC spheroids |
3D tumor imaging |
High SNR, time- gated imaging; selective labeling in tumor spheroids |
|
Dirheimer et al., 2024 |
CdSe/CdS/ZnS |
610 |
A20 peptide (non-specific) |
HSC-3 tongue SCC spheroids |
3D margin labeling |
Uptake driven by positive zeta potential, not specific binding |
|
Dapkute et al., 2017 |
CdSe/ZnS |
625 |
None (MSC delivery) |
Breast cancer (MDA-MB-231, mouse) |
Stem cell– mediated delivery |
Tumor-homing MSCs deliver QDs with high specificity; minimal off- target signal |
Table 3: Quantum Dot Platforms for Margin Visualization in Epithelial Tumors
Quantum Dot Imaging Platforms
QD-based imaging systems are emerging as transformative tools in dermatologic oncology, offering both in vivo and ex vivo platforms for enhanced tumor margin visualization. Their unmatched brightness, photostability, and multiplexing capabilities allow for dynamic imaging strategies tailored to the specific needs of skin cancer surgery. In vivo visualization leverages the fluorescence of surface-functionalized QDs administered systemically or locally to delineate tumor margins in real-time. Near-infrared QDs, such as QD800, enable deep tissue imaging with reduced background autofluorescence and have been successfully used to target integrins and EGFR in squamous cell carcinoma [8,9]. This facilitates fluorescence-guided surgery, where surgeons can visualize tumor borders intraoperatively using QD-labeled molecular markers. In murine models, EGFR-targeted QDs have demonstrated clear signal accumulation at cutaneous tumor edges within 15 minutes of injection, with optimal signal-to-noise ratios sustained up to 6 hours [8]. Ex vivo visualization platforms are equally valuable for margin assessment immediately after tissue excision. Liu et al. (2019) showed that fluorescent probes applied topically to excised NMSC specimens provided spatial resolution within 100 µm of histologically confirmed tumor margins [3]. Time-gated imaging systems, such as the LI-COR Odyssey scanner, further enhance QD signal clarity by suppressing autofluorescence and enabling spectral deconvolution, particularly when imaging cadmium- based or indium phosphide QDs. The integration of QDs with dermatoscopic and microscopic tools represents a promising frontier. QD-assisted fluorescence dermatoscopy could improve early diagnostic sensitivity for subclinical BCC and SCC, especially when using multiplexed QDs tuned to specific tumor and stromal biomarkers. Additionally, coupling QDs with reflectance confocal microscopy (RCM) or multiphoton fluorescence imaging allows high-resolution, depth-resolved imaging of tumor architecture, which is particularly relevant for margin control in ambiguous or infiltrative lesions. Confocal-compatible QD systems have been tested in spheroid models to visualize marker expression in live tissue analogs, and time-gated detection methods enhance visualization of QD-labeled tumor borders even in thick tissue slices [7,10]. Overall, quantum dot imaging platforms enable scalable and modular systems that can be adapted for real-time surgical navigation, rapid ex vivo pathology, and integrated preoperative mapping, all with the goal ofmaximizing precision and minimizing recurrence in NMSC resection.
Preclinical and Emerging Clinical Evidence
QDs have demonstrated strong promise in preclinical settings for delineating tumor margins in NMSC, particularly SCC and BCC (Table 4). Animal model studies, excised human specimen imaging, and comparative analyses with other fluorophores such as indocyanine green (ICG) provide converging evidence of QDs’ utility in surgical guidance. In vivo animal models have shown robust tumor labeling using QDs conjugated to EGFR antibodies or integrin-targeting peptides. In an SCC xenograft model, QD800-labeled anti-EGFR probes enabled real-time tumor margin visualization within 15 minutes post-injection [8]. Similarly, studies utilizing integrin-targeted QDs demonstrated specific accumulation in αvβ3- and αvβ6-overexpressing tumors, with significant signal enhancement and margin clarity [9,10]. Ex vivo imaging studies further support the role of QDs in surgical margin delineation. Applying a near-infrared cathepsin-activatable probe to excised SCC and BCC specimens, achieved spatial concordance with histopathologic margins within 100 µm [3]. While QDs were not used directly in the tissue samples, QD-loaded phantoms demonstrated sharper edge response and higher signal fidelity than traditional fluorescent agents, underscoring their potential in rapid intraoperative use. Comparative evaluations with standard fluorophores such as ICG reveal the advantages of QDs. ICG suffers from low photostability, rapid clearance, and broad tissue distribution, limiting its effectiveness for margin precision. QDs, in contrast, offer brighter emission, reduced photobleaching, and multiplex imaging capability [4,18]. Furthermore, time-gated detection enabled by QDs minimizes background autofluorescence and enhances tumor-to-margin contrast [10]. Collectively, these findings indicate that QDs outperform conventional contrast agents in specificity, durability, and multi-marker visualization. As cadmium-free QD formulations advance, clinical translation for real-time margin assessment during Mohs or wide local excision procedures appears increasingly feasible.
|
Delivery Method |
Carrier System |
Benefits |
|
Stem Cell-mediated |
Human skin-derived MSCs |
Tumor tropism, minimal off-target |
|
Direct IV injection |
QD + EGFR/RGD peptide |
Rapid tumor localization |
|
Topical + ex vivo scan |
Cathepsin-activated dye (control study) |
High margin resolution (~100 µm) |
|
Intratumoral injection |
QD-liposomes |
Controlled release, reduced toxicity |
|
Microneedle-assisted |
Gold or polymer QD analogs |
Enhanced dermal penetration |
Table 4: Delivery Strategies for Quantum Dots in Skin Cancer Models
Safety and Regulatory Considerations
Despite their promise, the clinical translation of QDs for surgical margin visualization is hindered by concerns over toxicity, biocompatibility, and regulatory approval. The heavy metal content of traditional cadmium-based QDs, such as CdSe/ZnS core–shell structures, has raised significant concerns regarding long-term tissue accumulation, oxidative stress, and potential carcinogenicity [5,6]. Studies have reported that cadmium ions released through degradation or oxidation can induce reactive oxygen species (ROS) production and mitochondrial damage, particularly in hepatic and renal systems [18]. To address these challenges, biodegradable and cadmium-free QDs are being actively developed. Carbon quantum dots (CQDs), graphene quantum dots (GQDs), and silicon-based QDs offer comparable photostability and fluorescence performance while reducing systemic toxicity [13,15]. For instance, CQDs derived from organic sources have demonstrated effective tumor labeling and minimal off-target effects in vivo, while GQDs have served as photosensitizers in photodynamic therapy with high efficacy and biocompatibility [6]. Additionally, ternary alloy QDs such as CuInSe/ZnS have been explored for their near-infrared emission and lower environmental risk [10]. Regulatory challenges remain a significant barrier to intraoperative adoption. While platforms like Cornell Dots (silica-based QDs) have progressed to clinical trials for melanoma imaging, no QD system has received full regulatory approval for surgical margin visualization in skin cancer [13]. Hurdles include stringent safety evaluations, unclear long-term biodistribution profiles, and limited standardization across imaging systems. Surface modifications such as PEGylation, zwitterionic coatings, and antibody conjugation have helped reduce immunogenicity, yet regulatory agencies continue to emphasize the need for robust pharmacokinetic, toxicity, and clearance data prior to approval [5,16].
Going forward, clinical translation will likely depend on: Transition to non-toxic, biodegradable QD systems; Integration with existing FDA-approved surgical platforms; and Evidence from large-scale animal and early-phase human studies confirming safety and accuracy. These developments are critical to ensure that QDs can move from promising preclinical tools to practical, regulatory- compliant agents for intraoperative use in dermatologic oncology.
Discussion
The next frontier in QD research for NMSC margin visualization lies in smart, multiplexed platforms that not only illuminate tumors but dynamically guide clinical decision-making. Innovations in QD design, targeting strategies, and computational integration are shaping a translational roadmap toward personalized fluorescence- guided surgery (FGS). Multiplexed QD systems, capable of emitting at distinct wavelengths, offer the ability to simultaneously label tumor cells, stromal elements, immune infiltrates, and neovasculature within a single surgical field [4,5]. For example, QDs conjugated to EGFR, αvβ6 integrin, and E-cadherin have been used to delineate cancerous epithelium, tumor-stroma interfaces, and epithelial-to-mesenchymal transition zones, providing a comprehensive tumor map in real-time [10,18]. Emerging “smart” QDs are engineered to respond to biochemical stimuli in the tumor microenvironment, such as pH changes, enzymatic activity (e.g., cathepsin cleavage), or oxidative stress, to enhance contrast only in malignant zones [3]. These context-sensitive probes could allow adaptive margin visualization, where fluorescence intensity or emission spectrum shifts based on tissue pathology, helping surgeons distinguish between inflamed, dysplastic, and malignant tissue. Integration with artificial intelligence (AI) further extends the potential of QDs beyond simple imaging. Machine learning algorithms trained on spectral and spatial data can assist in intraoperative classification of tissue types, margin adequacy prediction, and even alerting surgeons of high-risk zones in real time [13]. AI-enhanced platforms could also calibrate QD signal thresholds, minimize false-positives from autofluorescence, and enable precision resection with minimal healthy tissue loss.
Clinical translation of these tools will depend on:
• Development of cadmium-free, FDA-compliant QDs with standardized emission profiles
• Real-world validation in Mohs surgery and excisional procedures
• Incorporation into AI-augmented imaging systems and portable dermatoscopic devices
In this evolving paradigm, QDs are poised not merely as fluorescent tags, but as intelligent surgical co-pilots, navigating the invisible boundaries between normal and neoplastic skin. QDs represent a transformative innovation in the visualization of surgical margins for non-melanoma skin cancers. Their unique optical properties, including high photostability, narrow emission spectra, and tunable fluorescence, make them ideal for real-time, high-resolution imaging in cutaneous oncology. Whether used in vivo during fluorescence-guided surgery or ex vivo on resected specimens, QDs offer unparalleled precision in defining tumor boundaries, potentially reducing recurrence rates and improving patient outcomes. Despite these advantages, several barriers hinder their routine clinical adoption. Concerns about heavy metal toxicity, limited regulatory frameworks, and challenges in achieving targeted delivery without off-tissue accumulation remain unresolved. Nonetheless, the development of biodegradable QDs, improved bioconjugation strategies, and integration with artificial intelligence are actively addressing these limitations. Looking ahead, QD platforms are poised to move beyond static imaging and into the realm of intelligent, multiplexed, and adaptive systems that inform surgical decisions in real time (Table 5). With continued interdisciplinary research and regulatory support, quantum dots hold significant translational promise for enhancing the precision and safety of skin cancer surgery.
|
Platform |
Clinical Application |
Translation Stage |
Challenges Remaining |
|
Cornell Dots (C-dots) |
Melanoma imaging (SLN) |
Human trials |
Scale-up, targeting flexibility |
|
QD800-EGFR |
SCC margin delineation |
Preclinical |
Immunogenicity, cadmium content |
|
QD800-RGD |
Angiogenesis visualization |
Preclinical |
Need for biodegradable QDs |
|
ZnCuInSe/ZnS QDs |
HNSCC 3D imaging |
Preclinical |
Delivery in live tumors |
|
NAC-CdTe/CdS QDs |
DNA biosensing (in vitro) |
Early-stage |
Tissue-specific targeting needed |
Table 5: Clinical and Translational Potential of QDs in NMSC
References
- Hasan, N., Nadaf, A., Imran, M., Jiba, U., Sheikh, A., Almalki,W. H., ... & Ahmad, F. J. (2023). Skin cancer: understanding the journey of transformation from conventional to advanced treatment approaches. Molecular cancer, 22(1), 168.
- Khan, N. H., Mir, M., Qian, L., Baloch, M., Khan, M. F. A., Ngowi, E. E., ... & Ji, X. Y. (2022). Skin cancer biology and barriers to treatment: Recent applications of polymeric micro/ nanostructures. Journal of advanced research, 36, 223-247.
- Liu, Y., Walker, E., Iyer, S. R., Biro, M., Kim, I., Zhou, B.,... & Wilson, D. L. (2019). Molecular imaging and validation of margins in surgically excised nonmelanoma skin cancer specimens. Journal of Medical Imaging, 6(1), 016001- 016001.
- Hu, Z., Qian, G., Müller, S., Xu, J., Saba, N. F., Kim, S., ... & Chen, Z. G. (2016). Biomarker quantification by multiplexed quantum dot technology for predicting lymph node metastasis and prognosis in head and neck cancer. Oncotarget, 7(28), 44676.
- Fang, M., Peng, C. W., Pang, D. W., & Li, Y. (2012). Quantum dots for cancer research: current status, remaining issues, and future perspectives. Cancer Biology & Medicine, 9(3), 151- 163.
- Sarkar, S., Srivastava, T. P., Sahoo, O. S., Shankar, A., Rai, A., Pethusamy, K., ... & Karmakar, S. (2024). Applications of quantum dots in preventive oncology. Asian Pacific Journal of Cancer Prevention: APJCP, 25(3), 747.
- Dirheimer, L., Pons, T., Marchal, F., & Bezdetnaya, L. (2022). Quantum dots mediated imaging and phototherapy in cancer spheroid models: state of the art and perspectives. Pharmaceutics, 14(10), 2136.
- Yang, K., Zhang, F. J., Tang, H., Zhao, C., Cao, Y. A., Lv, X. Q., ... & Li, Y. D. (2011). In-vivo imaging of oral squamous cell carcinoma by EGFR monoclonal antibody conjugated near-infrared quantum dots in mice. International journal of nanomedicine, 1739-1745.
- Huang, H., Bai, Y. L., Yang, K., Tang, H., & Wang, Y. W. (2013). Optical imaging of head and neck squamous cell carcinoma in vivo using arginine-glycine-aspartic acid peptide conjugated near-infrared quantum dots. OncoTargets and therapy, 1779-1787.
- Yakavets, I., Francois, A., Guiot, M., Lequeux, N., Fragola, A., Pons, T., ... & Marchal, F. (2020). NIR imaging of the integrin-rich head and neck squamous cell carcinoma using ternary copper indium selenide/zinc sulfide-based quantum dots. Cancers, 12(12), 3727.
- Dirheimer, L., Pons, T., François, A., Lamy, L., Marchal, F., Dolivet, G., ... & Bezdetnaya, L. (2024). Peptide-mediated targeting of Quantum Dots in a 3D model of head and neck cancer. Photodiagnosis and Photodynamic Therapy, 49, 104337.
- Dapkute, D., Steponkiene, S., Bulotiene, D., Saulite, L., Riekstina, U., & Rotomskis, R. (2017). Skin-derived mesenchymal stem cells as quantum dot vehicles to tumors. International Journal of Nanomedicine, 8129-8142.
- Samathoti, P., Kumarachari, R. K., Bukke, S. P. N., Rajasekhar, E. S. K., Jaiswal, A. A., & Eftekhari, Z. (2025). The role of nanomedicine and artificial intelligence in cancer health care: individual applications and emerging integrations—a narrative review. Discover Oncology, 16(1), 1-35.
- Fadhil, E. A., Lafta, F. M., & Abdullah, M. M. (2024). Synthesis of NA cysteine-capped CdTe QDs for optical biosensing. Journal of Optics, 1-8.
- Mohanty, S., Desai, V. M., Jain, R., Agrawal, M., Dubey, S. K., & Singhvi, G. (2024). Unveiling the potential of photodynamic therapy with nanocarriers as a compelling therapeutic approach for skin cancer treatment: current explorations and insights. RSC advances, 14(30), 21915-21937.
- Kapare, H., Bhosale, M., & Bhole, R. (2024). Navigating the future: advancements in monoclonal antibody nanoparticle therapy for cancer. Journal of Drug Delivery Science and Technology, 106495.
- Adamus-Grabicka, A. A., Hikisz, P., & Sikora, J. (2024). Nanotechnology as a Promising Method in the Treatment of Skin Cancer. International Journal of Molecular Sciences, 25(4), 2165.
- Shetty, B., Chauhan, R. S., Tirupathi, S., Krishnapriya, N., Patil, L., & Rathi, N. (2024). Quantum dots in noninvasive imaging of oral squamous cell carcinomas: A scoping literature review. Journal of Cancer Research and Therapeutics, 20(3), 745-749.


